Abstract
Pulmonary capillary hemangiomatosis (PCH) is a very rare and refractory disease characterized by capillary angioproliferation. The updated classification of pulmonary hypertension categorizes PCH into a subgroup of pulmonary arterial hypertension (PAH) alongside pulmonary veno-occlusive disease (PVOD). However, the definitive diagnosis of PCH only with noninvasive tools remains difficult. The aim of this study was to elucidate the radiological and physiological characteristics of PCH. We searched for cases of pathologically confirmed PCH in the English literature published between 2000 and 2018. We identified 26 cases among 39 studies. Then, we extracted and evaluated the relevant clinical information in all cases with available data. On chest computed tomography (CT), ground-glass opacities (GGOs) were observed in 92% of the cases, in which poorly defined nodular pattern was the most common (88%). GGOs in a bat-wing distribution were observed in one case. Septal lines and lymph node enlargement were observed less frequently (each 19%, 12%). Seven cases (27%) had overlapping abnormalities. Diffusing capacity of the lung for carbon monoxide (DLCO) was remarkably decreased. Alveolar hemorrhage by histological findings or bronchoalveolar lavage (BAL) was observed in seven cases. The present study showed that the most characteristic findings of CT in PCH was centrilobular GGOs with a poorly defined nodular pattern, and septal lines and lymph node enlargement were seen less frequently. Alveolar hemorrhage detected by BAL and decreased DLCO may also be helpful to recognize the possibility of PCH like PVOD.
Keywords
Introduction
Pulmonary capillary hemangiomatosis (PCH) is a rare form of pulmonary hypertension (PH) (<100 reported cases) that is characterized by capillary angioproliferation in the alveolar septa. 1 Although histological findings are required for the definitive diagnosis of PCH, obtaining adequate lung samples is usually difficult due to severe PH. Additionally, since its clinical characteristics are poorly defined, patients with PCH are frequently misdiagnosed, 2 and diagnosis is made after death or lung transplantation. 3 Nevertheless, the prognosis is poor, and median survival from the onset of the initial symptom is reported as approximately three years. 2 Furthermore, as the common PCH symptoms, such as progressive dyspnea, cough, chest pain, and hemoptysis, 4 are nonspecific, diagnostic clues for PCH by noninvasive modalities are much needed.
In the updated classification of PH from Sixth World Symposium on Pulmonary Hypertension (2018), PCH is categorized in the World Health Organization group 1.6, namely “pulmonary arterial hypertension (PAH) with overt features of venous/capillaries involvement” alongside pulmonary veno-occlusive disease (PVOD), because PVOD/PCH and PAH have been considered as a continuum of pulmonary vascular disease. 5 Recently, some reports of PVOD/PCH have demonstrated that high-resolution chest tomography (HRCT) images, pulmonary function tests (PFTs), and bronchoalveolar lavage (BAL) can be useful modalities for noninvasive diagnostic examinations.4,6 The characteristic findings of PVOD/PCH in HRCT are centrilobular ground-glass opacities (GGOs), septal lines, and mediastinal lymph node enlargement. 7 PFTs, including forced expiratory volume in 1 second (FEV1) and FEV1/forced vital capacity (FVC) ratio, are normal, but the diffusing capacity of the lung for carbon monoxide (DLCO) is remarkably decreased. 8 Bronchoscopy is not usually performed, but BAL can reveal hemosiderin-laden macrophages in PVOD. 9 However, although the clinical findings of “PVOD” or “combined PVOD and PCH” are occasionally reported, previous reports of clinical characteristics which focus on PCH were very limited. Therefore, the features of these noninvasive examinations in the PCH remain uncertain.
The aim of this study was to elucidate the radiological and physiological characteristics of PCH. Developing noninvasive diagnostic tools may help to raise suspicion of the presence of PCH and lead to beneficial effects for the patient with PCH, such as early treatment and improved prognosis and outcomes.
Methods
Literature search
To identify PCH cases, we searched English literature published between 2000 and 2018 via PubMed/Medline, the Cochrane Central Register of Controlled Trials, and the Cochrane Database of Systematic Reviews, using the keywords “pulmonary capillary hemangiomatosis” and/or “PCH.” Other keywords, including “pulmonary veno-occlusive disease” and/or “PVOD,” related to PCH were also searched in these databases.
The study was conducted in accordance with the ethical principles of 1964 Helsinki Declaration and subsequent amendments. The requirement for the approval by the Ethical Review Board of the Graduate School of Medicine of Chiba University was waived by the ethics committee because the analysis of the present study was limited to preexisting literature data.
Selection criteria
Review of 26 selected cases with pulmonary capillary hemangiomatosis.
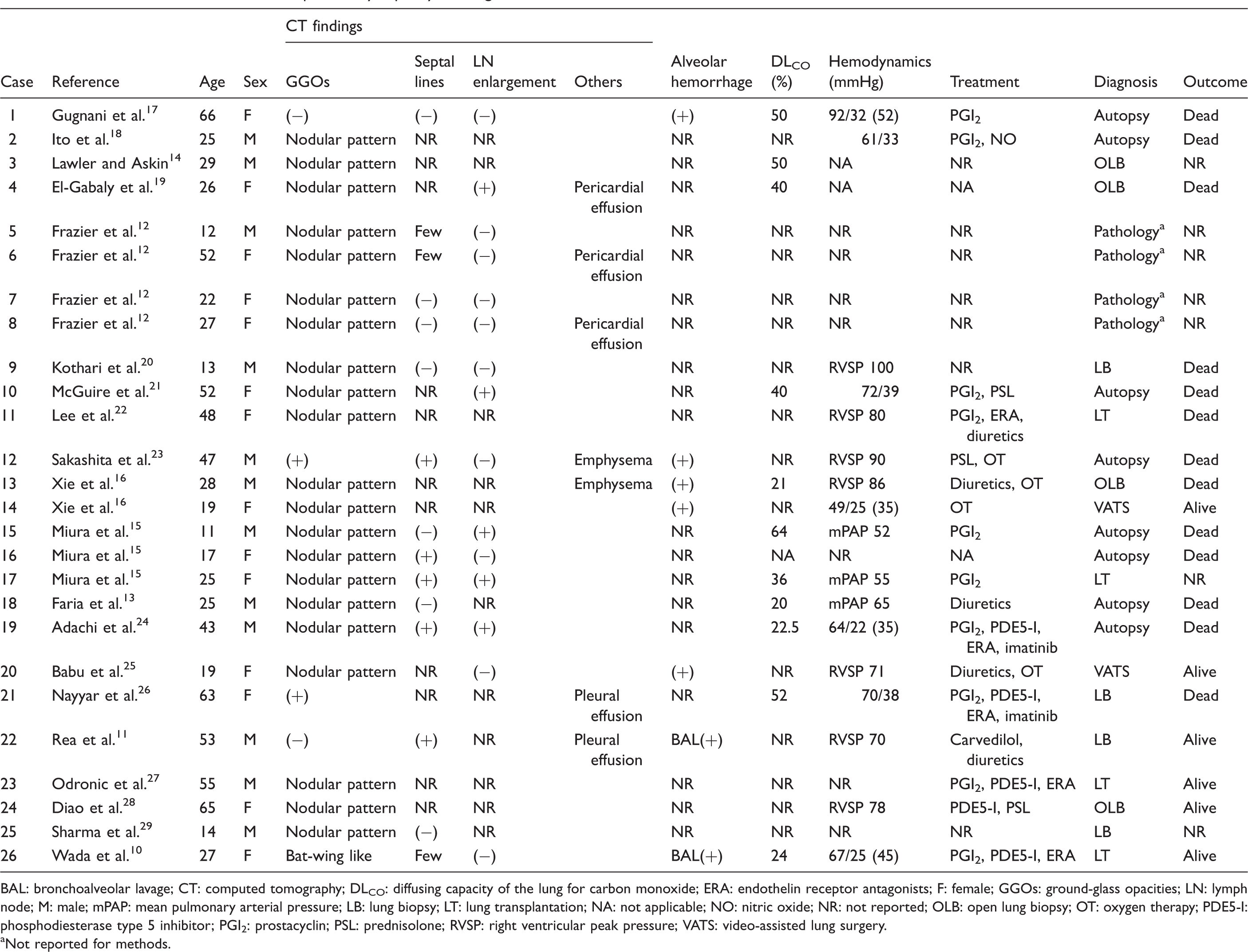
BAL: bronchoalveolar lavage; CT: computed tomography; DLCO: diffusing capacity of the lung for carbon monoxide; ERA: endothelin receptor antagonists; F: female; GGOs: ground-glass opacities; LN: lymph node; M: male; mPAP: mean pulmonary arterial pressure; LB: lung biopsy; LT: lung transplantation; NA: not applicable; NO: nitric oxide; NR: not reported; OLB: open lung biopsy; OT: oxygen therapy; PDE5-I: phosphodiesterase type 5 inhibitor; PGI2: prostacyclin; PSL: prednisolone; RVSP: right ventricular peak pressure; VATS: video-assisted lung surgery.
Not reported for methods.
Results
Clinical characteristics of PCH
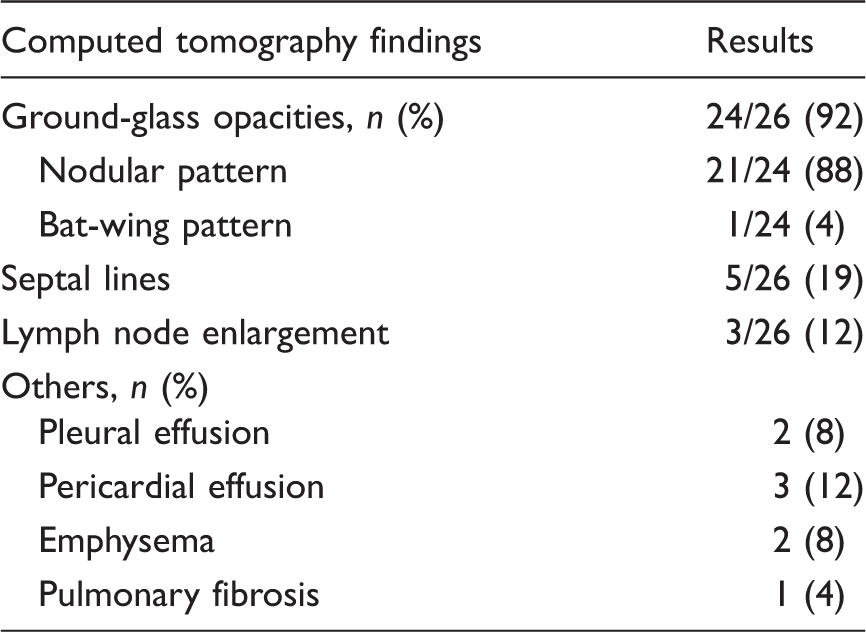
Clinical characteristics of 26 cases with pulmonary capillary hemangiomatosis.

DLCO: diffusing capacity of the lung for carbon monoxide; ERAs: endothelin receptor antagonists; PAP: pulmonary arterial pressure; PDE5: phosphodiesterase type 5; RVSP: right ventricular peak pressure.
Results are expressed as mean ± SD.
Radiological findings
Radiological characteristics of 26 cases with pulmonary capillary hemangiomatosis.
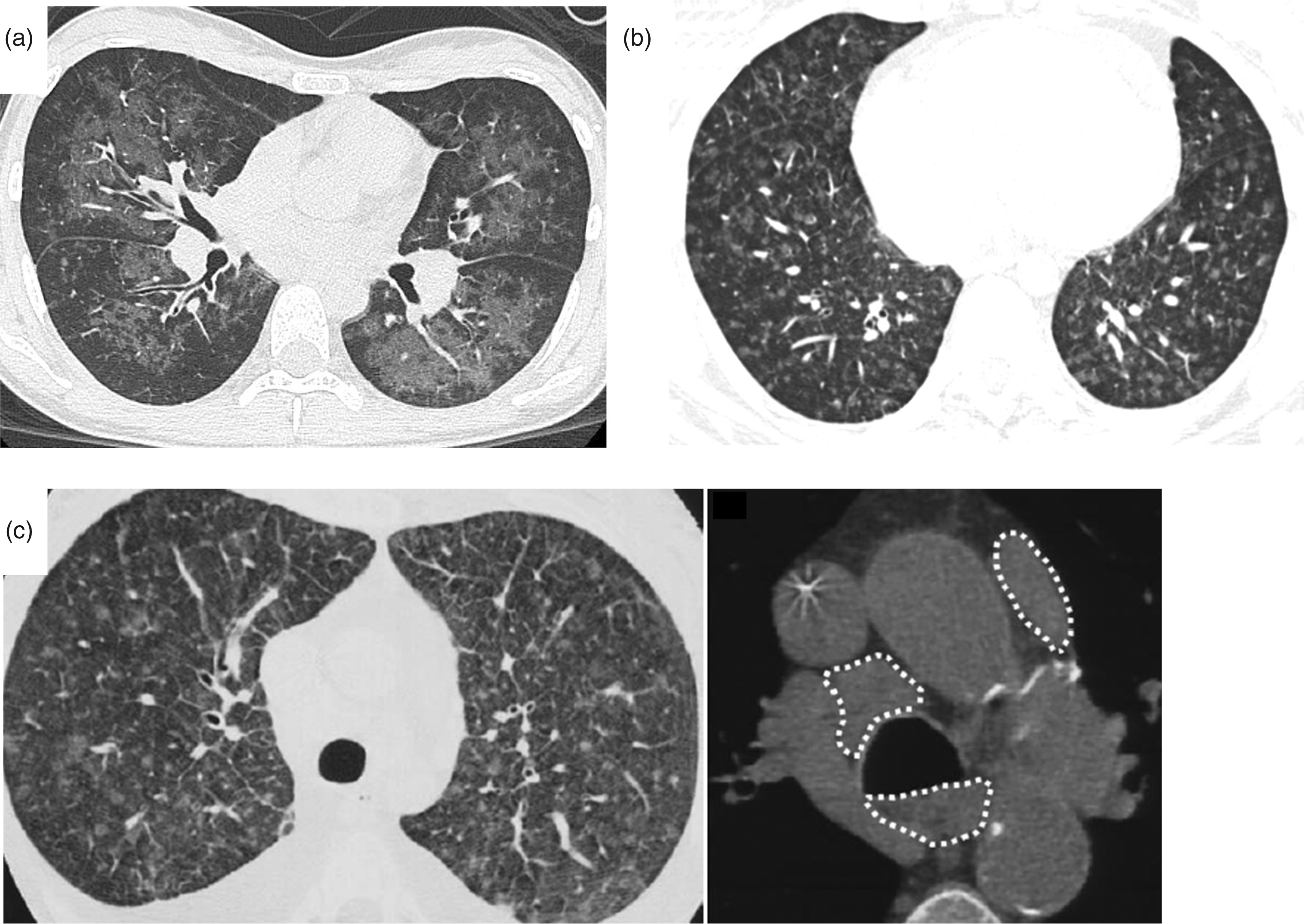
Regarding the bat-wing distribution of GGOs, this was observed in a patient with PCH reported from our hospital. HRCT of the chest showed a rare radiological form of GGOs distributed in a diffuse bat-wing pattern
10
in the lung field, but sparing the subpleural parenchyma (Fig. 1a). There were a few thickened interlobular septa in the lower lobes. Lymph node enlargement was not seen. BAL revealed hemosiderin-laden macrophages. Based on these clinical findings, we diagnosed as PVOD with secondary pulmonary hemosiderosis. She was treated with sildenafil, bosentan, and epoprostenol. After three years from the onset of symptoms, she underwent double-lung transplantation. Histological findings demonstrated capillary proliferation with multiple layers in patchy areas, arterial intimal fibrosis, medial hypertrophy, and hemosiderin-laden macrophages in the alveolar spaces. There were a few pulmonary venous occlusions. Because these pathological findings were consistent with PCH, she was finally diagnosed with PCH.
Typical pattern of CT in PCH and PVOD. (a) Unique pattern of PCH: GGOs distributed diffusely in a bat-wing pattern in the lung field, sparing subpleural parenchyma,
10
(b) typical pattern of PCH: diffuse centrilobular GGOs with poorly defined nodular pattern,
13
and (c) typical pattern of PVOD: centrilobular GGOs with poorly defined nodular pattern, septal lines, and mediastinal lymph node enlargement (white dotted lines).
6
Physiological findings (PFTs and BAL)
In the present analysis, the findings of remarkably decreased %DLCO were identified in 11 cases (38.1 ± 14.9%, range 20%–64%). Many cases with available data showed normal or mild decreased values of FEV1 and FEV1/FVC ratio. Additionally, hemosiderosis detected by histological findings or BAL was observed in 7/26 cases (Table 2), which might be related to diffuse or nodular GGOs in CT. Among these cases, only two cases reported the findings of BAL.10,11 Conversely, no cases reported transbronchial lung biopsy (TBLB). However, “lung biopsy” (not including open lung biopsy) was performed in four cases (Table 2).
Pathological findings
In the present study, histological confirmation of PCH was obtained in all cases, and detailed description of pathological findings was provided for 22 cases. All the 22 cases showed capillary proliferation. Pulmonary arterial changes with intimal hyperplasia and/or fibrosis without plexiform lesions were described in 9 cases. In addition, alveolar hemorrhage was shown in five cases, and no venous occlusion was confirmed in six cases.
Discussion
The present study explored the features of radiological and physiological findings in pathologically diagnosed PCH. The major findings of this study were that (1) the most characteristic findings of CT in PCH is centrilobular GGOs with poorly defined nodular pattern (21/26 cases, 81%), (2) septal lines and lymph node enlargement are less frequently observed (each 19%, 11%), and (3) evidence of alveolar hemorrhage using BAL (2/2, 100%) and decreased DLCO on PFTs (10/11 cases, 91%) also appear to be helpful noninvasive assessments for the recognition of PCH.
In 1989, Faber et al. 30 first reviewed the clinical and pathologic features of PCH. They described that a diffuse bilateral reticulonodular pattern was typical in chest radiographs of patients with PCH. 30 Then, several cases of PCH31,32 and literature about HRCT findings of PCH were recently reported.1,12 In these reports, the most common characteristics of PCH compared with PAH are centrilobular GGOs, septal lines, and lymph node enlargement in HRCT images. GGO patterns in HRCT of PCH patients might also have some specific characteristics, because GGOs with a poorly defined nodular pattern, diffusely, bilateral in distribution, and sparing of the periphery are reported in PCH cases13,31,32 (Fig. 1b). Frazier et al. 12 also mentioned that CT findings of PCH were characterized by widespread ill-defined centrilobular nodules of GGOs in addition to main pulmonary arterial enlargement. In the present study, centrilobular GGOs with nodular patterns was the most common radiological feature, which is consistent with these previous reports.13,31,32 Centrilobular GGOs may represent focal tumor-like capillary angioproliferation in the alveolar septa,14,32 locally increased perfusion to the lesion, and secondary alveolar hemorrhage (i.e., hemosiderosis) in the airspace. 30 Regarding bat-wing pattern of GGOs, this type of GGO is sometimes observed in pulmonary edema, interstitial lung disease, PH, lymphoproliferative disorder, and hemosiderosis. We speculate that this bat-wing pattern in which GGOs were distributed is in correlation with both the primary lesion of PCH and occult alveolar hemorrhages because histological findings of the transplanted lung showed capillary angioproliferation in patchy areas and abundant hemosiderin-laden macrophages (case 26; see Fig. 1a). In the present study, other CT findings, such as pleural effusion (8%), pericardial effusion (12%), and emphysema (8%) were less frequent. Therefore, these CT findings may be less helpful to recognize the possibility of PCH.
As the present study analyzed radiological and physiological characteristics of PCH only, and not PVOD, the difference in such findings between PCH and PVOD was not clarified. Regarding histological findings, PCH is characterized by abnormal capillary angioproliferation at least two layers thick and extends along septal lines.1,33 The major feature distinguishing PCH from other diseases is the infiltration of capillary vessels into other pulmonary structures, including the walls of arteries and veins, intralobular fibrous septa, and bronchi. 33 On the other hand, main lesion of PVOD is venous occlusion, and capillary angioproliferation is also found as secondary change of venous occlusion.3,6 In PVOD, the infiltration into other structures is not observed, and capillary angioproliferation is seen at single layer. 33 In the present study, three characteristics of HRCT images in PCH were shown: centrilobular GGOs, septal lines, and lymph node enlargement. Nonetheless, these are also known as characteristics of PVOD 34 (Fig. 1c). Patterns of GGOs in PVOD may vary from a minimal patchy appearance to an extensively diffuse one, 35 while in the present study, the typical pattern of GGOs in PCH comprised a poorly defined nodular pattern. Additionally, septal lines are observed in 93% of patients with PVOD and adenopathy, in 80% of those with PVOD. 34 When there are two or three common findings in HRCT, PVOD can be distinguished from PAH with a 75% sensitivity and an 84.6% specificity. 7 Conversely, our study of PCH cases demonstrated that septal lines and lymph node enlargement were observed less frequently (19% and 12%, respectively) than in PVOD. Therefore, when a patient with PH is suspected to have PVOD/PCH, we speculate that centrilobular GGOs with poorly defined nodular pattern and absence of septal lines or lymph node enlargement may be more suggestive of PCH than PVOD. However, as confirmed diagnostic cases of PCH are very limited, the difference in these CT findings between PCH and PVOD was uncertain, and further investigation is needed. Indeed, previous report indicated that patients with PCH show larger size of GGOs in HRCT compared to those with PVOD. 15 Histologically, PCH presents tumor-like capillary angioproliferation, and PVOD presents dilated capillary vessels due to changes secondary to pulmonary venous occlusion. Thus, the different condition of the capillaries between PCH and PVOD seems to lead to differences in the sizes of centrilobular GGOs that are observed via HRCT. 15
Among the physiological examinations based on noninvasive modalities, PFTs are known as a useful tool to recognize the possibility of PVOD/PCH. In PVOD, some previous reports have shown that the features of PFTs are normal values of FEV1 and FEV1/FVC and remarkably decreased DLCO (frequently DLCO < 55%).7,8 DLCO/VA in patients with PVOD is significantly decreased compared with PAH. 7 In PVOD, interstitial edema and/or pulmonary capillary angioproliferation due to chronic pulmonary venous obstruction may be associated with severe reduction of DLCO. 9 On the other hand, severe reduction of DLCO in PCH may be explained by the underlying pathophysiology; the proliferative capillaries and alveolar hemorrhage fill the alveolar septa in PCH. The gas exchange mechanism may be impaired in such pathophysiology. Previously, it has been reported that the gas exchange becomes more severe in PCH when there is pulmonary artery narrowing, venous occlusion; subsequently, alveolar hemorrhage appears in addition to capillary angioproliferation. 14 However, there are only few review articles specifically describing PFT findings in PCH. Almagro et al. 2 demonstrated that the findings of PFTs, FVC, FEV1, and FEV1/FVC in PCH were almost normal and that there was a marked reduction of DLCO, similar to those in PVOD. In the present study, the %DLCO decreased markedly in almost all cases. Therefore, we speculate that the severe reduction of DLCO may be one of a meaningful finding in PCH. Another noninvasive tool is bronchoscopy; however, this is not usually examined in patients with PAH, especially those with severe PH. Although the findings of CT are helpful for the diagnosis of PVOD/PCH, other lung diseases, such as interstitial lung disease, infections, or lymphoproliferative disorders, also present GGOs. Hemosiderin-laden macrophages in BAL correlate with occult alveolar hemorrhage. 6 Because the main lesion of PVOD is postcapillary pulmonary vessels, alveolar hemorrhage is a common characteristic in patients with PVOD. Reportedly, patients with PVOD present a higher percentage of hemosiderin-laden macrophages and Golde score compared with those with idiopathic PAH. 9 Therefore, BAL is one of the essential noninvasive tools for the detection of PVOD. Kano et al. reported that a child with PVOD showed GGOs distributed diffusely in a bat-wing pattern in the lung field but were sparing the lung periphery. That patient’s BAL also revealed hemosiderin-laden macrophages. 36 Bat-wing GGO is also observed in cases of idiopathic pulmonary hemosiderosis, which is characterized by hemoptysis and alveolar capillary hemorrhage. 37 It has been speculated that GGOs distributed diffusely in a bat-wing pattern were caused not only by PVOD itself but also by massive alveolar hemorrhage. Recently, it has been proposed that hemosiderin-laden macrophages in the sputum may also support the diagnosis of PVOD and distinguish PVOD from other forms of PH. The reported sensitivity was 100% and the specificity was 97% for the clinical diagnosis of PVOD. 38 Conversely, there is little evidence on whether BAL supports the clinical diagnosis of PCH. Xie et al. 16 reported that among 64 cases, some cases showed hemosiderin-laden macrophages in BAL, and BAL may be a useful tool for the diagnosis of PCH. In the present study, we only identified two cases in which pulmonary alveolar hemorrhage by BAL was detected. The main lesion in PCH, capillary angioproliferation, also seems to cause an alveolar hemorrhage. Therefore, alveolar hemorrhage detected by BAL may be considered as one of the useful noninvasive diagnostic tools for PCH. As the number of cases is very limited in the present study, further analysis with more number of cases is required to evaluate BAL in PCH cases. Conversely, TBLB (reported as “lung biopsy”) was performed in four cases (Table 2). As the histological evaluation to identify characteristic changes of PCH requires an extensive lung tissue evaluation (a minimum of five blocks from surgical lung biopsies), TBLB is not appropriate for adequate sampling. 3
This study has several limitations. First, most of the selected articles were single case reports, and the number of cases was very limited because confirmed PCH cases are rare. Second, we could not verify images of CT or other information (PFTs, BAL, hemodynamics, diagnostic tools, treatments, and outcomes) in all cases. Third, we did not analyze the findings of PCH compared to PVOD directly. Fourth, CT images were not scanned using the same equipment or in similar settings.
In conclusion, our study showed that the most characteristic findings of CT in PCH were centrilobular GGOs with a poorly defined nodular pattern, while septal lines and lymph node enlargement were seen less frequently. Severe reduction of DLCO on PFTs and alveolar hemorrhage detected by BAL may also help to establish the diagnosis of PCH like PVOD. However, if these findings are obtained, we always need to rule out other diseases to make a definitive diagnosis of PCH. Though the clinical diagnosis of PCH is difficult, developing noninvasive diagnostic tools may be useful in suggesting the diagnosis of PCH and lead to beneficial effects, such early detection and treatment, which may help improve the prognosis and outcomes of patients with PCH.
Footnotes
Acknowledgments
We sincerely thank Dr Takashi Ogura for his advice regarding the interpretation of HRCT in a case with PCH and Mrs Chieko Handa, Mrs Tamie Hirano, and Miki Sakurai for their technical assistance and general support.
Author contributions
All authors contributed to the conceptual design of the analysis. RA, JT, and AS collected data and wrote the paper. RA and JT performed the analysis. All authors took part in drafting and revising the article. All authors have read and approved this final manuscript.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This study was partly supported by the Japanese Ministry of Health, Labour and Welfare research grants specifically designated to the Respiratory Failure Research Group and Cardiovascular Diseases and the Pulmonary Hypertension Research Group from the Japan Agency for Medical Research and Development (No. 16ek0109127h0002). The funder had no role in the study’s design, collection of data or their analysis, decision to publish, or preparation of the manuscript.
