Abstract
Background:
Pulmonary hypertension (PH) is an underdiagnosed cause for chest pain in patients without significant coronary artery disease (CAD). Studies showed that enlarged pulmonary arterial (PA) and right ventricular chamber sizes correlate with the severity of PH. Therefore, we studied the association between chest pain, right ventricular dimensions (RVDs), and PA size on coronary coronary tomographic angiography (CCTA).
Methods:
The CCTA of 87 patients presenting with chest pain without evidence of obstructive CAD was examined. The PA diameter (PAD), right atrial dimension (RAD), and RVD were measured. A comparative control cohort included 31 patients who presented without cardiopulmonary complaints and underwent thoracic CT. The risk for obstructive sleep apnea (OSA) was assessed using STOP-BANG questionnaires.
Results:
Patients with chest pain without obstructive CAD showed markedly dilated right atrial and ventricular chambers compared with standard parameters (right atrium: 48 ± 6.4 mm; right ventricle long axis: 61 ± 9.5 mm). When comparing chest pain vs non-chest pain group, respectively, the mean PAD measured 25.92 ± 0.43 mm vs 22.89 ± 0.38 mm (P < .001), RAD2 measured 40.1423 ± 0.7108 mm vs 34.8800 ± 1.0245 mm (P = .0048), and RVD2 measured 31.7729 ± 0.7299 mm vs 27.6379 ± 1.6178 mm (P = .034). Chest pain was associated with higher PAD (odds ratio [OR]: 11.11, P < .05) after adjusting for age, sex, body mass index, history of hypertension, hyperlipidemia, congestive heart failure, chronic obstructive pulmonary disease, OSA, and smoking. The chest pain group had a mean STOP-BANG score of 3.9 ± 1.8 in all patients, and 3.62 ± 0.20 in patients without known history of OSA, representing an elevated risk index for the disease.
Conclusions:
In patients presenting with chest pain without obstructive CAD on CCTA, there is a strong association between the presence of chest pain and enlarged PAD. They also represent a high-risk group for OSA.
Keywords
Introduction
Chest pain accounts for 6 million annual visits to the emergency department (ED) in the United States, making it the second most common complaint nationwide. 1 Patients presenting to the ED with chest pain constitute an important diagnostic challenge. Many of these patients have a low intermediate probability of coronary artery disease (CAD) which can be ruled out by computerized coronary tomographic angiography (CCTA) based on evidence presented in clinical studies such as Rule Out Myocardial Infarction/Ischemia Using Computer Assisted Tomography (ROMICAT) trial. 2 Pulmonary hypertension (PH) is an uncommon cause of chest pain accompanying dyspnea, which resembles angina pectoris in many characteristics including location, radiation, intensity, quality, and even relation to exertion as the afferent nerves from the pulmonary artery (PA) enter the nervous system with similar pathways as those from the heart.3–6 These similarities can lead to an erroneous diagnosis of CAD. Based on the characteristics of their symptoms, many of these patients undergo a diagnostic gated CCTA for the evaluation of coronary arteries. This provides a unique opportunity to the interpreting physicians to evaluate the PA size, and to some extent, right ventricular (RV) size and morphology. This can determine whether this radiologic variable associated with PH in patients without significant CAD correlates with chest pain. Such approach can help early detection of the subset of chest pain patients who have possible PH as underlying cause of their chest pain, thus facilitating early treatment for PH.
Earlier detection of PH improves outcomes. 7 Therefore, screening algorithms have been proposed to facilitate the timely diagnosis of PH, using a combination of echocardiographic, physiologic (lung function), and radiologic noninvasive techniques, 8 before proceeding to the invasive yet confirmatory right heart (RH) catheterization. With the advancements in computed tomographic (CT) scan technology, there have been attempts to address the utility of CT to predict the presence of PH. Computed tomographic measurements of PA dimensions have shown a correlation with the presence of PH and its severity, 9 providing a potential for noninvasive methods for the diagnosis of the disease.
The PA as a vessel is more compliant than the systemic arterial system, making it more sensitive to pressure and volume changes. Consequently, an increase in mean PA pressure correlates with PA diameter (PAD). 9 Many PA dimensions have been studied for presence of an association between the presence and severity of PH, including the PAD, the cross-sectional area, the ratio of the diameter to the bronchus, the ratio of the diameter to the pulmonary vein, and the ratio of diameter to the aortic diameter, among others.10–13 Generally, the measurement of the main PA size using CT shows a moderate to strong correlation with PH. 9 Furthermore, PA dimensions might have prognostic clinical utility besides its ability to predict PH, as a study showed that in patients with inoperable chronic arterial or thromboembolic PH, the PA size was associated with unexpected death. 14
All causes of pulmonary arterial hypertension (PAH) (group 1) and non-PAH (groups 2-5) PH must be considered. 15 Obstructive sleep apnea (OSA) is an underdiagnosed chronic disorder that is associated with PH (World Health Organization [WHO] Group III). Various questionnaires have been used to triage patients with suspected OSA, of which the “STOP-BANG questionnaire” has demonstrated to have high sensitivity. 16 However, the diagnosis of OSA requires an overnight polysomnography to detect apneic events. Obstructive sleep apnea had been associated with noncardiac chest pain and angina. 17 In fact, with the use of continuous positive airway pressure in patients with OSA, the number of angina episodes has been decreased. 17
Therefore, this study was conducted to investigate the relationship between chest pain and radiologic parameters associated with PH such as PAD and right-sided cardiac morphology. This can discern the association between chest pain and PH, as well as its underlying causes including OSA.
Methods
Patient selection
At Buffalo General Medical Center, all identifiable patients were prospectively recruited from the February 1, 2015 to August 31, 2015. The study population was categorized into 2 groups: chest pain and non-chest pain (control) groups. The inclusion criteria for the chest pain group were as follows: (1) presented with chest pain, (2) age >18 years, and (3) underwent CCTA showing absence of obstructive CAD (defined as >50% coronary stenosis) and absence of detectable pulmonary embolism. The non-chest pain group includes patients who meet the following inclusion criteria: (1) absence of chest pain or cardiopulmonary symptoms at initial presentation, (2) age >18 years, and (3) received CT chest. Exclusion criteria for both groups include patients less than 18 years of age, or patients with prior history of obstructive CAD, coronary artery bypass surgery, percutaneous intervention, or acute coronary syndromes. The study protocol was approved by the institutional review board.
Data abstraction
Clinical data abstraction by chart review was performed independently by 2 abstractors (N.M. and A.C-R.). The discrepancy between the abstractors was settled by joint review and discussion. To examine the probability of OSA, abstractors also collected data from the STOP-BANG survey questionnaire for OSA, which was provided to the patients enrolled at the time of hospital visit or collected on phone interview postdischarge. Demographic and baseline characteristics collected include age, sex, race, body mass index (BMI), baseline left ventricular ejection fraction, past medical history of medical conditions including congestive heart failure (CHF), hypertension (HTN), hyperlipidemia (HLP), chronic obstructive pulmonary disease (COPD), OSA, and smoking. Presence of electrocardiographic (ECG) evidence of RH strain was also obtained in the chest pain group including P-pulmonale, RV hypertrophy, right bundle branch block, RV conduction delay (RVCD), and right axis deviation.
CT image analysis
Analysis of the CT images was performed independently by 2 abstractors (N.M. and A.C-R.) with interobserver agreement of measurement technique. Computerized coronary tomographic angiography image data include PA size, and diastolic right atrial dimension (RAD) and RV dimension (RVD). The CT chest of control group was used to measure PA size using similar methods; however, RAD and RVD could be measured accurately in only 15 patients due to non–ECG-gated nature of such studies that led to significant motion artifacts of RV chamber. Pulmonary artery size was measured at the level of arch of aorta as shown in Figure 1A. RAD1 and RAD2 dimensions and RVD1, RVD2, and RVD3 dimensions were measured as shown in Figure 1B. RVD1 is a basal diastolic diameter of RV cavity at the level of tricuspid annulus. RVD2 is the mid-RV cavity diastolic diameter. RVD3 is a measurement of the longitudinal extent of RV cavity that extends perpendicularly from tricuspid annulus to the RV apex. RAD1 is a maximal RA cavity diameter measured parallel to tricuspid annulus. RAD2 is the maximal RA diameter measured perpendicular to the tricuspid annulus.
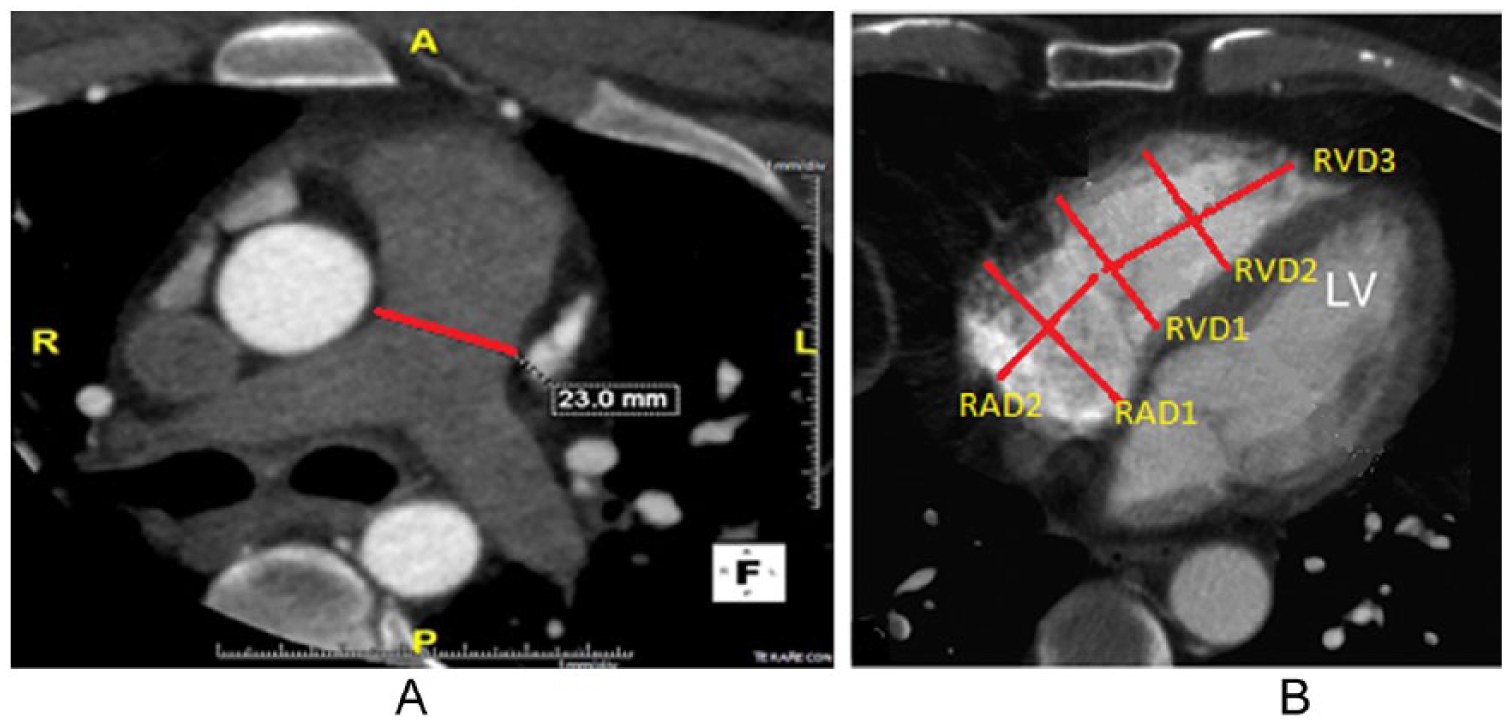
Illustration of measurement method of (A) PAD, (B) RAD and RVD on CCTA. RVD1 is a basal diastolic (images were taken during diastole but not necessarily end diastole) diameter of RV cavity at the level of tricuspid annulus. RVD2 is the mid-RV cavity diastolic diameter. RVD3 is a measurement of longitudinal extent of RV cavity that extends perpendicularly from tricuspid annulus to the RV apex. RAD1 is a maximal RA cavity diameter measured parallel to tricuspid annulus. RAD2 is the maximal RA diameter measured perpendicular to the tricuspid annulus. A indicates anterior; P, posterior; R, right; L, left; PAD, pulmonary artery diameter; RAD, right atrial dimension; RVD, right ventricular dimension.
Statistical analysis
The primary measurement parameter was the association between acute chest pain and PAD in patients without significant CAD. Secondary measures included the average RAD and RVD in the chest pain group of patients, and their risk of OSA-based STOP-BANG questionnaire score. Statistical analyses were performed using SAS 9.4 (SAS Institute Inc., Cary, NC, USA). Comparisons between chest pain and non-chest pain groups were done using contingency tables, χ2 test for dichotomous variables, and 2-tailed t test for continuous variables. The difference in the mean PAD was compared between the 2 study groups, and a subgroup analysis based on sex was also performed. In addition, PAD was categorized to be significantly high or low according to the study participants’ median value. Odds ratios (ORs) for the association between chest pain and PAD were adjusted for potential confounders (age, sex, BMI, history of HTN, HLP, OSA, CHF, COPD, and smoking) using logistic regression model. A P value of <.05 was considered to be statistically significant.
Results
Demographic and clinical characteristics
There were a total of 118 patients identified, including 87 in the chest pain and 31 in the non-chest pain group. The demographic characteristics of the study participants in chest pain and non-chest pain groups are listed in Table 1. The chest pain group had a significantly higher BMI 30.61 ± 0.84 vs 26.88 ± 0.71 (P = .001), were more likely to have a history of HTN (55.17% vs 17.24%, P < .001), HLP (26.44% vs 7.14%, P = .03), and smoking (26.44% vs 7.14%, P < .001). Of all the patients in the chest pain group, 15 had evidence of RH strain on ECG, 13 of these showed presence of RVCD. As illustrated in Table 2, no significant differences in PAD, RVDs, or RADs were found in patients with our without ECG evidence of RH strain.
Demographic characteristics of the study participants in chest pain and non-chest pain groups.

Abbreviations: HTN, hypertension; HLP, hyperlipidemia; CHF, congestive heart failure; COPD, chronic obstructive pulmonary disease; OSA, obstructive sleep apnea.
Results are expressed as percentage or mean ± SEM.
P < .05.
Comparison of PAD, RVD, and RAD in the chest pain group patients with or without evidence of RH strain on ECG.

Abbreviations: ECG, electrocardiography; PAD, pulmonary artery diameter; RAD, right atrial dimension; RH, right heart; RVD, right ventricular dimension.
Results are expressed mean ± SEM.
Pulmonary arterial size
The results of the comparison of mean PAD of the study participants between chest pain vs non-chest pain groups are listed in Table 3, which show that PAD was significantly greater in the chest pain group, even after adjusting for the sex differences. The overall mean PAD was measured as 25.92 ± 0.43 mm in the chest pain group and 22.89 ± 0.38 mm in the control group (P < .001). This remained significant when performing a subgroup analysis for sex. Men in the chest pain vs non-chest pain groups had PAD of 26.44 ± 0.58 mm vs 23.73 ± 0.39 mm, respectively (P = .0063), whereas women had PAD of 25.57 ± 0.61 mm vs 22.21 ± 0.58 mm (P = .0037), respectively. Odds ratio between chest pain and a significantly higher PAD was 11.11 (2.88-42.83, P < .05), 11.26 (2.10-60.38, P < .05) after adjusting for age, sex, BMI, history of HTN, HLP, CHF, COPD, OSA, and smoking.
The mean PAD, RVD, and RAD of the study participants compared between chest pain vs non-chest pain groups.
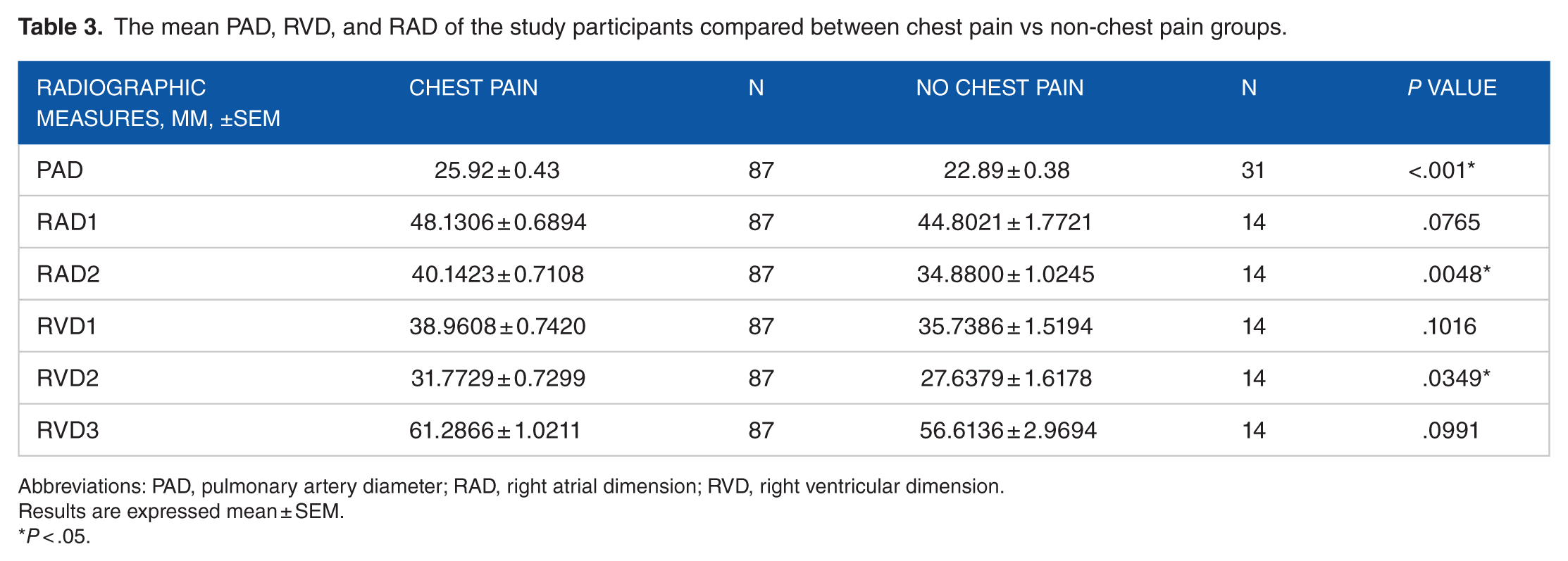
Abbreviations: PAD, pulmonary artery diameter; RAD, right atrial dimension; RVD, right ventricular dimension.
Results are expressed mean ± SEM.
P < .05.
RV chamber size
The chest pain group had an RAD1 of 48.13 ± 6.43 mm, RAD2 of 40.14 ± 6.63 mm, RVD1 of 38.96 ± 6.92 mm, RVD2 of 31.77 ± 6.81 mm, and RVD3 of 61.29 ± 9.52 mm. Based on the existing echocardiographic reference ranges, these measures fall within the upper limits of normal range. 18 When comparing chest pain vs non-chest pain group, respectively, the mean RAD2 measured 40.1423 ± 0.7108 mm vs 34.8800 ± 1.0245 mm (P = .0048), and the mean RVD2 measured 31.7729 ± 0.7299 mm vs 27.6379 ± 1.6178 (P = .034). The rest of the RV measurements, as outlined in Table 3, were not found to be significantly different between the 2 groups.
STOP-BANG questionnaires for the likelihood of OSA
Based on the STOP-BANG survey, the chest pain group for the risk of OSA showed a mean score of 3.9 ± 1.8, which falls under intermediate to high probability of OSA compared with previously reported standards. 16 In addition, the chest pain group with a history of OSA has a mean score of 5.56 ± 0.30, whereas without history of OSA has a mean score of 3.62 ± 0.20 (P < .01).
Discussion
The results of our study demonstrate an association between the presence of acute chest pain and enlarged PA dimensions. In agreement with previously reported studies, we also confirm that these patients represent higher risk group for the presence of OSA. Evaluation of RV has also shown elevated diastolic chamber dimensions in patients presenting with acute chest pain and without obstructive CAD on CCTA. Furthermore, the association between the chest pain of possibly noncoronary cause and enlarged main pulmonary dimensions was clearly discernible, even after correcting for potential confounding variables including age, sex, BMI, history of HTN, HLP, CHF, OSA, COPD, and history of smoking. Our results show that ECG evidence of RH strain does not translate into radiologic evidence of PA, RV, or RA enlargement. This may be due to the fact that most of the patients with positive RH strain on ECG was due to RVCD, which is nonspecific to PH and can have false positives,19–21 as one study showed that only the ventricular activation time in V1 correlated with RV dilation. 21
Many of the acute chest pain patients that present to the ED with low-to-intermediate probability of CAD which is ultimately ruled out by CCTA are left without an explanation to their clinical symptom or left with an umbrella diagnosis of “noncardiac chest pain” at disposition. Our study results illustrate the potential that a portion of these patients’ acute chest pain may be related to the increased RH dimensions, especially PAD. Although PAD is not the gold standard method of diagnosing PH, it can serve as a screening tool to identify patients that are high risk for developing PH, thus leading to an earlier diagnosis and identification of the underlying causes, such as OSA. Multiple prior studies have shown that an increased PA size is associated with PH with a specificity of up to 100%.22–27 One limitation to the outcome of our study is that the chest pain group had an average PAD of 25.92 ± 0.43 mm. However, most studies showing a high specificity for predicting PH using PAD used higher cutoffs.22–27 Although there are multiple causes of PH in the chest pain group (WHO groups I-V), our study examines whether OSA is a potential contributory variable for the increased PAD and acute chest pain. This is illustrated by the significant differences in RV and proximal PA dimensions in the chest pain vs non-chest pain groups (18.4% vs 0.0%, P = .0145) and also by the high STOP-BANG score in the chest pain group. However, the OR for the association between chest pain and PAD remained significant even after adjusting for history of OSA as a confounding variable which may indicate that other causes of PH may be the underlying confounders. Nonetheless, patients in the chest pain group without a history of OSA had a mean score of 3.62 ± 0.20, which demonstrates intermediate risk for the development of OSA, also indicating a possibility of undiagnosed OSA in this study group. Although we attempted to exclude patients with pulmonary emboli as a confounding variable, small pulmonary emboli not detected on CCTA may still be an underlying cause of PH in the chest pain group.
Limitations of our study include the inability to measure the RV and atrial dimensions in our control cohorts due to significant motion artifacts. We also were unable to obtain enough STOP-BANG surveys on patients in the non-chest pain to analyze the difference in scores between the 2 groups. Furthermore, further studies using gold standard methods of diagnosing PH and OSA are needed to demonstrate the direct association between chest pain in patients ruled out for obstructive CAD and these diseases. Nevertheless, this study strongly highlights the possibility of PH and OSA as potential contributors of acute chest pain in patients without obstructive CAD on CCTA. This is clinically relevant to health care providers as it encourages further diagnostic investigations for the disease in that subset of patients, which would allow for early the detection and treatment of PH and OSA. There was a large difference in the sample sizes between the groups; nonetheless, the study remains to be adequately powered within each cohort.
There is a paucity of studies to understand the clinical significance of PAD. Our study examines the unique cohorts of chest pain with low intermediate risk factor for CAD, which has been then excluded by CCTA obtained during their emergency room (ER) visit. This cohort represents a large proportion of ER visits with recurrent and often nonrelenting symptoms. Our study adds to the existing clinical knowledge by highlighting OSA as an alternative cause of these symptoms and thus emphasizing early identification and therapy of these patients. We also studied the right atrial and RV chamber sizes in the study group which where dilated compared with standard parameters, which were not properly studied previously.
Conclusions
In patients presenting to the ED with chest pain without evidence of obstructive CAD on CCTA, there is a strong association between the presence of chest pain and enlarged PAD. These patients also represented a high-risk group for the presence of OSA. The results of his novel study highlight the possibility of PH and OSA as potential contributors of acute chest pain in patients without obstructive CAD. Further studies using gold standard methods of diagnosing PH and OSA are encouraged to demonstrate the direct association between chest pain and these diseases in patients ruled out for obstructive CAD, with the potential for their early detection and treatment.
Footnotes
Author Contributions
Authors NM, WM, and US contributed to the conceptualization, analysis and interpretation of all study data. All authors contributed to the design and execution of study. WM, NM and US contributed in the manuscript drafting. All authors revised it critically for important intellectual content.
Disclosures and Ethics
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Funding:
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The expenses of research reported in this publication were defrayed by Dr. Mike Wilson Advanced Cardiac Imaging Foundation. U.C.S. is a recipient of Clinical and Translational Science -Mentored Career Development Award.
Declaration of conflicting interests:
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
