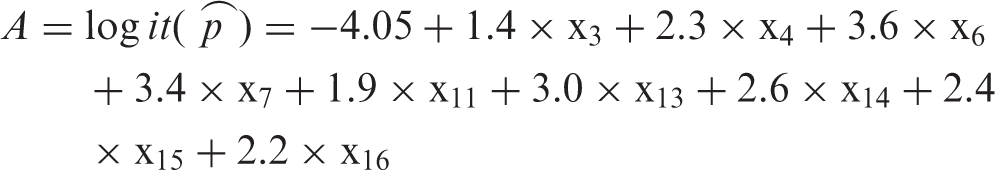Abstract
Background
A positive D-dimer test has high sensitivity but relatively poor specificity for the diagnosis of pulmonary embolism, causing difficulty for clinicians unskilled in pulmonary embolism diagnosis in determining whether a patient with a positive D-dimer test needs to undergo computed tomographic pulmonary angiography.
Objectives
We sought to develop a new clinical decision-making rule based on a positive D-dimer result to predict the probability of pulmonary embolism and to guide clinicians in making decisions regarding the need for computed tomographic pulmonary angiography.
Methods
We conducted a prospective, multicenter study in three hospitals in China. A total of 3014 inpatients with positive D-dimer results were included. In the derivation group, we built a multivariate logistic regression model and deduced a regression equation from which our score was derived. Finally, we validated the score in an independent cohort.
Results
Our score included nine variables (points): chest pain (1.4), chest tightness (2.3), shortness of breath (3.6), hemoptysis (3.4), heart rate ≥100 beats/min (3.6), blood gas analysis (2.9), electrocardiogram presenting a typical S1Q3T3 pattern (4.1), electrocardiogram findings (2.4), and ultrasonic cardiogram findings (3.7). The sensitivities and specificities were 100% and 86.94%, respectively, in the derivation group and 100% and 90.82%, respectively, in the validation group. Additionally, the observed and predicted proportions of patients who underwent computed tomographic pulmonary angiography were 16.82% and 10.76%, respectively, in the derivation group and 18.72% and 11.40%, respectively, in the validation group.
Conclusions
The new score can categorize inpatients with a positive D-dimer test as pulmonary embolism-likely or pulmonary embolism-unlikely, thus reducing unnecessary computed tomographic pulmonary angiography examinations.
Keywords
Introduction
Pulmonary embolism (PE) is a serious disease with high morbidity and mortality.1,2 In addition, because it has nonspecific clinical manifestations and lacks a specific auxiliary examination, PE is associated with high rates of misdiagnosis and missed diagnoses, which has been a common source of medical disputes. 3 Therefore, it is imperative to achieve an accurate and early diagnosis of PE.
Currently, clinicians rely on a variety of noninvasive diagnostic tests, such as plasma D-dimer measurements, blood gas analyses, lower limb proximal deep vein compression ultrasonography, and computed tomographic pulmonary angiography (CTPA), which is acknowledged as the gold standard method, to diagnose PE.4,5 Clinicians pay specific attention to patients with a positive D-dimer test because it has a sensitivity of approximately 97% for PE. However, the specificity of a positive D-dimer test for PE is only approximately 40%,6,7 causing difficulty for clinicians, especially clinicians inexperienced with PE diagnosis, in determining the probability of PE and whether the gold standard CTPA test is needed for an accurate diagnosis. In addition, it is not feasible to extensively use CTPA to reduce the risk of a missed diagnosis in PE. Only approximately 27% of patients who undergo CTPA have been reported to be subsequently diagnosed 8 ; therefore, increased use of CTPA represents a waste of medical resources, results in expensive investigations, and increases related risks.9,10 In addition, CTPA cannot be performed in suspected PE patients in many rural hospitals due to lack of equipment. Therefore, more appropriate and noninvasive approaches for PE diagnosis need to be explored and developed.
In response to these findings, a few clinically predictive models for PE have been developed to separate suspected PE patients into groups with different pretest probabilities.11,12 Two of the most validated and widely used clinical prediction tools are the Wells score and the revised Geneva score.12–14 Of these, the Wells score is more accurate than the revised Geneva score for diagnosing PE15,16; however, its utility is controversial because subjective criteria (e.g., “whether an alternative diagnosis is less likely than PE”) carry a great deal of weight in scoring 17 and limit its standardization, resulting in poor interobserver reproducibility. The Wells group further found that the combination of a low or intermediate clinical probability according to the Wells score and a normal D-dimer result can be inferred to safely exclude a diagnosis of PE.17–19 However, there is currently no explanation for why this combination is effective, and many clinicians are therefore confused when interpreting results. Above all, these scores are based on patients with suspected PE according to their clinical history rather than positive D-dimer results and are intended to predict the likelihood of PE, aiming to help clinicians who are inexperienced with PE diagnosis make further clinical decisions related to confirming or excluding a PE diagnosis. Thus, in this study, we analyze hospitalized patients who have already undergone testing for D-dimer with positive results (i.e., not all symptomatic patients are analyzed) and explore a new clinical prediction rule that is based on positive D-dimer results and objective clinical variables. We hope that this system will guide clinicians in making appropriate decisions for patients with positive D-dimer results and reduce unnecessary CTPA examinations.
In the present study, we developed a clinical decision-making rule that is based on positive D-dimer results. We combined clinical variables that can be observed in inpatients in real-time, including symptoms, signs and findings on frequently performed texts (e.g., electrocardiography, electrocardiogram (ECG), echocardiography, ultrasonic cardiogram (UCG), and blood gas levels). 20 Moreover, our rule is derived from a study involving a large multicenter cohort of inpatients with positive D-dimer results, and our test passed an external retrospective validation, demonstrating that it is credible and valid.
Methods
Study Design and Patients
We designed a multicenter prospective outcome study with the aim of developing a clinical decision-making rule to aid in PE diagnosis. We conducted the study at the in-patient departments of three general and teaching hospitals (The Second Affiliated Hospital of Chongqing Medical University, China; The Second People’s Hospital of Chengdu, China; and The First Affiliated Hospital of Kunming Medical College, China) from August 2016 to May 2017. Clinicians assessed inpatients’ conditions and determined whether they needed to undergo a plasma D-dimer test, and we selected consecutive inpatients with elevated plasma D-dimer who were older than 18 years as the eligible study population. The following exclusion criteria were applied: (1) ongoing anticoagulant treatment, (2) contraindications to CT (e.g., a known allergy to contrast iodine agents, a risk of allergic reaction, creatinine clearance of 0.50 mL/s (30 mL/min), 21 or pregnancy), (3) an estimated life expectancy of less than one month, and (4) younger than 18 years. We obtained written informed consent from all enrolled patients in addition to the approval of the ethics committee of our institution. Between August 1, 2016 and May 31, 2017, we screened 3101 patients with a positive plasma D-dimer result and excluded 87 patients because of a contraindication for CT (n = 52) or protocol violations (n = 35). In total, we enrolled 3014 patients in this study.
Once the patients were enrolled, they underwent a sequential diagnostic work-up, including an arterial blood gas analysis (while breathing room air), ECG, UCG, and chest radiography. The clinicians in charge of the patients performed the clinical evaluations, including recording demographic characteristics on a standardized data form (Form 1); recording clinical signs and symptoms of PE; and recording the results of arterial blood gas analysis tests, ECG, UCG, and chest radiography. We did not interfere with the clinicians’ decisions regarding diagnostic and treatment strategies and merely noted which patients underwent CTPA and the discharge diagnosis.
Collect the basic clinical data of inpatients.
Diagnostic Strategy
We considered patients PE positive if they met at least one of the following conditions: a positive pulmonary angiogram in patients with a high clinical probability, a positive helical CT scan, a high-probability VQ scan plus a moderate or high pretest probability 22 or a venous thromboembolic event within the one-month follow-up period. Most of the patients were diagnosed by CTPA, which is the most commonly used imaging test to confirm PE because of its diagnostic accuracy and wide availability. After the patients were discharged from the hospital, we tracked them for up to one month by telephone. During this month, we estimated the incidence of confirmed symptomatic thromboembolic events as the rate of missed diagnoses of PE during hospitalization.
Score derivation
The patients who were hospitalized between August 1, 2016 and March 31, 2017 were defined as the derivation sample. We evaluated all the clinical variables (15 variables referred to here as X1–X15, see Table 2) in our database that are known to be potentially associated with PE and performed a univariate analysis to select predictive variables (defined as those with p values <0.05, indicating significance) for a logistic stepwise regression model. We included the variables that were significantly associated with PE in a logistic stepwise regression analysis, which was performed using the least squares principle (α = 0.1). The strength of the association between each candidate predictor and PE was determined using odds ratios. We established a multiple linear regression model and derived the regression equation
Score validation
We validated the score in an independent cohort of inpatients who were enrolled between April 1, 2017 and May 31, 2017 were not recruited into the derivation set and were recruited from two hospitals (The Second Affiliated Hospital of Chongqing Medical University, China, and The Second People’s Hospital of Chengdu, China). The diagnostic algorithm and the diagnostic criteria used to determine the presence of PE were identical to those used in the derivation sample study. We collected the variables that were significantly associated with PE and retrospectively calculated the score for each patient. All patients were then followed for one month after they were discharged from the hospital.
Statistical analysis
We assessed the validity and value of our score by calculating its sensitivity and specificity, agreement rate, positive and negative predictive values, false-positive rate, and false-negative rate in both the derivation group and the validation group. We then compared the above data between the derivation and validation samples. In addition, we compared the proportions of observed and predicted patients who underwent CTPA in both the derivation and the validation group using a chi-square test. A p value <0.05 indicated a significant discrepancy between the observed and predicted events. We performed all statistical analyses using SPSS software.
Results
The basic characteristics of the participants.

ECG: electrocardiogram; UCG: ultrasonic cardiogram; PAP: pulmonary arterial pressure; TRPG: tricuspid regurgitation peak systolic gradient.
Variables enrolled in and their associations with pulmonary embolism in the univariate analysis.
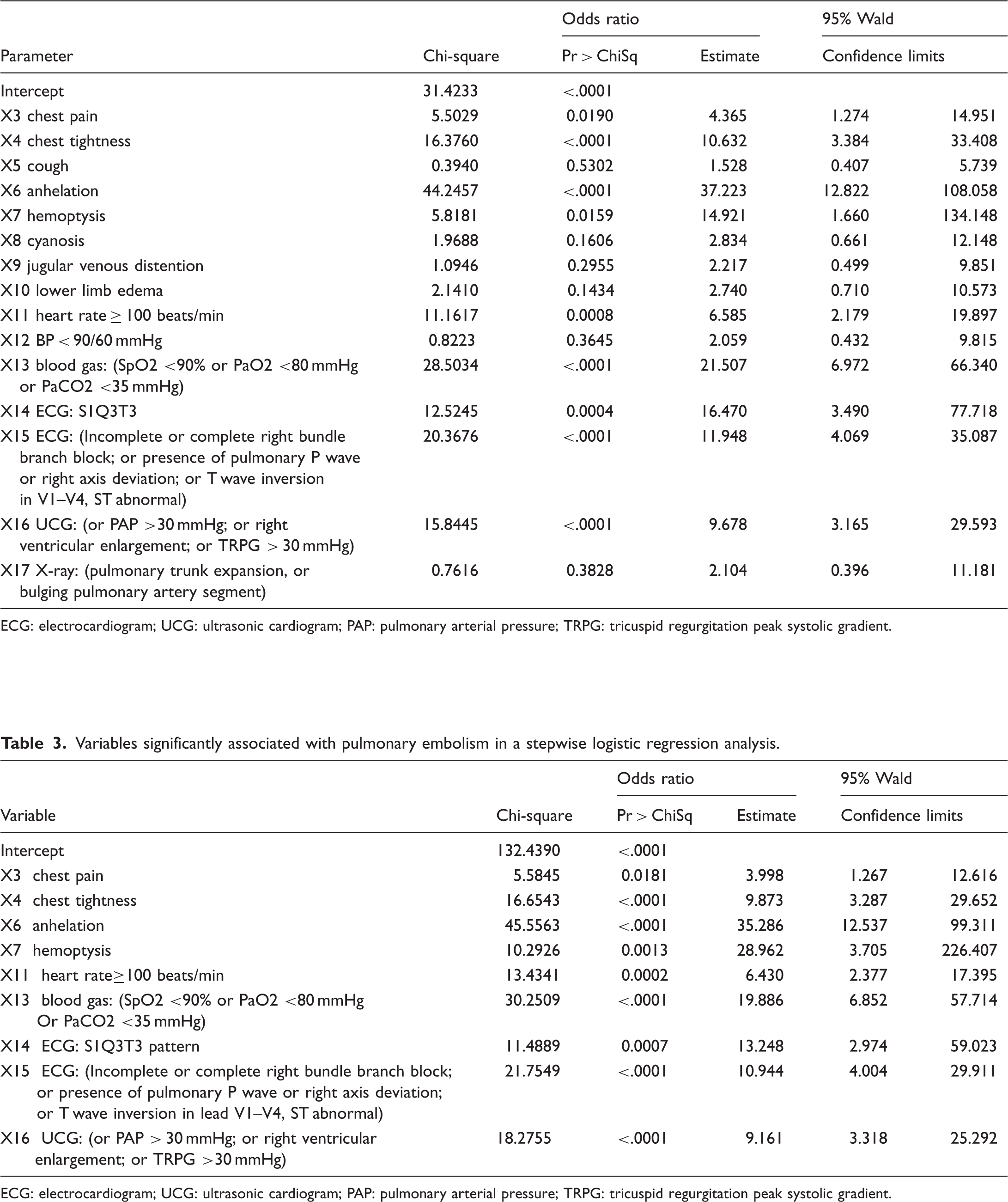
ECG: electrocardiogram; UCG: ultrasonic cardiogram; PAP: pulmonary arterial pressure; TRPG: tricuspid regurgitation peak systolic gradient.
Variables significantly associated with pulmonary embolism in a stepwise logistic regression analysis.
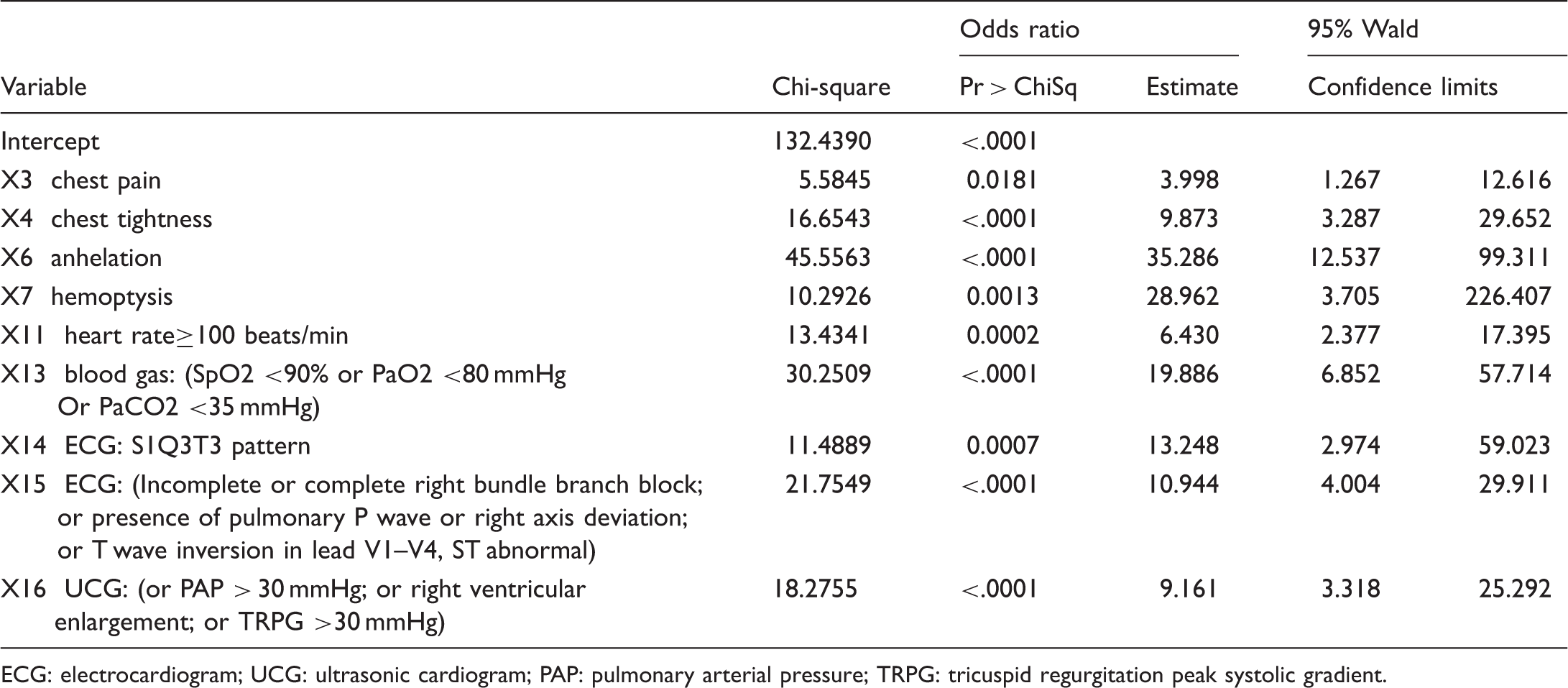
ECG: electrocardiogram; UCG: ultrasonic cardiogram; PAP: pulmonary arterial pressure; TRPG: tricuspid regurgitation peak systolic gradient.
Final score.

ECG: electrocardiogram; UCG: ultrasonic cardiogram; PAP: pulmonary arterial pressure; TRPG: tricuspid regurgitation peak systolic gradient.
Validity and value of the score
Proportion of patients in each category and the validity and values of the score.
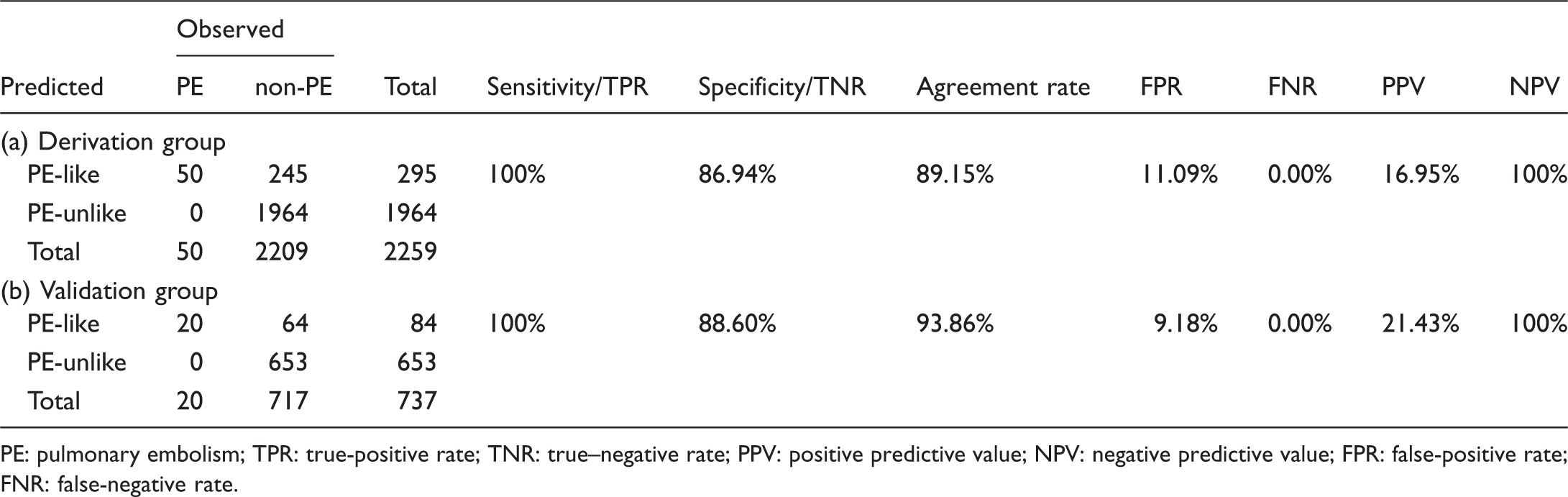
PE: pulmonary embolism; TPR: true-positive rate; TNR: true–negative rate; PPV: positive predictive value; NPV: negative predictive value; FPR: false-positive rate; FNR: false-negative rate.
Observed and predicted proportions of patients undergoing CTPA in both derivation group and validation group.
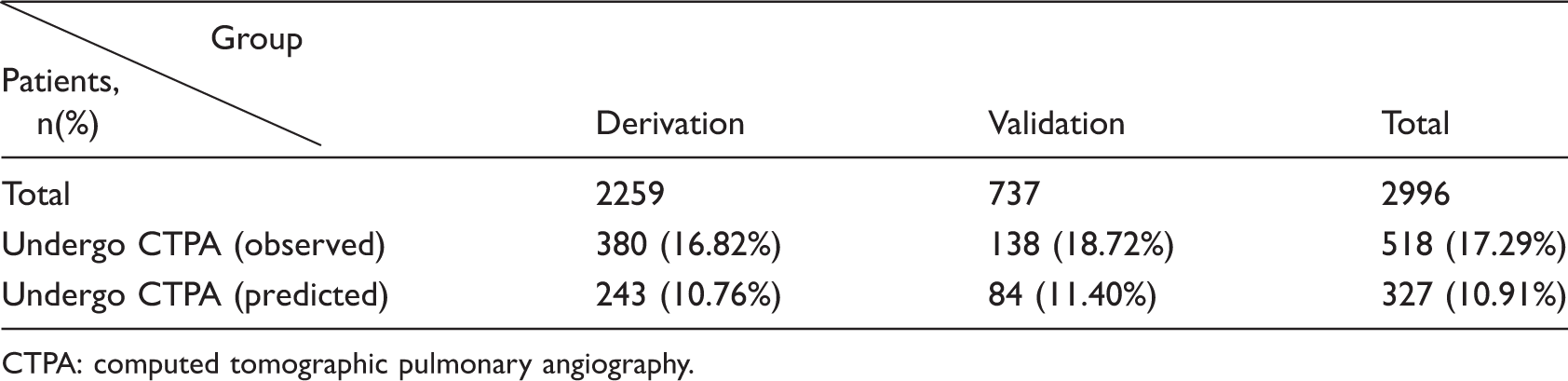
CTPA: computed tomographic pulmonary angiography.
Discussion
In this study, we present a clinical decision-making rule that was developed using a large multicenter cohort of consecutive inpatients with positive plasma D-dimer results. The score, which was derived using a recommended method, was standardized and based on clinical information that was collected from the patients in real-time. The score allows clinicians to make appropriate decisions for patients with positive D-dimer results and is expected to reduce unnecessary CTPA examinations.
Clinicians always view a positive plasma D-dimer test as a cautionary sign of PE because of its high sensitivity for PE. However, the specificity of D-dimer is only approximately 30%–40%,6,7 causing difficulty for clinicians who are inexperienced in diagnosing PE in determining the probability of PE and whether a further diagnostic strategy is needed based on a positive D-dimer result. 23 We therefore developed a new rule for predicting PE in which we combined a positive D-dimer result with other clinical features. We selected several items significantly associated with PE for inclusion into the score, including symptoms and signs commonly encountered in PE and the results of related clinical examinations (blood gases, ECG, UCG).24–26 The data related to the clinical manifestations included in the score are easy to obtain from the patient’s history and a physical examination. In addition, the clinical examinations required are relatively noninvasive and simple to perform, which is acceptable for patients. Moreover, all the variables included in this score are reasonably straightforward and objective, and the scorer can easily come to a “black or white” conclusion (i.e., whether the patient is PE-likely or PE-unlikely). These factors make our score easy to compute and repeatable. Therefore, this scoring system is feasible for both clinicians and inpatients.
We clearly identified PE, the outcome of interest, and defined PE using accepted diagnostic criteria that were verified during a one-month follow-up period. 27 All the items included in this score were precisely defined and reasonably straightforward to obtain. In addition, we used classical statistical methods, including univariate analysis, to identify the variables that are significant associated with PE. This was followed by a logistic stepwise regression analysis that included all the identified significant predictors. In addition, the score was validated by different clinicians in a cohort of patients that was entirely nonoverlapping with the derivation sample. As shown in Table 5, our score also achieved favorable accuracy in both the derivation and validation sets. Indeed, the diagnostic criteria, follow-up mode, and diagnostic algorithm used in the two sets were identical. Although the validation samples were recruited from the same two centers (The Second Affiliated Hospital of Chongqing Medical University, China, and The Second People's Hospital of Chengdu, China) as that of derivation group, they were recruited during different periods (August 2016 to March 2017 and April 2017 to May 2017, respectively). However, a commonly accepted rule requires that there should be at least five test cases with the predicted outcome (PE) per independent variable. In our derivation sample group, 50 patients were confirmed to have PE, and the final score comprised only nine variables, for an average of 5.6 cases for per variable. We consider that this result may be due to our broad inclusion criteria (inpatients with elevated plasma D-dimer levels). The clinicians did not have strict inclusion criteria for performing the D-dimer test for hospitalized patients because the plasma D-dimer test is relatively inexpensive (approximately $12) and aids in the diagnosis of many diseases. Clinically, the D-dimer test can assist in the diagnosis of patients with suspected PE and suspected lower extremity deep venous thrombosis 28 and can be performed as a routine screening test before and after surgery and for elderly or bedridden patients as well as patients with atrial fibrillation or myocardial infarction.29,30 In addition, the use of the D-dimer test is promising in the diagnosis of periprosthetic joint infection and for predicting some types of cancer.31,32 Therefore, the group of patients with elevated D-dimer levels will inevitably include many patients with other diseases other than PE. However, this issue does not reduce the value of our score because our score is not only intended to predict PE but to help clinicians make appropriate diagnostic decisions for patients with elevated D-dimer levels. Unavoidably, our study has the limitation of missing data. In the derivation and validation groups, we missed 11 (0.48%) and 7 (0.94%) patients, respectively; 8 of them quit midway through the study for personal reasons, and the other 10 patients were lost to contact during the one month of follow-up. Nevertheless, 0.48% and 0.94% are quite low missing rates and do not weaken the reliability of our results. Although our research can help clinicians decide whether to perform CTPA to exclude PE in patients with positive D-dimer results, our model requires further examinations, such as ultrasound, ECG, and blood gas analysis, which are somewhat expensive. However, it can avoid excessive CTPA and missed diagnosis of PE and can help clinicians, especially those who are inexperienced with PE diagnosis, to make appropriate clinical decisions for patients with elevated plasma D-dimer levels.
Many clinicians, especially nonspecialists (e.g., nonrespiratory or cardiovascular physicians) who diagnose PE, commonly run into problems when they encounter patients with a positive plasma D-dimer result. In cases of missed PE diagnosis, some clinicians manage these patients radically by broadly assigning examinations, such as CTPA, VA scans, chest 64–detector row CT, or even anticoagulation therapy, as preventive measures. 33 In contrast, some clinicians choose to temporarily wait when inpatients have a positive D-dimer result and reexamine their plasma D-dimer levels after a few days. However, the patient’s symptoms could become aggravated during this time, resulting in further necessary measures. Our score can be used in inpatients with a positive plasma D-dimer result to enable clinicians to evaluate the likelihood of PE and predict which patients require further examinations. A patient with a positive D-dimer test and a score of at least 4.05 points on our scoring system has a high probability of having PE. We therefore recommend that these patients, who were classified as PE-likely by our system, undergo the gold standard test, CTPA. As shown in Table 6, the rate at which CTPA examinations were recommended by our scoring system and the rate observed in an actual clinical setting were 10.91% and 17.29%, respectively (p < 0.05). Hence, this scoring system, to some extent, could be used to reduce the number of unnecessary CTPA examinations. Hence, our scoring system not only lightens the burden on patients but also helps clinicians to make timely decisions regarding the diagnosis of and therapeutic strategy for inpatients with a positive D-dimer test. However, this system’s clinical usefulness and the adherence of both clinicians and patients should be further studied.
The present score is not a diagnostic criterion but rather a tool to help doctors, especially non-PE specialists, determine which patients with a positive D-dimer test need to undergo a CTPA examination. As the results show, our score resulted in a false-negative rate (rate of missed diagnoses) of 0.00% (p>0.05) in the both the derivation and validation groups. Its sensitivity (TPR) was correspondingly high at 100% in both the derivation and validation groups, indicating that the score accurately and effectively identified PE patients. Regardless of sensitivity or specificity, our score seems more favorable than the Wells score and the Geneva score. 34 A meta-analysis comparing Wells scores and Geneva score shows that the sensitivities ranged from 63.8% to 79.3% and 55.3% to 73.6%, respectively, and that the specificities ranged from 48.8% to 90.0% and 51.2% to 89%, respectively. 34 However, further appropriate statistical analysis is required for their comparison. However, to minimize the rate of missed diagnoses, false-positive and relatively low-specificity results are unavoidable (Table 5). In the derivation and validation groups, we achieved false-positive rates (the misdiagnosis rate) of 11.09% and 9.18%, respectively (p>0.05), and specificities of 86.94% and 90.82%, respectively (p>0.05). Our score did result in the misdiagnosis of some non-PE diseases. For instance, a patient who suffered from atrial fibrillation and had a rapid heart rate combined with an intracardiac mural thrombus and heart failure was regarded as PE-likely by our system. In addition, another patient with chest wall trauma accompanied by a reactive increase in plasma D-dimer levels was also classified as PE-likely by our scoring system because the patient suffered from chest pain and subsequently presented with a rapid heart rate (>100 beats/min) and overventilation (PCO2 <35 mmHg). Therefore, the presented rules could be further improved in the future.
The Wells rule is the most extensively validated and most widely used rule for making clinical decisions. It is mainly based on risk factors for PE, such as malignancy, surgery and previous DVT/PE. 7 The scoring system presented here relies far more on clinical information, including symptoms, signs, and the results of additional clinical examinations performed after a positive D-dimer test that can be obtained in real-time. We therefore suggest that our score and the Wells score may be complementary in clinical applications. In addition, the Wells rule includes one real-time clinical item, i.e., “whether an alternative diagnosis is less likely than PE.” 7 This item is important but is also subjective and dependent on the clinical experience of the practitioner. Our score can assist clinicians in assessing this type of subjective criterion by dividing patients into PE-likely and PE-unlikely categories. We therefore propose that our scoring system should be combined with the Wells rule to improve the accuracy of clinical diagnoses of PE. Such a combined system remains to be studied.
In summary, we developed the first PE-related decision-making rule that is specific to inpatients with a positive D-dimer result. Our aim is to assist clinicians, especially non-PE specialists, in deciding which patients require further CTPA examination. The score is intended to minimize the rate of missed diagnoses in PE and reduce unnecessary CTPA examinations. Furthermore, our score, which is based on clinical manifestations and simple auxiliary examinations, is straightforward to obtain, objective in nature, and easy for both patients and clinicians to comply with. We therefore expect it to be widely used.
Footnotes
Authors’ contribution
M. L. wrote the manuscript, performed the analyses, and participated in designing the study. D. J. supervised and participated in the analyses and participated in writing and designing the study. C. L. constructed the prediction models, performed the statistical analyses, and helped to write the study. Z. L., Z. X., J. L., and C. W. participated in gathering the data. Y. J. coordinated the handling of samples. All authors reviewed the manuscript carefully and approved the final version.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: D. J., the corresponding author of the present study, is supported by a grant from the National Natural Science Foundation of China (grant number 81650003).