Abstract
Pharmacokinetic data for riociguat in patients with chronic thromboembolic pulmonary hypertension (CTEPH) have previously been reported from randomized clinical trials, which may not fully reflect the population encountered in routine practice. The aim of the current study was to characterize the pharmacokinetic of riociguat and its metabolite M1 in the patients from routine clinical practice. A population pharmacokinetic model was developed in NONMEM 7.3, based on riociguat and its metabolite plasma concentrations from 49 patients with CTEPH. One sample with riociguat and M1 concentrations was available from each patient obtained at different time points after last dose. Age, bodyweight, sex, smoking status, concomitant medications, kidney and liver function markers were tested as potential covariates of pharmacokinetic of riociguat and its metabolite. Riociguat and M1 disposition was best described with one-compartment models. Apparent volume of distribution (Vd/F) for riociguat and M1 were assumed to be the same. Total bilirubin and creatinine clearance were the most predictive covariates for apparent riociguat metabolic clearance to M1 (CLf,M1/F) and for apparent riociguat clearance through remaining pathways (CLe,r/F), respectively. CLf,M1/F, CLe,r/F, Vd/F of riociguat and M1, and clearance of M1 (CLe,M1/F) for a typical individual with 70 mL/min creatinine clearance and 0.69 mg/dL total bilirubin were 0.665 L/h (relative standard error = 17%)), 0.66 (18%) L/h, 3.63 (15%) L and 1.47 (19%) L/h, respectively. Upon visual identification of six outlying individuals, an absorption lag-time of 2.95 (6%) h was estimated for these patients. In conclusion, the only clinical characteristics related to riociguat exposure in patients with CTEPH from routine clinical practice are total bilirubin and creatinine clearance. This confirms the findings of the previous population pharmacokinetic studies based on data from randomized clinical trials.
Chronic thromboembolic pulmonary hypertension (CTEPH) is a pulmonary vascular disease caused by the chronic thrombotic obstruction of pulmonary arteries and peripheral vascular remodeling. 1 The disease is characterized by elevation of pulmonary artery mean pressure (mPAP) ≥ 25 mmHg and increase of pulmonary vascular resistance (PVR), and ultimately results in death due to the right ventricular failure. 2
Most CTEPH patients are successfully treated with surgical pulmonary endarterectomy (PEA), which is the gold standard in management of CTEPH. However, up to 40% of patients with CTEPH are considered technically inoperable due to distal lesions that are not surgically accessible or due to comorbidities, and 20–30% of patients develop persistent/recurrent pulmonary hypertension after PEA. 3 Both patient groups are potential candidates for the treatment with riociguat – a novel medication that relaxes vascular smooth muscle by stimulation of soluble guanylate cyclase (sGC). Riociguat displays a dual mode of action: it stimulates sGC independently of nitric oxide (NO) and increases the sensitivity of sGC to NO, resulting in increased cyclic guanosine monophosphate (cGMP) levels.4,5
The main biotransformation pathway for riociguat is N-demethylation by cytochrome P450 enzymes, most importantly CYP1A1. 6 Desmethylriociguat, the major circulating active metabolite M1, which exhibits 1/10th to 1/3rd of the pharmacological activity of riociguat, is further metabolized to the pharmacologically inactive N-glucuronide. 7 The drug is eliminated in the urine (33–45%) and feces (48–59%). 7
Pharmacokinetic (PK) data for riociguat have previously been reported from the randomized clinical trials (RCT) CHEST-1, 2 and PATENT-1, 2 in CTEPH and pulmonary arterial hypertension (PAH) patients, respectively.8–10 RCTs are conducted in tightly controlled settings and include patients who meet stringent inclusion and exclusion criteria and may therefore not accurately reflect the population treated in clinical practice.11,12 Therefore, the aim of the current study was to characterize the PK of riociguat and its metabolite desmethylriociguat (M1) in patients with inoperable CTEPH or persistent/recurrent pulmonary hypertension after PEA from routine clinical practice and to identify and quantify significant covariates associated with riociguat and M1 exposure in these patients.
Methods
Research design
This observational PK study was conducted at the Second Department of Internal Medicine, General University Hospital and First Faculty of Medicine, Charles University in Prague, Czech Republic. It was conducted in accordance with the principles laid down in the 18th World Medical Assembly (Helsinki, 1964), including all subsequent amendments, and in compliance with all laws and regulations of the Czech Republic. The approval of retrospective data collection was provided by ethics committee of the General University Hospital in Prague (ID 1208/18 S-IV). Written informed consent was obtained from all participants.
Patients were included in the study if they were diagnosed with inoperable CTEPH or persistent/recurrent pulmonary hypertension after PEA and received a stable riociguat dose for at least three months before the enrolment. Inoperability status was previously assessed by an interdisciplinary CTEPH team, consisting of a pulmonary hypertension specialist, a PEA surgeon, an anesthesiologist, and a radiologist. Persistent/recurrent pulmonary hypertension was diagnosed invasively by the right heart catheterization at least six months after PEA and defined as persistent elevation of mPAP ≥ 25 mmHg and PVR > 3 Wood unit.
The following data were collected from the outpatient check-up visit: time of the last dose and sampling, demography, medical history, concomitant medications, vital signs, functional capacity, and 6-minute walking distance (6MWD). Blood samples were collected for determination of N-terminal pro b-type natriuretic peptide (NT-proBNP), laboratory biochemical parameters, and riociguat and M1 concentrations. Retrospective data from the last right heart catheterization performed before initiation of riociguat treatment were used for the description of hemodynamics.
Riociguat dosing
Riociguat was prescribed according to Adempas® Summary of Product Characteristics (SmPC) and 2015 European Respiratory Society/European Society of Cardiology (ERS/ESC) treatment guidelines including required initial dose adjustments. 1 Riociguat was adjusted from a starting dose of 1 mg three times daily according to systolic systemic arterial pressure and signs or symptoms of hypotension (final range: from 1.5 mg to 2.5 mg three times daily).
Bioanalytical assay
After collection, blood samples were allowed to clot for 30 min at room temperature, and serum was separated by centrifugation (1500 × g, 15 min, 4℃) and stored frozen at –80℃ until analysis. Riociguat and M1 serum concentrations were measured using liquid chromatography with tandem mass spectrometric detection in positive ESI mode (LC-MS/MS); penta-deuterated perampanel was used as internal standard (IS). Internal standard solution (10 µL, 5000 ng mL−1 in methanol) and 300 µL acetonitrile were added to 100 µL of the serum sample in a 1.5 mL Eppendorf tube. The solution was vortexed for 30 s and centrifuged (9600 × g, 3 min) and the supernatant (100 µL) was transferred to an autosampler vial. The method was developed using Nexera X2 Shimadzu HPLC (Nakagyo-ku, Kyoto, Japan) coupled with AB Sciex QTRAP 5500 (MA, USA). Mobile phase A consisted of 0.1% formic acid in water and mobile phase B consisted of acetonitrile. The analysis was performed on a Zorbax Eclipse XDB-C18 column (1.8 µm, 50 × 4.6 mm). The initial LC conditions had a flow rate of 0.5 mL min−1 at a mobile phase composition of 50:50 (A: B). At 30 s the mobile phase composition was ramped to 10:90 (A: B) within 60 s and held for 90 s and then returned to initial LC conditions. Quantitation was done using multiple reaction monitoring (MRM) mode to monitor protonated precursor → product ion transition of m/z 423.022 → 109.100 for riociguat, 409.027 → 109.000 for M1 and 355.029 → 220.000 for penta-deuterated perampanel. Method performance was evaluated for riociguat and M1 following the recommendations of the Scientific Working Group for Forensic Toxicology. 13 The test range of the assay was 5–1000 µg/L. Coefficient of variation of intra-assay was less than 11%.
Population PK analysis
The data analysis was performed using NONMEM version 7.3.0 (ICON Development Solutions, Ellicott City, MD) and PsN v3.4.2 both running under Pirana 2.9.0. The first-order estimation algorithm with interaction (FOCE-I) was used. R 3.3.2 was used for the visualization of the data and model diagnostics.
Model development was performed in three steps:
(1)
Based on visual inspection of raw data, six patients had unexpectedly high riociguat concentrations at relatively late times after their last dose (Figure 1). These patients’ data were initially excluded from model development. After the covariate analysis, these patients were reintroduced and analyzed together with the other individuals.
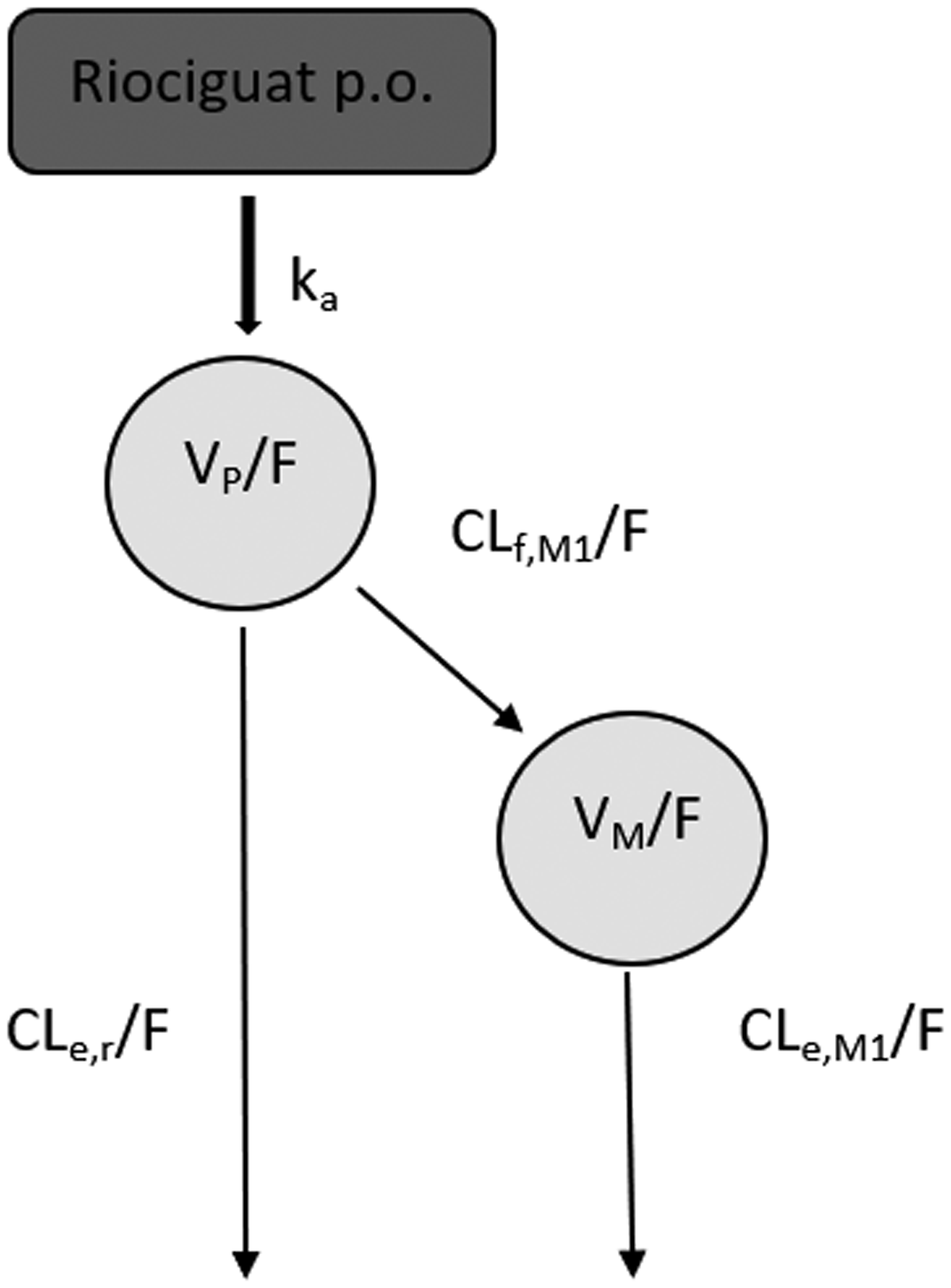
Serum concentrations plotted against time after the last dose. ID numbers from observations from individuals with outlying riociguat concentrations are indicated. (a) Riociguat; (b) Desmethylriociguat. Schematic representation of the pharmacokinetic model for riociguat and its metabolite in patients with chronic thromboembolic pulmonary hypertension. Pharmacokinetic parameters represent apparent values. Abbreviations: ka/F = apparent absorption rate constant, V/F = apparent distribution volume of the designated compartment (p = parent – riociguat, m = metabolite), CLf,M1/F = apparent riociguat metabolic formation clearance to M1, CLe,r/F = apparent riociguat clearance remaining after accounting for M1 formation, CLe,M1/F = apparent clearance of M1, F = bioavailability.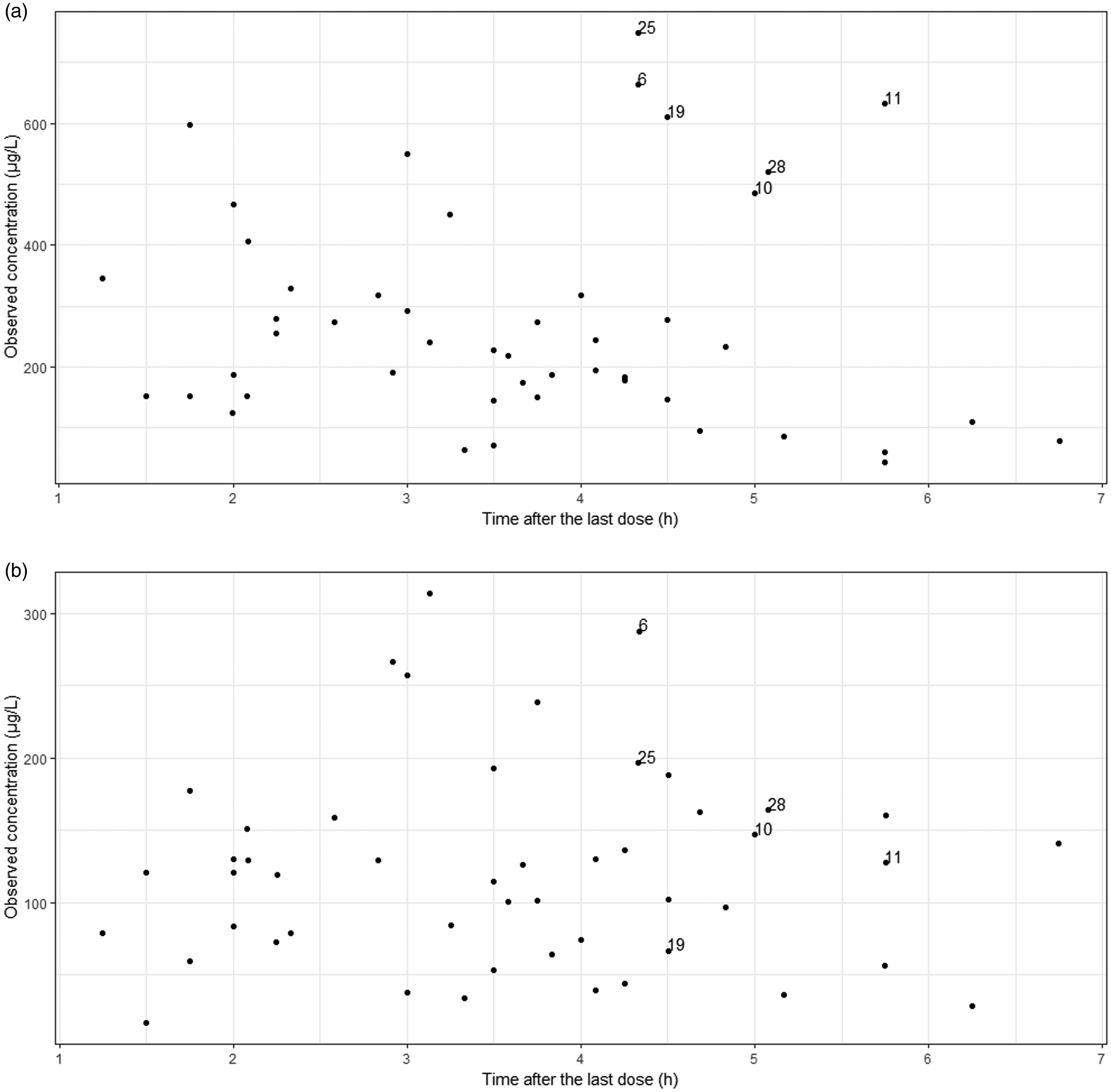
For model selection, a decrease in objective function of more than 6.63 points between nested models (p < 0.01) was considered statistically significant, assuming a χ2-distribution. Additional criteria for model selection were relative standard error (RSE) of the estimates of structural model parameters <50%, condition number calculated by dividing the largest and smallest eigenvalue from the model fit of <1000, physiological plausibility of the obtained parameter values, and absence of bias in goodness-of-fit (GOF) plots.
(2)
After the final covariate model was developed, the patients with outlying observations were reintroduced into the analysis. As previous reports suggested that food delays riociguat absorption for 3 h,7,18 it was investigated whether riociguat absorption in these patients was delayed. For this, both different ka values and a delayed onset of absorption as characterized by a lag-time (Tlag) were tested based on the criteria defined above. When it was observed that this procedure did not yield large differences in the obtained parameter values, the results from the fit which included all individuals were retained.
(3)
The predictive properties of the structural and statistical model were evaluated using normalized prediction distribution errors (NPDEs), a simulation-based diagnostics. For this, the dataset was simulated 500 times, after which the observed concentrations were compared to the range of simulated values using the NPDE package developed for R. 19
Results
Study population and data
Clinical characteristics of the study population.
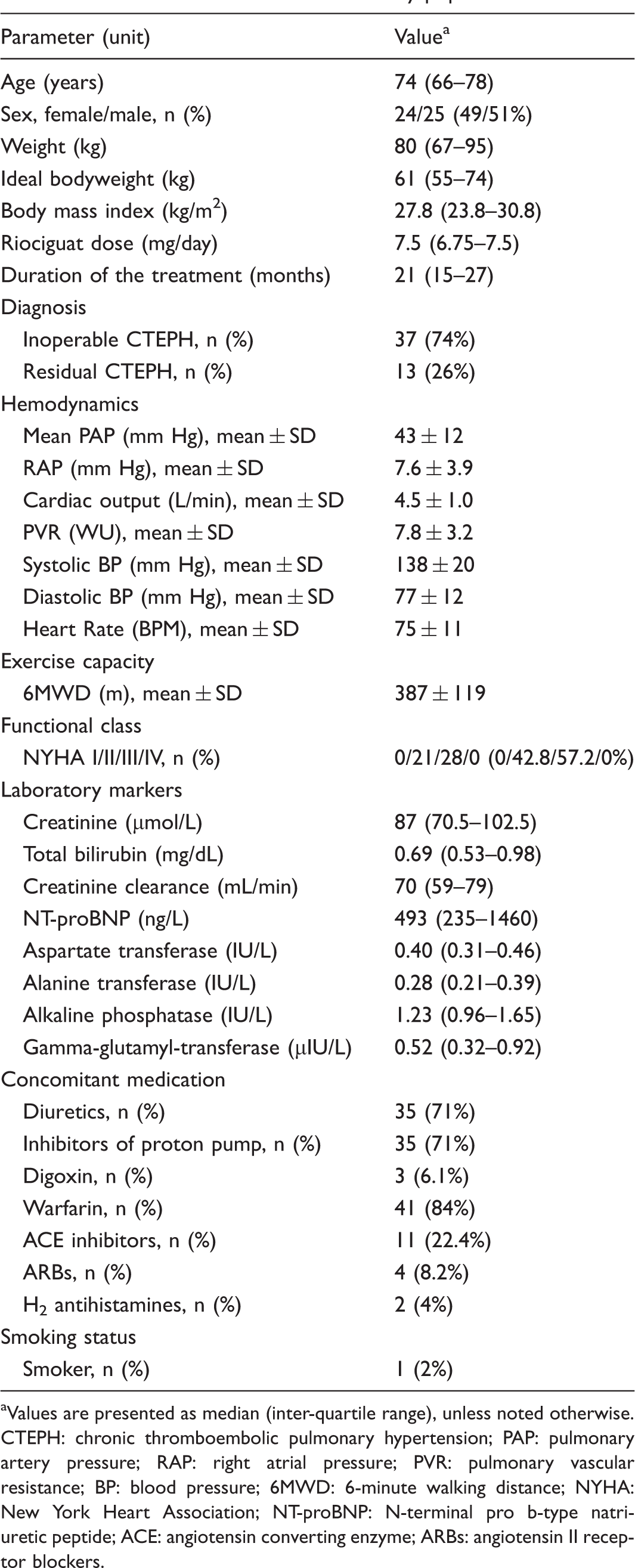
Values are presented as median (inter-quartile range), unless noted otherwise.
CTEPH: chronic thromboembolic pulmonary hypertension; PAP: pulmonary artery pressure; RAP: right atrial pressure; PVR: pulmonary vascular resistance; BP: blood pressure; 6MWD: 6-minute walking distance; NYHA: New York Heart Association; NT-proBNP: N-terminal pro b-type natriuretic peptide; ACE: angiotensin converting enzyme; ARBs: angiotensin II receptor blockers.
Population PK analysis
Observed riociguat and M1 serum concentrations were best described with one-compartment models and the same distribution volume was estimated for both compounds to achieve mathematical identifiability. Proportional residual error models provided the best description of the residual variability for both riociguat and M1 concentrations.
Figure 2 depicts a schematic representation of the obtained PK model. Creatinine clearance in a linear equation was found to be the most predictive covariate for CLe,r/F, was found to increase 0.009 L/h per unit (mL/min) creatinine clearance, as depicted in Figure 3(a). For CLf,M1/F, the most predictive covariate relationship was total bilirubin in an exponential equation with an estimated exponent of −0.463 (20%), as shown in Figure 3(b). After the inclusion of these covariate relationships, no other statistically significant covariates could be identified. CLf,M1/F, CLe,r/F, Vd/F of riociguat and M1, and CLe,M1/F for a typical individual of creatinine clearance (70 mL/min) and total bilirubin level (0.69 mg/dL) were 0.665 L/h (17%)), 0.66 (18%) L/h, 3.63 L (15%) and 1.47 (19%) L/h, respectively. Additionally, the analysis showed that the riociguat absorption in the six patients exhibiting high concentrations was delayed with a lag-time of 2.95 h (6%). The parameter values obtained in the final model fit as well as the median parameter values obtained in the bootstrap procedure are presented in Table 2.
Relationships between (a) total bilirubin and apparent riociguat metabolic formation clearance to M1 and (b) creatinine clearance and apparent riociguat clearance remaining after accounting for M1 formation in patients with CTEPH. Parameter estimates of the final model and their corresponding bootstrap estimates. Pharmacokinetic parameters represent apparent values. ka/F: apparent absorption rate constant; CL: apparent clearance of the designated pathway (see Figure 2 for the explanation of the symbols); CREACL: creatinine clearance in mL/min; BILTOT: total bilirubin level in mg/dL; θBILTOT: exponent for the covariate relationships between total bilirubin levels and CLf,M1/F; VP/F: apparent volume of distribution of the parent compound; VM/F: apparent volume of distribution of the M1 metabolite; TV: typical value of a parameter; Tlag: lag-time.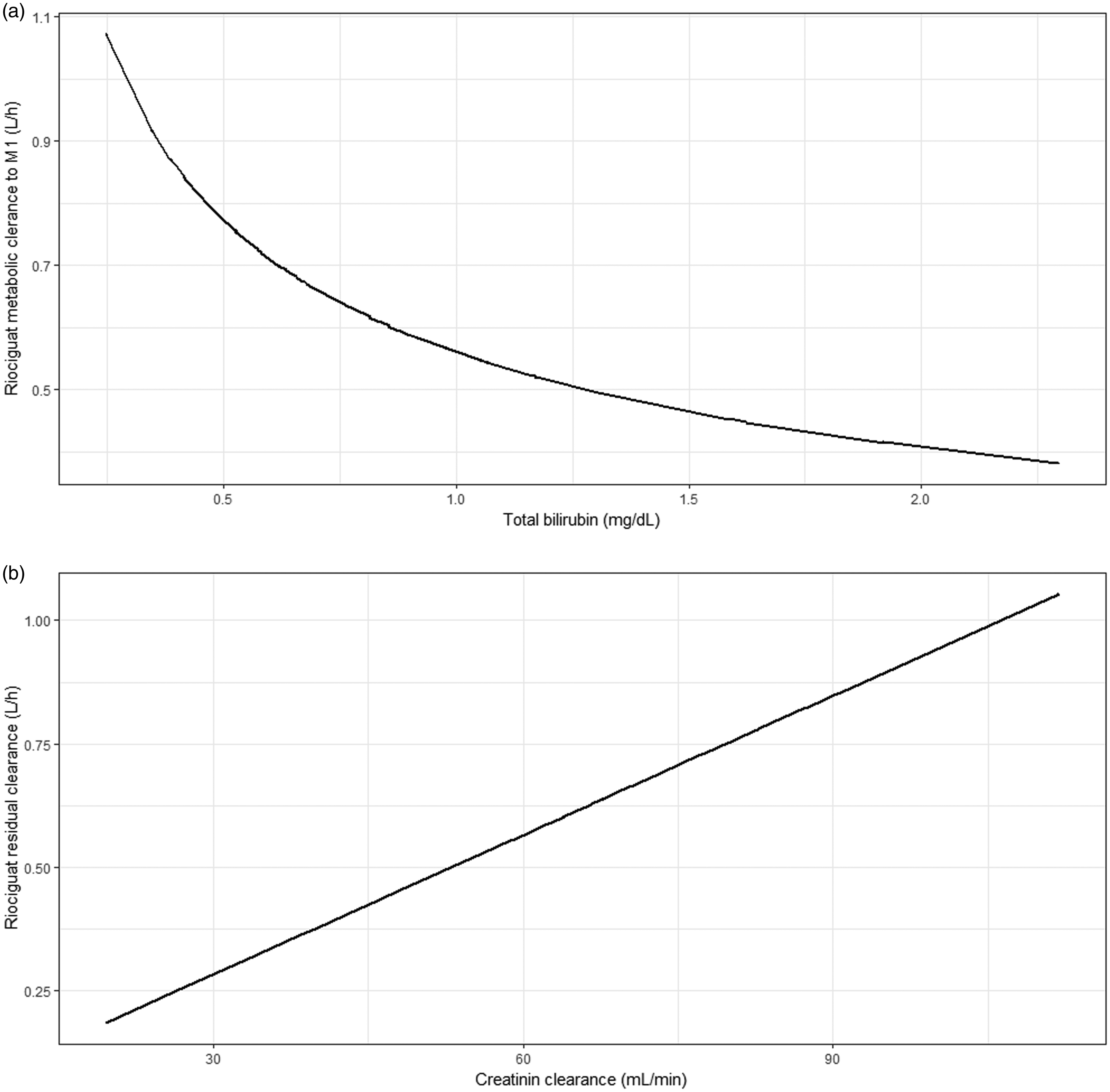
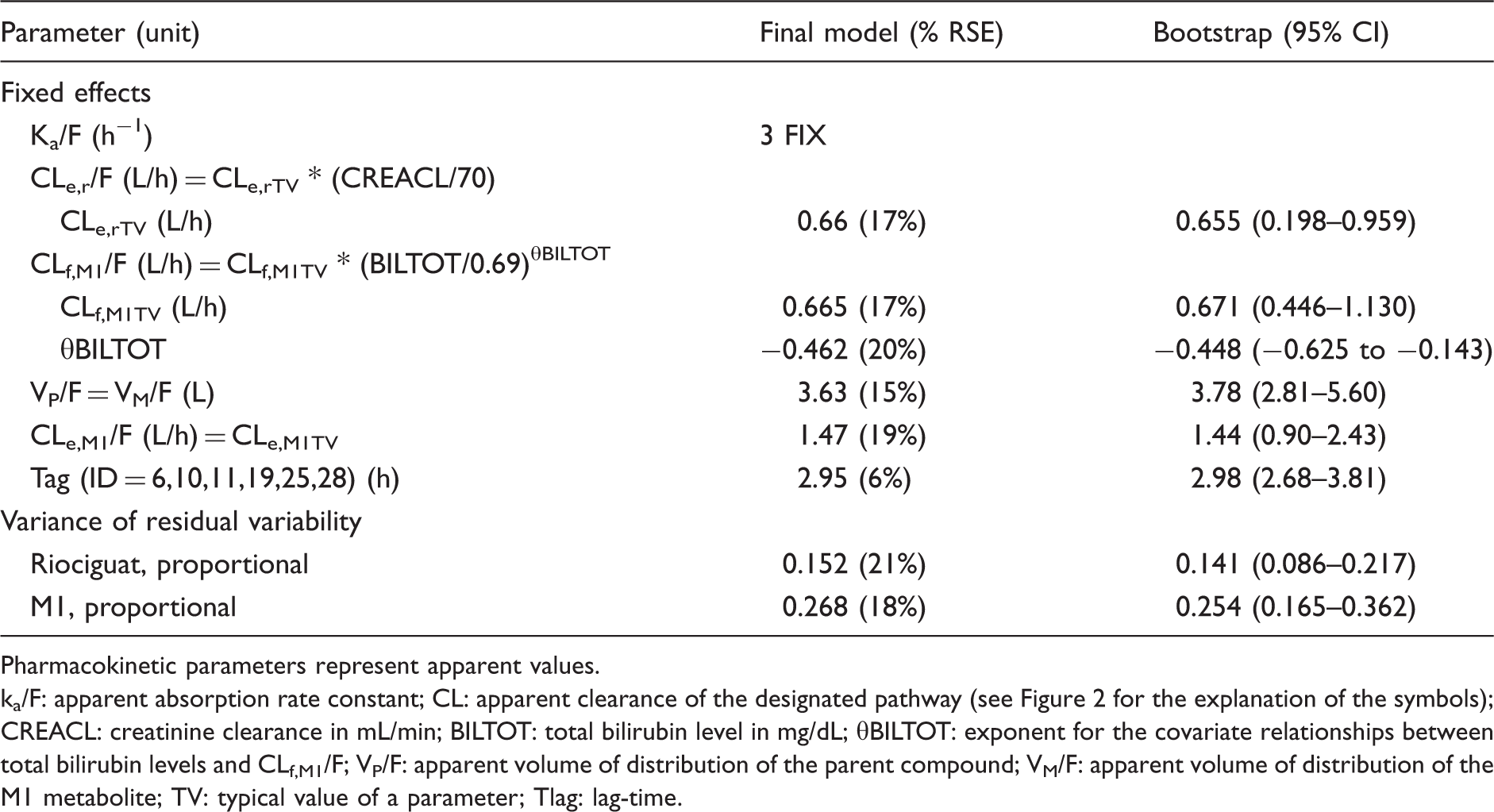
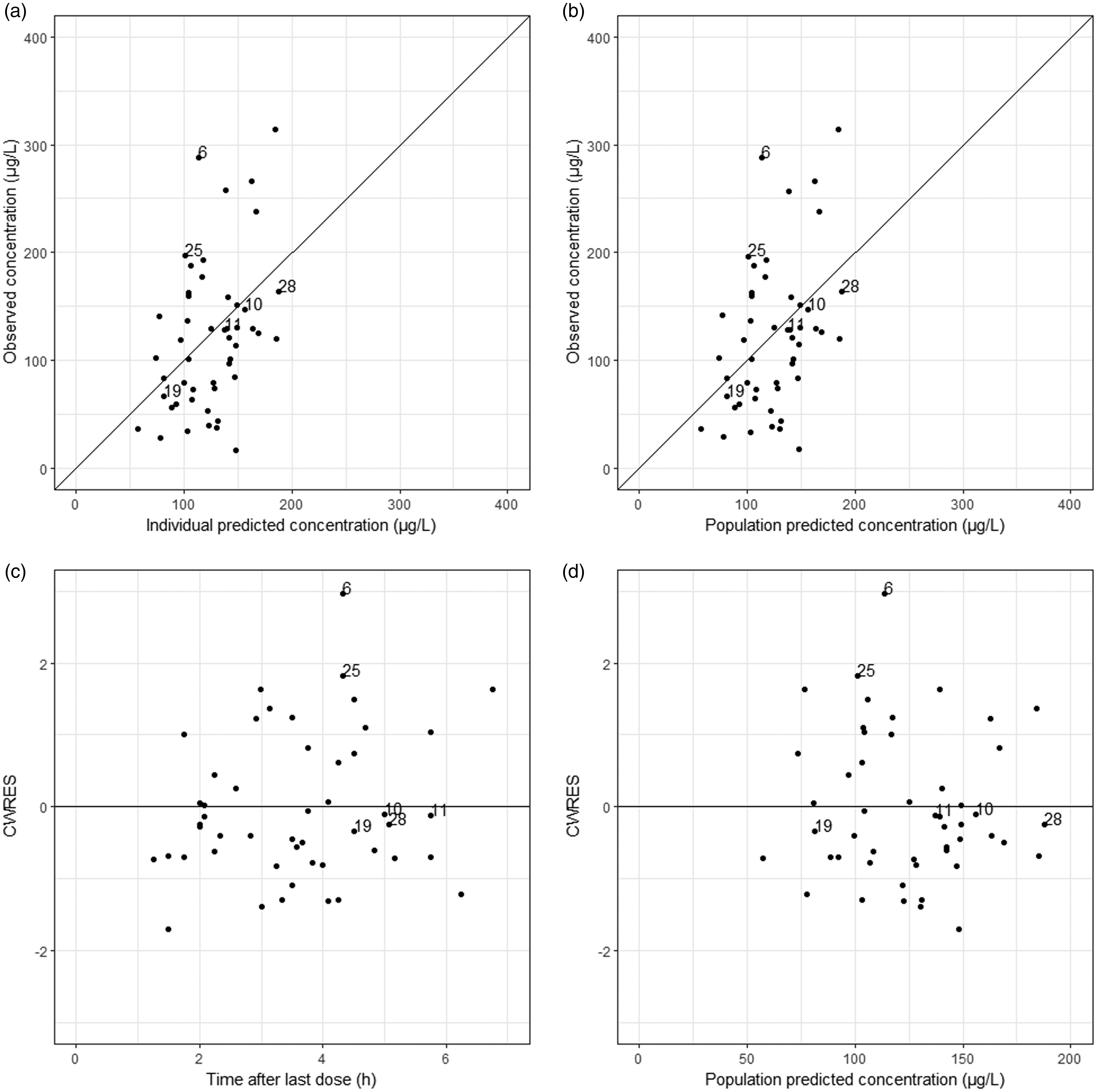
RSE values for the structural parameters were all below 30% in the final model, indicating good precision of the estimated parameters. All median parameter values in the bootstrap procedure were within 10% of the values obtained in the final model fit indicating that the model is robust. Figures 4 and 5 present the GOF plots for riociguat and M1. Absence of bias in these plots indicates that the final model describes the observed data accurately. Finally, there was no bias in NPDE, neither over time nor over concentration range, indicating that the predictive properties of this model are also accurate (Supplementary Figures 1S and 2S).
Goodness of fits (GOF) plots for the final model for riociguat pharmacokinetics in patients with chronic thromboembolic pulmonary hypertension. (a) Population predicted concentration vs. observed concentration. (b) Individual predicted concentration vs. observed concentration. (c) conditional weighted residuals (CWRES) vs. time after the last dose. (d) Conditional weighted residuals (CWRES) vs. population predicted concentration. ID numbers from observations from individuals with outlying riociguat concentrations are indicated. Goodness of fits (GOF) plots for the final model for M1 pharmacokinetics in patients with chronic thromboembolic pulmonary hypertension. (a) Population predicted concentration vs. observed concentration. (b) Individual predicted concentration vs. observed concentration. (c) conditional weighted residuals (CWRES) vs. time after the last dose. (d) Conditional weighted residuals (CWRES) vs. population predicted concentration. ID numbers from observations from individuals with outlying riociguat concentrations are indicated.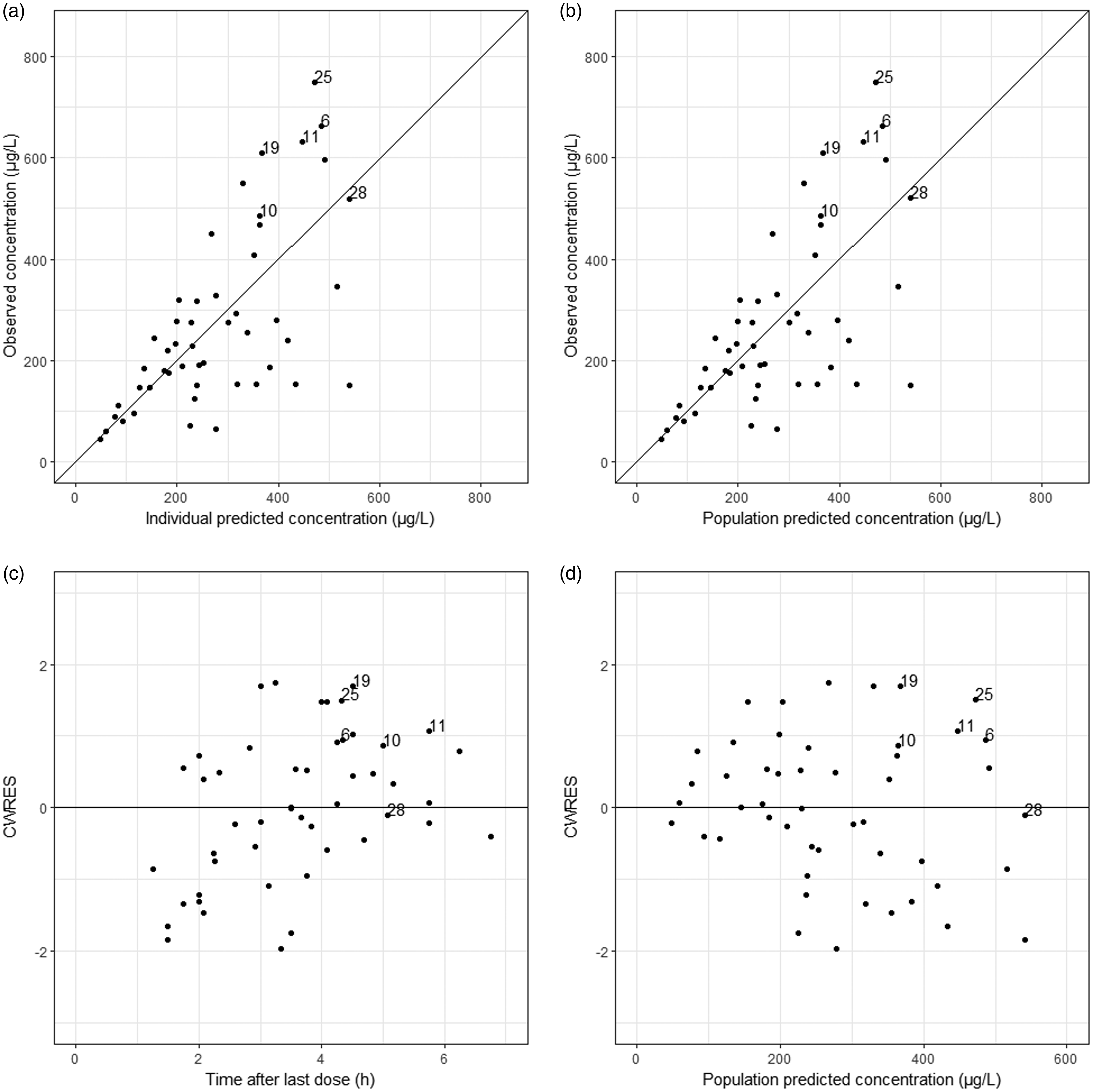
Discussion
This study used a population modelling approach to describe the PK of riociguat and its metabolite M1 in patients with CTEPH from routine clinical practice. The results of the study showed that riociguat metabolic clearance to M1 depends on the liver function, as characterized by the total bilirubin level. Creatinine clearance, a marker for kidney function, was found to be a predictive covariate for riociguat clearance remaining after accounting for M1 formation. These results indicate that impaired renal and hepatic function leads to reduced riociguat clearance and increased riociguat exposure.
Previous PK studies analyzed data obtained during RCTs, which are constrained only to patients that meet strict inclusion and exclusion criteria and may therefore not accurately reflect the population treated in the routine practice.11,12 The current analysis included data collected in everyday practice and may therefore provide more relevant clinical information. However, this type of data are usually not as “clean” as those from RCTs. In this study for example, six patients had unexpectedly high riociguat concentrations at the time of the observation that could not be explained (Figure 1(a)). Because of the inclusion of one sample per patient in the analysis, it was not possible to distinguish whether these patients or the particular observations were outliers. As previous reports however suggested that food might delay riociguat absorption for 3 h, 18 we investigated whether the observations could be explained by delayed riociguat absorption in these patients as characterized by a Tlag, or slowed down, as characterized by a different ka/F value. The analysis showed that data from these patients were best described with a delayed riociguat absorption with a Tlag of 2.95 h, which might be due to food impact, but the design of our study does not allow for conclusions to be drawn on this matter. An important clinical question is whether this delay in absorption would result in different riociguat exposure. If these patients indeed had a delayed riociguat absorption due to food, then changes in area under curve (AUC) would not be expected. However, if there is another reason for these concentrations to be so high, AUC could in fact be impacted. To make a final conclusion, we would need to have multiple observations per patient.
Although only one sample per patient was included in the analysis, samples were obtained within a wide range of time points after the last dose (Figure 1), which allowed us to develop a structural model and identify the predictive covariates for riociguat and M1 PK. PK parameter values could be obtained with acceptable precision, as reflected in low RSE (<30%) of the parameter estimates. In addition, extensive model validation showed that the model not only described the obtained data well (Figures 4 and 5), but also predicted the data well (Supplementary Figures 1S and 2S), meaning that the conclusions regarding parameter values and covariate effects in this model are well supported by the data.
An important advantage of the current analysis is the wide range of sampling time points after the last dose of riociguat in CTEPH patients, which covers a wide range than the previous multiple-dose population PK study, which included only trough samples from the PAH and CTEPH patients and additionally samples obtained 2–3 h after the first and second dose of drug only. 9
Overview of population pharmacokinetic studies evaluating pharmacokinetic parameters of riociguat and its metabolite.
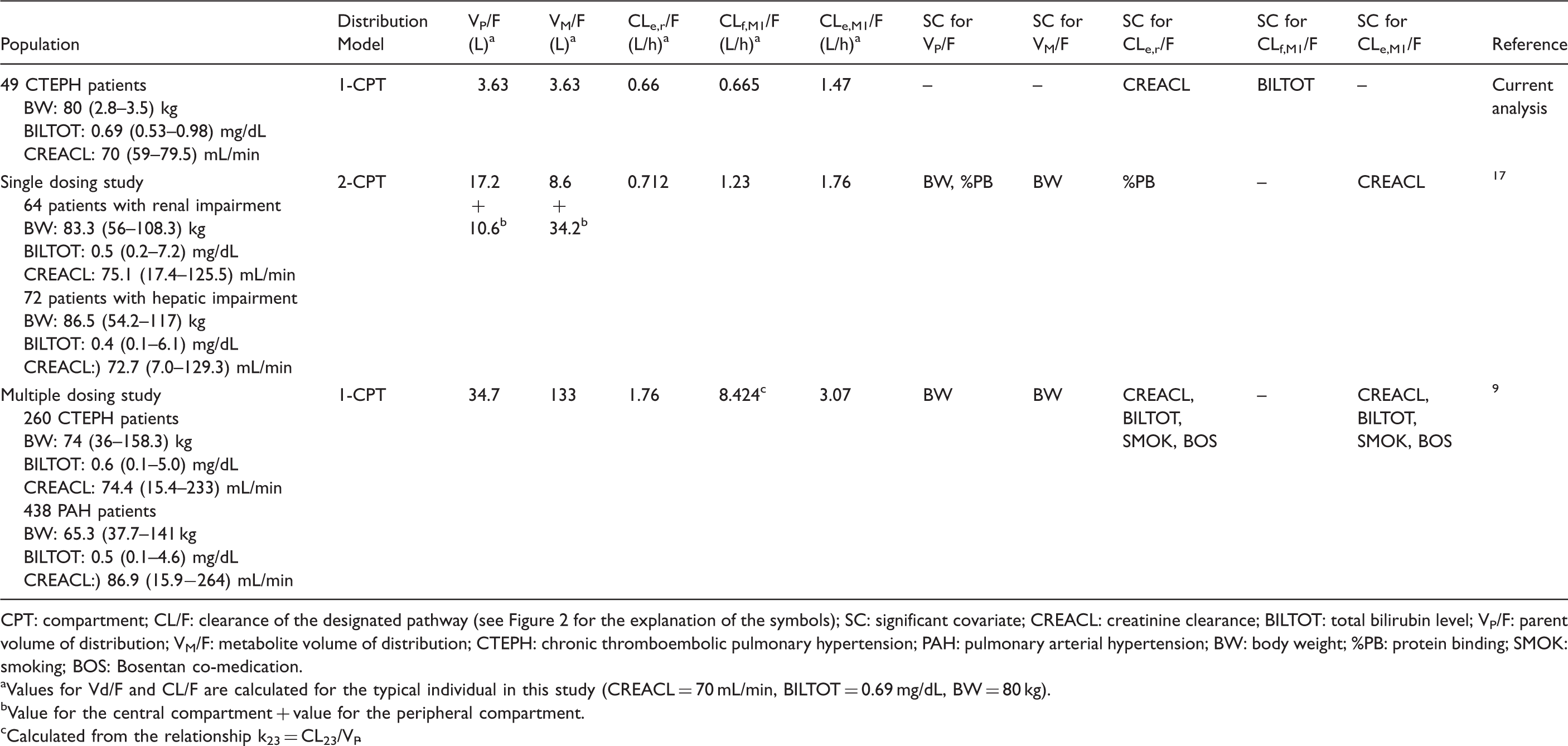
CPT: compartment; CL/F: clearance of the designated pathway (see Figure 2 for the explanation of the symbols); SC: significant covariate; CREACL: creatinine clearance; BILTOT: total bilirubin level; VP/F: parent volume of distribution; VM/F: metabolite volume of distribution; CTEPH: chronic thromboembolic pulmonary hypertension; PAH: pulmonary arterial hypertension; BW: body weight; %PB: protein binding; SMOK: smoking; BOS: Bosentan co-medication.
Values for Vd/F and CL/F are calculated for the typical individual in this study (CREACL = 70 mL/min, BILTOT = 0.69 mg/dL, BW = 80 kg).
Value for the central compartment + value for the peripheral compartment.
Calculated from the relationship k23 = CL23/VP.
It is important to note that not all PK parameters for M1 could be estimated without making assumptions. As data obtained after intravenous administration of the M1 metabolite, or data on the recovery of M1 in urine were not available, the value of Vd/F for M1 was assumed to be the same as the parent compound. The model validation confirmed that the model can accurately describe and predict the concentrations of riociguat and M1, but as a result of the assumption the absolute values of the parameters related to the metabolite should be considered in the context of the assumptions made in the current analysis. The conclusions regarding the impact of the covariates are not impacted by the assumptions.
In conclusion, we report on the PK of riociguat and its pharmacologically active metabolite desmethylriociguat in a cohort of CTEPH patients encountered in routine clinical practice. Our study confirms the findings from previous population PK studies based on data from RCTs, that the only clinical characteristics related to riociguat exposure in patients with CTEPH are total bilirubin levels and creatinine clearance.
Supplemental Material
PUL898031 Supplemental material - Supplemental material for Population pharmacokinetics of riociguat and its metabolite in patients with chronic thromboembolic pulmonary hypertension from routine clinical practice
Supplemental material, PUL898031 Supplemental material for Population pharmacokinetics of riociguat and its metabolite in patients with chronic thromboembolic pulmonary hypertension from routine clinical practice by Danica Michaličková, Pavel Jansa, Miroslava Bursová, Tomáš Hložek, Radomír Čabala, Jan Miroslav Hartinger, David Ambrož, Michael Aschermann, Jaroslav Lindner, Aleš Linhart, Ondřej Slanař and Elke H.J. Krekels in Pulmonary Circulation
Footnotes
Acknowledgments
The authors would like to thank all the patients, nurses and physicians who were part of this study. The authors also thank Dr. Parth Upadhyay for code review.
Funding
This study was supported by the Charles University projects Progres Q25 and Q38 and by the project “International Mobility of Researchers at Charles University” CZ.02.2.69/0.0/0.0/16_027/0008495.
Availability of data and materials
Ethical approval
The approval of retrospective data collection was provided by ethics committee of the General University Hospital in Prague (ID 1208/18 S-IV). Written informed consent was obtained from all participants.
Guarantor
Not applicable.
Contributorship
D.M. analyzed the data and wrote the manuscript; P.J. conceived and designed the study, performed the clinical trial, and wrote the manuscript; M.A., J.L. and A.L. performed the clinical trial; O.S. wrote the manuscript; M.B., T.H., R.Č., and D.A. developed the analytical method and performed laboratory analyses; J.M.H. wrote the manuscript; E.H.J.K. supervised the data analysis and wrote the manuscript.
Conflict of interest
P.J. has received fees and grants from Actelion Pharmaceuticals Ltd, AOP Orphan, and MSD.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
