Abstract
Early identification of the causes of cholestasis is important for appropriate management of patients with hyperthyroidism. We report a patient who had hyperthyroidism and severe cholestasis after methimazole (MMI) treatment. The patient was diagnosed as having MMI-induced cholestatic hepatitis. Treatment with MMI was stopped at admission to hospital. However, his serum total bilirubin (TBil) level rose from 410.5 µmol/L to 519.9 µmol/L and prothrombin time activity (PTA) dropped from 81.0% to 52.2% in 10 days. To prevent further deterioration of his liver function, plasma exchange was performed three times, and dexamethasone (10 mg, intravenously) was used each time. His PTA rose to 101% and his TBil continued to increase to 669.8 µmol/L after plasma exchange. He was subsequently diagnosed as having thyrotoxicosis-induced cholestasis and treated with radioactive iodine (380 MBq) 2 weeks after admission. His hyperthyroidism was significantly relieved, but the TBil level further increased to 776.8 µmol/L. Three weeks after admission, oral prednisone (30 mg/day) was used in this patient. Subsequently, his TBil levels gradually decreased and his liver function almost normalized within 3 months. We discuss the literature on cholestasis in the context of hyperthyroidism.
Keywords
Introduction
Hyperthyroidism is a common clinical syndrome in which the thyroid produces and releases excess thyroid hormones. The most common cause of hyperthyroidism is Graves’ disease. The treatment for hyperthyroidism includes surgery, antithyroid drugs (ATDs), and radioiodine therapy. Methimazole (MMI) and propylthiouracil (PTU) as oral ATDs are widely used to treat hyperthyroidism. Liver injury is a common complication in patients with hyperthyroidism.1,2 Severe cholestasis—either induced by thyrotoxicosis or as an adverse effect of ATD—has been increasingly reported in patients with hyperthyroidism.3–6 Additionally, patients with Graves’ disease may also have concomitant complications in the form of other autoimmune diseases, such as autoimmune hepatitis 7 and primary biliary cirrhosis, 8 which can also present as cholestasis. Therefore, identification of the causes of cholestasis is important for appropriate management of patients with hyperthyroidism. We report here a patient with cholestasis and provide a brief review on the diagnosis and treatment of cholestasis in patients with hyperthyroidism.
Case report
A 41-year-old man had taken MMI intermittently since he was diagnosed with Graves’ hyperthyroidism 6 years previously. Ten days before admission to hospital on 16 December 2015, he had scleral icterus and passed dark urine after taking MMI (30 mg/day) intermittently for 2 months. One week before admission, an ultrasound scan of the neck showed defuse enlargement of the thyroid gland with no nodules. Thyroid-stimulating hormone receptor antibodies were positive with a titer of 5.18 U/L (positive >1.75 U/L).
On admission, he had nausea, fatigue, vomiting, agitation, tremulousness, and abdominal pain. A physical examination showed that his temperature was 36.3°C and blood pressure was 119/85 mmHg. He had severe icterus of the sclerae and skin. Additionally, his thyroid was diffusely enlarged. However, his respiratory and cardiovascular systems were normal, with a heart rate of 100 beats/minute, and an abdominal examination was unremarkable. Results of the patient’s blood biochemical indicators are shown in Table 1.
Laboratory findings of the patient with hyperthyroidism at the time of admission.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; ALP, alkaline phosphatase; GGT, gamma glutamyl transpeptidase; TBiL, total bilirubin; PTA, prothrombin time activity; INR, international normalized ratio; WBC, white blood cells; PLT, platelets; Cr, creatinine; TSH, thyroid-stimulating hormone; FT3, free triiodothyronine; FT4, free thyroxine.
To exclude other causes of liver injury, additional studies showed that the patient was negative for immunoglobulin M antibodies for Epstein–Barr virus and hepatitis A and E viruses. Tests for hepatitis B virus surface antigen and core antibody, and hepatitis C virus antibody were negative. Tests for antinuclear antibody, double-stranded DNA antibody, anti-liver-kidney microsomal antibody, anti-smooth muscle antibody, and anti-mitochondrial antibody M2 were also negative. The patient’s serum ceruloplasmin level was normal. The patient also had no history of liver disease. Abdominal ultrasonography, which was performed on admission, showed unremarkable changes of the liver, pancreas, and spleen, and no evidence of lithiasis in the common bile duct or gallbladder. Furthermore, magnetic resonance cholangiopancreatography on post-admission day 2 did not show any evidence of lithiasis, strictures, or masses in the bile duct. The patient refused to undergo percutaneous liver biopsy. The patient had no medical history, except for hyperthyroidism, and had never consumed alcohol.
After admission, because the Roussel Uclaf Causality Assessment Method (RUCAM) scale score for MMI was 7 in our patient, he was diagnosed as having MMI-induced liver injury. The R-value (alanine aminotransferase [ALT]/upper limit of normal value (ULN)/alkaline phosphatase [ALP]/ULN) in this patient was 0.667, which indicated cholestatic hepatitis. Treatment with MMI was stopped at the time of admission. The patient received standard conservative therapy, including bed rest, nutritional and energy supplements, and intravenous infusion of water–electrolyte and acid–base equilibrium maintenance. Propranolol therapy was used to relieve his symptoms. His serum total bilirubin (TBil) level rose from 410.5 to 519.9 µmol/L and prothrombin time activity (PTA) dropped from 81.0% to 52.2% in 10 days. To prevent further deterioration of his liver function, plasma exchange was performed three times, and dexamethasone (10 mg, intravenously) and 2000 to 2500 mL of frozen plasma was used each time. No hepatic encephalopathy was found. After plasma exchange, his PTA rose to 101%; however, his TBil level continued to increase to 669.8 µmol/L.
Assuming that thyrotoxicosis could also cause liver injury, we added radioactive iodine treatment (380 MBq) to control his hyperthyroidism 2 weeks after admission. His hyperthyroidism was greatly relieved after radioactive iodine therapy (Figure 1), but the TBil level further increased to 776.8 µmol/L (Figure 2). Three weeks after admission, oral prednisone (30 mg/day) was administered to this patient, and his TBil level gradually decreased. The dosage of prednisone was gradually tapered (Figure 1). His liver function almost normalized after 3 months. During the follow-up period 3 months after discharge, the patient remained asymptomatic and showed no evidence of ongoing hepatitis or sequelae.
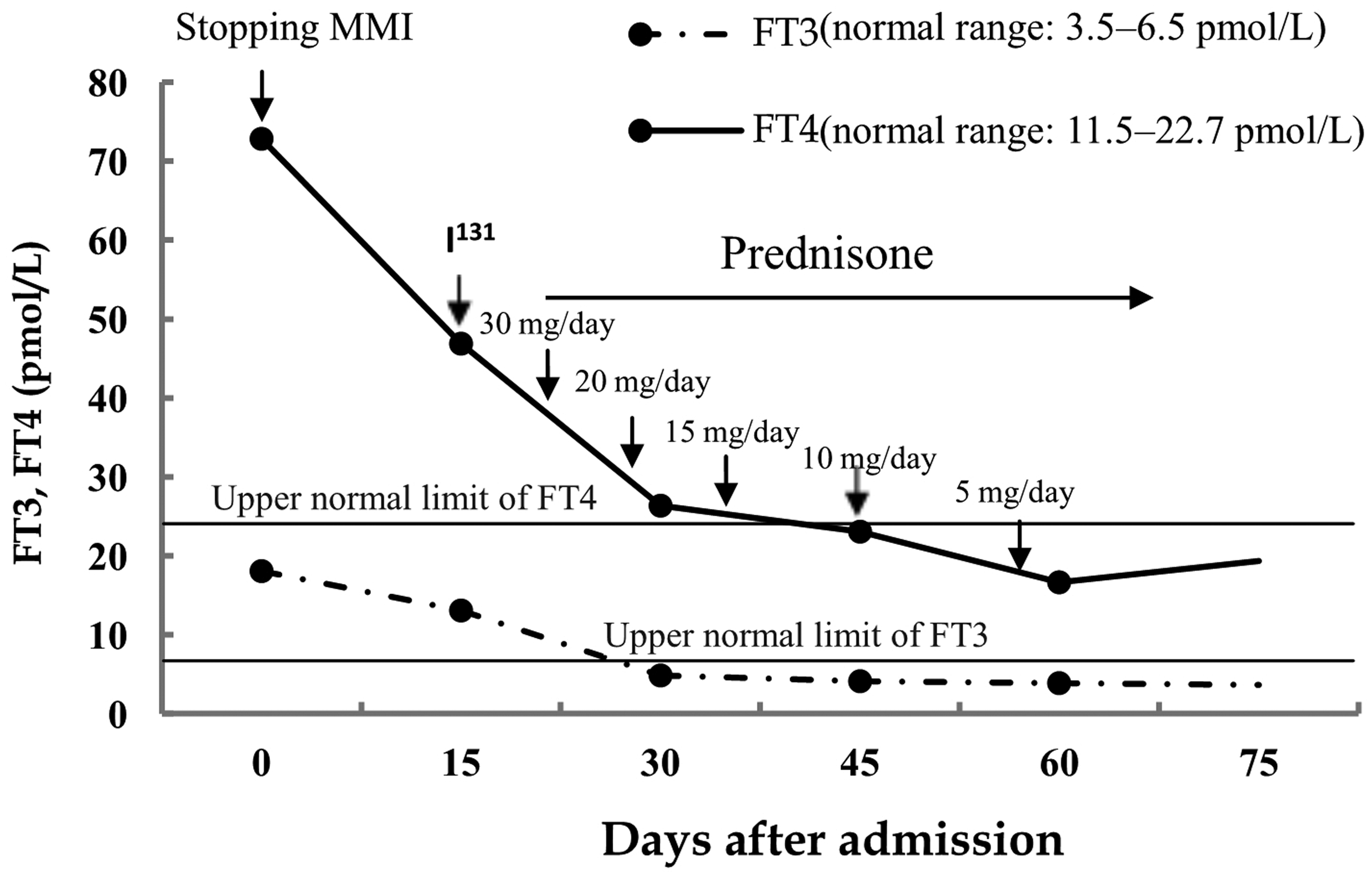
FT3, FT4, and TSH levels over time (days) after the patient’s admission. FT3, free triiodothyronine; FT4, free thyroxine; TSH, thyroid-stimulating hormone; I131, radioactive iodine therapy; MMI, methimazole.
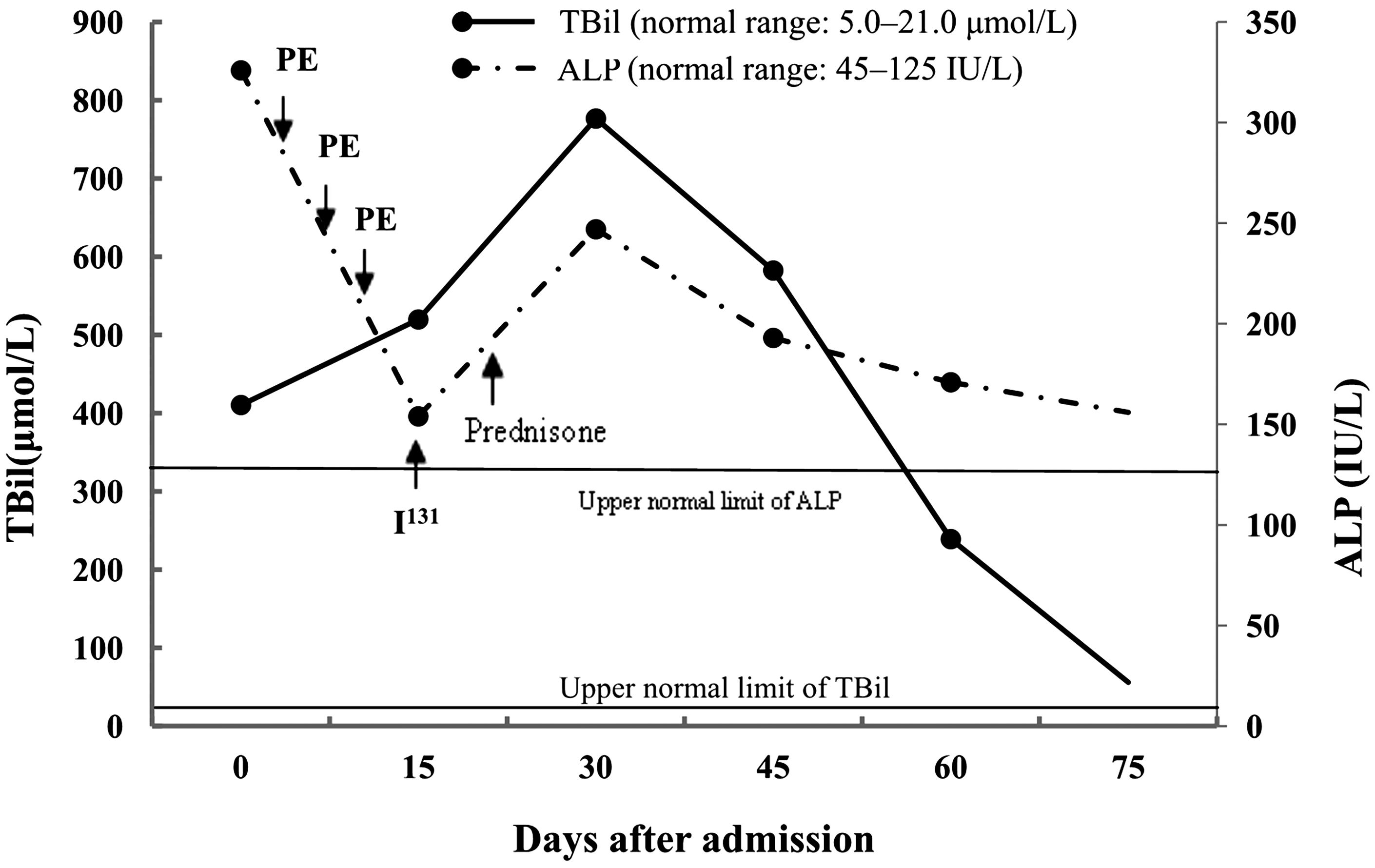
TBil and ALP levels over time (days) after admission of the patient. TBil, total bilirubin; ALP, alkaline phosphatase; I131, radioactive iodine therapy; PE, plasma exchange.
This case report did not require ethics committee approval because it did not involve animal or human studies. Written informed consent for publication was obtained from the patient.
Discussion
Cholestasis in patients with hyperthyroidism is a rare, but complicated, severe disease. Severe cholestasis may progress to liver dysfunction and require urgent and accurate management. However, in clinical practice, identifying the causes of cholestasis in patients with hyperthyroidism is difficult.
Previous studies have shown a wide range in the incidence of abnormalities of liver function in patients with untreated hyperthyroidism. The most common abnormality of liver function is an increase in ALP levels.1,9 However, bone isoenzyme of ALP is responsible for an elevation in ALP levels in patients with hyperthyroidism. 10 Elevation of gamma glutamyl transpeptidase (GGT) levels varies from 14% to 62% in patients with hyperthyroidism.11,12 Therefore, the true prevalence of cholestasis in patients with hyperthyroidism remains unknown. Severe cholestasis and liver failure have been increasingly reported in case reports of thyroid disorders4,5,13–19 (Table 2). A recent study showed that 65% of patients with Graves’ hyperthyroidism were associated with different degrees of hepatic dysfunction. Among them, 32.4% were classified as the bile stasis type, and severe liver injury (defined as ALT or aspartate aminotransferase levels ≥20 ULN, GGT levels ≥10 ULN, ALP levels ≥5 ULN, and/or TBiL, direct bilirubin (DBiL) levels ≥5 ULN) found in 6.6% of patients before ATD treatment. 9 Although, initially, serum thyroid-stimulating hormone levels ≤0.02 mIU/L were found to be a risk factor for abnormal liver biochemistry, 1 other studies showed that the severity of hyperthyroidism was not associated with the occurrence of liver injury. 20 Additionally, intrahepatic cholestasis has been found in patients with subclinical hyperthyroidism. 21 The mechanisms involved in development of cholestasis in patients with hyperthyroidism are not fully understood.22,23 Cholestasis is related to hyperthyroidism complicated by congestive heart failure. However, cholestasis is also found in patients with uncomplicated hyperthyroidism. 13 Other studies have suggested that increased hepatic oxygen consumption without a parallel increase in hepatic blood flow in the hypermetabolic state of hyperthyroidism may result in insufficient oxygen supply in the centrilobular zones, which may interfere with bile transport and result in cholestasis. 24 Recent studies have shown that hyperthyroidism increases mitochondrial oxygen consumption and production of free oxygen radicals. Oxidative stress of hepatocytes results in apoptosis of hepatocytes.25,26
Previous case reports of thyrotoxicosis-induced severe cholestasis.

M, male; F, female; ALT, alanine aminotransferase; ALP, alkaline phosphatase; GGT, gamma glutamyl transpeptidase; TBiL, total bilirubin; TSH, thyroid-stimulating hormone.
PTU and MMI can induce liver injury in 0.1% to 0.2% of patients with hyperthyroidism.27–29 Previous studies showed that PTU mostly caused hepatocellular injury and MMI often caused cholestasis.27,30 However, the incidence of MMI-induced cholestasis varies among reports.28,30,31 In a study that included 37,370 patients with hyperthyroidism treated with MMI, cholestasis occurred at a rate of 0.24/1000 person-years. 29 A recent study showed that in patients with ATD-induced severe hepatotoxicity, the frequency of the cholestatic type in the MMI group (35.3%) was higher than that in the PTU group (17.9%). 32 Severe liver injury or severe cholestasis induced by MMI is also being increasingly reported in case reports.33–37 Liver injury usually occurs within 2 days to 3 months after taking MMI.27,38 An older age of patients and a higher dose of MMI are associated with occurrence of cholestasis. 27 However, in a recent study, cholestasis was found to be dose independent of MMI. 29 Therefore, MMI-induced cholestasis is idiopathic and unpredictable, and autoimmune mechanisms may be involved in its occurrence. 29
Concomitant autoimmune liver diseases, including autoimmune hepatitis and primary biliary cirrhosis, as causes of cholestasis have only been reported in a few case reports describing hyperthyroidism in patients.7,8,39,40 This indicates that concomitant autoimmune liver diseases are not a common cause of cholestasis in these patients. However, other underlying liver diseases should also be excluded in patients with hyperthyroidism and cholestasis, especially in high epidemic countries of hepatitis B or C virus. Kang et al. reported MMI-induced acute liver failure in a patient who was a chronic hepatitis B virus carrier. 41
As shown in Table 3, patients in most previous case reports of MMI-associated severe cholestasis also had hyperthyroidism. Therefore, determining the causes of cholestasis in patients with MMI-treated hyperthyroidism is difficult, based solely on the clinical and biochemical presentation. A previous case report suggested that the RUCAM scale provided a more accurate measurement in determining the causality of MMI-induced hepatotoxicity than other methods. 42 However, this scale needs to be verified in larger number of patients. Additionally, the RUCAM scale is based on ALP from hepatic origin only. 43 As stated above, bone isoenzymes of ALP are significantly increased in patients with hyperthyroidism, indicating that the RUCAM scale is also inaccurate in determining the causality of ATD-induced cholestasis. The RUCAM scale score for MMI was 7 in our patient, but his liver function worsened after MMI was discontinued. Therefore, we could not exclude thyrotoxicosis-induced cholestasis. TBil levels rapidly decreased after his hyperthyroidism resolved, which further supports the causality between thyrotoxicosis and cholestasis in our patient.
Previous case reports of methimazole-induced severe cholestasis.

M, male; F, female; ALT, alanine aminotransferase; ALP, alkaline phosphatase; TBiL, total bilirubin; DBiL, direct bilirubin; TSH, thyroid-stimulating hormone.
Treatment of cholestasis in patients with hyperthyroidism remains a significant challenge in clinical practice. For patients with suspected ATD-induced cholestasis, ATDs should be discontinued. For patients with thyrotoxicosis-induced cholestasis, rapid restoration of euthyroidism is important for the recovery of liver function. 9
Previous studies have shown that control of hyperthyroidism is important for recovery of cholestasis, induced by MMI or hyperthyroidism.9,28 However, the choice of treatment methods for hyperthyroidism is complicated in clinical practice. Although there has been a report of successful switching to PTU after MMI-induced liver injury, 3 it was not recommended because of cross-allergic reactions that might exist between PTU and MMI. 44 ATDs may aggravate liver injury and occasionally induce liver failure. Therefore, use of ATDs in patients with thyrotoxicosis-induced severe cholestasis is risky. Urgent thyroidectomy is also considered risky in patients with thyrotoxicity and severe liver dysfunction. Most previous studies have shown that radioactive iodine therapy is a safe and effective treatment strategy in patients with MMI- or thyrotoxicosis-induced cholestasis,32,45 although radioactive iodine therapy has also been reported to cause hepatic injury on occasion.46,47
Glucocorticoids have been successfully used for treating cholestatic hepatitis caused by MMI and thyrotoxicosis. 48 Glucocorticoids reduce conversion of thyroxine to triiodothyronine in peripheral tissues and decrease serum thyroxine levels in patients with hyperthyroidism by reducing thyroxine secretion. 49 Additionally, previous studies have shown that corticosteroids are effective in reducing peripheral oxidative stress present in infiltrative Graves’ ophthalmopathy.50,51 In animal studies, other antioxidants, including vitamin E, curcumin, and L-carnitine, have been shown to alleviate L-thyroxine-induced oxidative stress in rat liver;52,53 however, their clinical efficacy remains to be demonstrated. An artificial liver support system has also been found to be effective in treating patients with ATD-induced cholestasis by decreasing thyroid hormone and bilirubin levels and promoting recovery of liver injury. 54
In conclusion, distinguishing between thyrotoxicosis- and MMI-induced severe cholestasis is difficult. Discontinuing MMI, treatment with glucocorticoids, and control of hyperthyroidism with radioactive iodine may be effective treatment methods for severe cholestasis induced by thyrotoxicosis or MMI in patients with hyperthyroidism.
