Abstract
Adherence to therapy for pulmonary arterial hypertension is essential to optimize patient outcomes, but data on real-world adherence to different pulmonary arterial hypertension drug classes are limited. This retrospective database analysis evaluated relationships between adherence, hospitalization, and healthcare costs in pulmonary arterial hypertension patients treated with endothelin receptor antagonists or phosphodiesterase type-5 inhibitors. From the IQVIA Adjudicated Health Plan Database, patients with pulmonary arterial hypertension were identified based on diagnostic codes and prescriptions for endothelin receptor antagonists (ambrisentan, bosentan, macitentan) or phosphodiesterase type-5 inhibitors (sildenafil, tadalafil) approved for pulmonary arterial hypertension. Patients were assigned to the class of their most recently initiated (index) pulmonary arterial hypertension therapy between 1 January 2009 and 30 June 2015. Medication adherence was measured by proportion of days covered; patients with proportion of days covered ≥80% were considered adherent. The proportion of adherent patients was higher for endothelin receptor antagonists (571/755; 75.6%) than for phosphodiesterase type-5 inhibitors (970/1578; 61.5%; P < 0.0001). In both groups, hospitalizations declined as proportion of days covered increased. Among adherent patients, those on endothelin receptor antagonists had a significantly lower hospitalization rate than those on phosphodiesterase type-5 inhibitors (23.1% versus 28.5%, P = 0. 0218), fewer hospitalizations (mean (standard deviation) 0.4 (0.8) versus 0.5 (0.9); P = 0.02), and mean hospitalization costs during the six-month post-index ($9510 versus $15,726, P = 0.0318). Increasing adherence reduced hospitalization risk more for endothelin receptor antagonists than for phosphodiesterase type-5 inhibitors (hazard ratio 0.176 versus 0.549, P = 0.001). Rates and numbers of rehospitalizations within 30 days post-discharge were similar between groups. Mean total costs were higher with endothelin receptor antagonists than phosphodiesterase type-5 inhibitors in all patients ($91,328 versus $72,401, P = 0.0003) and in adherent patients ($88,867 versus $56,300, P < 0.0001), driven by higher drug costs.
Keywords
Introduction
Disease progression in pulmonary arterial hypertension (PAH) leads to increasingly debilitating symptoms, high morbidity, frequent hospitalizations, and ultimately right heart failure and premature death.1–4 Delaying progression is thus an essential step in achieving the treatment goals for patients with PAH of achieving and maintaining good exercise capacity, quality of life and right-ventricular function, avoiding hospitalization, and improving survival.5–7
PAH-related hospitalization is an important measure of disease progression that has been demonstrated to predict increased mortality among patients in clinical practice 8 and in long-term, event-driven clinical trials. 1 Hospitalization for worsening PAH is a key component of the composite trial endpoint of time to clinical worsening formulated at the fourth World Symposium on Pulmonary Hypertension (WSPH) 9 and endorsed as a meaningful primary endpoint at the fifth WSPH. 10 In addition to their clinical burden, hospitalizations in patients with PAH are costly, and their healthcare impacts are amplified by the fact that readmission is common following discharge from hospital. 11
Given these considerations, it is important to determine how PAH-specific medications may reduce hospitalization in this patient population, including the influence of aspects of drug use such as patient adherence to prescribed therapy. Although data on the relationship between medication adherence and outcomes in PAH are limited, 12 evidence from other chronic diseases shows that nonadherence is associated with adverse outcomes, including hospitalization. 13 It is reasonable to expect that hospitalizations for worsening PAH may decrease with improved adherence to PAH therapy, 14 but this needs to be established in real-world practice.
Previous retrospective studies of PAH-specific therapies have indicated that adherence to PAH-specific drugs may vary by drug class, as high adherence to treatment has been reported with endothelin receptor antagonists (ERAs), 15 whereas relatively low adherence to treatment with phosphodiesterase type-5 inhibitors (PDE-5is) has been observed in other studies.16,17 ERAs and PDE-5is are readily comparable because all are oral agents that are often used in first-line therapy. 5 In contrast, PAH medications that target the prostacyclin pathway include intravenous, subcutaneous, inhaled, and oral agents, most of which are typically reserved for later in the treatment pathway and for patients at high risk for clinical worsening. 5 However, only two prior retrospective database studies have directly compared adherence to ERAs versus PDE-5is.18,19 Both of these studies indicated higher adherence with ERAs, but neither was reported in sufficient detail to permit a more complete evaluation.
To address these evidence gaps, we performed a retrospective database analysis on use of PAH-specific medications and hospitalizations in patients with healthcare claims indicative of PAH. The primary objectives of our study were to assess adherence to ERAs versus PDE-5is, and to explore the interactions between PAH-specific drug class and adherence in terms of their association with risk of hospitalization, healthcare resource use, and costs.
Methods
Data source
Data for this study were retrieved from the IQVIA Adjudicated Health Plan Database (previously known as PharMetrics Plus, P+), a fully adjudicated, longitudinal database of medical and pharmaceutical claims for more than 150 million unique US patients, with more than 40 million patients represented per year. The database includes detailed medical and pharmacy claims with cost information, linked at the individual patient level. All data used in these analyses have been anonymized and are fully compliant with the Health Insurance Portability and Accountability Act Privacy Rules; as such, Institutional Review Board review and approval were not required.
Study design and sample
Data were retrieved for the study period commencing 1 July 2008 and ending 31 December 2015. For the purposes of assigning patients to a drug class, the index drug was defined as the patient’s most recent PAH-specific ERA or PDE-5i within the index period of 1 January 2009 to 30 June 2015. This allowed for a six-month minimum pre- and post-index evaluation of the patient’s data. The index date was defined as the first date of receipt of the index drug during the index period. Drugs included in the ERA class were bosentan (Tracleer®; Actelion Pharmaceuticals US, Inc., South San Francisco, CA), ambrisentan (Letairis®; Gilead Sciences, Inc., Foster City, CA), and macitentan (Opsumit®; Actelion). Drugs included in the PDE-5i class were sildenafil citrate (Revatio®; Pfizer Inc., New York, NY) and tadalafil (Adcirca®; Eli Lilly and Company, Indianapolis, IN). PAH-specific drugs from other classes were not considered, namely prostanoids, the prostacyclin receptor agonist selexipag, and the soluble guanylate cyclase stimulator riociguat.
Because the claims database does not include linked medical charts that would have permitted confirmation of PAH diagnoses, patients were ascertained on the basis of a claims-based algorithm. Adult patients (at least 18 years of age at the index date) were included if they had at least one inpatient claim or at least two outpatient claims at least 30 days apart with an International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) diagnostic code for pulmonary hypertension, namely ICD-9-CM 416.0 (Primary pulmonary hypertension) or 416.8 (Other chronic pulmonary heart disease. Pulmonary hypertension, secondary), with the requirement that these claims must be within 90 days before the index date (pre-index baseline period) and 180 days after the index date (post-index follow-up period). Additional inclusion requirements were at least one claim for a PAH-specific ERA or PDE-5i (as listed above) during the index period, with continuous health plan enrollment for at least 90 days prior to the index date and at least 180 days following the index date. The analysis cohort was limited to patients with at least two claims for their index drug, for consistency with a recent retrospective claims database study of adherence to PAH medications. 19
Exclusion criteria were as follows: patients age ≥65 years at the index date not covered by a Medicare Advantage Prescription Drug plan, who typically have less complete capture of pharmacy claims data compared to Medicare Advantage beneficiaries; Medicare Cost coverage; one or more prescription claim for PDE-5i brands indicated for treatment of erectile dysfunction, namely sildenafil (Viagra®; Pfizer), tadalafil (Cialis®; Lilly), or vardenafil hydrochloride (Levitra®; Bayer HealthCare Pharmaceuticals Inc., Whippany, NJ); combination therapy (i.e. claims during the study period for more than one PAH-specific agent); a diagnosis of sickle cell anemia (because pulmonary hypertension associated with this condition was recognized at the fifth WSPH as being significantly different from PAH20); and data quality issues such as an invalid year of birth, gender, or health plan enrollment dates. Although bosentan is approved in Europe for reducing the number of new digital ulcers in patients with systemic sclerosis and ongoing digital ulcer disease, there are no FDA-approved medications for the treatment of digital ulcers in the United States; thus, it was not necessary to screen out drug claims for digital ulcers.
Demographic variables retrieved for each included patient were age, gender, geographic region, type of health plan, payer type, physician specialty associated with the index PAH prescription, and prior use of other PAH-related therapies (anticoagulants/heparin, calcium channel blockers, digoxin, diuretics). Clinical comorbidities of interest included congenital heart disease, connective tissue disorder, diabetes, drug or toxin ingestion, human immunodeficiency virus infection, liver disease, obesity, renal failure, right heart failure/cor pulmonale, and systemic hypertension (unconfirmed) (see Supplementary Table 1 in the online supplement for the complete list and diagnostic codes).
Outcomes
Outcomes assessed in this study were adherence to PAH-specific drugs, hospitalization and rehospitalizations, and healthcare resource use and associated costs. Adherence to index therapy was measured using the proportion of days covered (PDC) metric, calculated as the total days’ supply of the index therapy divided by 180 days (i.e. the duration of the post-index period). 21 For claims with days’ supply extending beyond day 180, only the portion of days’ supply captured within the 180-day window were included in PDC. Overlapping days from an early refill were counted only once from the end of the previous days’ supply. Patients were considered to be adherent to therapy if their PDC was ≥80%, the commonly accepted threshold for good adherence to PAH therapies16,17 and other pulmonary and cardiovascular medications.13,22
Hospitalizations and rehospitalizations within 30 days of discharge were quantified as rates (i.e. percentage of patients hospitalized), mean number of hospitalizations, and risk of hospitalization. Further details on the calculation of hospitalization risk are presented in the following “Statistical Analysis” section.
Healthcare resource use and costs were quantified for PAH-related therapies, emergency room visits, physician office visits, other outpatient visits, and hospital days. Cost data were adjusted for inflation using the medical care index of the Consumer Price Index and are reported as 2014 dollars.
Statistical analysis
Descriptive statistics are reported as counts and percentages for categorical variables, and mean and standard deviation (SD) for continuous variables. Univariate analyses for comparisons were performed using Chi-square or Fisher’s exact tests for categorical variables, and one-way ANOVAs, t-tests, or Wilcoxon Rank-Sum tests as appropriate for continuous variables.
The Charlson Comorbidity Index (CCI) was calculated using the updated methods of Quan et al., 23 based on medical activity within the 90 days prior to and including the index date.
Kaplan–Meier analysis was used to assess time to the first hospitalization that had diagnostic codes indicative of being PAH-related (ICD-9-CM 416.0 or 416.8) in either the first or second positions. Cox proportional hazards models were used to calculate adjusted hospitalization risk, with index treatment as the main effect and the following covariates: prior ERA or PDE-5i use, age, gender, region (Northeast, Midwest, South, West; Northeast as the reference), CCI (0 versus 1, 2–3 versus 1, ≥4 versus 1), treating physician specialty, pre-index hospitalization, and history of renal failure, congenital heart disease, respiratory disease, connective tissue disorder, clinical depression, and liver disease.
All statistical analyses were conducted using SAS® version 9.3 (SAS Institute Inc., Cary, NC).
Results
Patient characteristics
Ascertainment of the PAH patient cohort.
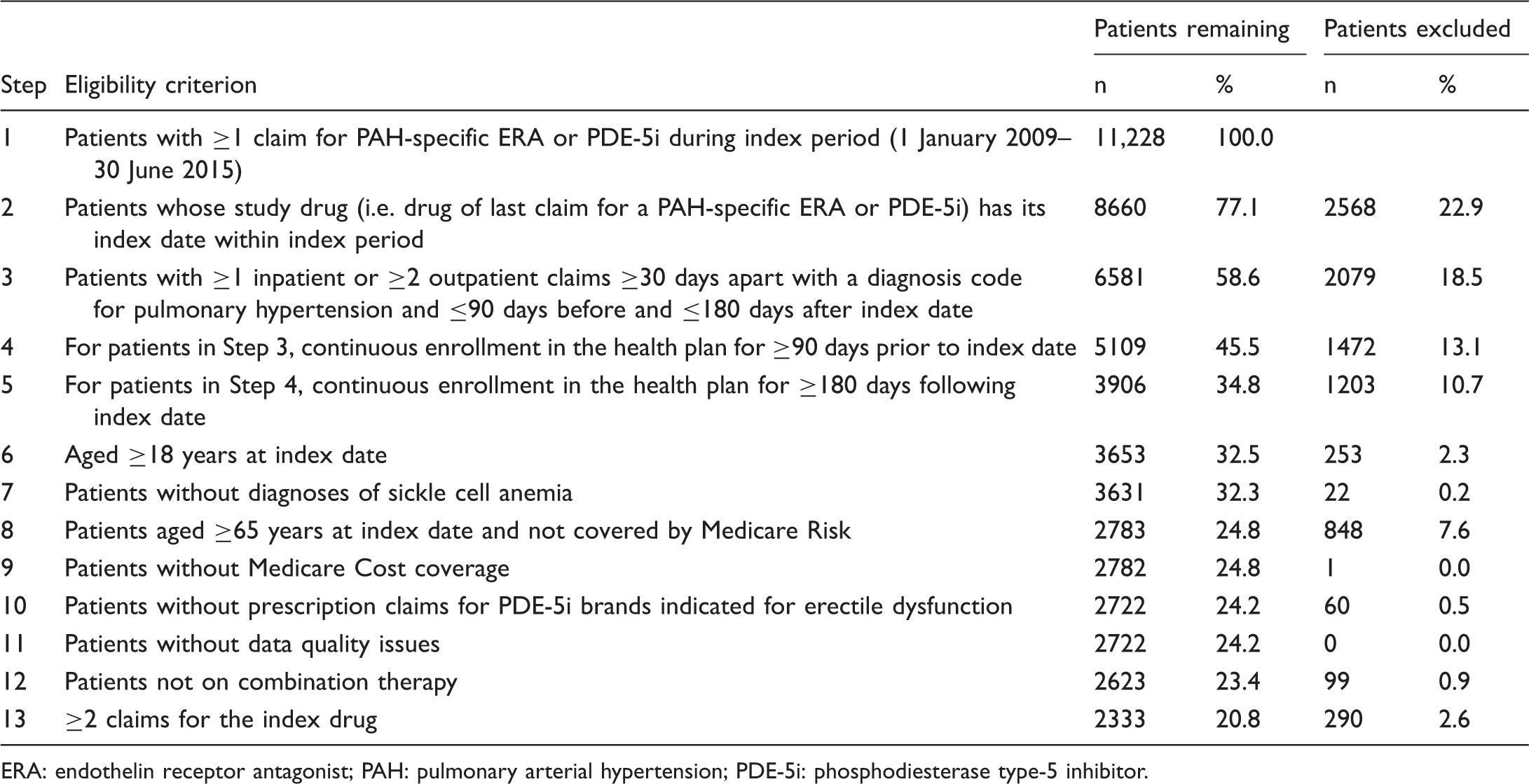
ERA: endothelin receptor antagonist; PAH: pulmonary arterial hypertension; PDE-5i: phosphodiesterase type-5 inhibitor.
Patient demographic characteristics.
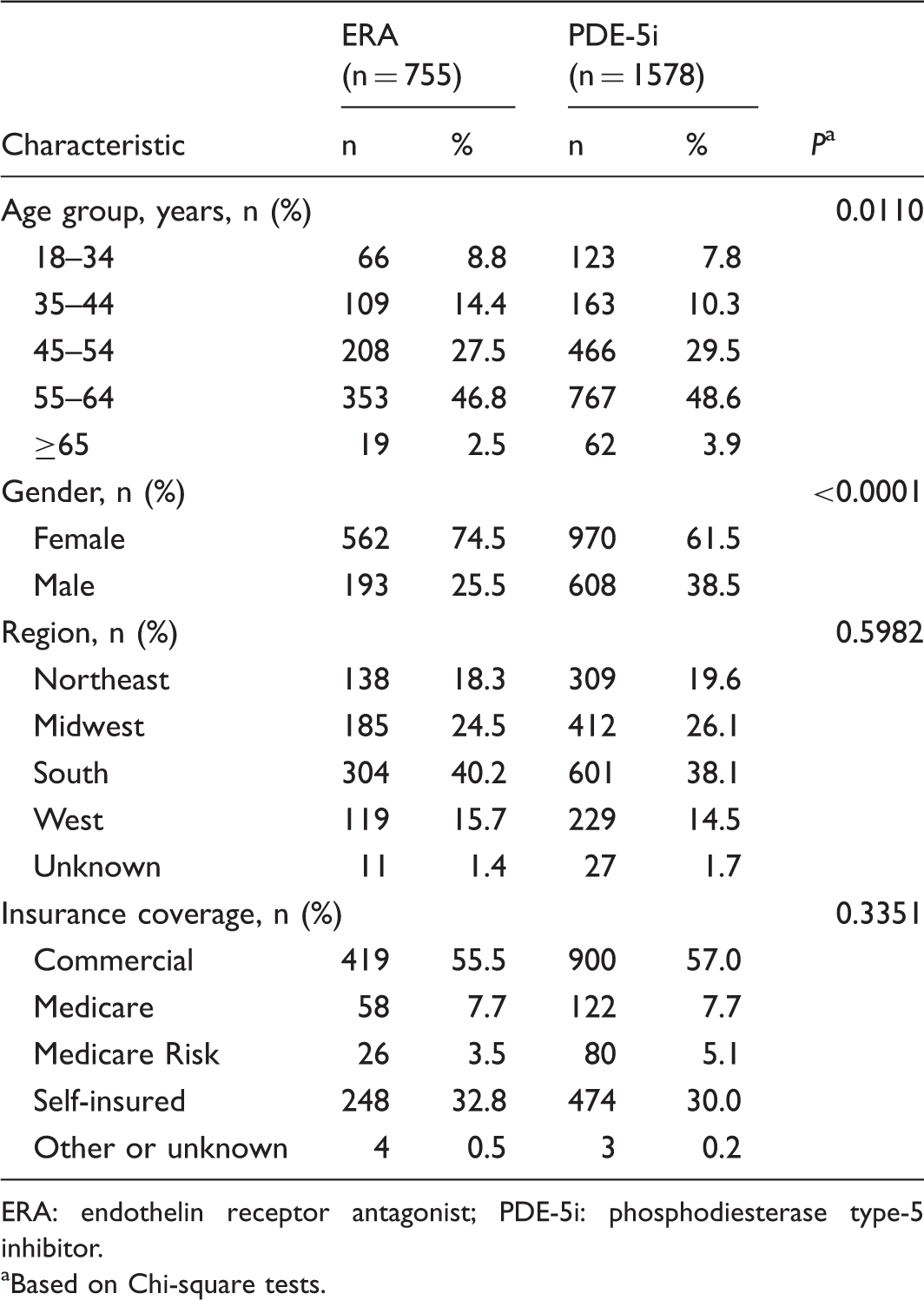
ERA: endothelin receptor antagonist; PDE-5i: phosphodiesterase type-5 inhibitor.
Based on Chi-square tests.
Patient comorbidities.
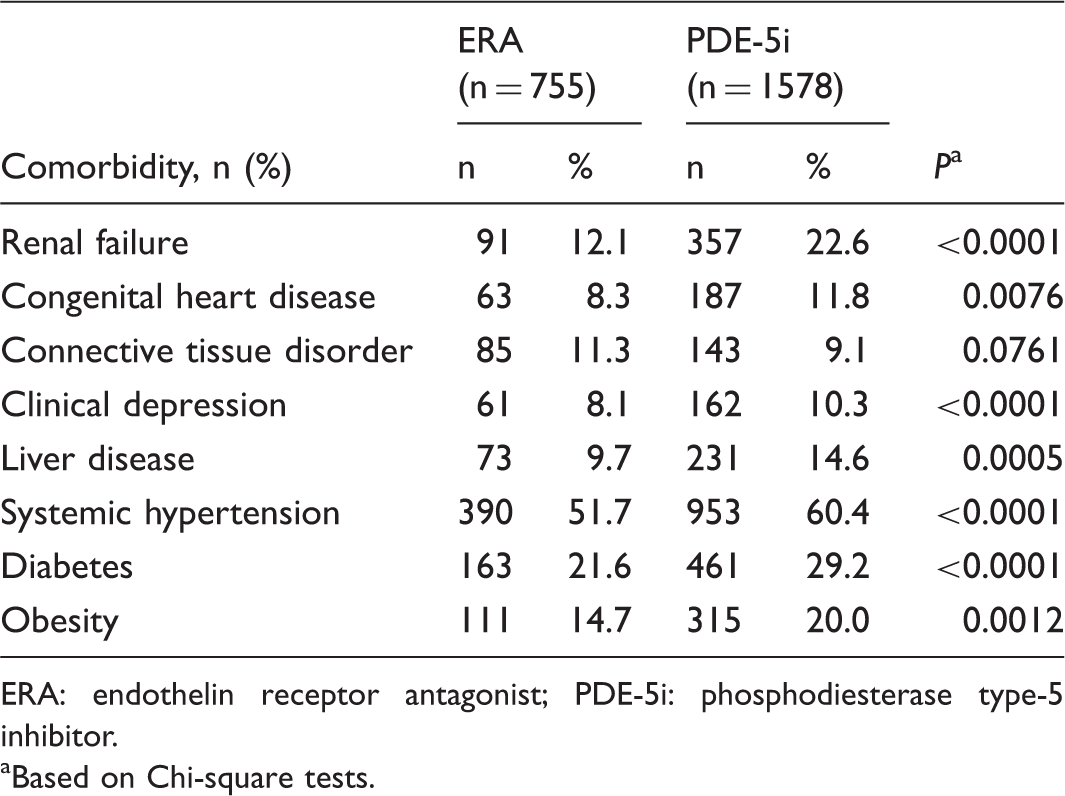
ERA: endothelin receptor antagonist; PDE-5i: phosphodiesterase type-5 inhibitor.
Based on Chi-square tests.
Adherence
As shown in Fig. 1, adherence to PAH-specific therapy differed between the two drug classes. In patients with ≥2 prescriptions for their index therapy, 75.6% (571/755) of patients with an index ERA were adherent (i.e. had PDC ≥80%) compared with 61.5% (970/1578) of patients receiving PDE-5is (P < 0.0001).
Distribution of patients by PDC in the two drug classes.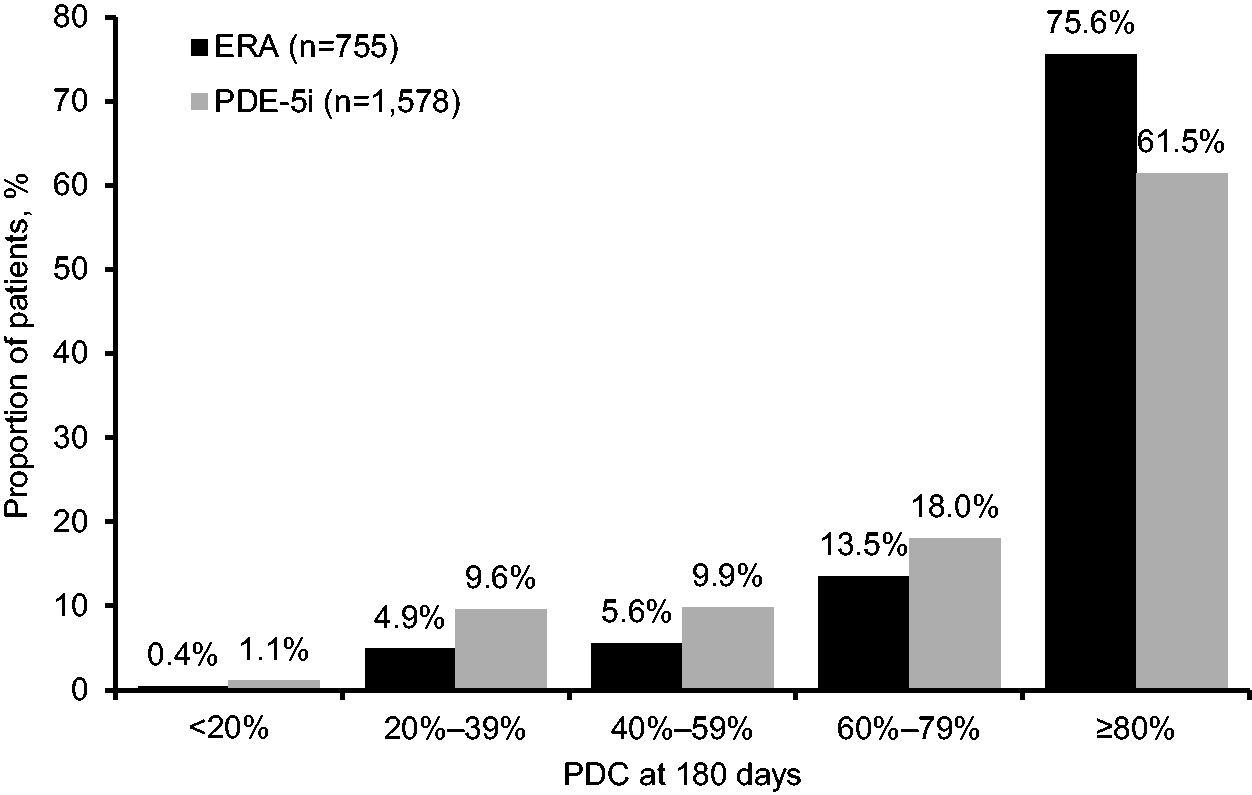
Hospitalization
Rate of hospitalizations and rehospitalizations within 30 days of discharge.
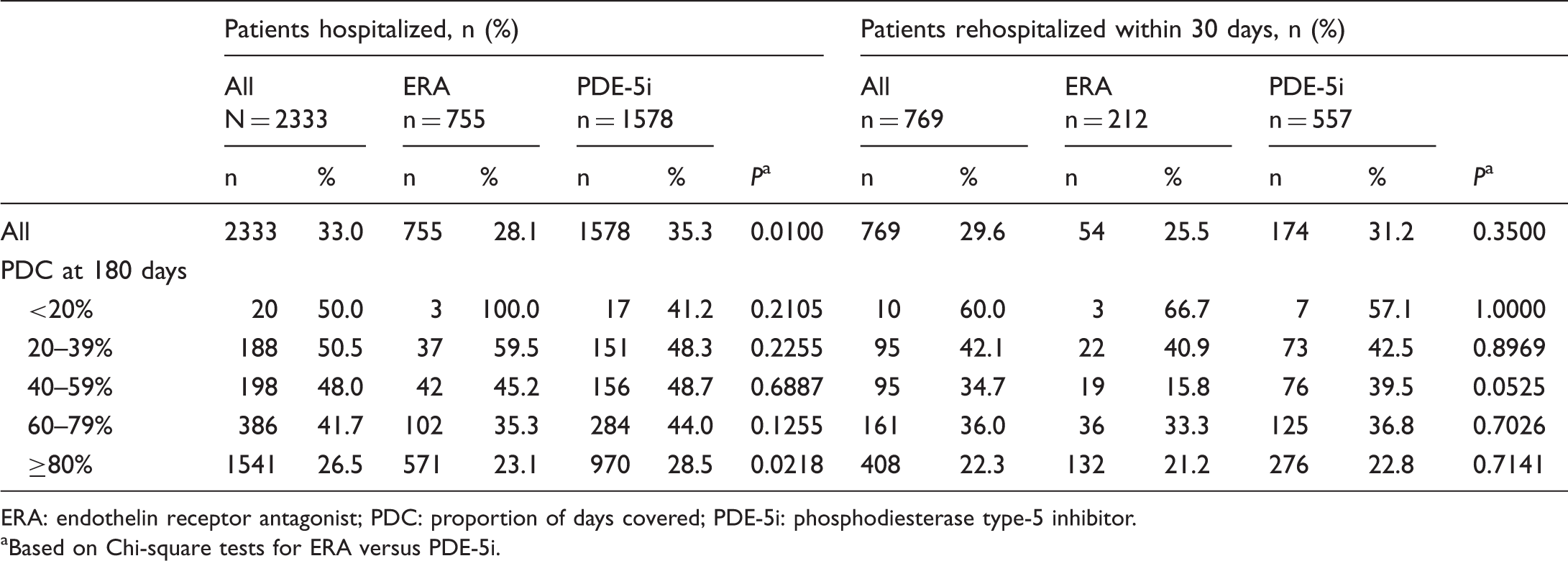
ERA: endothelin receptor antagonist; PDC: proportion of days covered; PDE-5i: phosphodiesterase type-5 inhibitor.
Based on Chi-square tests for ERA versus PDE-5i.
As reported in Table 4, among adherent patients, those on ERAs had a significantly lower hospitalization rate compared with those on PDE-5is (23.1% versus 28.5%, P = 0.0218). Based on the Cox model, an increase in PDC from 0.50 to 1.00 reduced risk of hospitalization by 58% for ERA patients and 26% for PDE-5i patients. Higher PDC was associated with a lower risk of hospitalization for ERA and PDE-5i, based on the hazard ratio (HR) of 0.549 for PDC (P = 0.018). Increasing adherence had a greater impact on reducing hospitalization risk in the ERA group than in the PDE-5i group (HR = 0.176 versus 0.549, P = 0.001), as calculated by multiplying the effect of higher PDC for PDE-5i patients (HR = 0.549) by the interaction effect for ERA versus PDE-5i (HR = 0.321).
Average number of hospitalizations and rehospitalizations within 30 days of discharge.
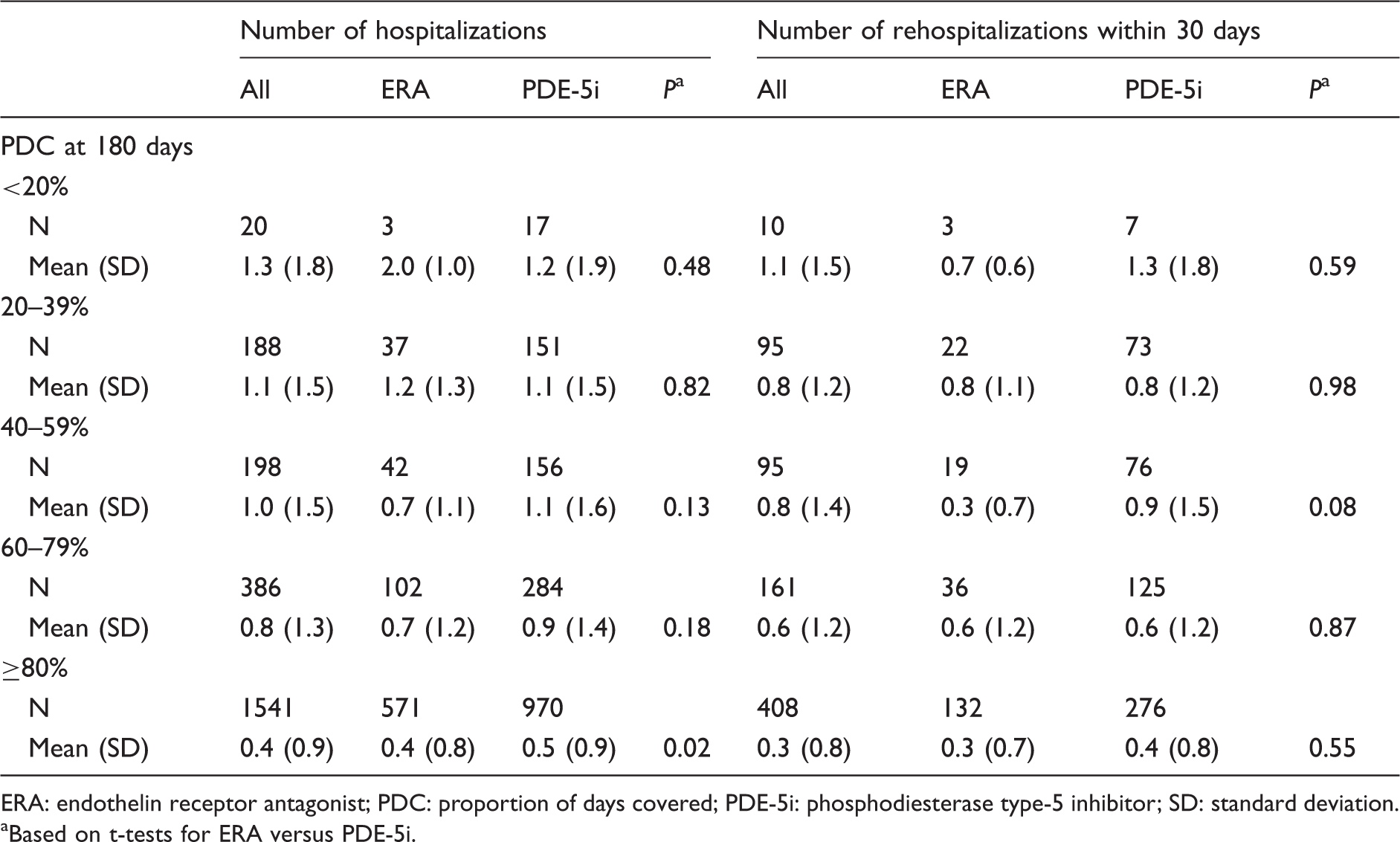
ERA: endothelin receptor antagonist; PDC: proportion of days covered; PDE-5i: phosphodiesterase type-5 inhibitor; SD: standard deviation.
Based on t-tests for ERA versus PDE-5i.
Rehospitalization
The percentage of patients rehospitalized within 30 days declined with increasing PDC, although in the ERA group this rate was lowest at 40–59% PDC (Table 4). Notably, this PDC category had a small sample size for the ERA group (n = 19). Rehospitalization rates were similar for ERA and PDE-5i patients, except in the 40–59% adherence category (Table 4).
The mean number of rehospitalizations generally decreased with increasing PDC, though for ERA patients it was the same for 40–59% and ≥80% PDC (Table 5). The mean number of rehospitalizations was generally similar for the ERA and PDE-5i groups (Table 5).
Healthcare resource use and costs
Relationship between adherence and healthcare resource utilization.
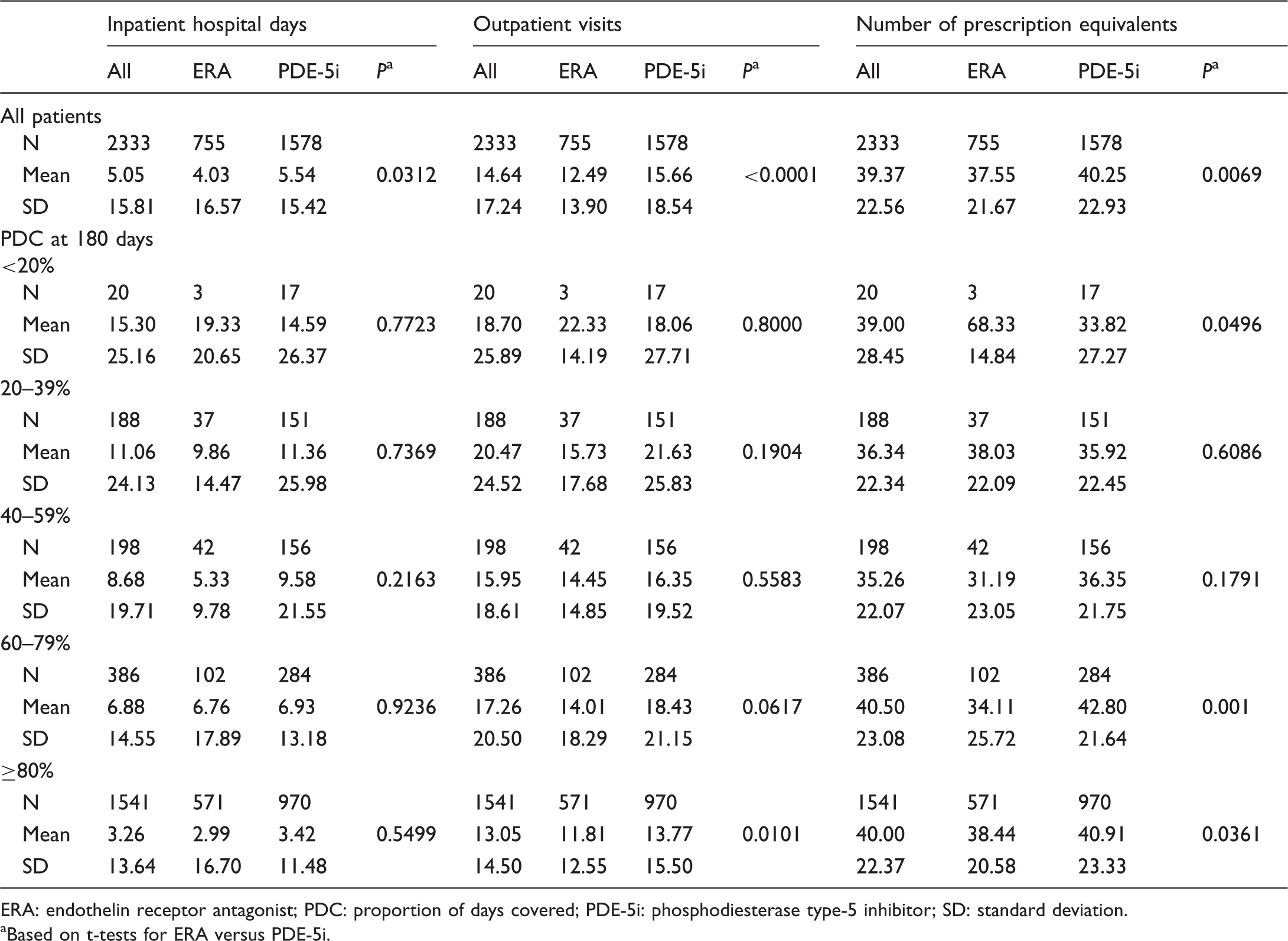
ERA: endothelin receptor antagonist; PDC: proportion of days covered; PDE-5i: phosphodiesterase type-5 inhibitor; SD: standard deviation.
Based on t-tests for ERA versus PDE-5i.
Healthcare costs are reported in Fig. 2. Mean total costs (medical + drugs) incurred during the six-month post-index period generally decreased with increasing PDC in the PDE-5i group (though with a smaller peak in the 60–70% category) but generally increased with increasing PDC in the ERA group (see Supplementary Table 2 in the online supplement for detailed results, including P values). Mean total costs during the six-month post-index period were higher in the ERA group than in the PDE-5i group in all patients ($91,328 versus $72,401, P = 0.0003) and in adherent patients ($88,867 versus $56,300, P < 0.0001), but did not differ significantly between drug classes in other PDC categories.
Healthcare costs by drug class and PDC.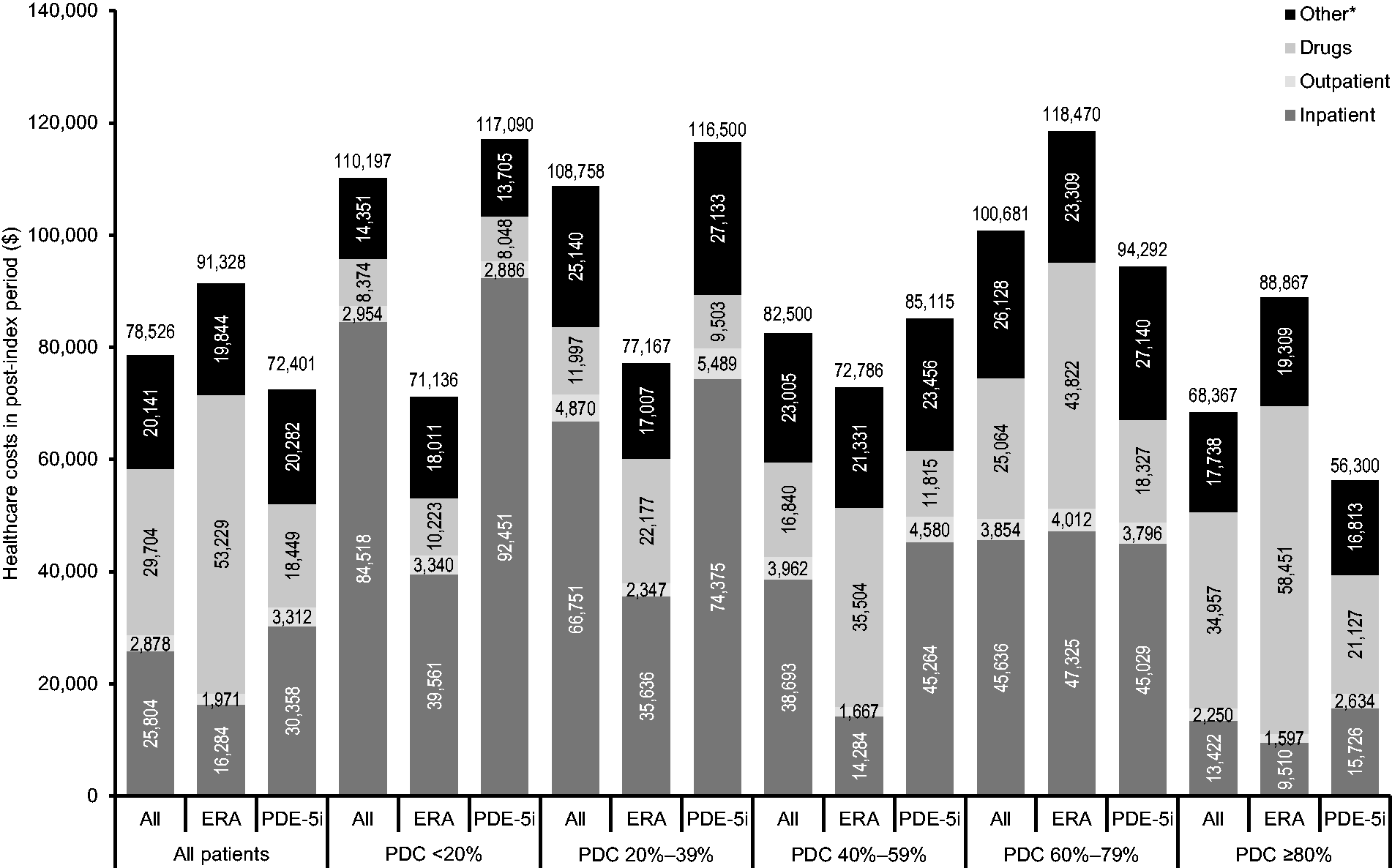
Mean hospitalization costs during the six-month post-index period were significantly lower in the ERA group than in the PDE-5i group for all patients ($16,284 versus $30,358, P = 0.0015) and for the subgroup of patients classified as adherent based on having PDC ≥ 80% ($9510 versus $15,726, P = 0.0318) (Fig. 2 and Supplementary Table 2). Conversely, mean drug costs were higher in the ERA group compared with the PDE-5i group for all patients ($53,229 versus $18,449, P < 0.0001) and for the subgroup of adherent patients ($58,451 versus $21,127, P < 0.0001). Mean outpatient costs incurred during the six-month post-index period were lower in the ERA group than in the PDE-5i group in all patients ($1971 versus $3312, P < 0.032), but did not differ significantly between drug classes in specific PDC categories. Other healthcare costs did not differ significantly between the two drug classes.
Discussion
These analyses suggest that ERAs and PDE-5is may be associated with differing levels of medication adherence and thus have different clinical and economic outcomes in real-world practice. Our findings of higher adherence with ERAs than with PDE-5is are consistent with those reported (thus far only in abstract form) in recent retrospective database analyses.18,19 An analysis by Hull et al. 18 of 2010–2015 data from the Optum Research Database estimated a mean PDC of 0.8 with ERAs versus 0.6 with PDE-5is (P < 0.001). A different analysis by Leo et al. 19 of 2010–2015 data from an unspecified database reported PDCs of 61.3–75.1% for individual PDE-5is and 67.4–97.0% for individual ERAs.
Our results also support findings reported by Leo et al. 19 that increasing adherence is associated with a reduction in hospital admissions. In that analysis, every 46% increase in PDC was associated with 0.5 fewer hospitalizations per patient. The present study also found that increasing adherence was associated with a reduction in the risk of hospitalization. Furthermore, we observed a greater reduction in risk of hospitalization for the same degree of improvement in adherence in patients receiving ERAs compared with those receiving PDE-5is. The reason for this difference in the magnitude of risk reduction with improved adherence is unclear, but may reflect the difference in comorbidities between the groups, with the PDE-5i group being older, more often male, and with higher prevalence of diabetes, obesity, and renal insufficiency. These risk factors may associate with greater presence of left heart disease complicating the pulmonary hypertension, making these patients less likely to benefit from PAH-specific therapies.
In addition, the prevalence of clinical depression was higher in the PDE-5i group compared with the ERA group. Comorbid depression has previously been shown to be associated with lower adherence to medications for other chronic diseases, including chronic obstructive pulmonary disease (COPD) and diabetes.24,25 Although our analyses adjusted for presence/absence of depression, they may have been influenced by unmeasured depression-related factors, such as adherence to antidepressants. A study of comorbid depression in patients with COPD found that high adherence (≥80% PDC) to antidepressants was associated with a lower risk of hospitalizations compared with no antidepressant use. 26
The reduction in hospitalization with higher PDC for ERAs and PDE-5is points to the potential benefit of removing barriers to adherence. Gaps in insurance coverage and high copayments for PAH drugs are potential financial barriers that can lead to treatment interruption. 14 In a retrospective database analysis, Waxman et al. 17 found that the rate of good adherence to PDE-5is (i.e. PDC ≥80%) was lower in PAH patients who had higher copayments. Patient out-of-pocket expenses depend on drug prices, the extent of insurance coverage (including reimbursement policies, copayment thresholds, and benefit caps), and any applicable patient support programs. 27 These considerations are of particular concern since a number of PAH-specific drugs have recently come off patent, which may trigger the ending of patient support programs from pharmaceutical manufacturers once generic versions of their products become available—generic manufacturers typically do not offer patient assistance programs. 28 Although underinsured patients may apply to copayment foundations for third-party assistance, 29 one such organization, the Caring Voice Coalition, is no longer providing financial support due to the decision by the US Department of Health and Human Services to rescind their Advisory Opinion. 30 Efforts may be underway to seek changes in the current regulations preventing pharmaceutical companies from offsetting copayments for Medicare recipients, which if instituted may improve adherence and reduce hospitalizations in these patients.
Hospitalizations and drug therapy were the main cost drivers in this study, accounting for 32.9 and 37.8% of total costs, respectively. Despite reduced hospitalization costs in the ERA group, higher drug costs resulted in higher total costs compared with the PDE-5i group, implying a net cost to achieving improved patient outcomes with ERAs.
Although hospitalization in patients with PAH has previously been shown to lead to readmission and thus higher costs later, 11 which would lead to the expectation that rehospitalizations should be less frequent with ERAs than with PDE-5is, we did not observe this trend. However, the follow-up in the present study was limited to six months, and thus all rehospitalizations could not be captured. Consequently, the true extent of rehospitalization and its associated cost may have been underestimated.
The primary limitation of this study is the lack of a unique ICD-9-CM code for PAH. 31 Consequently, patients with PAH had to be identified based on an algorithm using ICD-9-CM codes for pulmonary hypertension and drugs approved for treating PAH, which could have led to diagnostic misclassification. An analysis of medical charts for 43 patients ascertained to have PAH on the basis of ICD-9 codes and claims for PAH-specific drugs found that 42 (98%) had a documented PAH diagnosis. 32 However, a recent study by Papani et al., 33 in which claims-based algorithms were cross-checked against medical records for 683 patients, found that algorithms incorporating ICD-9-CM codes 416.0 and 416.8 plus at least one prescription for a PAH-specific medication had a specificity for hemodynamically diagnosed PAH of 82–87%, thus misidentifying a substantial percentage of patients with other forms of pulmonary hypertension. This potential misclassification could have led to violation of the assumption that both groups in our study (i.e. ERA and PDE-5i recipients) are equally likely to represent patients with true PAH diagnoses, i.e. patients in Group 1 of the WSPH classification scheme for pulmonary hypertension. 20
An important corollary of misidentification is that if the PDE-5i group had a higher proportion of patients with non-PAH forms of pulmonary hypertension, this could account in part for poorer adherence and higher hospitalization rates. Use of PAH-specific therapy for patients with other forms of pulmonary hypertension is common in the US, as shown by a 2011 survey in which 28 of 30 responding pulmonary hypertension specialty centers reported using PAH medications in at least some patients with non-Group 1 pulmonary hypertension. 34 However, the Choosing Wisely Top Five campaign of the American Thoracic Society and the American College of Chest Physicians recommends against routinely using advanced vasoactive agents approved only for PAH to patients with pulmonary hypertension resulting from left heart disease or hypoxemic lung diseases, because of potential harm and lack of established benefit in these patients (as well as substantial cost). 35 In real-world practice, inappropriate use would be expected to drive both treatment discontinuation and hospitalization. Furthermore, a higher proportion of patients with non-Group 1 pulmonary hypertension in the PDE-5i group than in the ERA group could partially explain why improving adherence brought less benefit in terms of reducing hospitalization in the PDE5i group.
Although it is not possible to quantify what percentage of patients in our analysis were misclassified, and thus to confirm greater representation of non-PAH forms of pulmonary hypertension in the PDE-5i group, a previous cross-sectional study at three US pulmonary hypertension centers in 2010–2011 found that a higher proportion of prescriptions for PDE-5is than for ERAs went to patients with non-Group 1 pulmonary hypertension: 42% versus 29%, respectively. 36 Potentially inappropriate prescribing may be even more common in other settings. For instance, in a retrospective analysis of phosphodiesterase-5 inhibitor prescribing for Group 1 PAH versus other forms of pulmonary hypertension in the Veterans Health Administration between 2005 and 2012, Kim et al. 37 estimated that approximately 80% of pulmonary-hypertension-related PDE-5i prescriptions went to patients with forms of pulmonary hypertension other than PAH. The authors noted that such use exposes patients to potential harm and imposes a financial burden on the healthcare system.
Our study was also subject to other limitations. Patients aged ≥65 years are under-represented in the database due to the exclusion of individuals in this age group with Medicare coverage other than Medicare Advantage commercial insurance, which may limit the generalizability of the findings. Furthermore, the claims data are collected for insurance payments rather than for research, and may be subject to coding error (we censored only patients with missing or bad data elements). Finally, as is true for all retrospective database analyses, it is not possible to draw firm conclusions regarding the causality of the associations we found.
In conclusion, this research indicates that higher adherence to ERAs and PDE-5is in patients with PAH is associated with a lower risk of hospitalization and rehospitalization, with a greater risk reduction seen in the patients who had better adherence to ERAs compared with PDE-5is. These results support maximizing adherence as an important strategy to improve outcomes and reduce hospitalization costs for these patients.
Supplemental Material
PUL880086 Supplemental material - Supplemental material for Medication adherence, hospitalization, and healthcare resource utilization and costs in patients with pulmonary arterial hypertension treated with endothelin receptor antagonists or phosphodiesterase type-5 inhibitors
Supplemental material, PUL880086 Supplemental material for Medication adherence, hospitalization, and healthcare resource utilization and costs in patients with pulmonary arterial hypertension treated with endothelin receptor antagonists or phosphodiesterase type-5 inhibitors by Robert P. Frantz, Jerrold W. Hill, Cassandra A. Lickert, Rolin L. Wade, Michele R. Cole, Yuen Tsang and William Drake III in Pulmonary Circulation
Footnotes
Acknowledgements
The authors would like to thank Jing He at IQVIA for data management and programming support.
Conflict of interest
RPF has steering committee and advisory board participation for Actelion Pharmaceuticals Ltd; steering committee and data and safety monitoring board participation for United Therapeutics Corporation; and has received consulting fees and/or coverage of travel expenses from Arena Pharmaceuticals, Bayer Corporation, and Abbott. RLW is an employee of IQVIA, a consultancy that received payment from Actelion Pharmaceuticals US, Inc. to conduct this research. JWH was an employee of IQVIA at the time this research was conducted. CAL, MRC, YT, and WD are employees of Actelion Pharmaceuticals US, Inc. CAL owns shares in Actelion.
Contributorship
JWH, CAL, RLW, MRC, and WD were involved in study conception and design. YT contributed to study design. JWH, RLW, and WD participated in data acquisition and analysis. RPF, JWH, CAL, MRC, YT, and WD contributed to data interpretation. All authors participated in drafting the manuscript and/or revising it critically for important intellectual content, approved the final version for publication, and agree to take public responsibility for appropriate portions of the content.
Ethical approval
This study is a retrospective claims database analysis and thus does not contain any experiments on human or animal subjects for which ethical approval is required.
Funding
This research was sponsored by Actelion Pharmaceuticals US, Inc. Actelion contracted IQVIA to analyze the data, and funded manuscript development and publication. The authors had the right to publish these data independent of the sponsor. Medical writing and editorial support were provided by W. Mark Roberts, PhD, Montréal, Québec, Canada, funded by Actelion Pharmaceuticals US, Inc.
Guarantor
RLW is the guarantor for this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
