Abstract
Despite the importance of preserved right ventricular structure and function with respect to outcome across the spectrum of lung, cardiac, and pulmonary vascular diseases, only recently have organized efforts developed to consider the pulmonary vascular–right ventricular apparatus as a specific unit within the larger context of cardiopulmonary pathophysiology. The Third International Right Heart Failure Summit (Boston, MA) was a multidisciplinary event dedicated to promoting a dialogue about the scientific and clinical basis of right heart disease. The current review provides a synopsis of key discussions presented during the section of the summit titled “Emerging Hemodynamic Signatures of the Right Heart.” Specifically, topics emphasized in this element of the symposium included (1) the effects of pulmonary vascular dysfunction at rest or provoked by exercise on the right ventricular pressure-volume relationship, (2) the role of pressure-volume loop analysis as a method to characterize right ventricular inefficiency and predict right heart failure, and (3) the importance of a systems biology approach to identifying novel factors that contribute to pathophenotypes associated with pulmonary arterial hypertension and/or right ventricular dysfunction. Collectively, these concepts frame a forward-thinking paradigm shift in the approach to right heart disease by emphasizing factors that regulate the transition from adaptive to maladaptive right ventricular–pulmonary vascular (patho)physiology.
Keywords
In the autumn of 2013, the Third International Right Heart Failure Summit convened in Boston, Massachusetts, to discuss contemporary concepts in the pathobiology and clinical management of right-sided cardiovascular disease. This symposium, which brought together international experts across a range of scientific and clinical fields, aimed to accomplish the following objectives: define a standardized vocabulary by which to discuss right heart disease, 1 present contemporary observations relevant to right ventricular (RV)–pulmonary vascular axis pathophysiology, and promote a dialogue about approaches to the care of patients with right heart failure syndromes. To accomplish these objectives, the summit was divided into three sections, titled (1) “Pulmonary Hypertension and the Right Ventricle—Thinking outside the Box,” (2) “Emerging Hemodynamic Signatures of the Right Heart,” and (3) “Transplantation in End-Stage Pulmonary Hypertension.” The salient scientific and clinical points of each section will be the feature of a review article series in Pulmonary Circulation, with section 2 being the focus of the current work.
EMERGING HEMODYNAMIC SIGNATURES OF THE RIGHT HEART
The normal RV pressure-volume relationship
This section of the symposium addressed the RV–pulmonary vascular relationship as a prism by which to study and predict maladaptive and clinically meaningful changes to right heart function.2,3 This discussion was initiated by Dr. Andrew Redington, who reviewed the significance of RV pressure-volume assessment as a tool to monitor the consequences of pulmonary vascular dysfunction with respect to RV performance.
The geometric and functional parameters of the normal left ventricle (LV) have been characterized thoroughly, and the effect of perturbations to LV systolic and diastolic efficiency on changes to functional capacity and longevity in patients has been described across virtually all forms of left-sided cardiovascular disease, including ischemic 4 and genetic 5 cardiomyopathies as well as pericardial disease. 6 By contrast, substantially fewer data on the RV pressure-volume relationship under (patho)physiological conditions are available. 7 This is due, in part, to the conformation of the RV chamber, which does not align with a simple geometric shape; thus, classical work using traditional non-invasive imaging (e.g., two-dimensional echocardiography, roentgenography) to calculate changes in RV cavitary volume across the cardiac cycle were largely confounded. 8 Even in the cardiac magnetic resonance (CMR) imaging era, monitoring with precision changes in RV volume or ejection fraction under different loading conditions characterizes global RV function in general, but most CMR indexes used in clinical practice are static measures that do not offer insight into the mechanistic underpinnings of RV contractile efficiency and/or the factors modulating a transition from adaptive to maladaptive RV remodeling.
Increased afterload mediates changes to the RV pressure-volume relationship
Seminal work in this field by Dr. Redington and colleagues characterized several key features unique to the normal RV pressure-volume relationship in humans (Fig. 1). 10 First, the pressure-volume loop is triangular in conformation. Second, blood ejection occurs early in the RV pressure upstroke, with ?4% of stroke volume being ejected prior to peak RV systolic pressure. Third, a majority of stroke volume is ejected after peak RV systolic pressure; thus, the isovolumic relaxation phase is substantially less well defined in the RV than the LV. These investigators would later characterize the effects of changes to loading conditions on the RV pressure-volume relationship in humans. Whereas increased volume loading does not change the loop shape or ejection pattern significantly, increased RV afterload (e.g., subpulmonic valvular stenosis, increased pulmonary vascular resistance [PVR]) results in a blunted rate of ejection during RV pressure decline and a distinct isovolumic relaxation phase.9,11
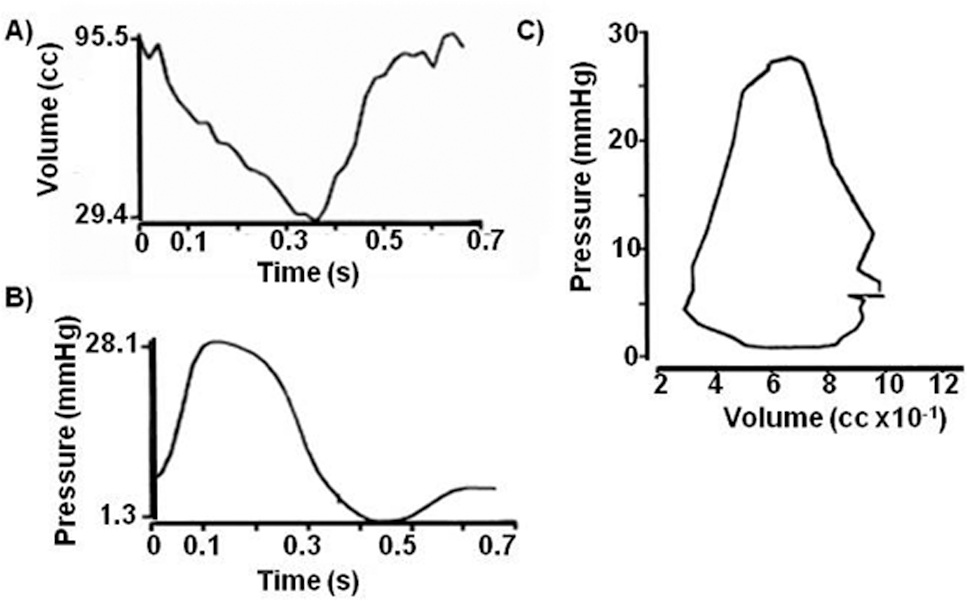
Normal human right ventricular (RV) pressure-volume loop. In seminal work by Redington and colleagues to characterize the normal RV pressure-volume loop, Simpson's rule was used to calculate the RV chamber volume from biplane ventriculograms that were derived from radiopaque casts of the human RV (A). Simultaneous pressure measurements were recorded using a micromanometer catheter placed in the apex of the RV (B), and the complete pressure-volume loop contour was created by superimposing volume and pressure measurements (C). Reproduced with permission from Redington et al. 9
These observations describe the fundamental characteristics that distinguish the RV pressure-volume relationship from the LV pressure-volume relationship under conditions of altered volume and pressure loading and establish a model by which to study RV efficiency with respect to pulmonary vascular function (i.e., RV–pulmonary arterial coupling).3,12 In fact, subsequent empiric efforts have established that predicting pathological changes to the RV pressure-volume loop contour hinges on the severity and time course of increases in RV afterload. In one study of experimental pulmonary embolism 13 in which swine were subjected to autologous pulmonary artery blot clot, RV conductance catheterization was performed to assess changes in pulmonary arterial elastance (Ea; i.e., RV afterload), RV end-systolic elastance (EES; i.e., load-independent contractility), and the EES ?Ea ratio, which is an assessment of RV–pulmonary arterial coupling (Fig. 2). In that study, a significant increase in Ea and EES was observed beginning 30 minutes after embolization, with a maximum peak within 4 hours. These changes resulted in an initial decrease in the EES ? Ea ratio and an attendant optimization of RV stroke volume. However, as predicted by Redington and colleagues,9–11 late responses to gradually increased RV afterload ultimately resulted in decreased RV stroke volume and maladaptive RV cavitary dilation.
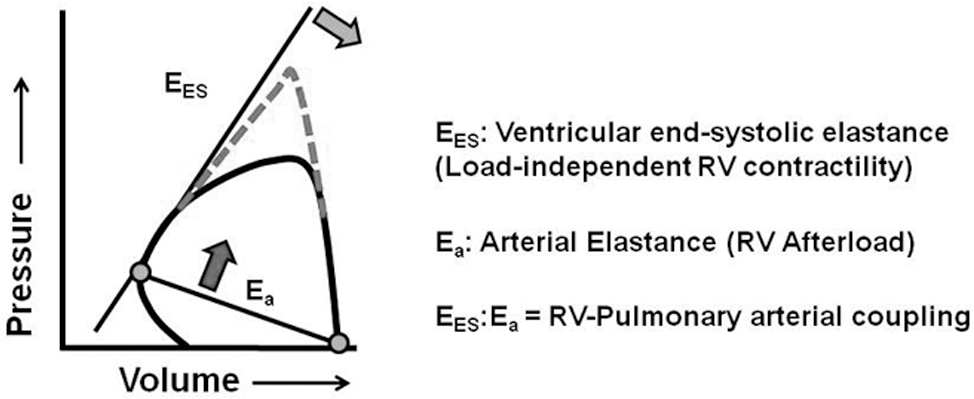
The pressure-volume diagram is used to calculate right ventricular (RV)–pulmonary vascular coupling. The pressure-volume diagram allows for the determination of RV end-systolic elastance (EES). EES is unaffected by changes to RV afterload (dashed line) and, therefore, is the best possible load-independent measurement of contractility. By contrast, arterial elastance (Ea) is proportional to pulmonary vascular resistance and is a measurement of RV afterload. The ratio of EES to Ea is a measure of the coupling of the respective ventricular and arterial loads. Thus, a decrease in EES (light gray arrow) or an increase in Ea (dark gray arrow) due to elevations in pulmonary vascular resistance, for example, disrupts normal coupling. Adapted with permission from Brimioulle et al. 14
These trends have been reproduced in studies of chronic right heart failure. In animals subjected to pulmonary arterial banding or treatment with monocrotaline to induce acute and chronic pulmonary hypertension, respectively, increased EES and preload recruitable RV stroke work were observed. 15 However, changes in these pressure-volume indexes are more pronounced in acute rather than chronic pulmonary hypertension and appear to be contingent largely on RV diastolic function rather than RV free-wall thickness. Along these lines, the importance of RV diastolic function to the pathophysiology of PAH in patients has been reported recently. For example, Rain et al. 16 performed single-beat RV pressure-volume analyses in 7 controls and 21 patients with PAH due largely to connective tissue and congenital heart disease. Their findings suggest that RV diastolic stiffness was increased significantly in PAH, possibly through a mechanism involving the protein titin in RV cardiomyocytes, which regulates sarcomeric stiffness.
Left heart disease and the RV pressure-volume relationship
The effect of increased left heart afterload on RV performance has generated provocative insight into the mechanism(s) that determine RV cardiac output. In one study of experimental chronic RV pressure overload in rabbits, aortic constriction decreased RV cavitary dimension and improved RV contractile performance. 17 Compared with the RV failure condition alone, aortic banding also attenuated histopathological evidence of adverse RV cardiomyocyte remodeling and decreased RV messenger RNA (mRNA) expression levels of matrix metalloproteinase 9 and transforming growth factor β, which are key signaling intermediaries involved in extracellular matrix metabolism and collagen deposition, respectively. 18 Overall, findings from this and other similar studies 19 illustrates the importance of interventricular dependence on RV geometry and performance and suggests that increased LV afterload promotes recruitment of the interventricular septum by the RV to maintain (or augment) RV stroke volume. The implications of these collective observations to patients continue to be explored, although technical factors associated with accurately measuring the RV pressure-volume relationship are an important consideration to the use of these methods in routine clinical practice. 20
HEMODYNAMIC PITFALLS AND PEARLS
In this segment of the symposium, Drs. Michael Landzberg and Alexander Opotowsky used a case-based approach to discuss strategies to synthesize a differential diagnosis and therapeutic approach to patients with various forms of right heart disease, particularly congenital heart 21 and exercise-induced pulmonary vascular 22 diseases. The speakers noted that a dilemma common to the management of this patient population is clinical decision making in asymptomatic or minimally symptomatic patients despite clinical evidence of active right heart disease. Additionally, despite the acuity and severity of many pulmonary vascular diseases, such as pulmonary arterial hypertension (PAH), clinicians must remain alert to alternative causes to patients' clinical presentations, such as competing cardiopulmonary diseases or other processes. To illustrate this latter point, a clinical vignette was presented in which strategies to characterize the contribution of aortic valve disease to exercise intolerance in a patient with bona fide PAH were reviewed.
Aortic valve disease in patients with pulmonary hypertension: cause or consequence?
The positive association between aortic valvular stenosis (AS) and pulmonary vascular dysfunction was originally reported more than 7 decades ago. 23 In one early study, the cardiopulmonary hemodynamic profile was evaluated in a cohort of 74 veterans with surgical AS, which demonstrated that at least moderate pulmonary hypertension was present in 54% of patients and that aortic valve area correlated inversely with pulmonary arterial pressure (PAP). 24 These findings are in agreement with subsequent observations reported in larger patient cohorts, including one study of 388 symptomatic AS patients undergoing right heart catheterization in which 63% had evidence of at least moderate pulmonary hypertension. 25
The prognostic relevance of pulmonary hypertension with respect to outcome in patients with AS is identified as an important factor determining eligibility for mechanical aortic valve replacement. In one recent prospective trial involving 1,116 patients with surgical AS, severe pulmonary hypertension was associated with increased age, more severe multivessel coronary disease, lower LV ejection fraction, and increased probability of recent myocardial infarction. 26 Furthermore, pulmonary hypertension of any degree was an independent predictor of mortality in the immediate postoperative period and over 5 years of follow-up. Indeed, pulmonary vascular disease severity has been raised as a potential clinical metric by which to predict clinical response to aortic valve treatment. Severe pulmonary hypertension (defined by the investigators as pulmonary arterial systolic pressure [PASP] of >60 mmHg) predicted increased mortality at 8 months after transcatheter aortic valve implantation (TAVI; hazard ratio [HR], 1.88 [95% confidence interval, 1.17–30]) in a prospective analysis of 345 aortic valve procedures, which was akin to other traditional high-risk clinical conditions, such as peri-procedural sepsis. 27
Ben-Dor et al. 28 studied 509 patients with severe AS evaluated for TAVI or balloon aortic valvuloplasty and observed that higher preprocedural PAP was associated with increased cardiovascular symptom burden, more severe renal insufficiency, and higher left ventricular end-diastolic pressure. In that study, pulmonary hypertension severity was abrogated immediately following TAVI (or surgical AV replacement), and this change was generally sustained 1 year after the procedure, although data from our institution (unpublished) and others 29 suggest a less consistent therapeutic benefit to the pulmonary vasculature following TAVI for aortic valve replacement.
Less well defined is the role of pulmonary vasoreactivity as a treatment target to improve outcome in patients with aortic valve disease. Among 20 patients with severe AS and pulmonary hypertension (mean PASP, 55 mmHg; PVR, 3.5 Wood units), the oral phosphodiesterase type 5 inhibitor sildenafil (40 or 80 mg) was well tolerated when administered as single dose and induced an immediate improvement in PASP, PVR, and LV stroke volume index by ?25% (P < 0.001), ?29% (P < 0.001), and +8% (P = 0.05), respectively. 30 Despite these encouraging data, larger studies are required to characterize further the potential benefits of pulmonary vasodilator therapy in patients treated medically for aortic stenosis or undergoing valve replacement. It is also worthwhile to note that patient fragility and low distance achieved in the 6-minute walk test 31 are established factors that predict outcome in patients undergoing TAVI or surgical aortic valve replacement, although the contribution of pulmonary vascular disease at rest or provoked by exertion to this effect is not known.
EXERCISE-INDUCED PULMONARY HYPERTENSION: WHEN DOES PHYSIOLOGY TRANSITION TO PATHOLOGY?
The factors that promote abnormal RV and pulmonary vascular response patterns to exercise are incompletely characterized, despite the critical importance of physical activity to the development of right heart failure symptoms provoked by exertion. In fact, although numerous reports have established metrics that define abnormal functional capacity and cardiovascular fitness occurring in concert with irregular exertional cardiopulmonary hemodynamics, a standardized hemodynamic definition for exercise-induced pulmonary hypertension remains lacking. 32 Traditional models have largely emphasized pulmonary vascular dysfunction due to abnormal blood vessel compliance, reactivity, and stiffness as critical determinants of developing exercise-induced PAH (EI-PAH). 33 However, changes to RV cavitary dimension and systolic function due to RV–pulmonary vascular uncoupling is increasingly recognized as a mediator of the EI-PAH pathophenotype 34 and was explored further in this section of the summit.
The pivotal role of RV function in determining cardiopulmonary responses to exercise
Observations from elite athletes identifies two forms of RV remodeling based on endurance (e.g., swimming, longdistance running) versus isometric (e.g., weight lifting) activities, which results in eccentric biventricular remodeling (including RV dilation) and concentric LV remodeling in the absence of significant changes to RV structure/function, respectively (Fig. 3). 35 The mechanism(s) underpinning these differences are unresolved; however, it has been hypothesized that the mitral and aortic valves function as resistors to protect the pulmonary vascular–RV circuit from afterload bursts generated by isometric activities. By contrast, RV involvement in the (patho)phenotype of endurance athletes is ascribed, in part, to the effects of subchronic elevations in cardiac output associated with repetitive aerobic activity on pulmonary vascular function that increases RV cavitary dimension and PAP. 36
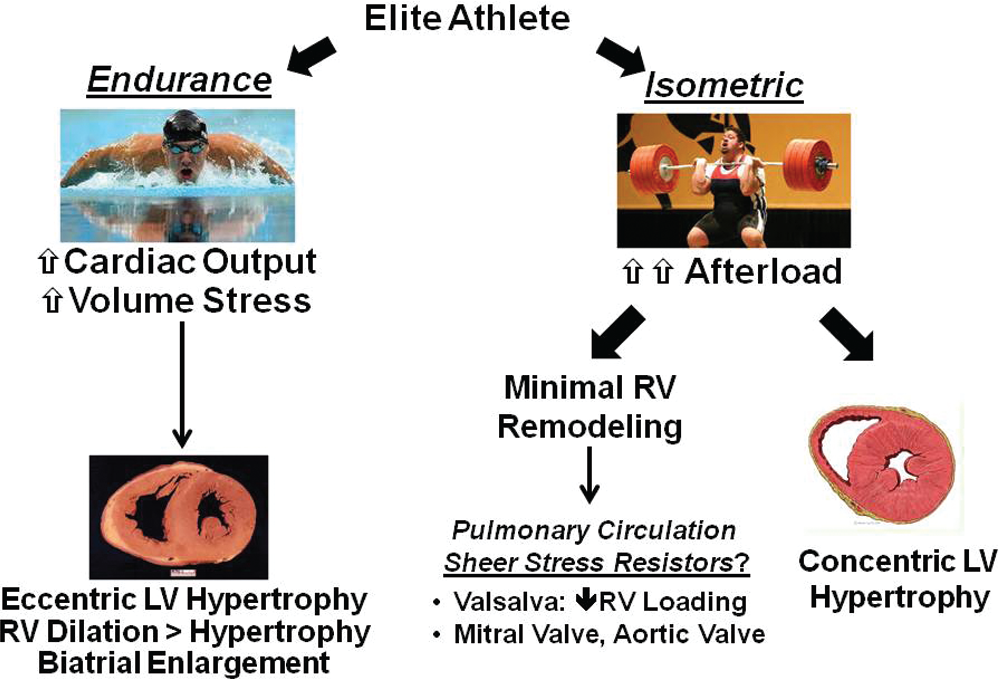
Differential effect on right heart adaptation of endurance or isometric exercise. Exercise-induced remodeling patterns tend to be associated with athletic activity type, which is due to activity-specific changes in cardiopulmonary physiology. For example, isometric exercises (e.g., competitive weight lifting), characterized by sudden bursts of increased afterload, increase left ventricular (LV) pressure-loading conditions and promote LV hypertrophy. By contrast, the right ventricle (RV) is generally spared from remodeling in isometric exercise, an effect hypothesized to occur as a consequence of left heart valve–mediated protection of the pulmonary circulation and RV from increased systemic pressure. In endurance exercise, sustained increases in cardiac output alters volume-loading conditions to both the LV and the RV, resulting in RV dilation and, to a lesser degree, hypertrophy that occurs in addition to left heart remodeling.
Recent reports suggest further that RV cardiomyocyte recovery kinetics may modulate, in part, the right heart response to exercise.37,38 La Gerche et al. 37 used CMR imaging to characterize the effects of ultraendurance athletic activity on RV performance. Their observations suggest that, compared with the preparticipation state, RV function is significantly impaired in the time period immediately following extremely long endurance events, such as marathon running (3 hours in duration) and ultratriathlons (11 hours in duration). Interestingly, they also identified a correlation between event duration and severity of RV functional impairment but noted that RV recovery was observed in most athletes by 12 days after the competition (Fig. 4). Presumably, these reversible changes to RV structure and function in elite athletes describe adaptive or only minimally maladaptive changes to cardiovascular function provoked by physical activity. Taken together, these observations illustrate the importance of characterizing further factors that promote divergence from normal RV exercise-response patterns.
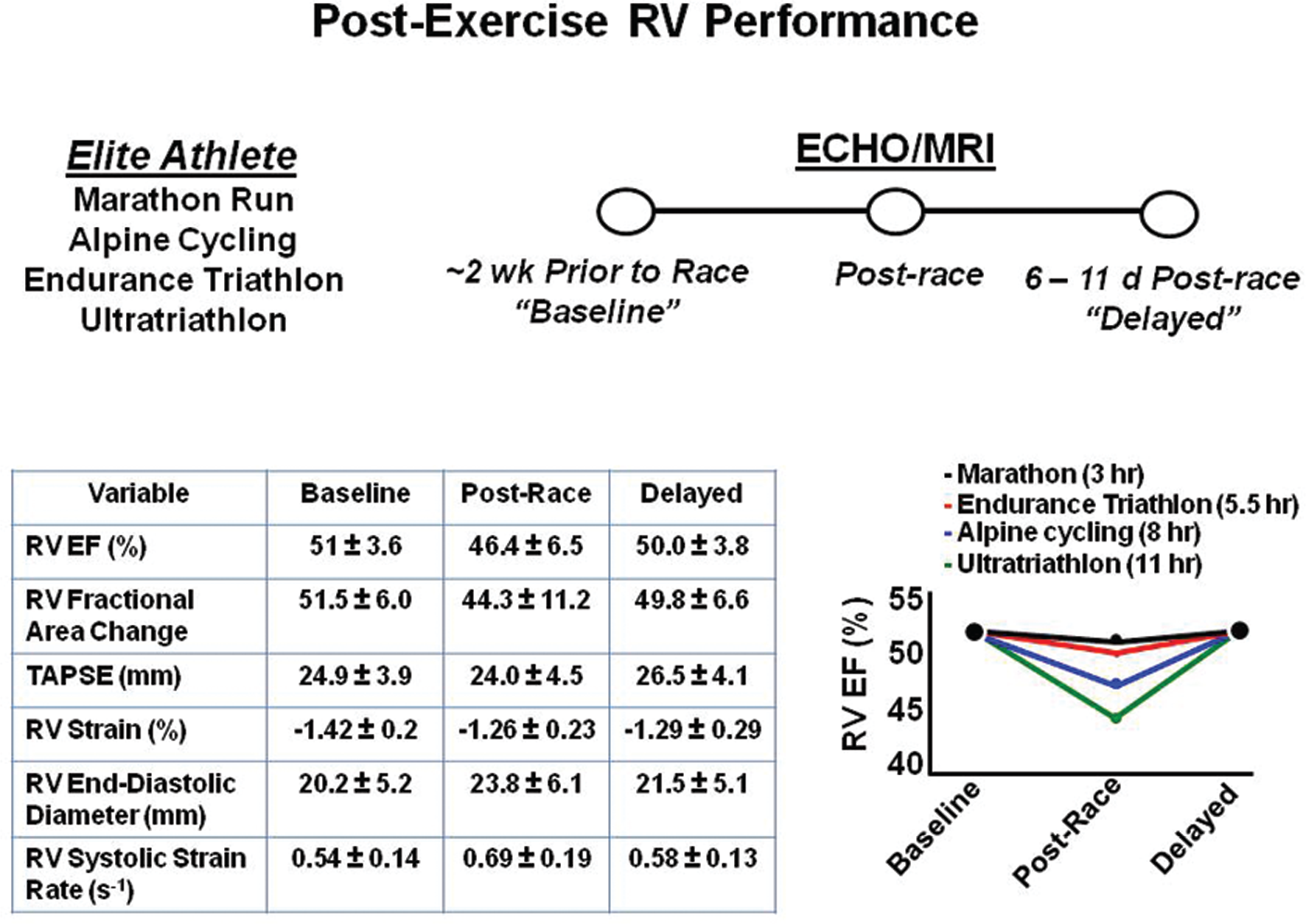
Changes to right ventricular geometry and performance as a function of time following extreme athletic participation. La Gerche et al. 37 recently reported changes to right ventricular (RV) structure and function in a group of marathon runners, alpine cyclers, endurance triathletes, and ultratriathletes, assessed by echocardiography (ECHO) or cardiac magnetic resonance imaging (MRI) within 2 weeks of competition (baseline), immediately after the completion of competition (postrace), and within 6–11 days after the competition (delayed). Overall, these data raise speculation that differences in RV cardiomyocyte recovery kinetics may influence the right heart response patterns to exercise. EF: ejection fraction; TAPSE: tricuspid annular plane systolic excursion. Adapted with permission from La Gerche et al. 37
Defining determinants of RV work: hydraulic power and RV–pulmonary vascular coupling
Even subtle perturbations to RV loading conditions are associated with unfavorable changes to right heart function. 39 Yet increased RV afterload is tolerated to varying degrees across the spectrum of cardiopulmonary disease,40,41 while assessing RV systolic function, such as ejection fraction, is not synonymous with measuring RV efficiency. To accomplish this, it may be useful to quantify the mechanical pump properties of the RV chamber in the context of pulmonary circulatory function. 42
One method to accomplish this is by defining RV hydraulic power as the energy per unit of time required to produce a steady flow of blood through the pulmonary circuit. This is quantitated by the product of cardiac output and mean PAP (mPAP).43,44 Within this construct, flow through the pulmonary vascular circuit will require a mean and oscillatory fraction of hydraulic power, which, in turn, exerts differing functionalities to the RV–pulmonary vascular relationship. On the one hand, the mean oscillatory fraction describes the contribution of RV hydraulic power to the biologically relevant component(s) of the system: blood flow and, thus, oxygen transport. On the other hand, the oscillatory fraction of hydraulic power describes the contribution of RV hydraulic power to the mechanical arm of the system, which is pulmonary arterial pulsatility and blood pressure. Under experimental conditions, the RV oscillatory fraction is ?23%, which is substantially higher than that reported for the LV (10%). 43
It reasons that factors that alter normal RV loading conditions—such as the pulmonary vascular fibrosis, inflammation, and blood vessel thickening that occur in PAH, for example—will potentiate a shift in the RV hydraulic power dynamic in favor of an increased oscillatory fraction. 45 To illustrate this further, consider Figure 5, which illustrates pulmonary vascular distensibility as a nonlinear relationship between cardiac output and mPAP. Under experimental conditions, cardiac output associates positively with pulmonary vascular distensibility up to ?30 L/min. In healthy individuals, the distensibility coefficient is ?2%, which protects the RV somewhat against an increase in afterload during activity. 46 The extent to which intrinsic disease of the pulmonary vasculature alters the slope of this curve, however, has not yet been fully characterized. Nevertheless, it is clear that, at least in the diseased state, pulmonary arterial compliance correlates inversely with RV function, as suggested by numerous clinical reports in patients with various forms of pulmonary hypertension.47,48 From these collective observations, the interrelatedness of pulmonary circulatory and RV function comes into focus.
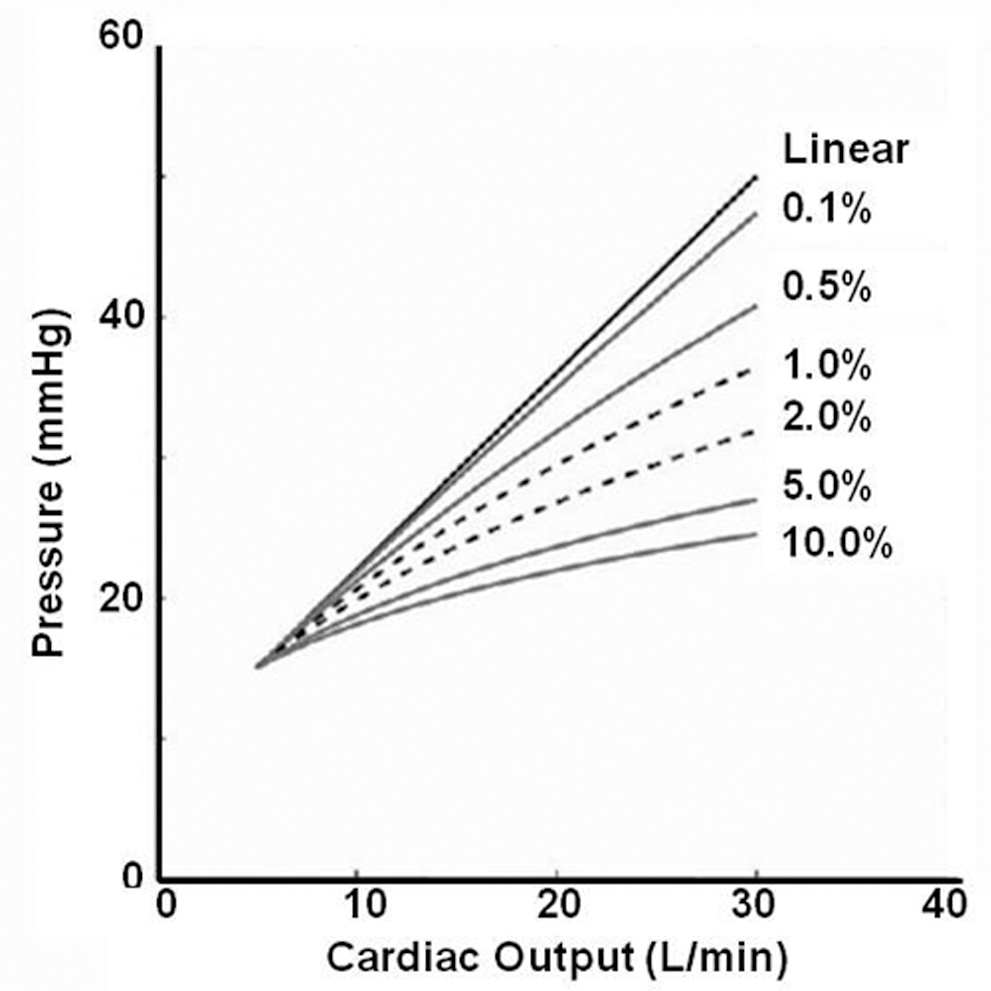
Pulmonary vascular distensibility curve. Modeled mean pulmonary arterial pressure-flow relationships at progressively increased pulmonary vascular distensibility is expressed as percent increase in diameter per millimeter of mercury of pressure. Reproduced with permission from Naeije et al. 46
Assessing RV efficiency through the pressure-volume loop model
The extent to which a disruption in the balance between the mean and oscillatory fractions of hydraulic work influences normal RV performance may be expressed vis-à-vis changes to the pressure-volume relationship (Fig. 2). As discussed by Dr. Redington (see the section “The normal RV pressure-volume relationship” above), the RV end-systolic elastance (EES) is a load-independent measure of myocardial contractility and is defined as the maximal pressure generated by the RV at a particular volume. Arterial elastance (Ea), which is calculated by the ratio of RV end-systolic pressure to stroke volume, is proportional to PVR and, thus, operates as a measurement of RV afterload. 49 Taken together, the ratio of EES to Ea is a measure of the coupling of ventricular contractility and pulmonary arterial load. Thus, a decrease in EES (i.e., contractility) or an increase in Ea (i.e., due to elevated PVR) disrupts normal coupling.
To investigate the physiological relevance of these relationships, Brimioulle et al. 14 performed a series of clever experiments in dogs to assess the RV end-systolic pressure-volume relationship in response to various stimuli designed to mimic exercise. In that study, pharmacological activation of β-adrenergic signaling with dobutamine increased the EES ? Ea ratio, primarily through effects on RV contractility rather than PVR. Thus, under conditions of enhanced RV contractility, improvements to cardiac output may occur at the expense of RV hydraulic efficiency, although proving this in the exercising human requires further investigation.
While interrogating the consequences of exercise with respect to RV–pulmonary vascular coupling by pressure-volume analysis remains a difficult task practically, Tolle et al.
50
demonstrated that among a cohort of patients undergoing invasive exercise hemodynamic monitoring, three subphenotypes merged: normal, in whom the cardiopulmonary hemodynamics were normal at rest and a >50% decrease in PVR was observed at peak exercise; patients meeting hemodynamic criteria for PAH at rest, in whom PVR declined slightly or was increased with exercise; and EI-PAH patients, in whom hemodynamics were normal at rest but functional capacity was diminished and PVR decline at rest was below anticipated levels. These dynamic cardiopulmonary hemodynamic changes were not contingent on changes in LV end-diastolic pressure, indicating intrinsic disease of the pulmonary vasculature. In that study, the downstream consequences of increased PVR on RV–pulmonary arterial coupling was examined indirectly by measuring the relationship between mPAP and maximal volume of oxygen consumption (
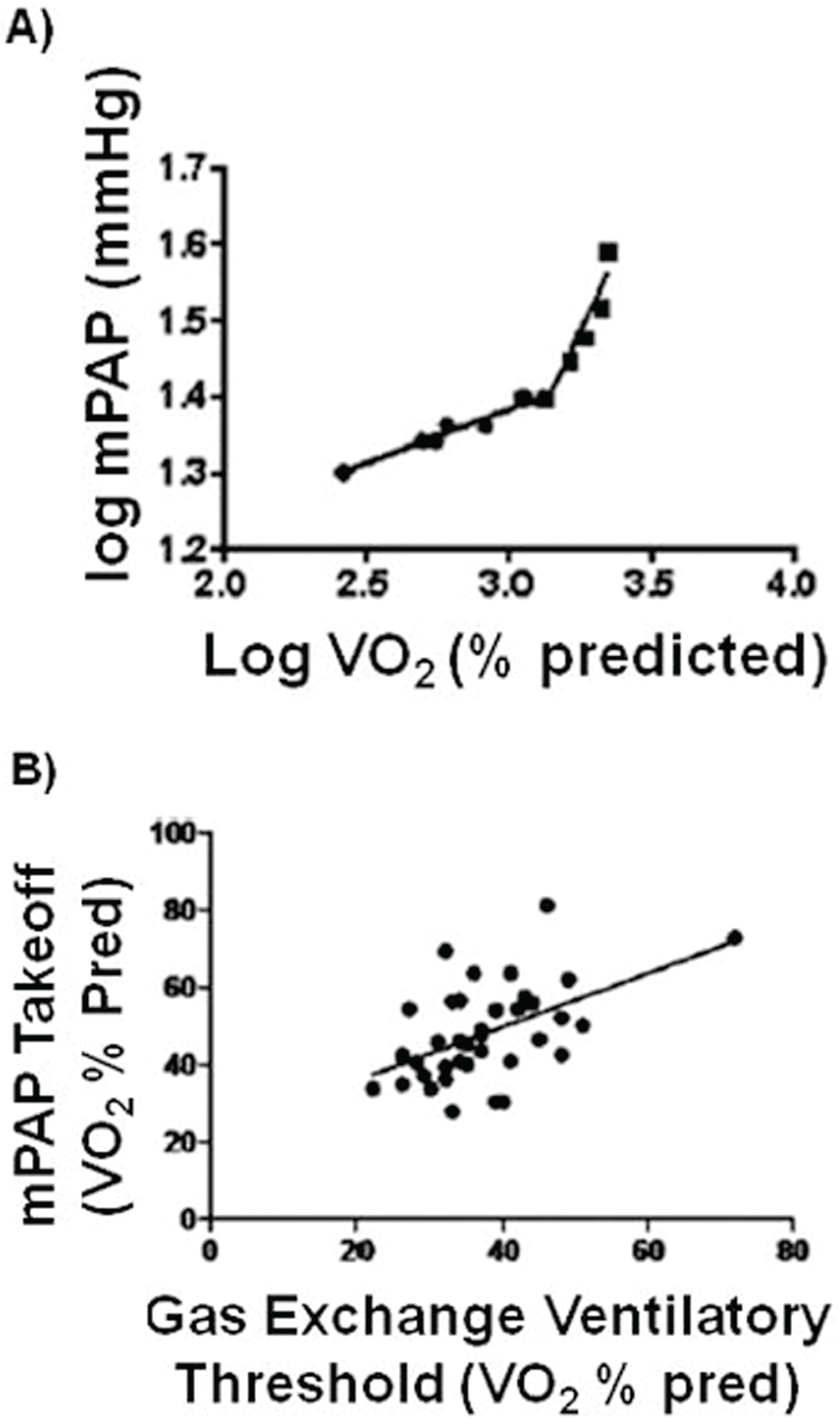
The relationship between mean pulmonary arterial pressure (mPAP) and peak volume of oxygen consumption (
THE SYSTEMS PATHOBIOLOGY OF PAH: A NETWORK MEDICINE APPROACH
Dr. Joseph Loscalzo discussed an evolving paradigm shift in the methods by which human diseases, including pulmonary vascular disease, are studied, analyzed, and categorized. The traditional method for classifying human disease is an organ-based approach that segregates phenotypes largely along pathophysiological lines. 51 This time-honored strategy is derived from the classical era of medicine, in which microscopy or physical diagnosis alone was sufficient to diagnose diseases for the primary purpose of prognosis. 52 However, in the genomic and molecular era of science, the possibility of developing target-specific therapies, such as monoclonal antibodies or small-molecule inhibitors, is increasingly realized; thus, methods to conceptualize diseases that consider a more precise pathophenotype classification are desirable. This has particular relevance to the field of pulmonary vascular disease, in which a swath of candidate molecules are implicated in the development of PAH, defined (somewhat vaguely) as a unifying end phenotype according to general cardiopulmonary hemodynamic criteria. In turn, delineating with greater precision the basic factors that define PAH clinical expression is anticipated to provide enhanced clarity over current methods for matching affected patients to disease-specific therapies. 53
There is substantial genetic overlap between selected, common human disease pathophenotypes, 54 which has been demonstrated using genome-wide association studies (GWASs) 55 and systems biology network methods. 56 The GWASs, which are performed using vast numbers of patients sharing a common disease, have identified genetic variability (i.e., single-nucleotide polymorphisms) in association with disease prevalence. The GWAS strategy, therefore, leverages large-scale epidemiological and genetic methods to provide, as Dr. Loscalzo stated, a “linear narrative of disease” through the identification of chromosomal loci implicated in incident disease occurrence. By contrast, a systems biology approach to defining disease pathophenotype is multidimensional: a network of molecular interactions (protein-protein, mRNA-mRNA, etc.) regulating the development of an intermediate pathophenotype is generated mathematically, on the basis of the curated literature or biological data sets, to identify novel targets involved in disease expression (reviewed recently by Silverman and Loscalzo 57 ).
The interactome and human disease
An overarching objective of network theory when applied to biology is to depict quantitatively the interactions between proteins (or other key cellular components), clinical phenotypes, and other variables relevant to understanding disease pathophenotypes. This is based on the principle that the functional effects of any single protein, for example, involve targets residing in both the intracellular and the extracellular compartment as well as at remote tissue/organ sites. 58 Thus, the effect of a specific genetic abnormality is likely to involve a range of other functionally relevant interactions, which forms the links between targets and, ultimately, the interactome. In fact, several published data sets support the assertion that biological networks are not random and generate a pattern of clustering nodes.58,59 The organization of node clusters may be grouped further into protein modules, which represent highly linked variables. Overall, this framework unveils connectivity between biologically relevant factors and/or clinical phenotypes.
In light of the observation that of 25,000 genes only ?1,700 are implicated in human disease, 60 the importance of functional connectivity between genes and gene products is more clearly realized. This may be depicted schematically by the centralization of fetally lethal genes and the distribution of nonessential genes to the periphery of the human interactome (Fig. 7). Building on this concept, disease modules are developed to express important biological factors according to interactivity, functionality, or relevance, as defined by association with a specific disease in the presence of an expression-altering effector (environmental factors, de novo mutation, etc.).62,63 Collectively, the network construct is a truly “ground-up” approach to classifying disease: key molecular interactions associating with disease pathophenotype are identified, which, in turn, link basic substrate abnormality to clinical pathophenotype.
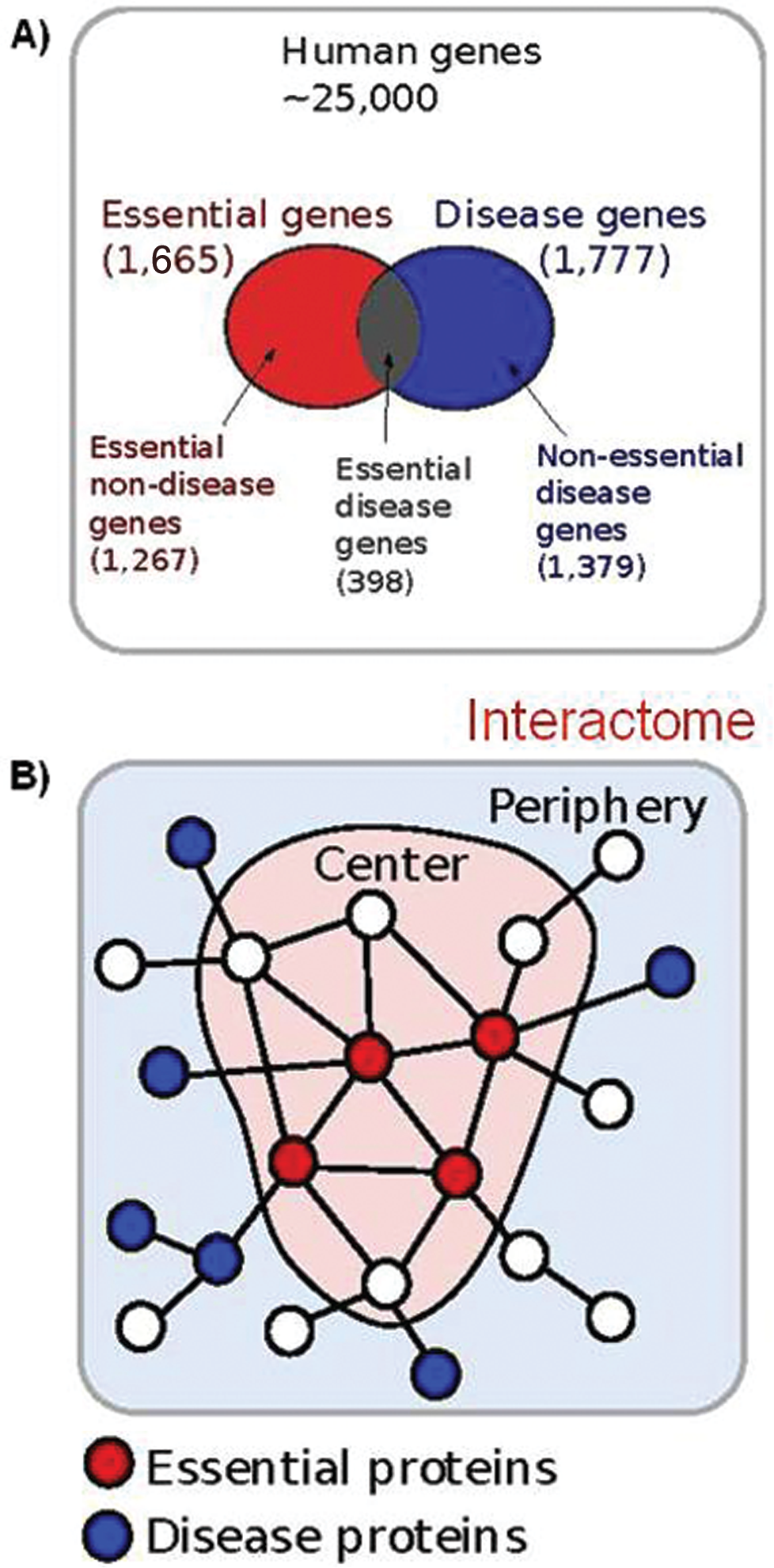
Distribution of genes in the human interactome. A, Depiction of the distribution of in utero essential versus known disease genes identified among the ?25,000 human genes. B, Schematic diagram of the differences between essential and nonessential disease genes. Nonessential disease genes (shown as blue nodes) are found to segregate at the network periphery, whereas in utero essential genes (shown as red nodes) tend to be at the functional center (encoding hubs and expressed in many tissues) of the interactome. A is reproduced with permission from Goh et al. 61 B is reproduced with permission from Barabási et al. 62
Systems biology and PAH
PAH is a heterogeneous disease in many respects, including the involvement of numerous molecular mechanisms identified in the development of pulmonary arterial thickening, fibrosis, and dysregulated pulmonary endothelial and smooth muscle cell apoptosis. In this regard, PAH is an excellent disease model for network biology analysis, which is, in part, the subject of an ongoing National Institutes of Health–sponsored consortium investigation (MAPGen; http://www.mapgenprogram.org). Parikh et al. 64 constructed a PAH network consisting of 131 nodes and 26 functional pathways that, when subjected to mathematical modeling, resulted in the development of a consolidated PAH interactome consisting of 115 nodes and 255 edges (i.e., interactions between nodes; Fig. 8). A subset of microRNAs (miRNAs), which are noncanonical and highly conserved noncoding ribonucleic acids, 65 were identified on the basis of their previously established role in mediating cellular responses to hypoxia, inflammation, and tissue growth factor β signaling. Of this group, miRNA-21 was studied further owing to its association with cellular phenotypes implicated in PAH and the regulatory influence observed by this particular miRNA on the network. Findings in cultured pulmonary arterial endothelial cells and in experimental PAH in vivo validated miRNA-21 involvement in the PAH pathophenotype via regulation of bone morphogenetic protein receptor type 2 and Rho kinase signaling pathways. Taken together, these findings demonstrate the potential for network biology to identify novel molecular links involved in the pathobiology of PAH.
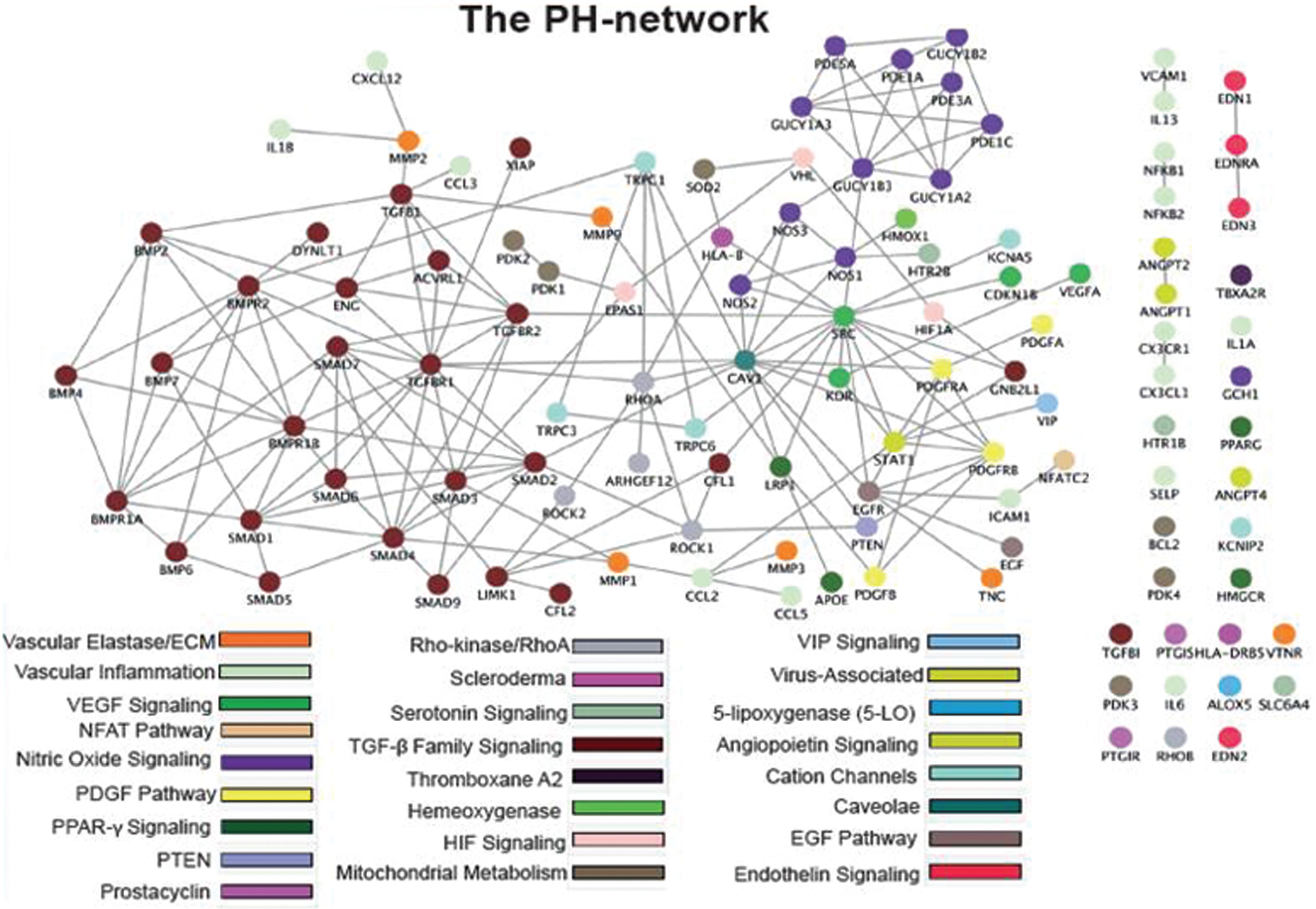
A proposed pulmonary hypertension (PH) network. The known first-degree interactions (indicated by gray lines) are displayed among the genes in the PH module. There exist a total of 115 nodes and 255 edges (interconnections). Reproduced with some modification with permission from Parikh et al. 64
CONCLUSIONS
A wide-ranging gamut of topics relevant to understanding RV–pulmonary vascular (patho) physiology in the current era was discussed in this section of the Third International Right Heart Failure Summit. Classical studies analyzing the RV pressure-volume relationship were reviewed and evaluated in the context of contemporary data, particularly regarding the effects of increased RV afterload and exercise on RV work efficiency. Novel reports characterizing changes to RV structure and function following competitive athletic participation has generated important insight into the potential contribution of RV myocyte recovery kinetics to determining adaptive versus maladaptive RV exercise response patterns in humans. Future work is likely to continue to leverage a systems biology approach to understanding further the molecular underpinnings that regulate RV and pulmonary vascular function and modulate the development of right heart pathophenotypes, particularly PAH and RV failure.
Footnotes
ACKNOWLEDGMENTS
I would like to acknowledge the expert technical assistance of Stephanie Tribuna in the preparation of the manuscript.
