Abstract
Pulmonary hypertension is associated with pronounced exercise intolerance (decreased V ċ O2 max) that can significantly impact quality of life. The cause of exercise intolerance in pulmonary hypertension remains unclear. Mitochondrial supercomplexes are large respiratory assemblies of individual electron transport chain complexes which can promote more efficient respiration. In this study, we examined pulmonary hypertension and exercise-induced changes in skeletal muscle electron transport chain protein expression and supercomplex assembly. Pulmonary arterial hypertension was induced in rats with the Sugen/Hypoxia model (10% FiO2, three weeks). Pulmonary arterial hypertension and control rats were assigned to an exercise training protocol group or kept sedentary for one month. Cardiac function and V ċ O2 max were assessed at the beginning and end of exercise training. Red (Type 1—oxidative muscle) and white (Type 2—glycolytic muscle) gastrocnemius were assessed for changes in electron transport chain complex protein expression and supercomplex assembly via SDS- and Blue Native-PAGE. Results showed that pulmonary arterial hypertension caused a significant decrease in V ċ O2 max via treadmill testing that was improved with exercise (P < 0.01). Decreases in cardiac output and pulmonary acceleration time due to pulmonary arterial hypertension were not improved with exercise. Pulmonary arterial hypertension reduced expression in individual electron transport chain complex protein expression (NDUFB8 (CI), SDHB (CII), Cox IV (CIV), but not UQCRC2 (CIII), or ATP5a (CV)) in red gastrocnemius muscle. Both red gastrocnemius and white gastrocnemius electron transport chain expression was unaffected by exercise. However, non-denaturing Blue Native-PAGE analysis of mitochondrial supercomplexes demonstrated increases with exercise training in pulmonary arterial hypertension in the red gastrocnemius but not white gastrocnemius muscle. Pulmonary arterial hypertension-induced exercise intolerance is improved with exercise and is associated with muscle type specific alteration in mitochondrial supercomplex assembly and expression of mitochondrial electron transport chain proteins.
Introduction
Pulmonary hypertension (PH) and subsequent right heart failure is associated with poor functional capacity and pronounced exercise intolerance.1,2 Despite pharmacological intervention to treat PH, patients still report low quality of life and worsening functional capacity concomitant with dyspnea and fatigue.3–6 There are currently no pharmacologic interventions to treat PH-associated exercise intolerance. However, PH patients have demonstrated functional improvements in response to exercise training (EXT).6–8 The mechanism of PH-induced exercise intolerance as well as the mechanism of exercise-mediated improvements are currently not well understood.
Impaired exercise tolerance in PH patients is not entirely explained by changes in central or peripheral hemodynamics,7,9 but may be associated with direct physiologic and biochemical alterations in skeletal muscle (SkM).2,9–11 SkM abnormalities have been identified in PH patients including reduced systemic oxygen extraction, reduced speed and strength of contraction, alterations in signaling cascades associated with muscle hypertrophy, and decreases in the proportion of oxidative fiber types.2,9–13 PH patients also exhibit decreases in mitochondrial volume and expression of electron transport chain (ETC) proteins, 14 which may contribute to reduced oxidative capacity and diminished exercise capacity (i.e. V ċ O2 max) in PH. In contrast, EXT-mediated improvements in SkM oxidative capacity are associated with increased mitochondrial biogenesis and SkM hypertrophy.15,16
Mitochondrial ETC supercomplex (SC) (a.k.a respirasomes) formation has recently emerged as an important regulator of mitochondrial respiration. Mitochondrial SCs are dynamic assemblies of individual ETC complexes consisting of complex I and varying copies of complexes III and IV in physical association to form discrete functional respiratory units. 17 SCs are thought to provide more efficient respiration which should be capable of improving mitochondrial function and potentially improving V ċ O2 max.18,19 Decreases in SC assembly have been demonstrated in human diaphragm SkM in diabetes. 20 In addition, EXT has been shown to enhance SC assembly in healthy humans in response to exercise. 21
It is unknown if SkM SC assembly is altered in diseases with chronic exercise intolerance such as heart failure and PH, and if these changes may be reversed with EXT. In this study, we evaluated exercise intolerance and the effect of chronic EXT in a rat model of pulmonary arterial hypertension (PAH). Rats were subjected to three weeks normoxia or SU5416 treatment + hypoxia (three weeks 10% FiO2; PAH) followed by one month with or without EXT. We evaluated changes in V ċ O2 max, SkM ETC protein expression, and changes in SkM SC assembly in oxidative and glycolytic SkM. PAH was associated with reduced ETC protein expression and impaired V ċ O2 max. EXT-improved V ċ O2 max in PAH animals and was associated with increased SC assembly.
Materials and methods
All materials were obtained from Sigma-Aldrich (St. Louis, MO) unless otherwise noted.
Animals and experimental model of PAH
All procedures were approved by the Institutional Animal Care and Use Committee at the Providence Veterans Affairs Medical Center (Providence, RI) and conformed to the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health.
Adult male Sprague-Dawley rats (n = 43, ∼200 g; Charles River Laboratories, Wilmington, MA) were used in this study. Animals were randomly assigned to either a control/normotensive (CON; n = 23) group, or a PH (PAH; n = 20) group using the chronic hypoxia model in combination with the VEGF receptor inhibitor, SU5416.
22
Animals assigned to the PAH group received SU5416 (Cayman Chemical, Ann Arbor, MI), at a dose of 20 mg/kg, subcutaneously, and were housed in a normobaric hypoxia chamber (10% FiO2; Biospherix A chamber + ProOx controller, Biospherix Ltd., Parish, NY) for three weeks, and then returned to normoxic (21% FiO2), room-air for the remainder of the experimental protocol. CON animals received a subcutaneous vehicle (diluent) and were housed in normoxic, room-air for the entire duration of the experimental protocol (Fig. 1). All animals were housed in pairs at 23℃, maintained on a 12:12-h light/dark cycle, and were provided standard rat chow and water, ad libitum.
Experimental design and timeline of PAH and exercise training regimen.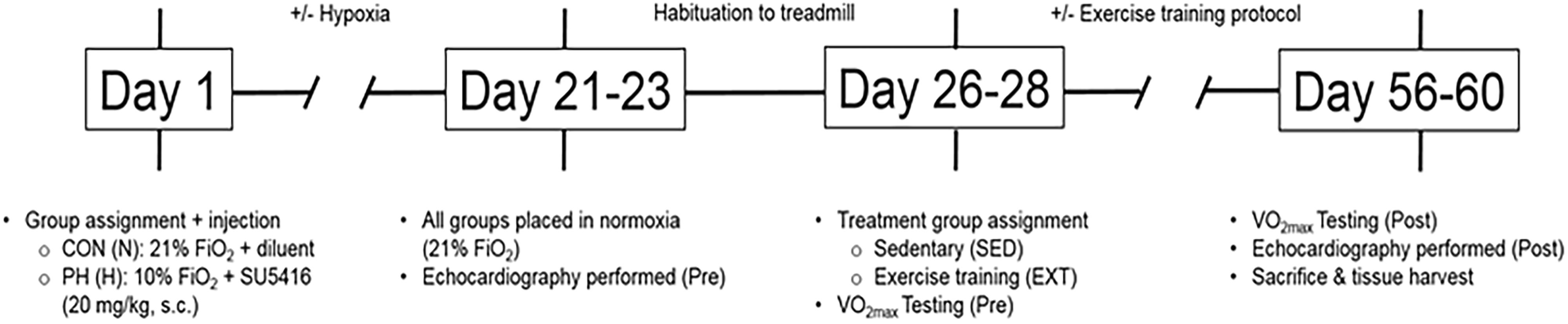
Echocardiography
Rats were anesthetized with isoflurane (2%/O2 balance) via nose cone and a transthoracic echocardiography (Vevo 2100, FUJIFILM Visualsonics Inc., Toronto, Canada) was performed (blinded) with a 40 MHz linear array transducer in accordance with previously published methods.23,24 The following measurements were obtained: pulmonary acceleration time (PAT) on pulse-wave Doppler recording at the right ventricular (RV) outflow tract, two-dimensional M-mode recordings from parasternal views were used to assess left ventricular (LV) stroke volume, cardiac output (CO), ejection fraction, and heart rate. Echocardiography was performed at the beginning of Week 4 (Pre) prior to habituation/exercise testing sessions, and was repeated at the end of Week 8 (Post) following ∼5 weeks of EXT or sedentary (SED) living. All echocardiography was performed ≥48 h after the last EXT session, when applicable. Data analysis was performed by a blinded individual.
V ċ O2 max testing and EXT protocol
At the end of three weeks of normoxic/hypoxic living, animals were again randomly assigned to one of four experimental groups: (i) sedentary normotensive (control; CON-SED; n = 11); (ii) sedentary pulmonary hypertensive (PAH-SED; n = 9); (iii) exercise-trained normotensive (CON-EXT; n = 12); or (iv) exercise-trained pulmonary hypertensive (PAH-EXT; n = 11). All exercise testing and training protocols were performed on a four-lane modular rodent treadmill (AccuPacer, Omnitech Electronics Inc., Columbus, OH) equipped with individual metabolic chambers and a gas analyzer system that simultaneously monitors and records individual metabolic profiles (i.e. V ċ O2, respiratory quotient, etc.) in real-time. Prior to any exercise testing or training, all groups were habituated to the treadmill through a gradual increase in running intensity to a final speed of 15 m/min at a 15° incline for 10 min/day. The habituation phase occurred over a one-week period and was used to assess running proficiency of individual animals, while minimizing the potential for an aerobic training effect that can occur with longer habituation periods.25,26 The EXT protocol, and subsequent criteria for fatigue, was designed to match the recommendations for exercise testing in rodents as set forth by the American Physiological Society’s Resource Book for the Design of Animal Exercise Protocols. 27 The study was designed utilizing continuous aerobic exercise (60 min/bout), not interval training. Animals assigned to EXT performed continuous, steady-state treadmill running at submaximal intensities that equated to 55–60% relative V ċ O2 max of the PAH-SED group (15 m/min, 15° incline, five days/week for 60 min/day) (Fig. 1).
A modified maximal exercise testing protocol (as described by Bedford et al. 28 ) was used to determine exercise capacity (measured by maximal aerobic capacity; V ċ O2 max) using open-circuit spirometry (flow rate = 2–3 L/min), and consisted of incremental increases in speed and/or incline every 3–5 min. True V ċ O2 max was defined as (i) the point at which V ċ O2 does not change despite changes in work load during steady, consistent running and (ii) a steady-state V ċ O2 that is sustained for a minimum of 1 min. Maximal exercise tests were performed on all rats at the beginning of Week 5 (baseline; Pre), and were repeated at the end of Week 8 (Post) following one month of EXT or SED living. All V ċ O2 max testing was performed ≥48 h after the last echocardiography/habituation/EXT session, when applicable.
Sacrifice and tissue harvest
At the completion of the experimental protocol, rats were euthanized with pentobarbital sodium (100 mg/kg) and heart and SkM tissues were excised, weighed, and harvested for molecular analysis. The red gastrocnemius (RG) and white gastrocnemius (WG) were identified by their color and anatomic location (RG: deep and WG: superficial) and carefully dissected and frozen for later biochemical analysis. RG and WG fiber typing has been carried out previously by multiple groups where it was demonstrated that RG contain a higher proportion of oxidative Type I fibers and Type IIA/D fibers (∼90%) versus WG which contains minimal Type I fibers and is predominantly glycolytic type IIB fibers (85–90%).29,30
Western blot
Rat muscle tissue (35–50 mg) was homogenized in Radioimmunoprecipitation assay buffer (RIPA) buffer (50 mM HEPES, pH 7.5, 150 mM NaCl, 5 mM ethylenediaminetetraacetic acid (EDTA), 5 mM ethelyne glycol tetraacetic acid (EGTA), 20 mM NaF, 20 mM Na+-pyrophosphate, 0.5% Na+-deoxycholate, 0.1% SDS, 1% Triton X-100) supplemented with protease inhibitors (ThermoFisher) and phosphatase inhibitors (Boston BioProducts, Cambridge MA), and centrifuged at 10,000 g for 10 min at 4℃. One-dimensional electrophoresis in SDS-PAGE was performed using 50 µg of protein. Separated proteins were transferred to membranes that were processed by immunodetection, using OXPHOS cocktail (Abcam, 1:1000) followed by strip and reprobe for vinculin (Sigma-Aldrich, 1:3000).
Mitochondrial DNA
Total cellular DNA was isolated from the soleus muscle using DNeasy Blood and Tissue kit (Qiagen, Valencia, CA). A master mix was prepared, which composed of 12.5 µL SYBR Green PCR Master Mix (BioRad), 8.5 µL nuclease free H2O, and 2 µL of 1:1 Forward:Reverse primer per reaction. Mitochondrial DNA primers and Actin primers were prepared in different master mix solutions, but ran on the same PCR plate. Using a 96-well PCR plate, each well was loaded with 23 µL of the master mix followed by the addition of 2 µL of isolated DNA (3 ng/µL). Extracted DNA was used to measure relative mitochondrial DNA content by PCR analysis 31 (StepOnePlus; Applied Biosystems, Foster City, CA) using the following primers for mt-cytb: Forward 5’-CCT CCC ATT CAT TAT CGC CGC CCT TGC-3’; Reverse 5’-GTC TGG GTC TCC TAG TAG GTC TGG GAA-3’, and was normalized to Actin, Forward 5’-GTC CAG CCC AGC CCT TCA GCA G-3’; Reverse 5’-CCG GAC CGG GCC GTA TAT GGA G-3’. To quantify the mitochondrial DNA content, nuclear DNA was used to determine ΔCT = (nucDNA CT – mtDNA CT), and relative mitochondrial DNA was determined by equation 2 × 2(ΔCT). 31
Blue Native PAGE
Approximately 50 mg of muscle tissue was minced and placed in 1 mL mitochondria isolation buffer (MIB: 0.28 M sucrose, 10 mM HEPES, pH 7.4, 2 mM EDTA) supplemented with protease and phosphatase inhibitors. Tissue was homogenized in MIB with a Dounce homogenizer, using 10 strokes each of loose and tight pestles. Homogenates were subjected to a 5 min, 2000 g spin to pellet nuclei and the resulting supernatant was subjected to 13,000 g spin followed by two washes in 1 mL MIB. Mitochondrial pellets were suspended in 100 µL MIB and subjected to bicinchoninic acid, (BCA) protein assay (Pierce). Equal amounts of mitochondrial protein ∼30 µg were solubilized in 60 µL 1× native-PAGE sample buffer (Invitrogen) supplemented with 1% Digitonin (Invitrogen). Following solubilization, a second BCA assay was performed and equal amounts of protein (∼10 µg) in 1× sample buffer with 0.25% Coomassie G250 were loaded on Invitrogen 4–16% NativePAGE gels. Gels were run for 1 h at 150 V followed by 250 V for 2.5 h. Gels were blotted to nitrocellulose membranes and probed for complex IV (Cox IV; Abcam), complex I (Ndufa9; Abcam), and complex III (UQCRFS1; Abcam), in that order with stripping in between with standard SDS/mercaptoethanol stripping buffer.
Statistical analyses
All data are presented as mean ± SEM and a threshold of significance was set at P ≤ 0.05. Unless otherwise stated, a Two-Way ANOVA with Student–Neuman–Keuls post-hoc test was used to determine significance using GraphPad Prism 6.01 (GraphPad Software, La Jolla, CA).
Results
EXT does not improve hemodynamics in PAH but does improve V ċ O2 max
The experimental protocol to initiate PAH and subsequent EXT are shown in Fig. 1. Development of PAH in rats was confirmed with echocardiography by decreased PAT and CO (Fig. 2a and b, respectively), as well as the development of RV hypertrophy as shown by Fulton Index (RV/LV + S) (Fig. 2c). There were no changes in LV:body weight (BW) ratios (not shown) or changes in body weight (Fig. 2d). EXT did not have any effect on PAH-mediated decreases in PAT, CO, or increases in RV hypertrophy (Fig. 2a–c). Differences in hemodynamic parameters before and after EXT are shown in supplementary Table 1.
Exercise training is not associated with improvements in right heart function, cardiac output, or RV hypertrophy. (a) Pulmonary acceleration time (PAT) and (b) cardiac output measured echocardiographically in sedentary animals (SED) or with 30 days exercise training (EXT) with (PAH) or without (CON) SUH-induced PAH. n = 5–11. (c) Heart weights showing RV hypertrophy (RV/LV + S) in PAH groups and (d) no effect of EXT or PAH on body weights. n = 8–11 *P < .05 Two-Way ANOVA SNK post-hoc test.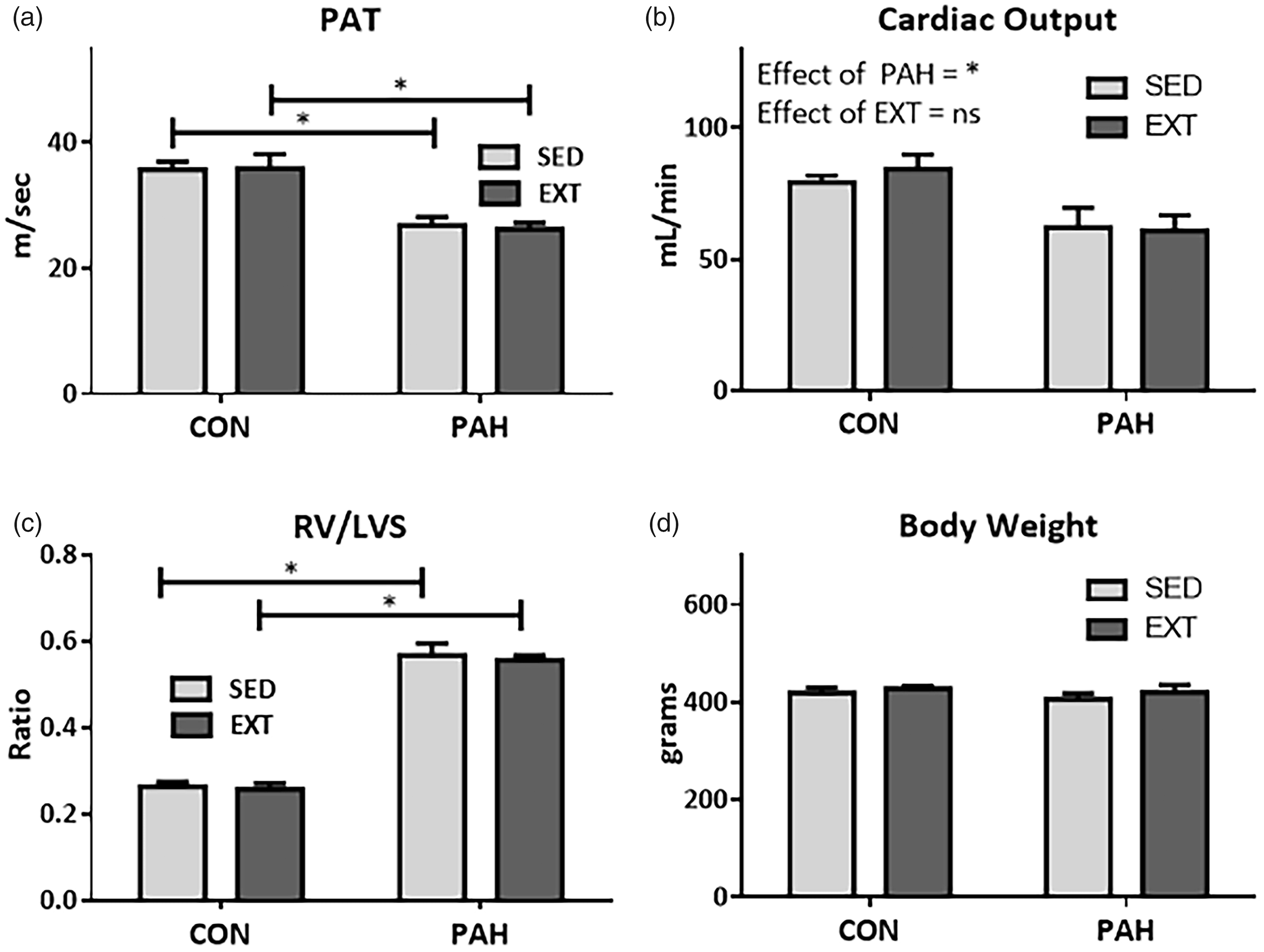
Sedentary PAH animals had reduced V ċ O2 max compared to the sedentary normoxic control group (Fig. 3). In the exercise groups, EXT (60% V ċ O2 max of PAH animals at one week) resulted in significantly higher V ċ O2 max in PAH + EXT animals (Fig. 3), despite no associated changes in central hemodynamics (Fig. 2).
PAH results in reduced V ċ O2 max which is improved with EXT. Metabolic treadmill testing was performed at the conclusion of the study following six weeks exercise training (EXT) or sedentary living (SED). Sedentary animals with PAH (SED-PAH) had decreased V ċ O2 max which was improved in the PAH + EXT group (n = 6–7), P < .05 Two-Way ANOVA, SNK post-hoc test.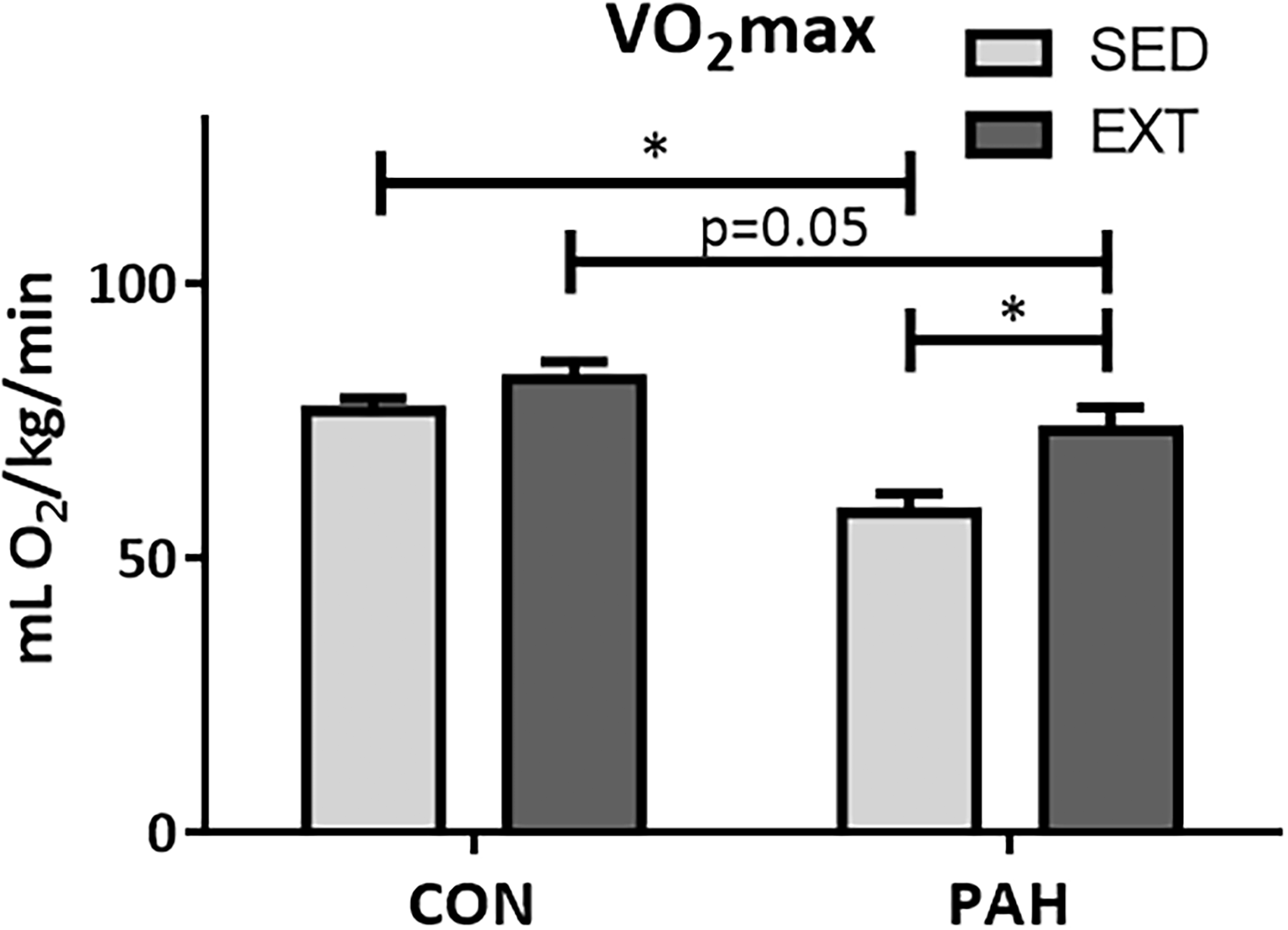
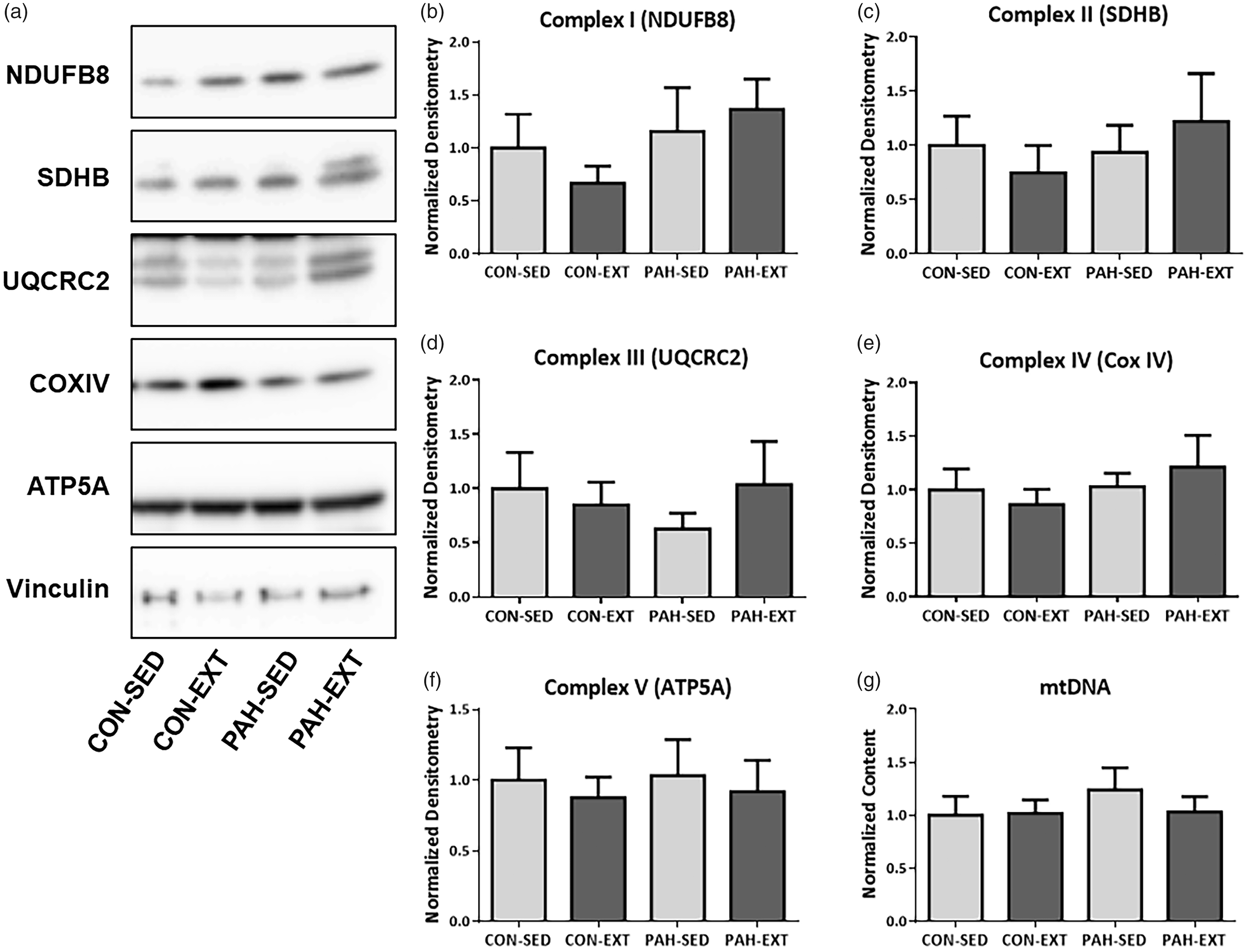
PAH alters SkM ETC protein expression specifically in Type I oxidative muscle
We examined both oxidative and glycolytic muscle types and assessed expression of mitochondrial DNA and ETC complex subunit expression in RG and WG muscle to determine if changes in mitochondrial biogenesis or ETC protein expression could explain changes in improved V ċ O2 max in PH animals. Representative immunoblots are shown in Fig. 4a. There were significant decreases with PAH in expression of complex I, II, and IV representative subunits (Fig. 4b, c, and e) as determined by Two-Way ANOVA, but no effect on complexes III and V (Fig. 4d and f). There was a main effect of EXT only in expression of the complex IV subunit MTCO1 (Fig. 4e). There was no effect of EXT in any of the other complex subunits. There were no changes in mitochondrial DNA with PAH or EXT (Fig. 4g). Predominantly glycolytic WG muscle did not have any changes in mitochondrial DNA or any of the ETC complex proteins (Fig. 5a–g).
PAH alters ETC protein expression in Red Gastrocnemius (RG) muscle which is unaffected by exercise training. (a) Western blot analysis of mitochondrial ETC protein subunits in RG whole tissue lysate. Representative blots are shown of NDUFB8 (complex 1), SDHB (complex II), UQCRC2 (complex III), MTCO1 (Complex IV), ATP5a (complex V), and vinculin. Blots were loaded with equal amount of whole lysate protein. (b–f) Quantification of western blots in (a) normalized to vinculin. (b) NDUFB8 and (c) SDHB were reduced in the SED-PAH group compared to SED-CON. There was no effect of EXT. UQCRC2 (d) was unaffected by PAH or EXT. MTCO1 (e) was reduced with PAH and improved with EXT as shown by main effects with Two-Way ANOVA; however, no groups were significantly different in post-hoc analysis. (f) ATP5a was unchanged by PAH or EXT. (g) mtDNA was assessed with qRT-PCR and there were no changes between groups. n = 6–10. *P < .05, Two-Way ANOVA, SNK post hoc analysis. Neither PAH or EXT alters ETC protein expression in White Gastrocnemius (WG) muscle. (a) Western blot analysis of mitochondrial ETC protein subunits in WG whole tissue lysate. Representative blots are shown of NDUFB8 (complex 1), SDHB (complex II), UQCRC2 (complex III), MTCO1 (complex IV), ATP5a (complex V), and vinculin. Blots were loaded with equal amount of whole lysate protein. (b–f) Quantification of western blots in A normalized to vinculin. (b) NDUFB8, (c) SDHB, (d) UQCRC2, (e) COX IV, and (f) ATP5A showed no changes between groups and were unaffected by either PAH or EXT. (g) mtDNA was assessed with qRT-PCR and there were no changes between groups. n = 6. *P < .05, Two-Way ANOVA, SNK post hoc analysis.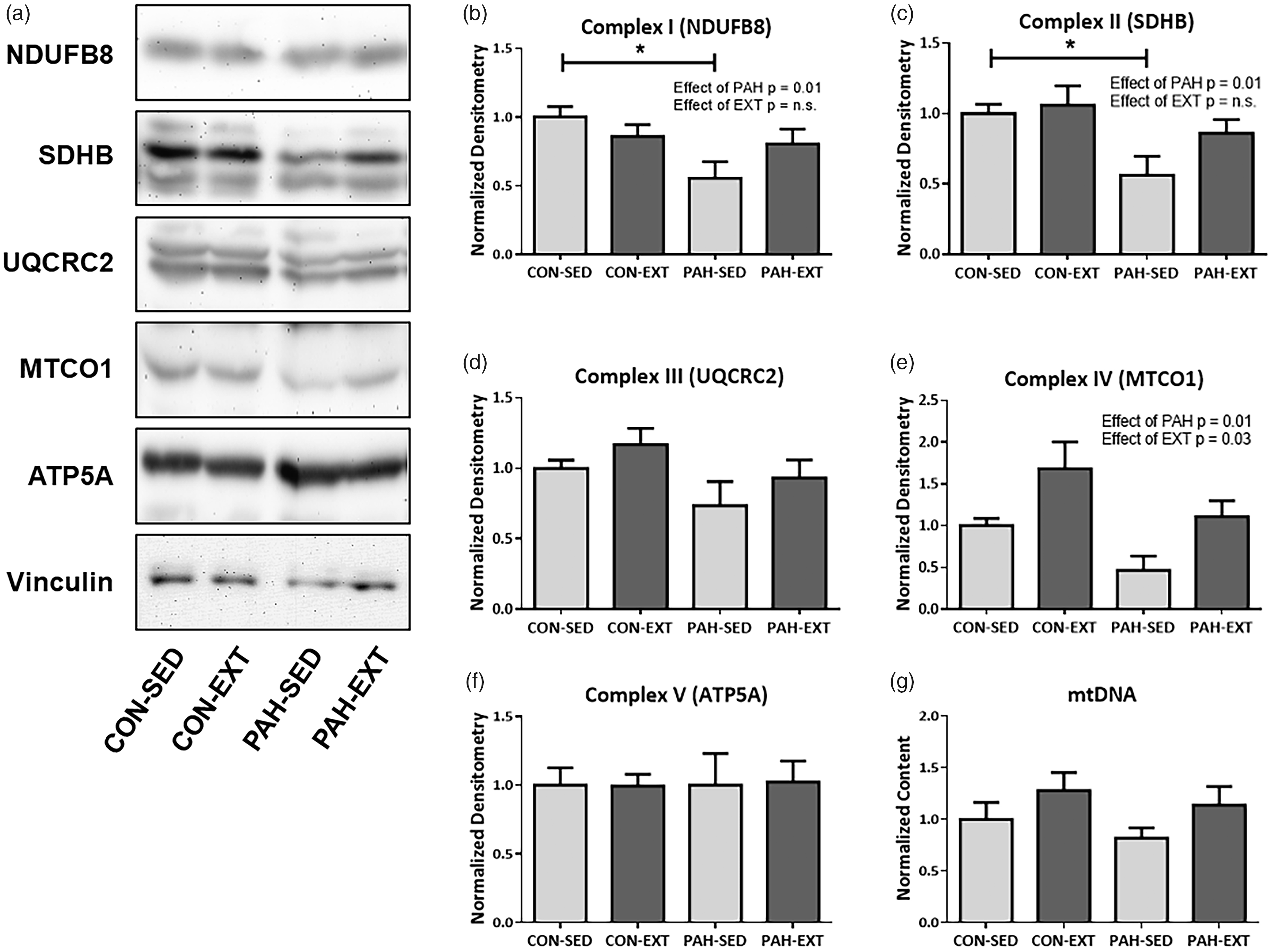
EXT enhances mitochondrial SCs in PAH
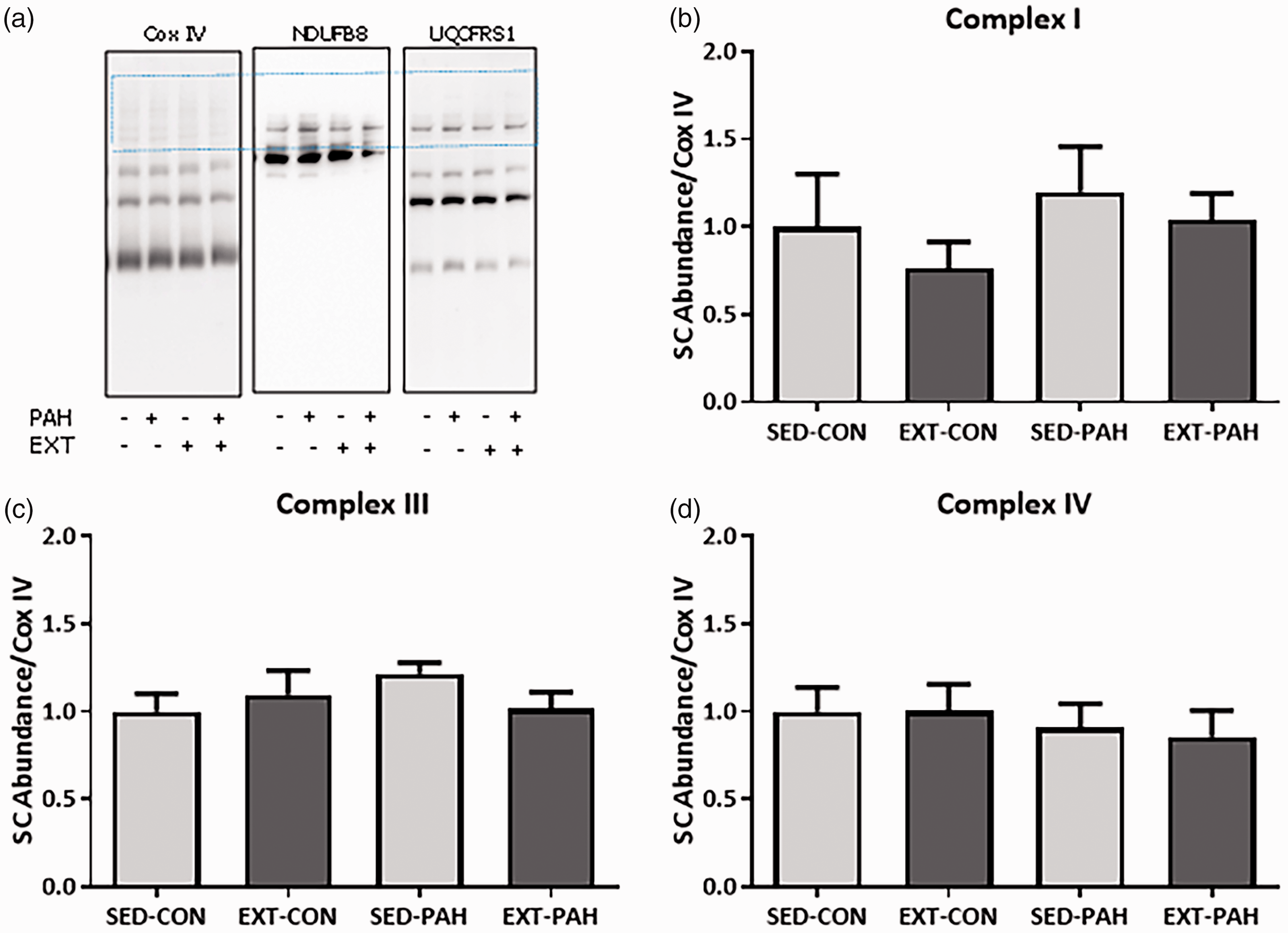
In contrast to minimal changes observed in ETC protein expression, we found that EXT robustly increases mitochondrial ETC SC assembly in oxidative SkM in rats with PAH. A representative Blue Native (BN)-PAGE for complexes I, III, and IV is shown in Fig. 6a. The boxed region shows the area quantitated as SCs which was normalized versus the entire Cox IV signal. There was an effect of EXT as determined by Two-Way ANOVA in complex I (Fig. 6b), complex III (Fig. 6c), and complex IV (Fig. 6d). There was no effect of PAH. There were significant changes in SC assembly incorporating complexes I and III in the PAH + EXT animals compared with PAH alone and a strong trend for increased SC assembly incorporating complex IV (P = .06) (Two-Way ANOVA, SNK post hoc test). There were no observable changes in SC assembly either due to PAH or EXT in WG muscle (Fig. 7a–d).
EXT increases formation of mitochondrial supercomplexes in oxidative RG muscle. (a) Representative BN-PAGE of isolated mitochondria solubilized with 1% Digitonin probed for NDUFB8 (complex I), UQCRFS1 (complex III), and Cox IV (complex IV). (b–d) Densitometric quantification of the boxed region in A normalized to the entire lane of complex IV. (b) NDUFB8 showed significant incorporation of SCs in EXT vs SED groups (c) UQCRFS1 demonstrated a significant increase in SC incorporation in PAH-EXT vs PAH-SED (P = .05). (d) Cox IV showed significant increase in SC assembly in PAH-EXT vs PAH-SED (P < .05). n = 4–5, *P < .05 Two-Way ANOVA, SNK post-hoc test. Neither PAH or EXT increase formation of mitochondrial supercomplexes in glycolytic WG muscle. (a) Representative BN-PAGE of isolated mitochondria solubilized with 1% Digitonin probed for NDUFB8 (complex I), UQCRFS1 (complex III), and Cox IV (complex IV). (b–d) Densitometric quantification of the boxed region in A normalized to the entire lane of complex IV. Neither (b) NDUFB8, (c) UQCRFS1, or (d) Cox IV showed any changes in SC assembly in PAH-EXT vs PAH-SED (P < .05). n = 6, *P < .05. Two-Way ANOVA, SNK post-hoc test.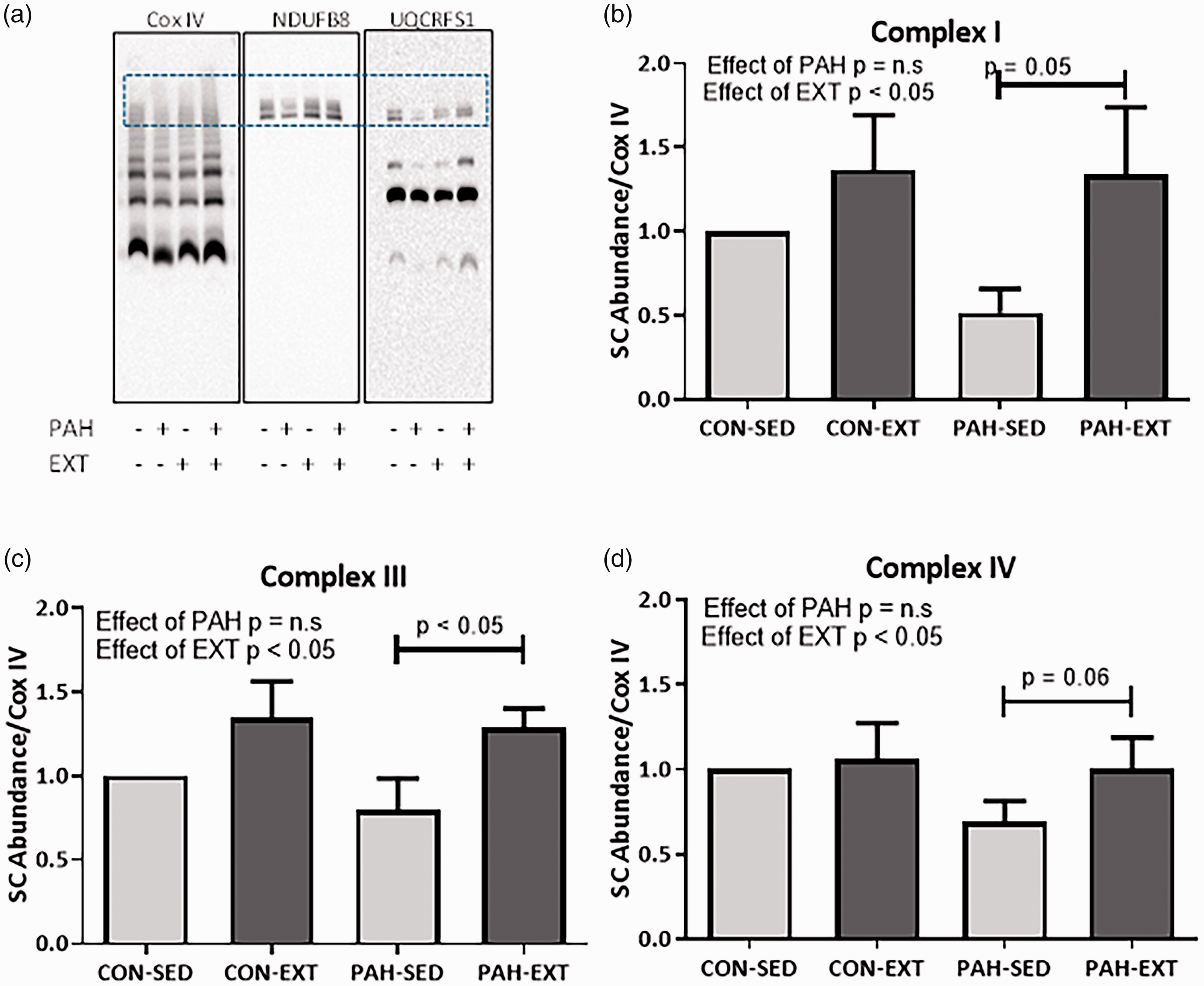
Discussion
In the present study, we have demonstrated that PAH in a preclinical rat model is associated with exercise intolerance associated with decreases in expression of specific mitochondrial ETC protein subunit expression in predominantly oxidative RG SkM. EXT improved V ċ O2 max as determined by metabolic treadmill testing. While EXT did not improve hemodynamics, it significantly increased the proportion of ETC subunits in SC assemblies without affecting individual subunit expression.
It has been increasingly recognized that exercise intolerance associated with heart failure is attributable to metabolic and biochemical alterations in the SkM.10–12,32,33 Numerous studies associated with left heart failure have demonstrated that there are SkM-specific changes that are independent of blood flow, oxygen delivery, and cardiac function.9,33–35 Importantly, EXT can improve symptoms associated with exercise intolerance in PH including increasing V ċ O2 max and six-minute walk distance, but these improvements also appear to be independent of improved blood flow, cardiac function, and CO.6,7 SkM-specific alterations in mitochondrial function have been previously explored in various types of left heart failure in both patients and animal models. Factors associated with left heart failure-induced exercise intolerance and reduced V ċ O2 max include altered fiber type distribution, mitochondrial volume and cristae structure, increased reactive oxygen species (ROS), and reduced antioxidant capacity, reduced respiration, and alterations in mitochondrial fission and fusion dynamics.15,36–41 Exercise intolerance associated with right heart failure has been less well studied, but similar alterations have been described. In human and animal studies, these include impaired respiration, alterations in ETC expression, and muscle atrophy.10,11,14,42
We have found that exercise induced a robust increase in formation of mitochondrial SCs in PAH animals, which was associated with enhanced V ċ O2 max despite no changes in CO. Mitochondrial SC formation and function has only recently emerged as an important regulatory mechanism of ETC function and activity. Dynamic regulation of SC assembly is thought to allow enhanced respiration by promoting increased electron flux and subsequently greater ATP generation, while limiting inefficiency and ROS production.19,43 Reductions in SC assembly that correlate with SkM function have previously been described in the diaphragm of diabetic patients. 20 Also, EXT has been associated with increased SC assembly in humans. 21 To our knowledge, this is the first description of EXT-induced SkM SC formation in an experimental PAH model. Furthermore, we have found that EXT-induced changes in V ċ O2 max are unlikely to be mediated by gross expression changes in ETC complex proteins. In our study, we did not observe any EXT-mediated changes in representative ETC protein expression, mtDNA, or expression of the ATPase subunit ATP5a. These data suggest that there were no mitochondrial ETC expression changes or biogenesis that could explain the EXT-induced improvement in V ċ O2 max, and that the observed EXT-mediated improvements are more likely due to enhanced SC assembly. One limitation of our study is that we did not directly assess changes in mitochondrial respiration in isolated muscle fibers and rely solely on V ċ O2 max measurements. However, previous studies have shown that limiting SC formation in mouse SkM reduces oxygen consumption and respiratory capacity, 44 and that exercise-induced SC levels correlate with mitochondrial O2 consumption in human SkM. 21 Overall, our data suggests that improved respiration due to increased SC formation may be able to overcome PAH-induced SkM deficiencies such as reduced ETC protein expression, thus leading to improvements in V ċ O2 max. These data may also indicate that modulation of SC assembly may be one of the beneficial mechanisms associated with symptom-limited rehabilitation in PAH patients. Strategies to improve SC formation including exercise and possible pharmacologic manipulation may lead to potential novel therapies for exercise intolerance.
The exact mechanism of SC assembly is unknown and is an intense area of investigation. Multiple factors have been identified that may promote SC assembly, these include cristae structural components such as cardiolipin, and possible assembly factors including Cox7RP (Cox7a2L)44,45 and rcf1/HIG2A (yeast and mammal homologs), 46 although their role as dedicated SC assembly factors in different models is controversial. Compelling evidence comes from studies demonstrating that Cox7RP KO mice have pronounced exercise intolerance. 44 Future studies will need to assess the mechanism of SC assembly and if altered expression of known SC assembly factors may play a role in EXT-induced SC assembly, as well as if overexpression can lead to similar improvements as exercise in PAH.
The mechanism of decreased V ċ O2 max in the PAH model is also unclear and likely multifactorial. We observed consistent decreases in SC formation in PAH animals; however, this did not reach statistical significance. Our data on expression of specific ETC complex subunits would support a role for decreased expression of ETC proteins contributing to impaired V ċ O2 max at least in the oxidative RG muscle. PAH resulted in decreases in complex I, II, and IV, but although EXT resulted in elevations in expression, there were no significant differences from PAH alone. It is also possible muscle atrophy and mitochondrial structural alterations may play a role in reduced V ċ O2 max. In addition, we cannot rule out a contributing component of EXT-induced changes in expression of specific ETC proteins that were not measured in this study.
The changes observed in ETC protein expression and SC assembly were only present in oxidative RG muscle. There were no changes in the glycolytic, mainly Type II WG, muscle. The lack of EXT-induced changes in WG muscle may be explained by the aerobic EXT regimen which should have greater effect on oxidative muscle types, resulting in minimal changes in WG. However, we were still able to detect significant amounts of assembled mitochondrial SCs in WG muscle despite the significantly lower mitochondrial content. It is unknown why PAH-induced right heart failure specifically causes SC assembly derangements in the oxidative muscle type. These signaling differences and underlying mechanisms in the two muscle types will need to be explored in future studies. It is noteworthy that exercise alone in our model resulted in minimal changes to ETC protein expression and SC assembly in control animals. The endurance EXT regimen was designed to achieve 60% of the initial V ċ O2 max of the PAH animals in both control and PAH-EXT groups. The initial V ċ O2 max was considerably lower in PAH animals, therefore control animals had a lower targeted V ċ O2 during exercise in comparison to their maximum. Therefore, this training regimen in control animals may not have elicited any changes in mitochondrial expression or SC assembly. Future studies will need to determine if higher intensity EXT is associated with increased SC assembly and the associated mechanism in healthy controls.
A limitation of the current study is that changes in microcirculatory function in the SkM were not investigated. Exercise intolerance in PAH has been attributed to both an underlying global metabolic disorder affecting the muscles as well as microcirculatory perfusion alterations potentially related to central hemodynamic impairments.2,47,48 Decreased capillary density or rarefaction has been documented in the SkM of animal models of PAH as well as patients.2,48,49 In addition, the VEGFR inhibitor SU5416 required to initiate PAH may reduce angiogenic responses. The interplay between vascular impairments and altered mitochondrial and muscle function is complex and both likely contribute to exercise intolerance. There has been documented evidence of both mitochondrial alterations as well as reduced capillary density in PAH,2,14,50,51 and capillary density is correlated with muscle function in numerous disease states. In addition, EXT is associated with increased angiogenic responses, 52 and it is unknown if this is linked to improved VO2 max in our model. However, exercise is also known to stimulate mitochondrial biogenesis. It is currently not clear if microcirculatory alterations in PAH are the cause of reduced mitochondrial density and metabolic changes in SkM. Moreover, mitochondrial biogenesis signaling through peroxisome proliferator-activated receptor gamma coactivator 1-α (PGC-1α) in SkM is linked to increased angiogenic responses suggesting that a common pathway may regulate both mitochondrial function and perfusion. 53 In our study, it is unclear if capillary rarefaction may initiate or be secondary to mitochondrial changes or if these muscle alterations have independent etiologies. Notably, our observed improvements in exercise tolerance were not linked to improvements in RV function such as CO and PAT. Nevertheless, our data clearly demonstrates altered mitochondrial ETC expression and SC assembly are associated with PAH and exercise. Determining the causative factors for these alterations and any potential role of vascular remodeling will need to be a focus of future research.
In summary, we have found that endurance EXT improves exercise intolerance in an animal model of PAH without affecting severity of PAH as shown by PAT and CO. PAH-mediated decreases in V ċ O2 max are associated with reduced ETC protein expression but these changes are unaffected by EXT. In contrast EXT increases assembly of mitochondrial SCs. Therefore, EXT and/or approaches to increase SC assembly may be appropriate therapeutic strategies to improve quality of life in PH patients.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020925762 - Supplemental material for Endurance exercise training in pulmonary hypertension increases skeletal muscle electron transport chain supercomplex assembly
Supplemental material, sj-pdf-1-pul-10.1177_2045894020925762 for Endurance exercise training in pulmonary hypertension increases skeletal muscle electron transport chain supercomplex assembly by Danielle J. McCullough, Nouaying Kue, Thomas Mancini, Alexander Vang, Richard T. Clements and Gaurav Choudhary in Pulmonary Circulation
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
Supported with resources and the use of facilities at the Providence VA Medical Center. Support provided by a pilot award by National Institute of General Medical Science (NIGMS) P20GM103652 (McCullough), VA Merit Award I01CX001892-01A1(Choudhary) , National Heart Lung and Blood Institute (NHLBI) RO1HL128661(Choudhary), NHLBI RO1HL148727 (Choudhary), NHLBI RO1HL135236 (Clements), AHA GIA GRNT20460376 (Clements), and NIGMS U54GM115677 (Clements). We like to acknowledge assistance from core services supported by the National Institute of General Medical Science of the National Institutes of Health under grant number P20GM103652.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
