Abstract
Right heart function is an important predictor of morbidity and mortality in patients suffering from pulmonary arterial hypertension and congenital heart diseases. We investigated whether the prostacyclin analog iloprost has a direct inotropic effect in the pressure-overloaded hypertrophic and dysfunctional right ventricle (RV). Rats were randomized to monocrotaline injection (60 mg/kg; n = 8), pulmonary trunk banding (PTB; n = 8), or a sham operation (n = 8). RV function was evaluated with magnetic resonance imaging, echocardiography, and invasive pressure measurements at baseline, after intravenous administration of placebo, iloprost 10 ng/kg/min, or iloprost 100 ng/kg/min (Ilo100). Infusion of Ilo100 induced a 12% ± 4% (P = 0.022) increase in stroke volume in the sham group and a 12% ± 3% (P = 0.005) increase in the PTB group. RV dP=dtmax was elevated by 11% ± 5% (P = 0.039) in the sham group and by 12% ± 3% (P = 0.016) in the PTB group. An elevation in cardiac output of 14% ±3% (P = 0.0008) and an 11% ± 2% (P = 0.0021) increase in RV systolic pressure were found in the PTB group. Iloprost caused a decrease in mean arterial blood pressure (MAP) in all groups of animals. An equal reduction in MAP induced by the arterial vasodilator nitroprusside did not improve any of the measured parameters of RV function. We conclude that iloprost has inotropic properties directly improving ventricular function in the hypertrophic and dysfunctional right heart of the rat.
INTRODUCTION
Right heart function is an important predictor of morbidity and mortality in patients suffering from pulmonary arterial hypertension (PAH) and congenital heart diseases.1,2 Increasing afterload of the right ventricle (RV) leads to pressure overload, and the RV responds by myocardial hypertrophy to maintain cardiac output at a sufficient level. Over time, this leads to RV contractile dysfunction and dilation, causing decompensated right ventricular failure with rising filling pressures, diastolic dysfunction, and reduced cardiac output. 3 Current treatment of PAH is focusing on dilation and remodeling of the pulmonary vessels. Despite optimal drug therapy, PAH causes right ventricular hypertrophy and failure, with an estimated 3-year survival of only 55%. 1 Direct pharmacological support of the RV might be beneficial in the treatment of PAH and congenital heart diseases.
Patients suffering from PAH have dysregulated prostacyclin metabolic pathways and reduced prostacyclin synthase expression in the pulmonary arteries. Prostacyclins are well established as first-line treatment of patients suffering from PAH in functional classes III and IV. They work mainly by dilating the pulmonary vessels. 4 The effect of prostacyclins in the right heart has been much discussed. Right ventricular function is improved by a decrease in afterload, but it is still not clear whether these drugs also stimulate the RV directly. Recent ex vivo studies suggest a possible positive inotropic effect of prostacyclins through direct stimulation of the cardiomyocytes.5–8 In vivo animal studies investigating possible direct effects of iloprost in the left ventricle (LV) and in the healthy RV have been carried out, but not in the pressure-overloaded and dysfunctional right heart.9,10 We aimed to investigate whether the prostacyclin analog iloprost improves RV function through afterload reduction or through a direct inotropic effect in the pressure-overloaded, hypertrophic, and dysfunctional right heart.
MATERIAL AND METHODS
Study design
Male Wistar rats (M&B Taconic, Ry, Denmark) were randomized to monocrotaline (MCT) injection (n = 8), pulmonary trunk banding (PTB; n = 8), or a sham operation (n = 8). After establishment of RV hypertrophy and dysfunction, we evaluated the acute effects of iloprost by magnetic resonance imaging (MRI), echocardiography, and invasive pressure measurements.
All measurements were made at baseline and after inravenous infusion of a placebo, iloprost (Ilomedin, Bayer Schering Pharma) in a therapeutic dose (Ilo10), or iloprost in a high dose (Ilo100). Iloprost and the placebo were administered as 2 minutes of loading-dose infusion followed by an 8-minute infusion of a maintenance dose before the measurements were made. Loading doses were 140 and 1,400 ng/kg and maintenance doses were 10 and 100 ng/kg/min for Ilo10 and Ilo100, respectively. The total injected volume of iloprost and the placebo was kept at a minimum, approximately 0.15 mL/kg/min (Fig. 1).
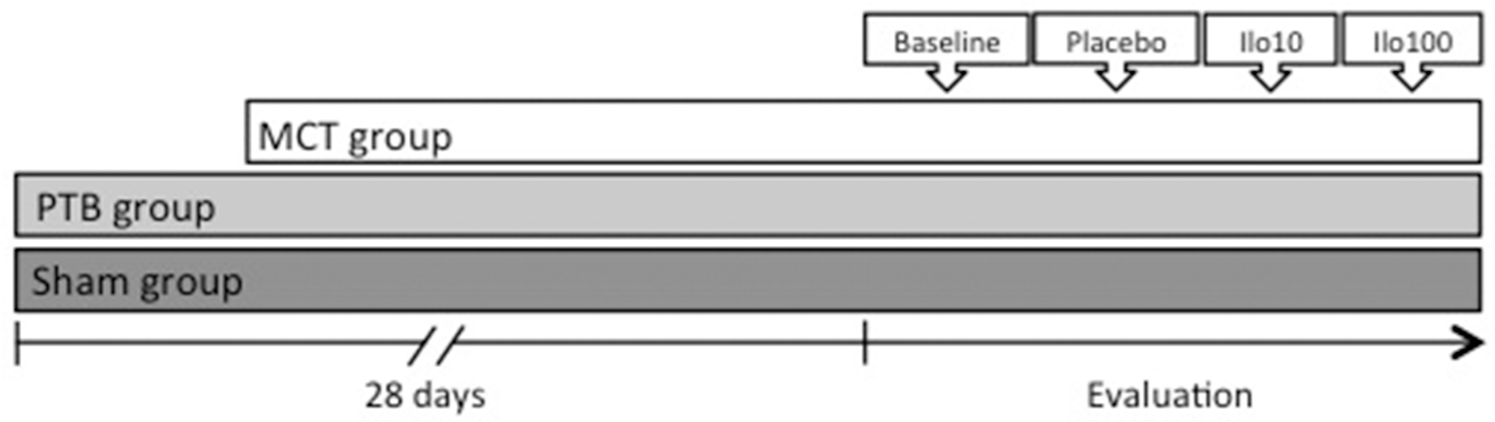
Schematic illustration of the study design. Animals in the PTB and sham groups were randomized and subjected to pulmonary trunk banding or sham operation, respectively, 4 weeks before evaluation. Animals in the MCT group were subjected to monocrotaline injection 3 weeks before evaluation. On day 1 of evaluation, magnetic resonance imaging was performed. On day 2, invasive pressure measurements and transthoracic echocardiography were performed. All measurements were made at baseline and after intravenous infusion of placebo, iloprost 10 ng/kg/min (Ilo10), or iloprost 100 ng/kg/min (Ilo100).
Rats were housed together in pairs in a room at 21°C with a 12-hour light-dark cycle and allowed free access to water and food. The entire study was conducted in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication 85–23, revised 1985) and was approved by the Danish Animal Experiments Inspectorate (authorization numbers 2006-561-1180 and 2010-561-1829). Echocardiography was done on the day of evaluation to include animals with sufficient right heart dysfunction. Only animals surviving the study protocol were included.
Pulmonary trunk banding
Rats were subjected to PTB, inducing right ventricular hypertrophy and dysfunction as described previously. 11 The rats were anesthetized with 3.5% sevoflurane (Abbott Scandinavia, Solna, Sweden) in a mixture with two-thirds O2 and one-third N2O. They were intubated and ventilated with a rodent ventilator (UB 7025, Hugo Sachs apparatus; Harvard Electronics, Hugstetten, Germany) at a respiration frequency 76 breaths/minute and a tidal volume of 3 mL. To avoid decrease in body temperature, the animals were placed on a heating pad during the procedure. A thoracotomy was made on the left side of the sternum to reveal the heart, and a surgical microscope was used to separate the pulmonary trunk from the aorta. The banding was done with a horizon applier (HZ137081; Weck Closure Systems, Durham, NC) premodified to compress a titanium clip (Horizon ligating clips, ref. 001200; Weck Closure Systems) to allow an inner diameter of 0.5 mm for the pulmonary trunk. Costae, muscles, and skin were closed in three separate layers, and a small tube was left to drain and evacuate air from the cavity. Then, 2 mL isotonic saline and buprenorphine, 0.12 mg/kg (Termgesic; RB Pharmaceuticals, Slough, UK), were injected subcutaneously, and the rats were placed in a cage with high oxygen concentration for up to 1 hour to recover. Buprenorphine was administered in the drinking water for 3 postoperative days (7.4 μg/mL). Sham-operated rats underwent the same procedure but without banding of the pulmonary trunk.
MCT treatment
MCT (Crotaline; Sigma-Aldrich, St. Louis, MO) was dissolved in 6 M HCl, and the pH was adjusted to 7.4 with 10 M NaOH. This MCT solution was diluted with saline and given as a single intraperitoneal injection (60 mg/kg) to the rats. Based on the results from pilot studies, the rats were left for 21 (±1) days to develop pulmonary hypertension and RV pressure overload-induced hypertrophy and dysfunction.
Transthoracic echocardiography
A Vivid 7 echocardiographic system (GE Healthcare, Wauwatosa, WI) with an 11-MHz phased-array transducer, operating at a rate of approximate 150 frames per second, was used to evaluate cardiac function. The animals were superficially anesthetized with sevoflurane, intubated, and ventilated. M-mode, two-dimensional, and pulsed Doppler recordings were obtained at the parasternal short-axis view and the apical 4-chamber view. Tricuspid regurgitation (TR) was evaluated with color Doppler. Tricuspid annular plane systolic excursion (TAPSE) was recorded in the apical 4-chamber view. TAPSE correlates well with prognosis and RV function in both rats and humans.12,13 Image analysis was performed offline with Echo-Pac software (GE Healthcare, Horten, Norway), with the observer blinded to the source of the samples. For each individual rat, 3 consecutive heart cycles were analyzed, and the mean was used as a representative value.
Invasive pressure measurements
Invasive pressures were recorded with Millar Mikro-Tip catheters (Millar Instruments, Houston, TX) connected to a signal-conditioning box (PCU-2000, Millar Instruments). Catheters were calibrated and presoaked for approximately 20 minutes in sterile saline before insertion. An injection of 50 units of heparin (Leo Pharma, Ballerup, Denmark) was given intramuscularly. The calibrated catheters were inserted into the carotid artery and the right jugular vein. The catheter in the jugular vein was advanced to the RV, optimal placement was ascertained by echocardiography, and the contour of the pressure wave was displayed on the screen. Hemodynamic data were recorded and analyzed with Notocord software (NOTOCORD Systems SAS, Midland, NC).
Magnetic resonance imaging
Cardiac MRI was performed with a 1.5-tesla whole-body scanner (Magnetom Avanto; Siemens, Erlangen, Germany) on spontaneously breathing rats. Superficial anesthesia was induced with 7% sevoflurane and maintained with 3% sevoflurane mixed in O2 (1 L/min) and N2O (0.5 L/min). The left femoral vein was catheterized, and the animal was placed in a supine position in a 4-element wrist coil. A pneumatic pillow was placed on the abdomen to monitor respiration, and subcutaneous electrocardiograph electrodes were inserted in the skin on the chest. An electro-optical converting box (Model 1025 system; SA Instruments, Stony Brook, NY) and a computer with PC-SAM software (SA Instruments) were used to monitor heart rate and respiration and to control gating of the signals. Image acquisition was gated to every second R wave and suspended during the inspiration phase.
The position and orientation of the LV long axis was determined by performing a number of cine (multiple-heart phase) acquisitions. Based on these, a stack of 6 contiguous slices encompassing the heart from base to apex in the cardiac short-axis orientation was acquired. We used a prospectively triggered spoiled gradient-echo cine sequence.
The imaging parameters were as follows: repetition time (TR) = 9.26 ms, echo time (TE) = 3.96 ms, flip angle = 15°, acquisition matrix = 128× 128, field of view = 60 mm × 60 mm, spatial in-plane resolution = 0.47 mm × 0.47 mm, slice thickness = 2 mm, number of heart phases = 21. These data were used for calculation of end-systolic and end-diastolic volumes and ejection fraction (EF) with Segment cardiac analysis software (http://segment.heiberg.se).
Flow measurements in the pulmonary artery were obtained with a phase contrast sequence and were used to measure stroke volume (SV) and cardiac output (CO). The measurement position was centered just above the pulmonary valve. The sequence parameters were TR = 12.7 ms, TE = 3.56 ms, flip angle = 15°, acquisition matrix = 128 × 128, field of view = 80 mm × 80 mm, spatial in-plane resolution = 0.63 mm × 0.63 mm, slice thickness = 3 mm, number of heart phases = 15. The velocity encoding (Venc) parameter was 100 cm/s. The data readout was finished after approximately 6 ms, while the remaining TR was “dead time.” To increase the temporal resolution, all flow measurements were repeated twice, with the second delayed by 6 ms relative to the first. The two data sets were combined, and flow values were obtained with dedicated analysis software (Siswin).
Right ventricular hypertrophy
Ascites and pleural fluid were evacuated and weighed (weights from Sartorius, Göttingen, Germany) after the animals were euthanized. The heart was dissected, separating the RV from the LV + septum (LV+S), and weighed separately. Liver, kidneys, spleen, and tibia were removed. The organs were weighed and the tibia length measured. An estimated value of pulmonary vascular resistance (PVR) was calculated as PVR = RVSP=CO (RVSP: right ventricular systolic pressure; CO: cardiac output).
Histology
The RV was fixated in 4% formaldehyde buffer, embedded in paraffin, and cut into 2-μm slices on a rotary microtome (Microm HM 360; Brock & Michelsen, Birkerød, Denmark). The slices were stained with collagen-specific Sirius Red. To evaluate the mean area of fibrosis, color images were captured through a polarized-light microscope (ColorViewII; Soft Imaging System, Münster, Germany) using the 4× objective (BX50F4; Olympus, Tokyo). The images were converted to 8-bit grayscale and analyzed with ImageJ software (US National Institutes of Health, Bethesda, MD, http://rsbinfo.nih.gov/ij/). Particle analysis was used to determine the area fraction of fibrosis, calculated as an average percentage of 3 randomly selected images with the observer blinded to the source of the sample.
Nitroprusside
We infused nitroprusside into a separate set of rats subjected to a sham operation (n = 6), the PTB operation (n = 6), or MCT injection (n = 6). This was done to confirm that the effects of iloprost were due to a direct effect in the right heart and not caused by systemic vasodilaõtion. The rats were anesthetized and intubated, and invasive pressures were monitored as described above. After placebo infusion, sodium nitroprusside, diluted in 5% glucose to a concentration of 50 μg/mL, was infused through the femoral vein at an initial rate of 0.1 mL/min. The infusion rate was increased by 0.1 mL/min every 2 minutes until mean arterial blood pressure (MAP) reached the same level as observed during iloprost infusion.
Data analysis and statistics
All quantitative data are expressed as mean ± standard error of mean. Significance of difference was evaluated with one-way repeated-measures ANOVA, followed by a relevant post hoc analysis. Differences between the groups of animals were tested by an unpaired t test. Observations not normally distributed were tested with equivalent nonparametric tests. All P values are based on the absolute values; P < 0.05 was considered statistically significant. All data were analyzed with GraphPad Prism (ver. 5.0; GraphPad Software, San Diego, CA).
RESULTS
Right ventricular hypertrophy and dysfunction
Compared with the sham operation, the PTB procedure and MCT treatment raised RV systolic pressure (Table 1) and induced RV hypertrophy measured by an increase in the RV weight/tibia length ratio (Fig. 2A). MCT animals had a higher lung weight than both sham and PTB animals (Table 1). The amount of interstitial myocardial fibrosis was elevated in the PTB group compared to that in sham animals (Table 1). Volume measurements by MRI in the PTB and MCT groups showed RV dilation (Figs. 2B, 3). TAPSE and EF were markedly decreased, demonstrating impairment of RV function (Fig. 2C, 2D). In the PTB and MCT groups, most animals had tricuspid regurgitation (Table 1). PVR was markedly elevated in the MCT animals compared to that in the sham animals (Table 1).
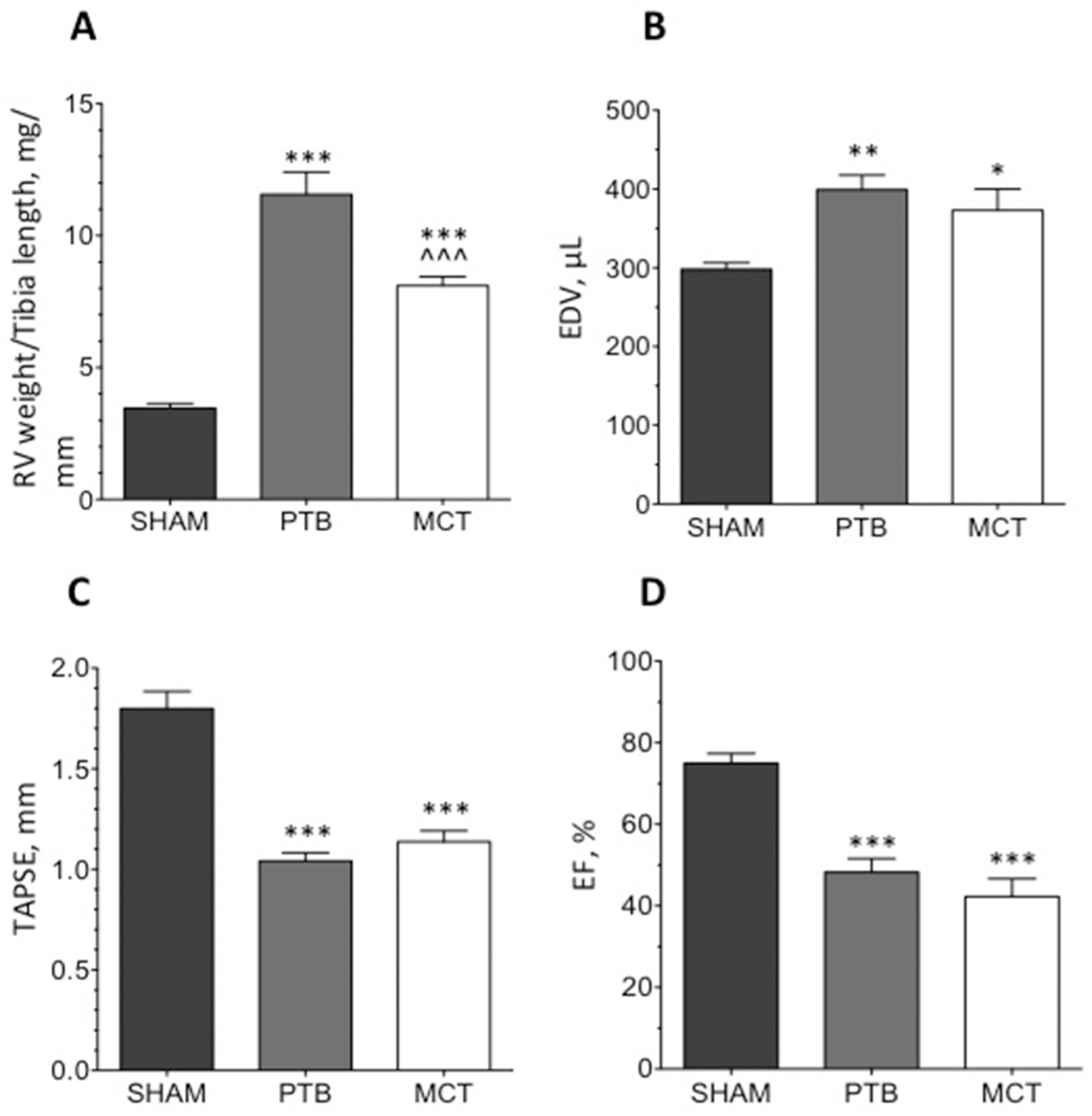
A, RV weight/tibia length. B, End-diastolic volume (EDV) measured at baseline. C, Tricuspid annular plane systolic excursion (TAPSE) measured at baseline. D, Ejection fraction (EF) measured at baseline. SHAM: sham-operation group; PTB: pulmonary trunk banding group; MCT: monocrotaline group. Single asterisk: P < 0.05, compared to SHAM; double asterisk: P < 0.01 compared to SHAM; triple asterisk: P < 0.001 compared to SHAM; triple caret (^^^): P < 0.001 compared to PTB.
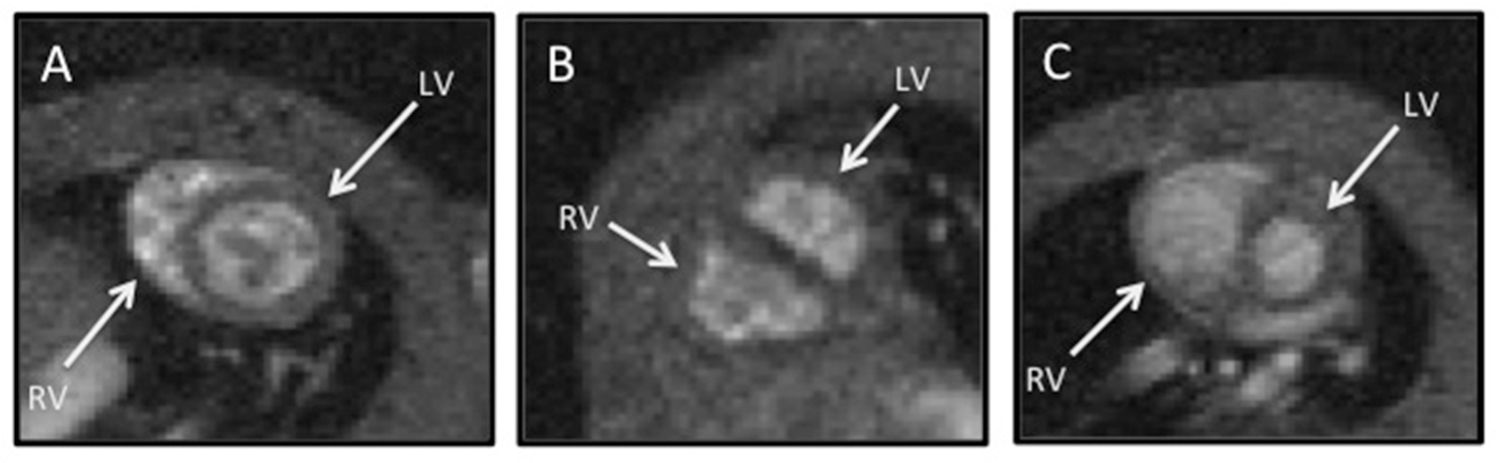
Magnetic resonance images made in the cardiac short-axis orientation, showing end-diastolic volumes from a sham-operation group animal (A), a pulmonary trunk banding group (PTB) animal (B), and a monocrotaline group (MCT) animal (C). The PTB and MCT animals present a large and dilated right ventricle (RV), which compromises the filling of the left ventricle (LV).
Animal characteristics and baseline measurements
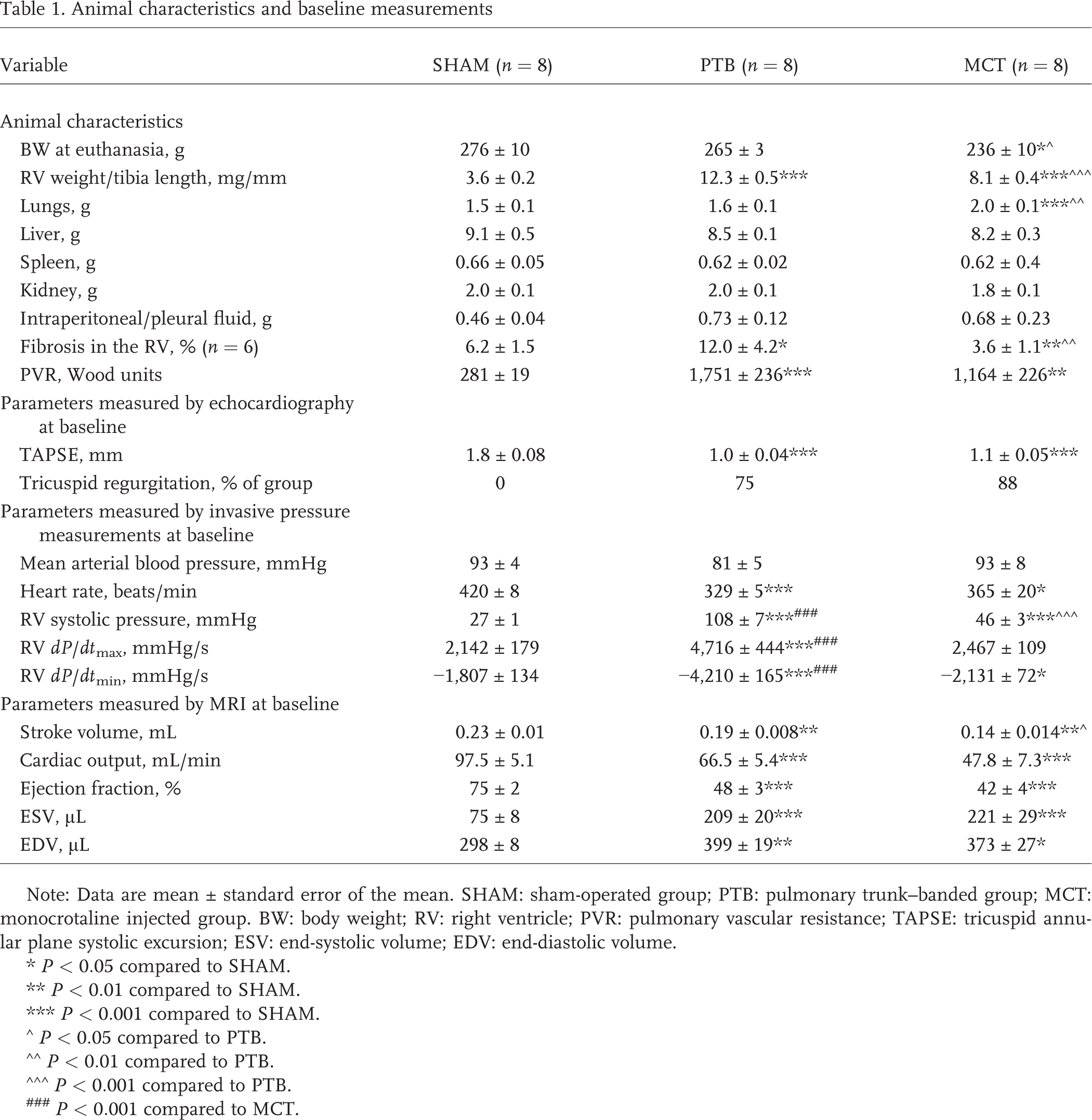
Note: Data are mean ± standard error of the mean. SHAM: sham-operated group; PTB: pulmonary trunk–banded group; MCT: monocrotaline injected group. BW: body weight; RV: right ventricle; PVR: pulmonary vascular resistance; TAPSE: tricuspid annular plane systolic excursion; ESV: end-systolic volume; EDV: end-diastolic volume.
P < 0.05 compared to SHAM.
P < 0.01 compared to SHAM.
P < 0.001 compared to SHAM.
P < 0.05 compared to PTB.
P < 0.01 compared to PTB.
P < 0.001 compared to PTB.
P < 0.001 compared to MCT.
Acute effects of iloprost infusion
Infusion of Ilo100 improved SV in sham and PTB animals (placebo vs. Ilo100: sham: 0.25 ± 0.01 vs. 0.28 ± 0.01 mL, P = 0.022; PTB: 0.19 ± 0.01 vs. 0.21 ± 0.01 mL, P = 0.005; Fig. 4A) and caused an elevation of CO in the PTB group (placebo vs. Ilo100: 63.0 ± 4.8 vs. 71.4 ± 5.1 mL/min, P = 0.0008; Fig. 4B). In the MCT animals, nonsignificant tendencies toward improvement of SV by 18% ± 6% (P = 0.17) and of CO by 15% ± 11% (P = 0.45) were observed (Fig. 4).
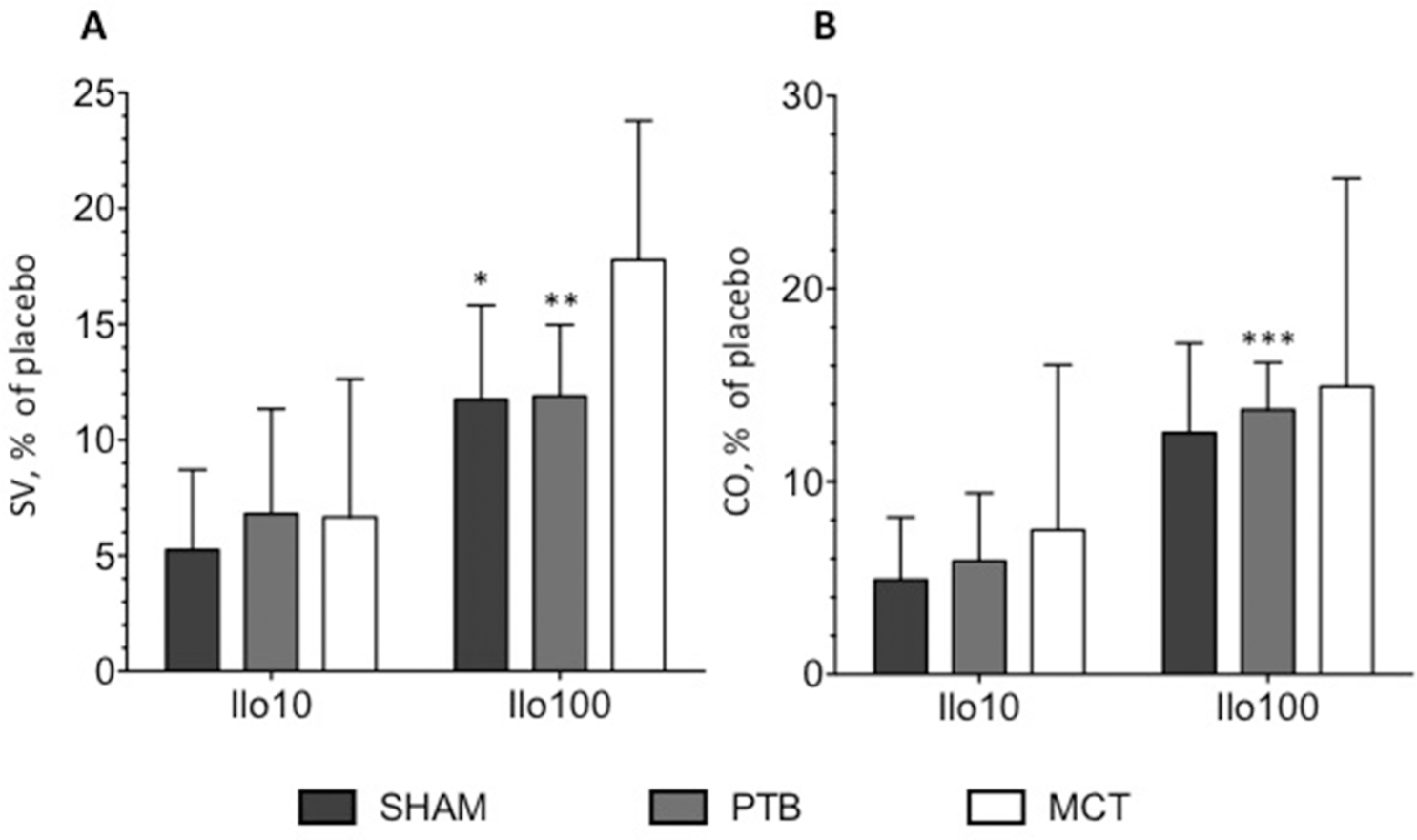
Stroke volume (SV; A) and cardiac output (CO; B) in percent difference from placebo treatment after injection of Ilo10 and Ilo100. Ilo10: iloprost 10 ng/kg/min; Ilo100: iloprost 100 ng/kg/min. SHAM: sham-operation group; PTB: pulmonary trunk banding group; MCT: monocrotaline group. Single asterisk: P < 0.05; double asterisk: P < 0.01; triple asterisk: P < 0.001; all compared to placebo.
Infusion of iloprost caused systemic vasodilation, demonstrated by a reduction of MAP in all groups of animals (placebo vs. Ilo100: sham: 84 ± 2 vs. 78 ± 2 mmHg, P = 0.005; PTB: 76 ± 4 vs. 70 ± 4 mmHg, P = 0.044; MCT: 71 ± 3 vs. 61 ± 2 mmHg, P = 0.001; Fig. 5A), but had no significant effect on heart rate. RVSP was markedly increased in PTB animals (placebo vs. Ilo100: 110 ± 6 vs. 121 ± 6 mmHg, P = 0.002), and a tendency toward a raised RVSP was observed in the sham group (P = 0.055; Fig. 5B). This was accompanied by an elevation of dP=dtmax in both the PTB and sham groups (placebo vs. Ilo100: PTB: 4, 730 ± 451 vs. 5, 338 ± 605 mmHg/s, P = 0.016; sham: 2, 218 ± 218 vs. 2, 521 ± 386 mmHg/s, P = 0.039; Fig. 5C). No effect on dP=dtmax was observed in the MCT group; however, a tendency toward a decrease in RVSP was observed (P = 0.075), indicating RV afterload reduction. Injection of the placebo did not change any of the measure parameters relative to baseline measurements.
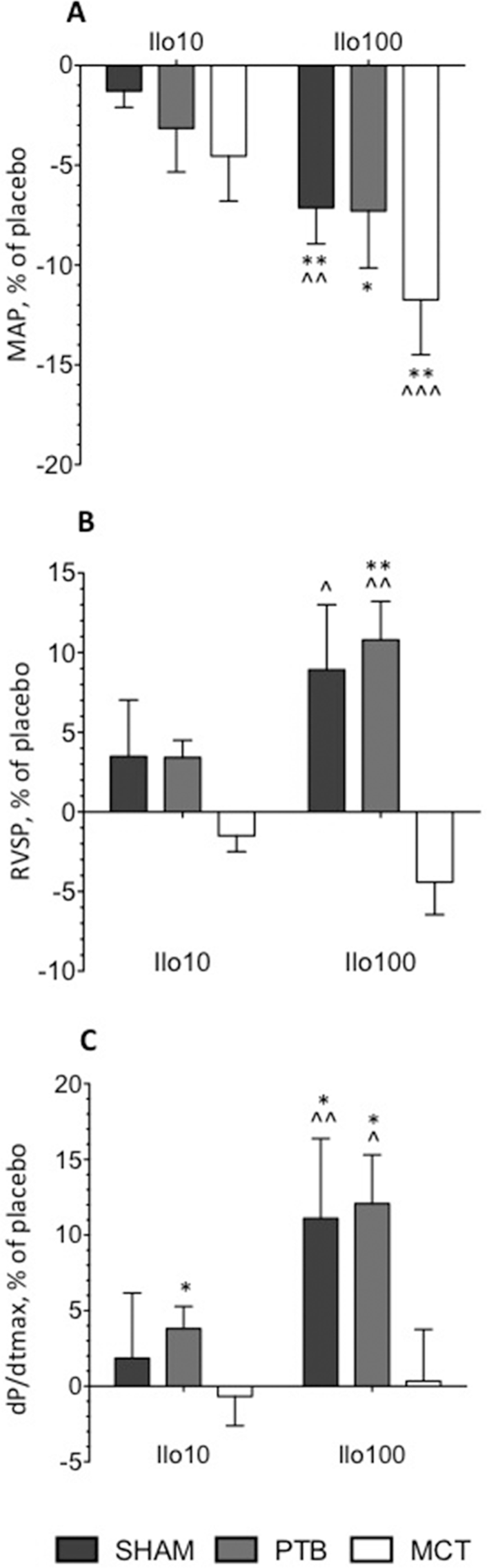
Mean arterial blood pressure (MAP; A), right ventricular systolic pressure (RVSP; B), and dP=dtmax (C) in percent difference from placebo treatment after injection of Ilo10 and Ilo100. Ilo10: iloprost 10 ng/kg/min; Ilo100: iloprost 100 ng/kg/min. SHAM: sham-operation group; PTB: pulmonary trunk banding group; MCT: monocrotaline group. Single asterisk: P < 0.05 compared to placebo; double asterisk: P < 0.01 compared to placebo; single caret (^): P < 0.05 compared to Ilo10; double caret: P < 0.01 compared to Ilo10; triple caret: P < 0.001 compared to Ilo10.
Nitroprusside
Infusion of Ilo100 caused decreases in MAP of 7% ± 2% (P = 0.005), 8% ± 3% (P = 0.044), and 14% ± 3% (P = 0.001) in the sham group, the PTB group, and the MCT group, respectively. Infusion of nitroprusside decreased blood pressure to the same level as infusion of iloprost (Fig. 6A). Compared to the placebo, nitroprusside caused a decrease in RVSP in all groups of animals (Fig. 6B). RV dP=dtmax was either unchanged or slightly reduced by nitroprusside (Fig. 6C).
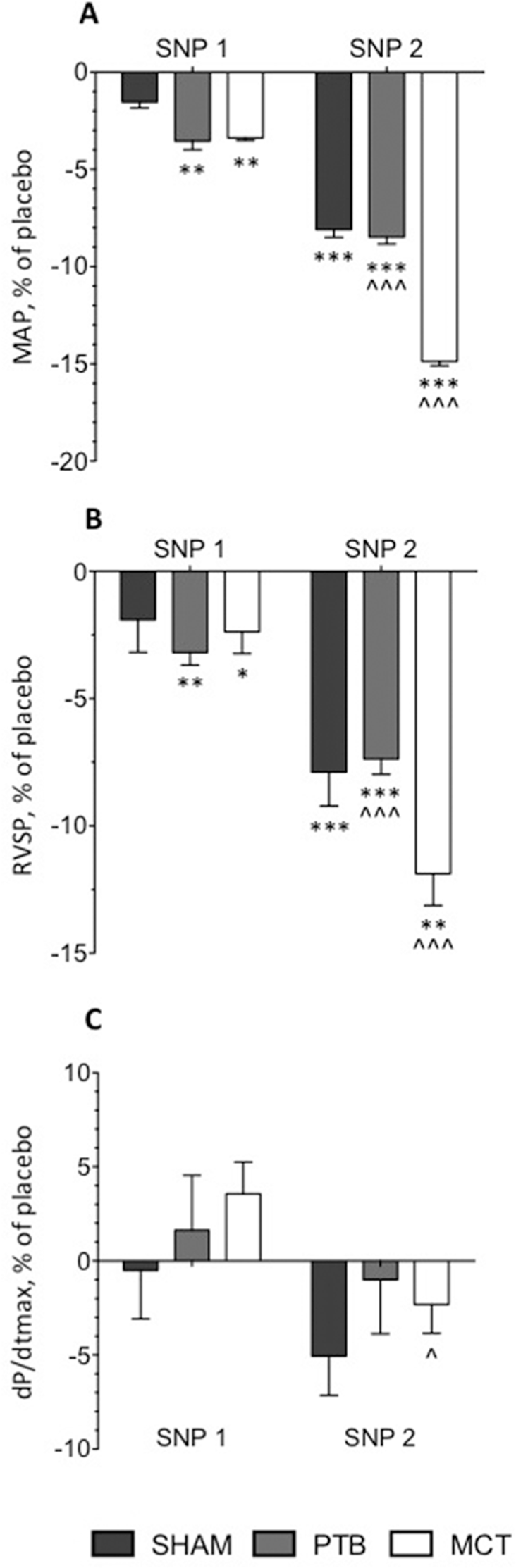
Mean arterial blood pressure (MAP; A), right ventricular systolic pressure (RVSP; B), and dP=dtmax (C) in percent of placebo after injection of SNP1 and SNP2. SNP1: sodium nitroprusside in a dose causing a decrease in MAP equal to that caused by Ilo10 (iloprost 10 ng/kg/min); SNP2: sodium nitroprusside in a dose causing a decrease in MAP equal to that caused by Ilo100 (iloprost 100 ng/kg/min). SHAM: sham-operation group; PTB: pulmonary trunk banding group; MCT: monocrotaline group. Single asterisk: P < 0.05 compared to placebo; double asterisk: P < 0.01 compared to placebo; triple asterisk: P < 0.001 compared to placebo; single caret (^): P < 0.05 compared to SNP1; triple caret: P < 0.001 compared to SNP1.
DISCUSSION
We demonstrated that iloprost exerts a direct positive inotropic effect in vivo, independent of pulmonary vasodilation, in the hypertrophic and functionally impaired right heart.
RV hypertrophy and dysfunction
The PTB procedure and MCT injections effectively induced hypertrophy, pressure overload, and functional impairment of the RV. The MCT group showed markedly elevated lung and RV weights compared to those in sham animals, in concordance with previous findings. 14 The PTB group demonstrated a more pronounced increase in RV weight, to more than 3 times that of the sham animals. Both groups demonstrated tricuspid valve regurgitation and dilation of the RV, confirmed by elevated RV end-diastolic and end-systolic volumes. Baseline SV, CO, EF, and TAPSE were significantly lowered, as seen in PAH patients with impaired RV function.13,15
The PTB procedure caused RVSP exceeding 100 mmHg, which coincides with previous findings. 16 Despite a lower RVSP in the MCT group than in the PTB animals, RV functional impairment was similar in the two groups of animals. This is in concordance with previous findings, showing that pressure overload alone is not sufficient to explain the deterioration of RV function. 17 The MCT model is complex, involving metabolites that cause toxic lesions in the lungs and liver of the rat. Previous studies have shown MCT to cause a direct cardiotoxic effect, myocardial inflammation, and coronary arteriolar medial thickening in the rat, unrelated to the degree of PAH.18,19
Acute effects of iloprost
Infusion of Ilo100 significantly improved RV function in sham-operated and PTB rats. This was demonstrated by an increase of SV and an elevation of dP/dtmax in both the sham group and the PTB group. In addition, increases in CO and RVSP were observed in the PTB group.
In the PTB animals, the RV afterload was fixed because of the pulmonary artery band, and these results therefore indicate that iloprost has a direct inotropic effect in the hypertrophic, dysfunctional right heart. Previous studies have shown that prostacyclins improve hemodynamic parameters in patients suffering from pulmonary hypertension.4,20 Zhang et al. 21 recently described the acute hemodynamic responses to inhalation of iloprost in patients with pulmonary hypertension.
Our findings contrast recent in vivo animal studies reporting a close arterial-ventricular coupling without any separate increase in RV contractility during prostacyclin administration.22–24 In our PTB animals, in which iloprost was unable to decrease RV afterload, we observed an inotropic effect. This was also seen in the work by Wauthy et al., 24 in which an increase in RV contractility was observed in animals failing to reduce RV afterload in response to prostacyclin. In the MCT group, we found a trend toward an increase in SV and CO and a decrease in RVSP when iloprost was administered, in agreement with previously reported observations. 14 The improvement in SV and CO without an increase in RVSP and dP/dtmax in the MCT animals is likely explained by unloading of the RV by pulmonary vasodilation.
Reasons for discrepancy between this and previous studies may also rely on animal species and choice of prostacyclin analog. Wauthy 23 and Kerbaul 22 both tested the effects of epoprostenol in pigs. Prostacyclin may not exert the same effects in pigs as in rats, and iloprost may have inotropic properties that epoprostenol does not. This is supported by several studies showing differential effects of prostacyclin analogs in different animal species.5,8,25 Furthermore, current research has revealed that prostacyclin analogs exert their actions through activation of several different prostanoid receptors and display different affinities and selectivities for each receptor subtype. This may potentially explain why different prostacyclin analogs show dissimilar effects on the RV.25,26
Ilo10 did not significantly improve the function of the RV. The receptors and pathways responsible for the inotropic effect of iloprost might be activated at higher doses. This is in concordance with Rex et al., 22 who tested a low dose of iloprost in pigs subjected to acute pulmonary hypertension and did not show any improvement in RV function. This is also supported by Kisch-Wedel et al., 10 who reported a positive inotropic response in the LV of the pig during administration of iloprost at a supratherapeutic dose.
Nitroprusside, which is a systemic arterial vasodilator, was titrated to produce the same drop in MAP as was achieved with iloprost infusion. At a MAP level equal to that caused by iloprost, no improvement in RV function was seen in any of the groups. This supports a direct effect of iloprost on RV performance unrelated to systemic vasodilation and a baroreceptor reflex activation. These results coincide with findings in heart failure patients showing infusion of iloprost to induce a more pronounced elevation of CO and SV than does nitroprusside infusion.27,28 Also, in the LV of the pig, iloprost and epoprostenol have shown positive inotropic effects, while no inotropic effect was observed during nitroprusside infusion. 10
CONCLUSION
Iloprost acutely improves RV function in the hypertrophic and dysfunctional RV of the rat in vivo. This was independent of pulmonary and systemic vasodilation, suggesting that iloprost has direct positive inotropic properties in the right heart.
