Abstract
Adenosine is a potent signaling molecule that has paradoxical effects on lung diseases. We have previously demonstrated that sustained adenosine exposure by inhibition of adenosine degradation impairs lung endothelial barrier integrity and causes intrinsic apoptosis through equilibrative nucleoside transporter1/2-mediated intracellular adenosine signaling. In this study, we further demonstrated that sustained adenosine exposure increased mitochondrial reactive oxygen species and reduced mitochondrial respiration via equilibrative nucleoside transporter1/2, but not via adenosine receptor-mediated signaling. We have previously shown that sustained adenosine exposure activates p38 and c-Jun N-terminal kinases in mitochondria. Here, we show that activation of p38 and JNK partially contributed to sustained adenosine-induced mitochondrial reactive oxygen species production. We also found that sustained adenosine exposure promoted mitochondrial fission and increased mitophagy. Finally, mitochondria-targeted antioxidants prevented sustained adenosine exposure-induced mitochondrial fission and improved cell survival. Our results suggest that inhibition of equilibrative nucleoside transporter1/2 and mitochondria-targeted antioxidants may be potential therapeutic approaches for lung diseases associated with sustained elevated adenosine.
Introduction
Adenosine is a purine nucleoside and a potent signaling molecule. Extracellular adenosine is produced by action of extracellular ectonucleotidases, CD39 and CD73, and modulates multiple cellular and physiological functions via G protein-coupled adenosine receptors (ARs).1,2 Among four types of ARs, endothelial cells (EC) predominantly express A2AR and A2BR.3,4 Extracellular adenosine can also be rapidly metabolized by cell surface adenosine deaminase (ADA). Dysfunction or inhibition of ADA can cause accumulation of extracellular adenosine, which can be taken up into cells by equilibrative nucleoside transporters (ENTs) and/or concentrative nucleoside transporters (CNTs). ENTs are diffusion-limited channels. Among four types of ENTs, EC predominantly express ENT1 and ENT2, with ENT1 being expressed at twice the level of ENT2. 5 ENT1 has a 2.8-fold higher affinity for adenosine than ENT2. 5 CNTs have very low affinity for adenosine and limited expression in EC. 5 Expression levels and adenosine affinity suggest that ENT1/2 plays a major role in adenosine uptake in EC. Upon uptake into cells, adenosine can be metabolized by intracellular ADA and/or adenosine kinase (AK).6,7 Adenosine also reacts with homocysteine (HC) to generate S-adenosyl-L-homocysteine (SAH). 6 SAH is a product of S-adenosyl-L-methionine (SAM) de-methylation. The balance of SAH/SAM can affect protein and DNA methylation.8–12 Although adenosine signaling through ARs has been extensively studied in the past decades, little is known about the role of intracellular adenosine signaling.
The concentrations of extracellular adenosine are about 40–600 nM in homeostatic conditions. 13 However, adenosine concentrations are elevated in response to inflammatory stimuli and tissue injury caused by acute hypoxia, high tide volume ventilation, endotoxin, and bleomycin.14–17 Adenosine has paradoxical effects on pathophysiology. We and others have demonstrated that acutely elevated adenosine enhances EC barrier function and protects against lung inflammation and injury in several animal models of acute lung injury (ALI), suggesting that acute exposure to adenosine is beneficial.3,18,19
In contrast, chronically elevated adenosine can be detrimental. ADA deficiency in humans causes accumulation of adenosine associated with severe combined immunodeficiency disease and lung inflammation. 20 Studies of ADA-deficient mice demonstrated that sustained elevated adenosine is responsible for increased permeability lung edema, pulmonary fibrosis, and emphysema.21,22 ADA enzyme replacement is a lifesaving strategy effective in the treatment of ADA-deficient patients and animals. 23 These findings clearly demonstrate a detrimental effect on the lungs of sustained adenosine exposure. Adenosine is also associated with other pathological conditions. Adenosine is elevated in plasma of patients with sepsis-induced ALI.24,25 Adenosine is also elevated in patients with chronic lung diseases. For example, adenosine is increased in bronchoalveolar lavage fluid and exhaled breath condensate of patients with asthma and in sputum of patients with cystic fibrosis.26,27 Expression of the ectonucleotidase, CD73, is elevated, and ADA activity is decreased in the lungs of patients with chronic obstructive pulmonary disease (COPD) and idiopathic pulmonary fibrosis (IPF).28–30 Plasma ADA activity is decreased in rats exposed to cigarette smoke (CS) for four weeks. 31 We have previously shown that lung adenosine levels were elevated in mice exposed to CS for three weeks. 32 These reports suggest that adenosine metabolism is altered in some lung diseases and that adenosine signaling potentially plays important pathophysiological roles.
Previous studies on sustained elevated adenosine-induced lung injury have focused on inflammatory cells and fibroblasts33,34; but little is known about the effects of sustained adenosine exposure on EC. Our previous report demonstrated that sustained exposure to adenosine by ADA inhibition causes endothelial barrier dysfunction and apoptosis, through ENT1/2-facilitated adenosine uptake.32,35 However, the mechanism(s) by which intracellular adenosine causes endothelial dysfunction is not known.
Mitochondria supply most ATP required for cellular function and survival through oxidative phosphorylation. 36 Mitochondria continually undergo mitochondrial fission and fusion to form elongated and interconnecting tubular networks in response to environmental changes.37–39 Dynamic mitochondrial fusion and fission are essential for mitochondria homeostasis. 40 Mitochondrial fission segregates damaged mitochondria, which are subsequently eliminated by mitochondrial autophagy (mitophagy), whereas mitochondrial fusion is critical for dilution of damaged or injurious contents among individual mitochondria, protecting mitochondrial DNA stability and respiratory functions. 41 Key proteins involved in mitochondrial fission, such as dynamin-related protein 1 (Drp1), and mitochondrial fusion, such as Mitofusin 1 and 2 (Mfn1 and Mfn2), are essential for cell survival.39,41 Mice deficient in either Mfn1 and Mfn2 die in midgestation, and interruption of mitochondrial fusion results in loss of inner mitochondrial membrane potential. 42 Conversely, overexpression of Mfn2 in heart muscle cells 43 or vascular smooth muscle cells 44 induces apoptosis. On the other hand, overexpression of Drp1 also induces the release of cytochrome C and subsequent apoptosis,45,46 but Drp1 knockout mice die during embryonic development. 47 Thus, mitochondrial fission and fusion maintain intact mitochondrial quality control. Our previous report suggested that sustained elevated adenosine caused by ADA inhibition caused mitochondrial injury, as evidenced by altered mitochondrial morphology and membrane potential. 32 In this study, we characterized the effect of sustained adenosine exposure on mitochondria fusion and fission and function. We found that sustained adenosine exposure elevated mitochondrial oxidative stress, increased mitochondrial fission, and reduced mitochondrial respiration, leading to endothelial injury, via ENT1/2-facilitated intracellular adenosine signaling. Our data suggest that inhibition of ENT1/2 and mitochondria-targeted antioxidant may be potential therapeutic approaches for adenosine-associated lung diseases.
Material and methods
Cells and reagents
Primary bovine pulmonary artery endothelial cells (PAECs) were purchased from Vec Technologies (Rensselaer, NY) and used between passages 3 and 7 in this report. The PAECs were cultured in minimum essential medium Eagle containing 10% of fetal bovine serum and 1 mM of sodium pyruvate. Adenosine, dipyridamole (DPM), S-(4-Nitrobenzyl)-6-thioinosine (NBTI), SB203580 (SB), 1,3-dipropyl-7-methylxanthine (DPMX), 8-(4{[(4-cyano)phenylcarbamoylmethyl]oxy}epheyl)-1,3-di-(n-propyl)xanthine (MRS1754), mitoTEMPO, 5′-iodotubercidin (ITU), and N-acetyl-cysteine (NAC) were purchased from Sigma Aldrich (St. Louis, MO). 2′-Deoxycoformycin (DCF) and SP600125 (SP) were purchased from Tocris Bioscience (Minneapolis, MN). Antibodies directed against Mfn1, Drp1, and Parkin were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Antibodies used to detect Pink1 and Vinculin were from Abgent (San Diego, CA) and Sigma Aldrich (St. Louis, MO), respectively. Hoechst 33342 and MitoSOX Red were purchased from Life Technologies (Carlsbad, CA). MitoTracker Green which visualizes mitochondria was purchased from Invitrogen (Grand Island, NY). Seahorse XF Cell Mito Stress Test kits (including oligomycin, carbonyl cyanide-p-trifluoromethox- yphenyl-hydrazon (FCCP), and a mixture of Antimycin A and Rotenone) were from Agilent Seahorse Bioscience (Billerica, MA). MTS assay kits were purchased from Abcam (Cambridge, MA).
Mitochondrial morphology and mitochondrial reactive oxygen species assays using fluorescence microscopy
PAECs were seeded on a coverslip in a 24-well plate. The cells were treated with vehicle or 50 µM of adenosine plus 50 µM of DCF for up to 48 h, a treatment that we have previously shown to increase EC adenosine levels.32,35 The cells were washed with the culture media twice followed by incubation with MitoTracker Green to stain mitochondria in the media for 20 min at 37 ℃. The media were removed, and the cells were washed twice with fresh media. The cells were then co-stained with MitoSOX Red to label mitochondrial reactive oxygen species (ROS) in the culture media for 20 min at 37 ℃. After washing twice with culture media, the coverslips were mounted onto microscope slides with a drop of warm media and sealed with nail polish. Images were captured by a fluorescence microscope. For the experiments where MitoTEMPO was used, the PAECs were pre-treated with 5 µM of MitoTEMPO or vehicle for 1 h. After pretreatment, the cells were treated with adenosine plus DCF (AD) in the presence or absence of MitoTEMPO for 30 h. The cells were then stained with MitoTracker green and processed for fluorescent microscopy.
Measurement and quantification of mitochondrial ROS in cultured EC using spectroscopy
PAECs were seeded on a black 96-well plate and grown to 80% confluency. The cells were treated with 50 µM adenosine plus 50 µM DCF (AD) for 5, 24, 30, and 48 h. The cells were then washed twice with Dulbecco's phosphate buffered saline (DPBS), which contains Ca2+ and Mg2+, to maintain cell adherence and were stained with Hoechst 33342 for 20 min. Afterward, the cells were washed with DPBS twice and then stained with MitoSOX Red for 20 min. After washing twice, fresh DPBS was added to both cell-containing wells and empty wells, which were used as blank. The fluorescence intensities of MitoSOX Red and Hoechst 33342 were captured by fluorescence spectrometer. The MitoSOX Red signals were normalized to Hoechst 33342. For the experiment where multiple inhibitors were used to dissect the signaling pathways leading to production of mitochondrial ROS, the cells were pre-treated with specific inhibitors for 1 h followed by incubation with AD in the presence or absence of the inhibitors for 48 h.
Assessment of mitochondrial oxygen consumption rate
PAECs (3.0 × 104 cells per well) were seeded on wells in a specialized 96-well plate designed for the XF96 Seahorse mitochondrial analyzer. The cells were pre-treated with multiple inhibitors for 1 h prior to the treatment with adenosine and DCF. Then, the cells were treated with vehicle or 50 µM of adenosine plus 50 µM of DCF (AD) in the presence or absence of inhibitors for 30 h. After 30 h, the cells were washed with Seahorse media twice and oxygen consumption rate (OCR) was measured by Seahorse mitochondrial analyzer. Oligomycin, FCCP, and a mixture of rotenone and antimycin A were automatically injected into the wells by the analyzer to assess mitochondrial basal and maximal OCR, ATP production, and spare capacity.
MTS cell viability assay
The PAECs were pre-incubated with 5 µM MitoTEMPO for 1 h and then treated with 50 µM of adenosine plus 50 µM of DCF (AD) in the presence or absence of 5 µM MitoTEMPO for 48 h. Cell morphology was captured by an inverted microscope. These cells were then processed for an MTS assay following the manufacturer's protocol.
Western blot analysis
PAECs were treated with adenosine and DCF (AD) for 0, 5, 24, 30, and 48 h. After the treatment, the cells were harvested using radioimmunoprecipitation assay buffer (RIPA buffer, 20 mM Tris-HCl in pH 7.5, 150 mM NaCl, 1 mM Na2EDTA, 1 mM EGTA, 1% NP-40, 1% sodium deoxycholate, 2.5 mM sodium pyrophosphate, 1 mM b-glycerophosphate, 1 mM Na3VO4, and 1 µg/ml leupeptin). The cell pellets in RIPA were centrifuged at 12,000 r/min for 15 min and the supernatant was collected for BioRad DC protein assay (BioRad, Philadelphia, PA). The protein lysate was run in an SDS-PAGE followed by wet-transfer to Polyvinylidene difluoride (PVDF) membranes at 100V for 1 h at 4 ℃. The membranes were blocked with 5% milk for 30 min followed by incubation with primary antibodies overnight at 4 ℃. After incubation overnight, the membranes were washed and incubated with secondary horseradish peroxidase (HRP)-conjugated antibodies for development.
Statistics
Two tailed t-test was performed for experiments comparing two groups. For experiments comparing multiple groups, one-way ANOVA with Tukey's multiple comparison test was performed. Data were displayed with mean ± standard deviation (SD). p < 0.05 is considered as statistically significant.
Results
Sustained adenosine exposure increased mitochondrial ROS
We previously demonstrated that adenosine plus ADA inhibition by DCF caused sustained increases in EC adenosine and that sustained adenosine exposure decreased mitochondrial membrane potential and activated the intrinsic pathway of apoptosis.32,35 Mitochondrial oxidative stress can lead to mitochondrial dysfunction.
36
Therefore, we examined whether adenosine can induce mitochondrial ROS in vitro. PAECs were treated with adenosine in the presence of DCF, which irreversibly inhibits ADA, thus maintaining adenosine levels,32,35 for up to 48 h and then subjected to assessments of mitochondrial morphology by MitoTracker green staining and mitochondrial ROS by MitoSOX Red staining. The cells treated with AD for 30 and 48 h had significantly elevated levels of mitochondrial ROS and lost characteristic mitochondrial morphology (Fig. 1a). To validate this result in a more quantitative fashion, the cells were stained with MitoSOX Red and normalized to cell numbers using Hoechst 33342 staining. The fluorescence intensities of mitochondrial ROS and Hoechst 33342 were measured and quantified by a fluorescence spectrometer. Consistent with the data from fluorescence microscopy, treatment of cells with AD significantly elevated mitochondrial ROS at 30 h and 48 h (Fig. 1b).
Sustained adenosine exposure increased mitochondrial ROS in lung EC. PAECs were treated with vehicle (V) or 50 µM adenosine plus 50 µM DCF (AD) for 5, 24, 30, and 48 h. Following treatment, the cells were washed and then incubated with MitoTracker Green to stain mitochondria and MitoSOX Red to label mitochondrial ROS. The cells were visualized by fluorescence microscopy. Panel a represents three independent experiments. Panel b: after treatment, the cells were washed twice and then stained with Hoechst 33342 for 20 min to quantify cell numbers. The cells were then washed twice and stained with MitoSOX Red for 20 min. The fluorescence intensity of Hoechst 33342 and MitoSOX Red were captured by a fluorescence spectrometer with Ex 360 nm/Em 460 nm and Ex 510 nm/Em 580 nm, respectively. The relative numbers of cells in each well were normalized by the fluorescence intensity of Hoechst 33342. The fluorescence intensity of MitoSOX Red in each well was then adjusted by the relative numbers of cells. The levels of mitochondrial ROS were expressed as fold changes of the fluorescence intensity of MitoSOX Red relative to that of untreated cells. The data are presented as mean ± SD of four independent experiments (n = 4). *p < 0.05 vs. control cells.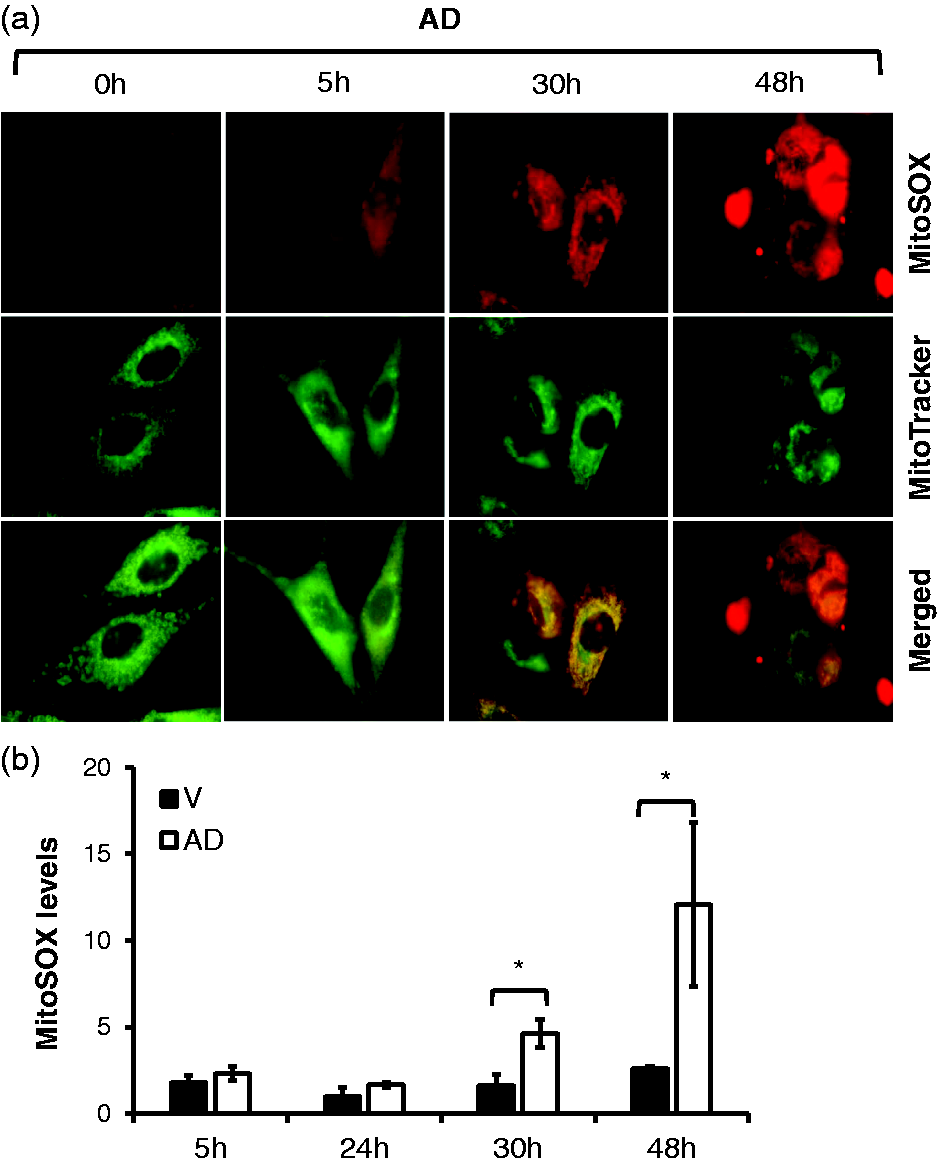
ENT1/2 and MAP kinases mediated sustained adenosine-induced mitochondrial oxidative stress
We have previously shown that inhibition of ENT1/2 by DPM and NBTI prevents sustained adenosine-induced apoptosis and barrier dysfunction in lung EC.32,35 Here, we further asked whether ENT1/2 and AR play a role in sustained adenosine-induced mitochondrial ROS generation. We found that sustained adenosine-induced production of mitochondrial ROS was completely prevented by the ENT1/2 inhibitors, DPM and NBTI, but not by either A2AR antagonist, DPMX, or A2BR antagonist, MRS1754 (Fig. 2a). These results demonstrate that sustained adenosine exposure elevates mitochondrial ROS via ENT1/2-dependent intracellular adenosine signaling in EC.
ENT1/2 and MAK kinases mediated sustained adenosine-induced mitochondrial oxidative stress. PAECs were pre-treated with ENT1/2 inhibitors, DPM (10 µM) or NBTI (10 µM), an A2AR antagonist, DPMX (10 µM), or an A2BR antagonist, MRS1754 (10 µM), for 1 h and then incubated with 50 µM adenosine and 50 µM DCF (AD) in the absence or presence of individual inhibitor for 30 h (Panel a). Similarly, the cells were pre-treated with a cytosolic antioxidant, NAC (12.5 mM), a mitochondrial ROS scavenger, mitoTEMPO (5 µM), a p38 inhibitor, SB203580 (SB, 10 µM), or a JNK inhibitor, SP600125 (SP, 10 µM), and then incubated with 50 µM adenosine and 50 µM DCF (AD) in the absence or presence of individual inhibitors for 30 h (Panel b). Following the treatments, the cells were washed twice and incubated with Hoechst 33342 and MitoSOX Red to quantify cell numbers and mitochondrial ROS levels. The relative numbers of cells in each well were normalized by the fluorescence intensity of Hoechst 33342. The fluorescence intensity of MitoSOX Red in each well was then adjusted by the relative numbers of cells. The levels of mitochondrial ROS were expressed as fold changes of the fluorescence intensity of MitoSOX Red relative to that of untreated cells. The data are presented as mean ± SD of three independent experiments (n = 3). *p < 0.05.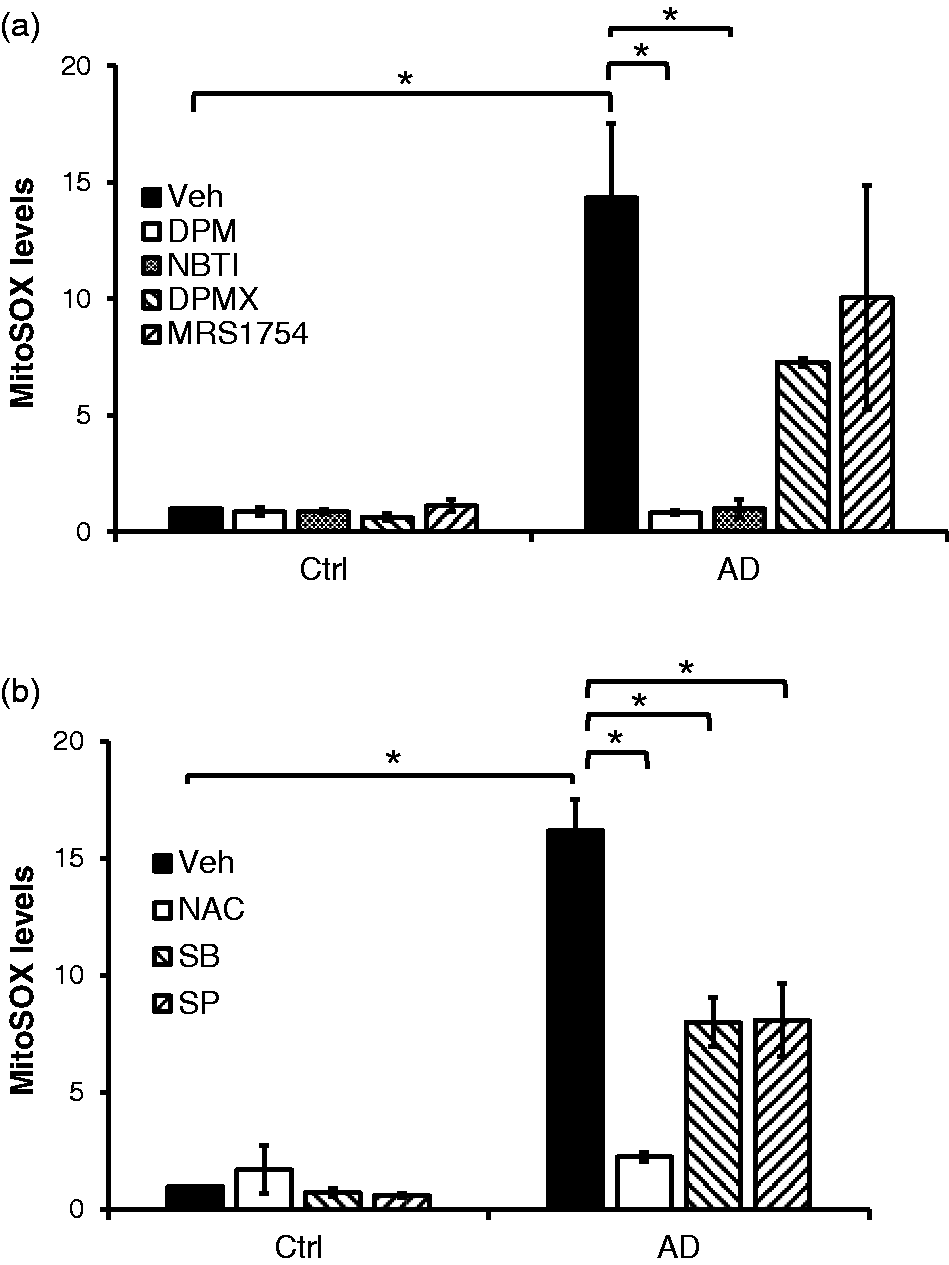
Intracellular ROS can trigger activation of p38 and JNK pathways leading to apoptosis.48–50 However, it is unclear whether p38 and JNK mediate mitochondrial ROS production. It is also unclear whether cytosolic ROS affects the generation of mitochondrial ROS. Therefore, we characterized the possible pathways leading to mitochondrial ROS production using a cytosolic antioxidant, NAC; a p38 inhibitor, SB203580; and a JNK inhibitor, SP600125. As expected, sustained adenosine-induced mitochondrial ROS production was completed prevented by NAC (Fig. 2b). These results suggest that cytosolic ROS is an upstream mediator driving mitochondrial ROS generation. Thus, replenishing NADPH (Nicotinamide adenine dinucleotide phosphate) via NAC can blunt mitochondrial ROS production. 51 We also found that inhibition of p38 and JNK by SB203580 and SP600125, respectively, attenuated sustained adenosine exposure-induced generation of mitochondrial ROS in EC (Fig. 2b). These data collectively suggest that sustained exposure to adenosine causes mitochondrial ROS via ENT1/2-facilitated intracellular adenosine uptake and subsequent cytosolic ROS and p38/JNK kinase pathways.
Sustained adenosine exposure reduced mitochondrial respiration via ENT1/2-dependent signaling
Abnormal mitochondrial morphology and elevated mitochondrial ROS may trigger abnormal mitochondrial function. To test this, we examined whether sustained elevated adenosine changed mitochondrial OCR in EC. We found that sustained adenosine exposure of EC decreased multiple parameters of mitochondrial metabolism, including basal respiration, ATP production, maximal respiration, and spare capacity, in EC (Fig. 3a). The reduced mitochondrial maximal respiration and spare capacity were completely prevented by ENT1/2 inhibitors, DPM and NBTI (Fig. 3a). These results demonstrate that sustained adenosine exposure reduced mitochondrial respiration through an ENT1/2-dependent pathway.
Sustained adenosine exposure impaired mitochondrial respiration via ENT1/2-mediated intracellular signaling. PAECs were pre-treated with ENT1/2 inhibitors, DPM (10 µM) or NBTI (10 µM) for 1 h and then incubated with 50 µM adenosine and 50 µM DCF (AD) in the absence or presence of individual inhibitor for 30 h (Panel a). PAECs were pre-treated with an A2AR antagonist, DPMX (10 µM), or an A2BR antagonist, MRS1754 (10 µM), for 1 h and then incubated with 50 µM adenosine and 50 µM DCF (AD) in the absence or presence of DPM (10 µM) with or without DPMX (10 µM) or MRS1754 (10 µM) for 30 h (Panel b). PAECs were pre-treated with AK inhibitor, ITU (10 µM) for 1 h and then incubated with 50 µM adenosine and 50 µM DCF (AD) in the absence or presence of ITU (10 µM) for 30 h (Panel c). The cells were then washed twice with basal seahorse media and subjected to assessments of OCR using XF96 seahorse analyzer. Oligomycin, FCCP, and a mixture of antimycin A and rotenone were added to the system to assess OCR response to those inhibitors for oxidative phosphorylation. The data are presented as mean ± SD of three independent experiments (n = 3). *p < 0.05.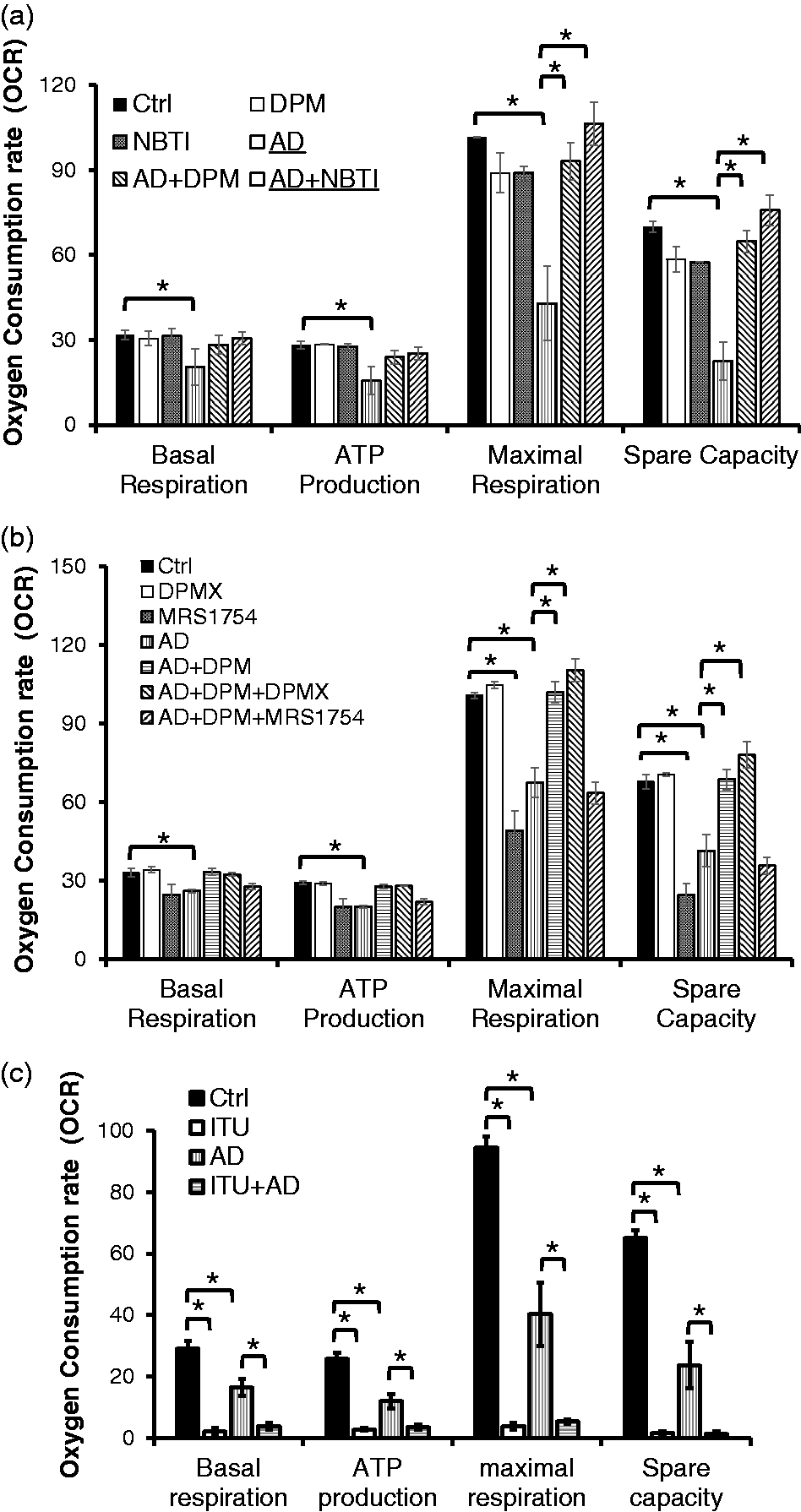
Inhibition of ENT1/2 has been shown to promote signaling of A2AR and A2BR.52–54 Thus, it is possible that inhibition of ENT1/2 protects against sustained adenosine-induced reduction of mitochondrial respiration by activating A2AR and/or A2BR signaling, but not due to inhibition of intracellular adenosine signaling. To address this possibility, we tested if the protective effect of ENT1/2 inhibitors could be abrogated by an A2AR antagonist, DPMX, or an A2BR antagonist, MRS1754. We found that DPMX had no effect on the protection of ENT1/2 inhibitors against sustained adenosine-induced inhibition of mitochondrial respiration (Fig. 3b). This result suggests that inhibition of ENT1/2 protects against sustained adenosine-induced reduction of mitochondrial respiration, independent of A2AR. The A2BR antagonist, MRS1754, significantly reduced mitochondrial basal respiration, ATP production, maximal respiration, and spare capacity, in the absence of sustained adenosine (Fig. 3b). In the presence of MRS1754, ENT1/2 inhibitors no longer protected against sustained adenosine-induced reduction of mitochondrial respiration (Fig. 3b). These results suggest that the A2BR signaling is essential for maintenance of mitochondrial respiration of lung EC.
AK is important in regulating the intracellular adenosine concentrations by converting adenosine to adenosine 5′-monophosphate (AMP). To explore the possible involvement of AK-dependent signaling in mediating sustained adenosine-induced endothelial injury, we tested the effect of the AK inhibitor, adenosine ITU on sustained adenosine-induced reduction of mitochondrial respiration. We found that ITU was more effective than sustained adenosine as an inhibitor of mitochondrial basal respiration, adenosine triphosphate (ATP) production, maximal respiration, and spare capacity in lung EC (Fig. 3c).
Sustained adenosine exposure impaired mitochondrial quality control system
The abnormal morphology of mitochondria and altered mitochondrial respiration suggest impairment of the mitochondrial quality control system. To test this notion, we examined the markers of mitochondrial fission and fusion, Drp1 and Mfn1. When EC were treated with adenosine and DCF for 30 h, the cells had significantly higher levels of Drp1 expression, with no changes in Mfn1 (Fig. 4a). Densitometry analysis confirmed that the elevation of Drp1 was statistically significant (Fig. 4b). These results suggest that sustained exposure to adenosine in EC promotes mitochondrial fission, leading to the altered mitochondrial morphology. Fragmentated mitochondria are removed by mitophagy. We evaluated the integrity of the mitochondrial quality control system by assessing the markers of mitophagy, Pink1 and Parkin. Sustained adenosine exposure upregulated the expression of Parkin in EC after 30 h of incubation, without changing Pink1 (Fig. 4c). Densitometry analysis further confirmed that the upregulation of Parkin following sustained adenosine treatment was statistically significant. These results indicate that sustained adenosine exposure in EC leads to mitophagy.
Sustained adenosine exposure increased mitochondrial fission and mitophagy. PAECs were treated with vehicle (Ctrl) or 50 µM adenosine plus 50 µM DCF (AD) for 5, 30, and 48 h. The cell lysates were harvested for western blot analysis (Panels a and c). Densitometry analysis was performed with three independent experimental repeats (Panels b and d). Vinculin was used as a protein loading control. The data are presented as mean ± SD of three independent experiments (n = 3). *p < 0.05.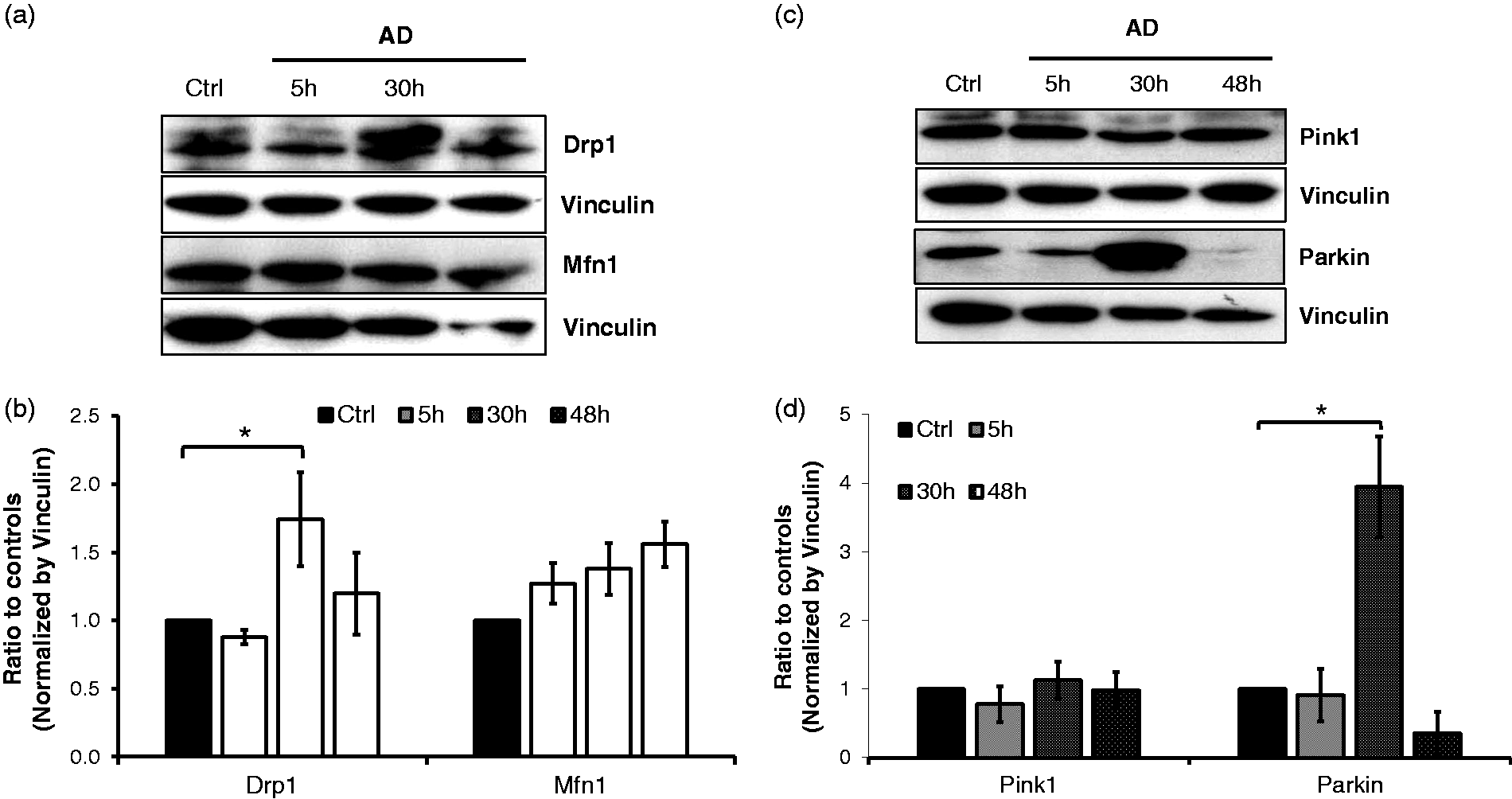
Sustained adenosine exposure increased mitochondrial fission and decreased cell viability via mitochondrial ROS
To examine whether depletion of mitochondrial ROS rescues the damaged morphology of mitochondria induced by sustained adenosine exposure, we utilized mitoTEMPO, a scavenger of mitochondrial ROS. As shown in Fig. 5a, sustained adenosine treatment caused fragmentation of mitochondria as compared to vehicle, confirming that sustained adenosine promoted mitochondrial fission. These morphology data are consistent with increased expression of Drp1 shown by western blot analysis (Fig. 4a). MitoTEMPO attenuated the fragmentation of mitochondria induced by sustained adenosine (Fig. 5a). Thus, sustained adenosine-induced mitochondrial fission and disrupted morphology are mediated through mitochondrial ROS and antagonizing mitochondrial ROS blunts sustained adenosine-associated EC injury.
Sustained adenosine exposure increased mitochondrial fission and promoted cell death via mitochondrial ROS. PAECs were pre-treated with 5 µM MitoTEMPO (MT) for 1 h followed by incubation with 50 µM adenosine and 50 µM DCF (AD) in the presence or absence of 5 µM MitoTEMPO for 30 h or 48 h. The cells were then stained with MitoTracker green to visualize mitochondrial morphology (Panel a), processed for cellular morphology (Panel b), or a MTS cell viability assay (Panel c). Panels a and b are representative images from three independent experiments. In panel c, the data are presented as mean ± SD of three independent experiments (n = 3). *p < 0.05.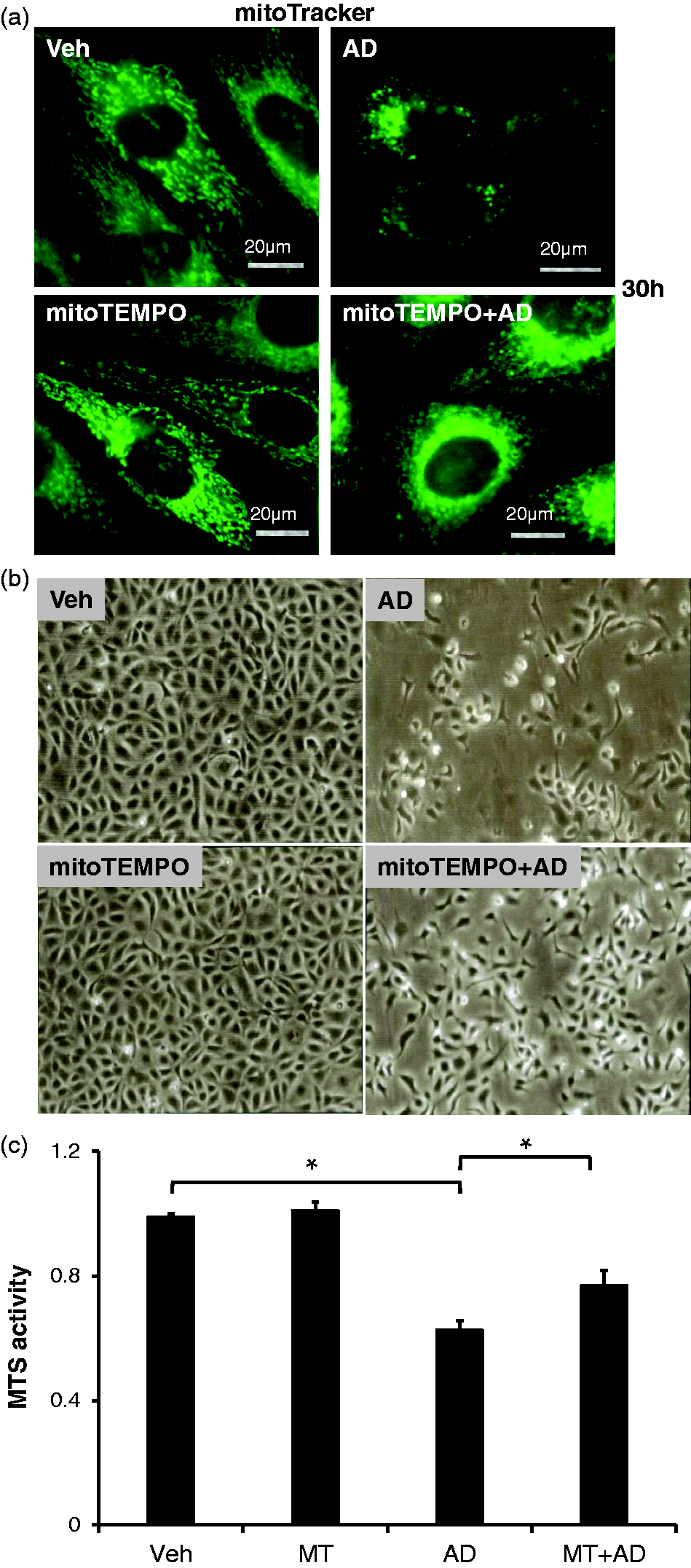
To test the therapeutic effect of targeting mitochondrial ROS, we cultured the ECs with adenosine and DCF in the presence or absence of mitoTEMPO. Our inverted microscopy analysis revealed that the cells treated with AD were less confluent, suggesting that sustained adenosine treatment induced cell death or inhibited cell proliferation (Fig. 5b). MitoTEMPO treatment increased EC confluency (Fig. 5b). We have previously shown that sustained adenosine exposure causes endothelial apoptosis. 32 To test if targeting mitochondrial ROS could attenuate endothelial death induced by sustained adenosine, we performed an MTS assay, which assesses cell viability. The MTS assay revealed that sustained adenosine treatment significantly reduced EC viability, and mitoTEMPO restored viability (Fig. 5c). These results suggest that the cell death induced by sustained adenosine exposure is, at least in part, mediated through mitochondrial ROS and that targeting mitochondrial ROS can be a therapeutic option for vascular injuries caused by sustained adenosine exposure.
Discussion
AR signaling has been studied in multiple lung diseases, such as ALI, IPF, and pulmonary hypertension.
2
However, there is limited understanding of the role of adenosine transport-facilitated events in lung diseases. Our previous studies have demonstrated that sustained adenosine exposure causes EC apoptosis and barrier dysfunction via ENT1/2 uptake into cells.32,35 In this study, we report that sustained adenosine exposure of pulmonary EC causes mitochondrial oxidative stress and promotes mitochondrial fission and dysfunction via an ENT1/2-dependent intracellular adenosine signaling. Fig. 6 summarizes our interpretation of these results.
Proposed model for sustained adenosine-induced endothelial mitochondrial dysfunction. Cellular injuries lead to accumulation of extracellular adenosine. Extracellular adenosine enters endothelial cells via equilibrative nucleoside transporters 1/2 (ENT1/2). Intracellular adenosine can be degraded by adenosine kinase (AK) or adenosine deaminase (ADA) to adenosine 5′-monophosphate (AMP) and inosine, respectively. Adenosine can also react with homocysteine (HC) upon inhibition of S-adenosyl-L-homocysteine (SAH) hydrolase to produce SAH, which is also a product of demethylation of S-adenosyl-L-methionine (SAM) by methyltransferases. SAM is synthesized exclusively in the cytosol and transported into mitochondria to service as a precursor to generate glutathione (GSH). Here, we propose that excessive generation of SAH leads to inhibition of SAM translocation from cytosol into mitochondria. SAM deficiency in mitochondria would impair generation of GSH, leading to increase in mitochondrial reactive oxygen species (mtROS). mtROS may further amplify mitochondrial oxidative stress via p38/JNK-dependent feedback loop. Activation of these pathways ultimately causes mitochondrial imbalance between fission and fusion, leading to mitochondrial dysfunction and endothelial injuries. Solid and dotted lines indicate defined and proposed pathways, respectively.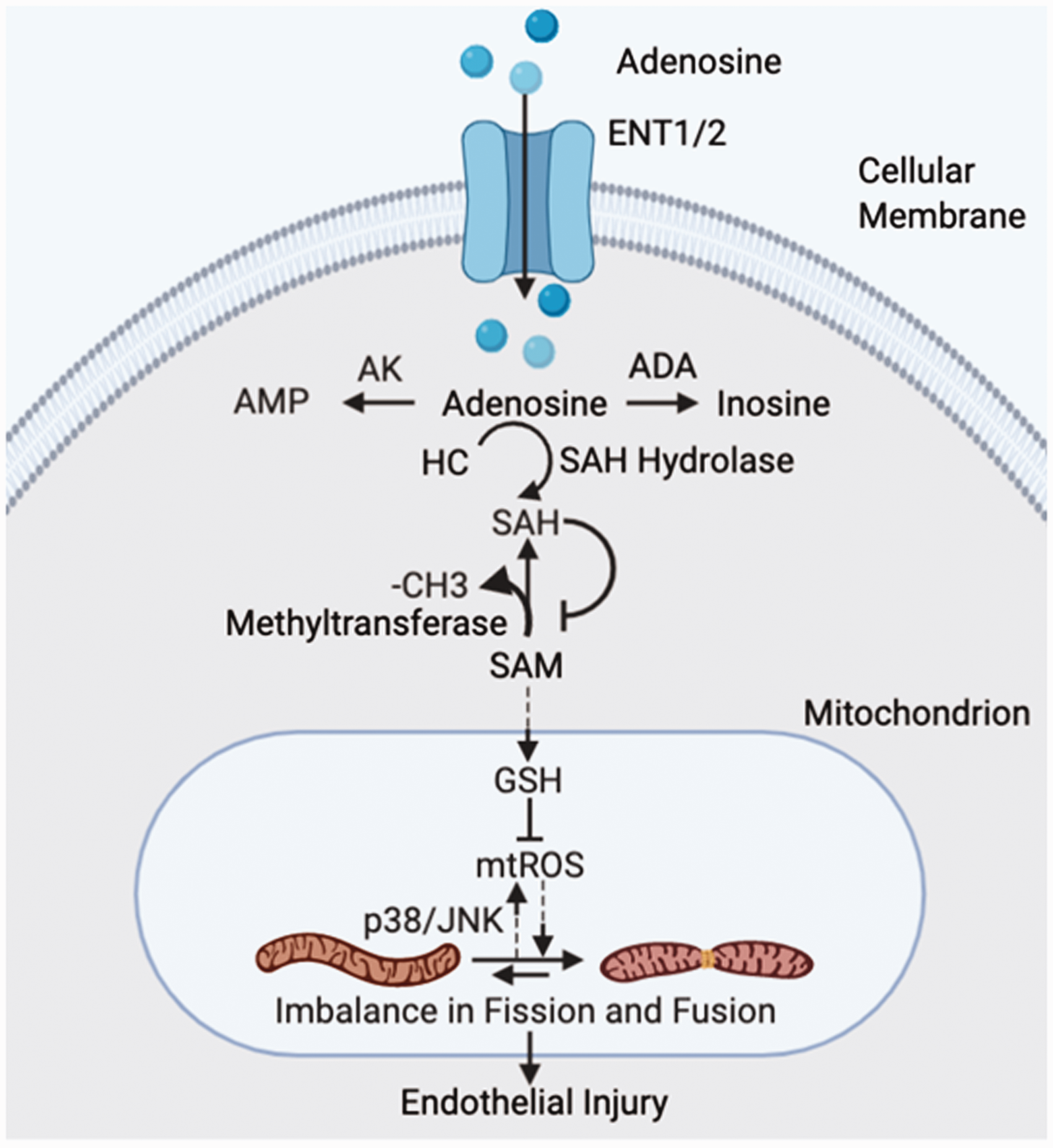
Adenosine is a paradoxical signaling molecule in that adenosine can protect against lung injury,18,55–58 but sustained adenosine exposure can cause lung diseases. 59 These paradoxical effects of adenosine are partially reconciled by previous studies showing that prolonged adenosine exposure causes lung EC apoptosis and barrier dysfunction via ENT1/2-mediated intracellular adenosine uptake,32,35 whereas acute exposure is protective against lung injury via activation of ARs by extracellular adenosine. 18 The current study demonstrates that sustained elevated adenosine increased EC mitochondrial ROS via ENT1/2-mediated intracellular adenosine signaling, but not via AR-dependent pathways. Inhibition of ENT1/2, but not of A2AR or A2BR, prevented sustained adenosine-induced elevation of mitochondrial ROS (Fig. 2a). Mitochondrial oxidative stress may impair mitochondrial respiration. We demonstrated that sustained adenosine exposure significantly diminished mitochondrial basal and maximal respiration, ATP production, and spare capacity; effects that were abolished by inhibitors of ENT1/2 (Fig. 3a). Inhibition of ENTs has been shown to promote A2AR- or A2BR-mediated signaling.52,53 We found that the protective effect of ENT1/2 inhibitors against sustained adenosine-induced suppression of mitochondrial respiration was not affected by an A2AR antagonist (Fig. 3b). This result suggests that the protective effect of ENT1/2 inhibition is independent of A2AR. This result further suggests that sustained adenosine exposure causes mitochondrial oxidative stress and subsequent impairment of mitochondrial respiration via ENT1/2-mediated intracellular adenosine signaling. We noted that the A2BR antagonist, MRS1754, alone significantly reduced basal mitochondrial respiration, ATP production, maximal respiration, and spare respiration of lung EC (Fig. 3b). These data suggest that A2BR signaling may play a role in maintenance of baseline levels of lung endothelial mitochondrial respiration. This concept requires further study.
How sustained adenosine exposure causes mitochondrial oxidative stress is not clear. Intracellular adenosine is eliminated by ADA and AK (see Fig. 6). 60 Inefficient removal by these enzymes results in accumulation of intracellular adenosine, which can be used as a precursor to generate SAH by inhibition of SAH hydrolase (SAHH). 6 SAH is also a product of SAM demethylation and a potent inhibitor of methyltransferases. SAHH activity is inhibited by ADA deficiency and purine compounds. 61 Inhibition of SAHH induces endothelial dysfunction via epigenetic regulation of p66shc-mediated oxidative stress. 62 The results of this study support the possibility that sustained adenosine may increase cellular ROS via intracellular adenosine and subsequent inhibition of SAHH and excessive production of SAH (Fig. 6).
How intracellular adenosine increases mitochondrial ROS remains unknown. We have previously shown that adenosine plus HC increases the ratio of SAH to SAM in lung EC. 63 SAM is synthesized exclusively in the cytosol and then transported into mitochondria via a specific SAM transporter (SAMT). 64 Mitochondrial SAM is essential for maintenance of mitochondrial function due to its critical role as a precursor to glutathione (GSH). 65 SAM has been shown to elevate GSH levels in hepatocytes and prevent alcohol-induced mitochondrial dysfunction and liver injury via attenuation of oxidative stress in rodents.65,66 Whether SAH/SAM ratio inhibits translocation of SAM from cytosol to mitochondria via inhibition of SAMT remains unknown. Since increased SAH/SAM ratio suppresses protein and DNA methylation, it is possible that limitation of SAM translocation into mitochondria would relieve inhibition of protein and DNA methylation. We speculate that cells may use this mechanism of reducing SAM translocation into mitochondria (Fig. 6) to maintain gene transcription and enhance survival.
ADA and AK decrease cellular adenosine (Fig. 6). Since ADA activity is inhibited in our model of sustained adenosine exposure, AK becomes the major mechanism to eliminate intracellular adenosine. AK knockout mice develop neonatal hepatic steatosis, a fatal condition characterized by rapid microvascular fat infiltration, leading to early postnatal death. 12 In this study, we show that inhibition of AK by ITU, which likely increases intracellular adenosine, dramatically reduced mitochondrial respiration (Fig. 3c). These results further support that elevation of intracellular adenosine is detrimental to mitochondrial respiration. However, our data cannot exclude the possibility that inhibition of AK by ITU reduced mitochondrial respiration via depletion of AMP, a source for generation of ATP.
Mitochondrial fission and fusion and mitophagy are essential mechanisms to maintain mitochondrial hemostasis, but can also cause mitochondrial injury.67–69 Dysregulation of these mitochondrial quality control systems has been implicated in many types of human diseases, such as Alzheimer's and Parkinson's Diseases, cardiomyopathy, and cardiac hypertrophy.70–75 Over-production of mitochondrial ROS has been shown to promote mitochondrial fission. 76 Similarly, we found that sustained adenosine exposure causes both mitochondrial oxidative stress and increased mitochondrial fission and mitophagy. We also showed that mitochondrial targeted antioxidant, mitoTEMPO, attenuated sustained adenosine-induced mitochondrial fission and improved cell survival. Our data suggest that mitochondrial oxidative stress and subsequent excessive mitochondrial fission are major mechanisms underlying sustained adenosine-induced endothelial injury (Fig. 6). How mitochondrial oxidative stress promotes mitochondrial fission is unknown. Our previous studies32,35 have shown that sustained adenosine exposure activated both p38 and JNK at 24 h and 48 h; effects that were completely prevented by the ENT1/2 inhibitors, DPM and NBTI. We have also found that both p38 and JNK were activated by sustained adenosine in mitochondria and that inhibition of p38 and JNK prevented sustained adenosine-induced mitochondrial oxidative stress, mitochondrial defects, and increased endothelial permeability and apoptosis.32,35 These results suggest an important role of p38 and JNK signaling in sustained adenosine-induced ENT1/2-dependent endothelial injury. Oxidative stress can activate p38 and JNK. 48 In this study, we also found that inhibition of p38 or JNK attenuated sustained adenosine-induced mitochondrial ROS production. Future studies will address whether p38 and JNK are involved in sustained adenosine-induced mitochondrial fission via a positive feedback loop (Fig. 6).
This study has some limitations. Main pulmonary artery EC were used for experiments. Thus, the results may not reflect effects of sustained adenosine on other lung cells, such as microvascular endothelial, epithelial, inflammatory, or matrix cells. In addition, the results were not confirmed in human lung tissues. More biologic replicates could be used to strengthen the statistical significance of the findings.
In summary, this study shows that sustained adenosine exposure increases mitochondrial fission and impairs mitochondrial respiration through an ENT1/2-facilitated elevation of mitochondrial ROS and p38/JNK-dependent pathways in lung EC (Fig. 6). Our data also suggest that inhibition of ENT1/2 and mitochondria-targeted antioxidant may be potential therapeutic options for lung diseases associated with increased adenosine.
Footnotes
Acknowledgements
We thank the Providence VA Medical Center for the provision of facilities and institutional support.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
Funding for this work was derived from a predoctoral fellowship (18PRE33960076) from American Heart Association (J.K.), NHLBI HL130230 (Q.L.), NIGMS P20 GM103652 (S.R. and Project 1 to Q.L.), and VA Merit Review (S.R.).
