Abstract
Limited evidence is available on outcomes associated with currently available medications from the endothelin receptor antagonist drug class (bosentan, ambrisentan, and macitentan) in elderly patients with pulmonary arterial hypertension. We evaluated mortality in predominantly elderly patients with pulmonary arterial hypertension in the US taking endothelin receptor antagonists. A retrospective administrative claims study was conducted using the Centers for Medicare and Medicaid Services national Medicare database. Patients with pulmonary arterial hypertension were identified using diagnostic codes. Cohort inclusion required age ≥18 years; ≥1 claim for macitentan, ambrisentan, or bosentan between 1 January 2014 and 31 December 2015 (index date and index endothelin receptor antagonist defined by first such claim); continuous enrollment for ≥12 months before and after the index date; and ≥80% of days covered for the index endothelin receptor antagonist. Follow-up was from index date until the earliest of Medicare disenrollment, death, or 31 December 2016. Multivariable Cox proportional hazards regression models were used to estimate mortality hazard ratios with 95% confidence intervals for macitentan vs. ambrisentan or bosentan, adjusting for potential confounders. The study cohort included 1628 patients on index macitentan, 2852 on ambrisentan, and 1972 on bosentan. Overall, 69% of patients were aged ≥65 years and most were females (76%). Macitentan was associated with an 18% lower risk for mortality than ambrisentan (hazard ratio: 0.82, 95% confidence interval: 0.72–0.93; P = 0.0026) and a 39% lower risk than bosentan (hazard ratio: 0.61, 95% confidence interval: 0.53–0.71; P < 0.0001). Variables that independently increased the risk of mortality included higher comorbidity index, increasing age, and inpatient hospitalizations at baseline.
Introduction
Pulmonary arterial hypertension (PAH) is a rare and debilitating chronic disease, characterized by vascular proliferation and remodeling of the small pulmonary arteries.1,2 The clinical profile of patients with PAH being treated in clinical practice has changed substantially over recent decades, with an increasing prevalence of elderly patients.3,4 Elderly patients with PAH have a higher rate of comorbid conditions compared with younger patients, introducing more complications into their treatment due to considerations such as polypharmacy and drug–drug interactions. 5 Although research has indicated that comorbidities negatively affect outcomes in patients with PAH, there is limited information regarding the effect of comorbidities on survival in elderly patients.6,7
Evidence is also sparse on the mortality benefit of PAH therapy in elderly patients not only because the low prevalence of PAH makes it unfeasible to recruit sufficient patients of any age to detect survival differences in a randomized controlled trial (RCT) but also because elderly patients with multiple comorbidities are typically excluded from PAH RCTs. 8 Additionally, even if elderly patients are included in PAH trials, few RCTs have been long enough to detect a difference in survival, and crossover of patients with clinical worsening to rescue therapy reduces the ability of these trials to demonstrate a difference in survival between treatment arms. 9 This represents an important evidence gap, because registry studies have demonstrated that advanced age is a determinant of poorer survival in PAH,7,10 and thus there is a clear need to evaluate the effect of PAH-specific therapies in older patients.
Despite this information gap, treatment goals for elderly patients are similar to those for younger patients with PAH and include delaying progression and achieving and maintaining good exercise capacity, quality of life, and right-ventricular function, with the ultimate aim of improving survival.11–13 Endothelin receptor antagonists (ERAs) are commonly used to treat patients diagnosed with PAH, and recommendations for their use are included in current clinical practice guidelines.11,14
Currently available ERAs differ in their endothelin receptor specificity and binding properties, tissue penetration, and other pharmacologic characteristics, which theoretically could be associated with different outcomes.15,16 However, survival was not assessed as a separate endpoint in the pivotal trials for the ERAs bosentan (Study 351 and BREATHE-1) and ambrisentan (ARIES-1 and -2).17–19 The primary endpoint for all of these trials was a change in exercise capacity (6-min walk distance). Time to clinical worsening was included as a composite secondary endpoint in BREATHE-1 and ARIES-1 and -2. SERAPHIN, the pivotal trial for the ERA macitentan, was a long-term, event-driven trial with a composite morbidity and mortality primary endpoint. 20 However, none of these ERA trials were powered to detect a survival benefit, and there have been no head-to-head RCTs comparing bosentan, ambrisentan, and macitentan on any endpoint. Therefore, the relative treatment effect of the three ERAs on mortality remains unknown.
To fill the evidentiary gap, real-world evidence such as data gathered from registries and claims databases is an important resource to examine survival and is increasingly being considered in regulatory decision-making.21–23 Such databases are important resources to examine survival in elderly PAH patients. For example, data from the REVEAL registry have been used to predict what survival in the SERAPHIN population would have been if patients had no access to macitentan, for comparison with observed survival in SERAPHIN to estimate the treatment effect of macitentan on survival. 9 Additionally, the OPUS registry provides valuable real-world data on the use of macitentan in a population over 65 years of age. 24 However, neither REVEAL nor OPUS allows a comparison of mortality rates among elderly patients as contemporary ERA therapy has evolved, or an assessment of whether there are potential differences in outcomes with different ERAs. Thus, we conducted a retrospective database study to examine the risk of mortality for predominantly elderly PAH patients treated with ERAs.
Methods
Data source
Data were retrieved from the Centers for Medicare and Medicaid Services (CMS) national Medicare database, which contains longitudinal medical and pharmaceutical administrative claims and death records for the US Medicare population, comprising approximately 56 million covered lives during the study period. All data used in these analyses have been anonymized and the database is fully compliant with the Health Insurance Portability and Accountability Act (HIPAA) Privacy Rules. This retrospective database analysis did not involve the collection, use, or transmittal of individual identifiable data. As such, a full institutional review board review to conduct this study was not required and the study is considered exempt according to 45CFR46.101(b)(4): Existing Data & Specimens—No Identifiers.
Study design and sample
Data were retrieved for the study period of 1 January 2013 through 31 December 2016 (Fig. 1). Our analysis did not include more recent data due to the two-year lag for the release of Medicare records by the CMS.
Study design. PH: pulmonary hypertension. *Date of first claim for macitentan, ambrisentan, or bosentan during the identification period.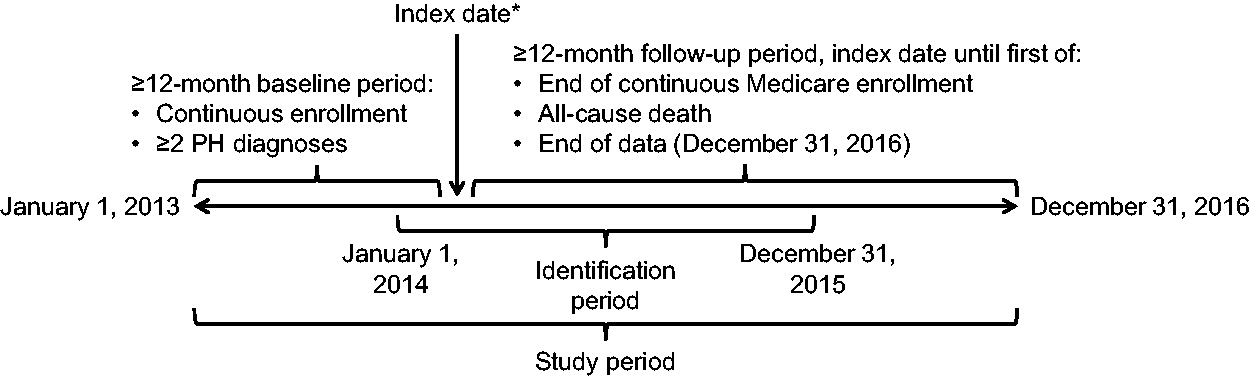
For each patient, the index date was defined as the first date of a claim for macitentan, ambrisentan, or bosentan within the identification period of 1 January 2014 to 31 December 2015, allowing a ≥12-month baseline period prior to index date and a ≥12-month follow-up period before the end of the study period. Follow-up for each patient was from the index date until the earliest of Medicare disenrollment, death, or end of study data (31 December 2016).
Eligible patients were aged at least 18 years at the index date (Medicare includes patients aged <65 years with disabilities and patients aged ≥65 years) and were required to have had continuous health plan enrollment with medical and pharmacy benefits (Part A, B, and D) during the baseline period and for at least 12 months after the index date. However, patients who died before 12 months of follow-up were also eligible.
To be included, patients had to have at least one pharmacy claim for macitentan, ambrisentan, or bosentan during the identification period, with the date of the first claim for one of these ERAs during the identification period designated as the index date and this ERA designated as the index drug. Patients were required to have a proportion of days covered (PDC) of at least 80% for the index ERA during the follow-up period. However, in cases where a patient was prescribed multiple ERAs during the identification period, one-year PDC was calculated for all the ERA prescriptions for that patient, and the patient was included in the ERA cohort with the highest PDC, which was still required to be at least 80%. The date of the first prescription claim for that cohort was then defined as the index date.
Additional inclusion criteria were at least two medical claims in the baseline period or on the index date with an International Classification of Diseases, Ninth or Tenth Revision, Clinical Modification diagnosis code (ICD-9-CM or ICD-10-CM, respectively) for pulmonary hypertension (PH), namely ICD-9-CM 416.0 (primary PH) or 416.8 (other chronic pulmonary heart disease. PH, secondary), or ICD-10-CM I27.0 (primary PH), I27.2 (other secondary PH), or I27.89 (other specified pulmonary heart diseases).25,26
Right heart catheterization (RHC) was not an inclusion criterion but was identified by Current Procedural Terminology code (Supplemental Table 1) and reported to describe the patient sample. Patients were excluded if they had any claim with a diagnosis code for left-sided heart failure during the baseline period (ICD-9-CM 428.1 or ICD-10-CM I50.1).
Patient baseline characteristics.
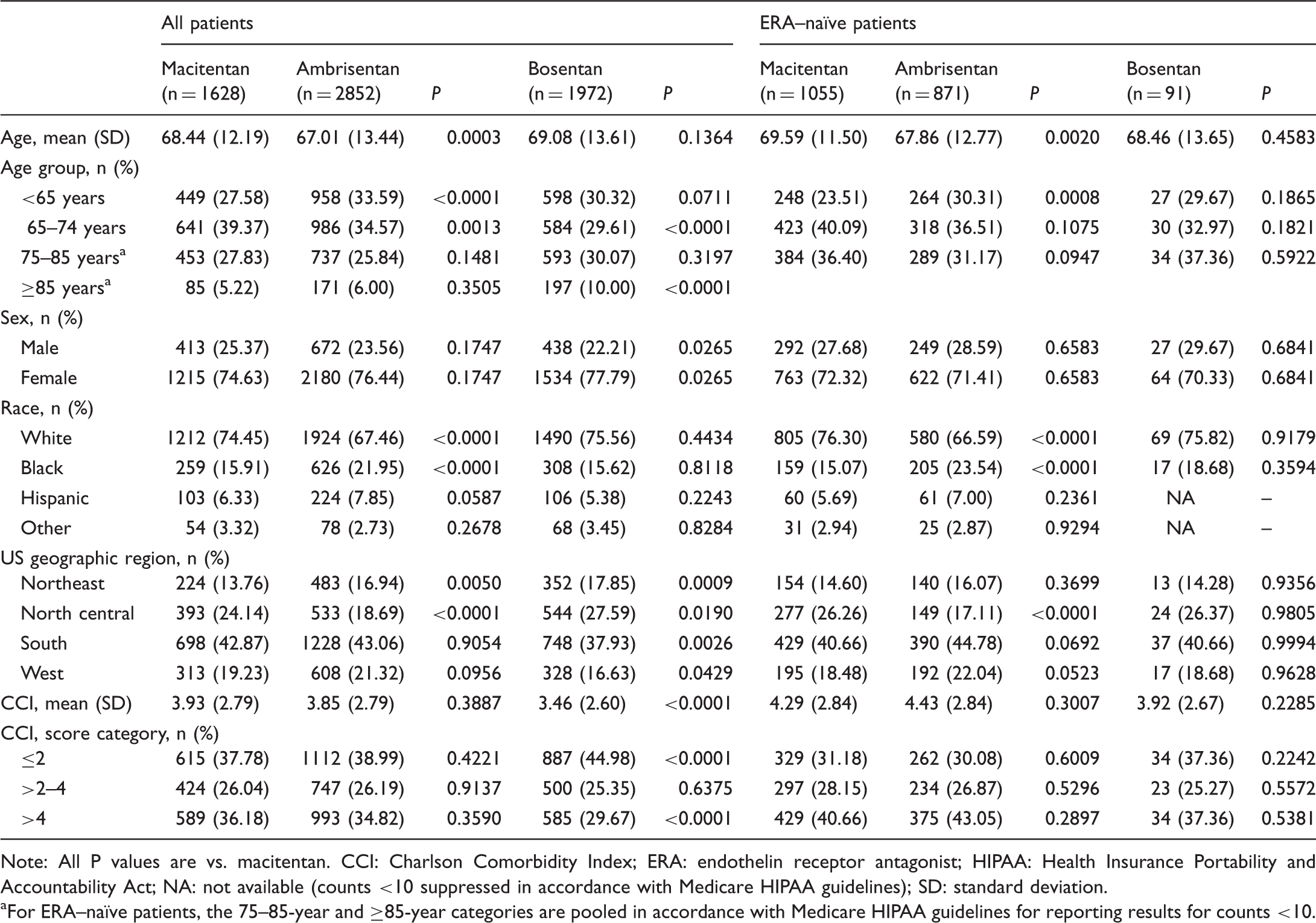
Note: All P values are vs. macitentan. CCI: Charlson Comorbidity Index; ERA: endothelin receptor antagonist; HIPAA: Health Insurance Portability and Accountability Act; NA: not available (counts <10 suppressed in accordance with Medicare HIPAA guidelines); SD: standard deviation.
For ERA–naïve patients, the 75–85-year and ≥85-year categories are pooled in accordance with Medicare HIPAA guidelines for reporting results for counts <10.
PAH medications of interest were the available ERAs (oral ambrisentan, bosentan, and macitentan), phosphodiesterase type 5 (PDE-5) inhibitors (oral sildenafil and tadalafil), prostacyclins (subcutaneous, intravenous, inhaled, and oral treprostinil, inhaled iloprost, intravenous epoprostenol), and the oral soluble guanylate cyclase stimulator riociguat. Drugs were identified from pharmacy claims by National Drug Code and from medical claims by Healthcare Common Procedure Coding System codes (Supplemental Table 1).
Outcomes
The outcome of interest in these analyses was mortality. All deaths during the follow-up period were counted (causes of death were not able to be identified). Mortality rate was calculated as events per 100 person-years. Time from the index date to death was compared between the three index-ERA cohorts. Results were analyzed for all patients and for patients who were not prescribed ERAs during the baseline period, referred to henceforth as the ERA–naïve population. Notably, ERA–naïve patients could either have been naïve to any PAH-specific therapy or have previously received PAH medications other than ERAs.
Statistical analysis
Descriptive statistics are reported for patient baseline characteristics. Mean and standard deviation are presented for continuous variables, and counts and percentages are presented for categorical variables. Counts of less than 11 were suppressed and reported as not available, in accordance with Medicare HIPAA guidelines for reporting results.
Between-group comparisons were performed using Student’s t-tests for continuous variables and chi-square tests for categorical variables.
Unadjusted Kaplan–Meier (KM) estimates of survival probabilities were plotted to assess the time to death. ERA medication discontinuation was not included as a censoring rule in the development of the KM curves because of the inclusion criterion requiring all patients in the analysis to have a PDC of at least 80%.
Multivariable Cox proportional hazards regression models were used to estimate hazard ratios (HRs) with 95% confidence intervals (CIs) for the risks of death in patients prescribed macitentan, ambrisentan, or bosentan after adjustment for potential confounders. Covariates included in the models were age, gender, geographic region, baseline comorbidities, and baseline healthcare resource utilization. Covariate selection for each model used a stepwise regression procedure. All statistical analyses were conducted using SAS® version 9.4 (SAS Institute Inc., Cary, NC).
Results
Patient characteristics
After the application of all eligibility criteria, 6452 patients were included in the overall population (Fig. 2): 1628 in the macitentan cohort, 2852 in the ambrisentan cohort, and 1972 in the bosentan cohort. The ERA–naïve population comprised 1055 patients in the macitentan cohort, 871 in the ambrisentan cohort, and 91 in the bosentan cohort. Patients were identified over a two-year period and followed for at least one year post-index date, resulting in a mean length of follow-up of 21 to 28 months across the different cohorts.
Ascertainment of the patient population. ERA: endothelin receptor antagonist; PAH: pulmonary arterial hypertension; PDC: proportion of days covered; PH: pulmonary hypertension. *Includes patients who died before 12 months of follow-up.
Patients were predominantly females (76%), and the proportion of women was slightly but significantly higher in the bosentan cohort than in the macitentan cohort (Table 1). Overall, 69% of patients were aged 65 years or older, and on average patients were slightly but significantly older in the macitentan cohort than in the ambrisentan cohort (Table 1). Most patients were white (72%), and the proportion of non-white patients was 7% higher in the ambrisentan cohort compared with the macitentan cohort (Table 1).
Detailed patient comorbidity results in the baseline period are reported in Fig. 3 and Supplemental Table 2. The most common comorbidity was hypertension, seen in 78% to 84% of patients across cohorts in the overall population and 79% to 89% in the ERA–naïve population. Compared with the bosentan cohort, the macitentan cohort had a significantly higher Charlson comorbidity score (Table 1).
Comorbidities during the 12-month baseline period in (a) the overall patient population and (b) the ERA–naïve subgroup. CAD: coronary artery disease; COPD: chronic obstructive pulmonary disease; CTD: connective tissue disorder; ERA: endothelin receptor antagonist; ILD: interstitial lung disease. *P < 0.05, macitentan vs. ambrisentan. †P < 0.05, macitentan vs. bosentan. ‡≥3 of the following: hypertension, diabetes, obesity, and CAD.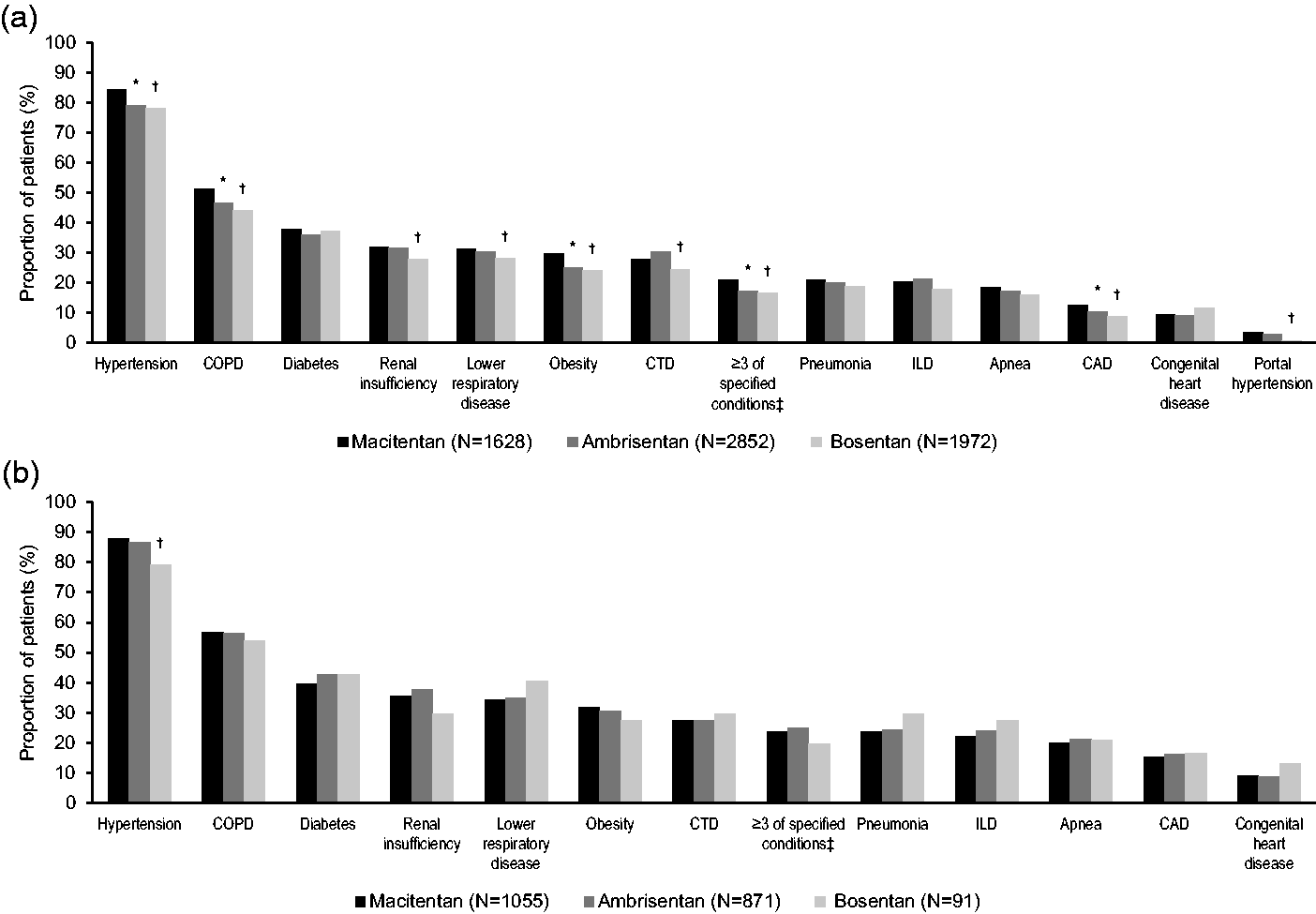
RHC during the baseline period appeared to be more common in the macitentan cohort (37%) than in the ambrisentan or bosentan cohorts (26% and 12%, respectively; Supplemental Table 3). However, it should be noted that we would not have captured RHC procedures performed before the study period. Higher rates of missing RHC would be expected in cohorts with a higher proportion of patients diagnosed earlier.
PH medication use during the baseline period
The most commonly used PAH medications during the 12-month baseline period were PDE-5 inhibitors, which were used by approximately 45% of patients (Fig. 4 and Supplemental Table 2). In the overall patient population, the use of any ERA before the index date was less common among patients with macitentan as their index ERA (35%), than among those with ambrisentan or bosentan as their index drug (69% and 95%, respectively), likely attributable to the more recent Food and Drug Administration approval of macitentan (2013) compared with ambrisentan (2007) and bosentan (2001
30
; Supplemental Table 3). Among patients with previous ERA use, bosentan had been used in the baseline period by more than 99% of patients with bosentan as their index drug, compared with 78% and 6% of those with macitentan and ambrisentan, respectively, as their index drug.
Utilization of pulmonary hypertension medications during the 12-month baseline period in (a) the overall patient population and (b) the ERA–naïve subgroup. ERA: endothelin receptor antagonist; PDE-5: phosphodiesterase type 5. *P < 0.05, macitentan vs. ambrisentan. †P < 0.05, macitentan vs. bosentan.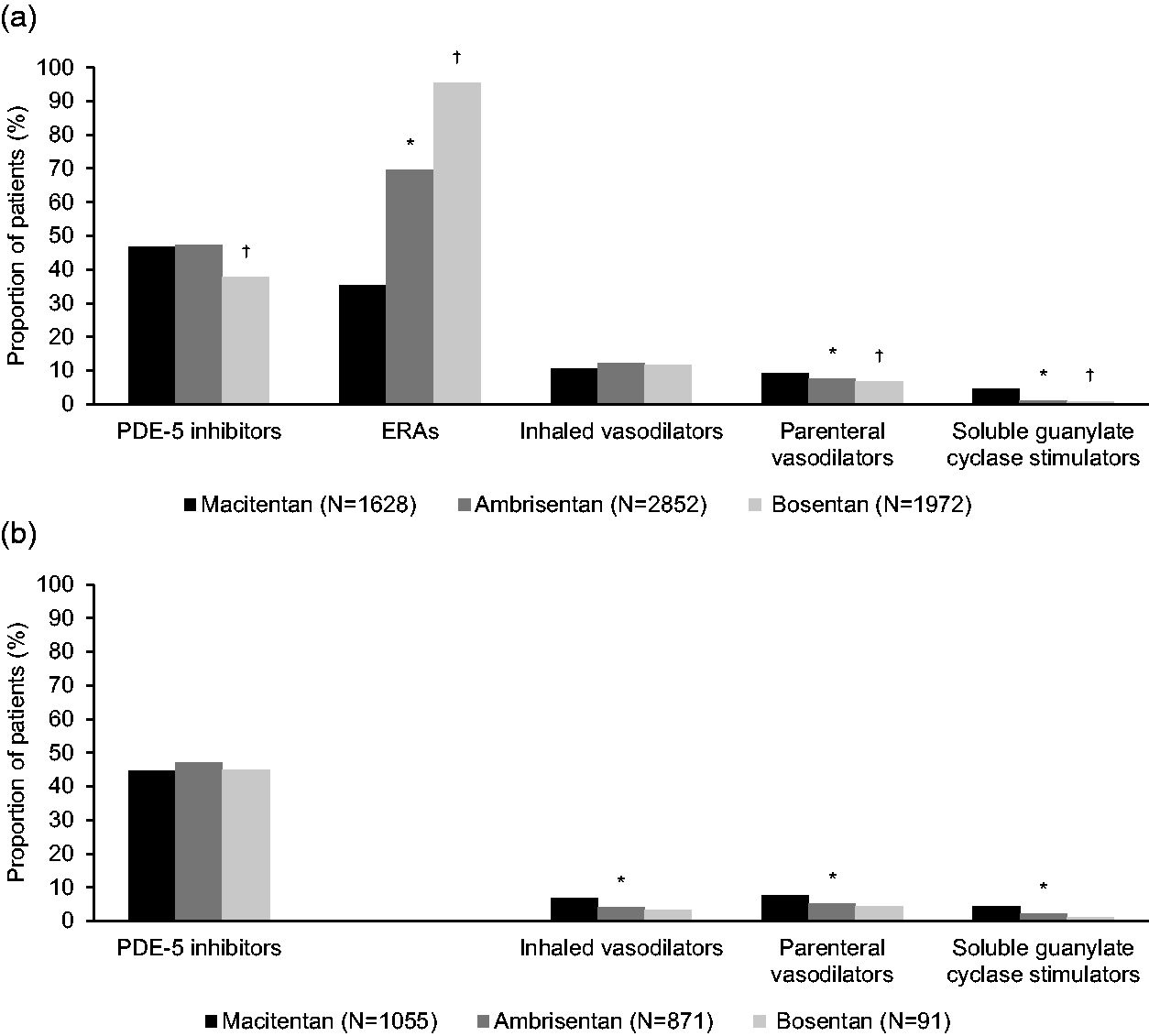
Mortality
A total of 1994 patients died during follow-up (Table 2). Independent risk factors for increased risk of death were increasing age, higher CCI, and inpatient hospitalization (Fig. 5a). For ERA–naïve patients, increasing age, CAD, renal insufficiency, pneumonia, portopulmonary hypertension, and COPD were associated with an increased risk of mortality (Fig. 5b).
Impact of covariates on time to death in the Cox proportional hazards models in (a) the overall patient population and (b) the ERA–naïve subgroup. For each between-group comparison, the group following “vs.” is the reference. CAD: coronary artery disease; CCI: Charlson Comorbidity Index; CI: confidence interval; COPD: chronic obstructive pulmonary disease; DME: durable medical equipment; ERA: endothelin receptor antagonist; HCRU: healthcare resource utilization; HR: hazard ratio; LOS: length of stay; PAH: pulmonary arterial hypertension; PDC: proportion of days covered; PDE-5: phosphodiesterase type 5; PH: pulmonary hypertension. Unadjusted mortality results. ERA: endothelin receptor antagonist.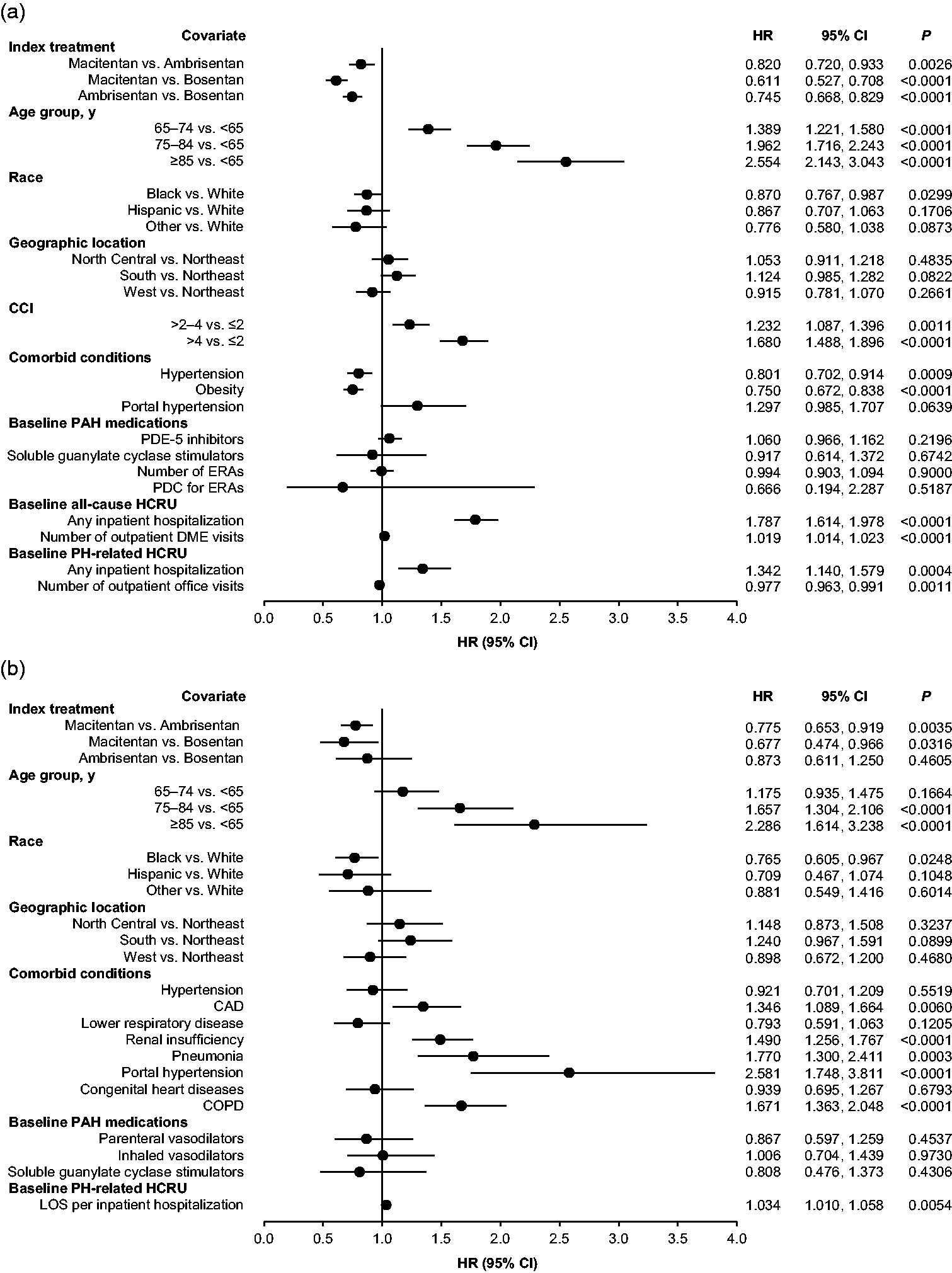
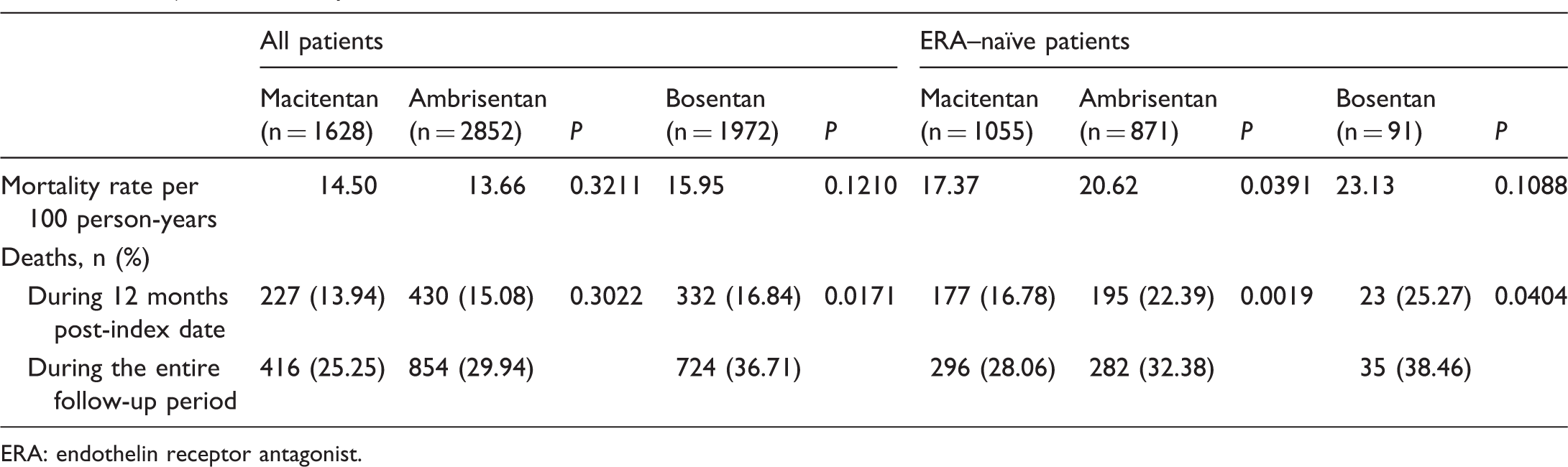
There was no significant difference in the unadjusted mortality rate in the overall patient population between the three ERAs, but macitentan had a significantly lower unadjusted mortality rate than ambrisentan in the ERA–naïve subgroup (17.37 vs. 20.62 per 100 person-years, respectively; P = 0.04). Kaplan–Meier curves of mortality in the overall patient population, which were not adjusted for confounding variables, indicated similar survival for macitentan and ambrisentan and lower survival for bosentan (Fig. 6a). However, unadjusted survival was consistently higher across the study period in the macitentan cohort than in the ambrisentan or bosentan cohorts within the separate ERA–naïve subpopulation as well as within the subpopulation of patients who were prescribed ERAs during the baseline period (i.e., ERA-experienced; Fig. 6b and c).
Kaplan–Meier survival estimates in (a) the overall patient population, (b) the ERA–naïve subgroup, and (c) the ERA-experienced subgroup.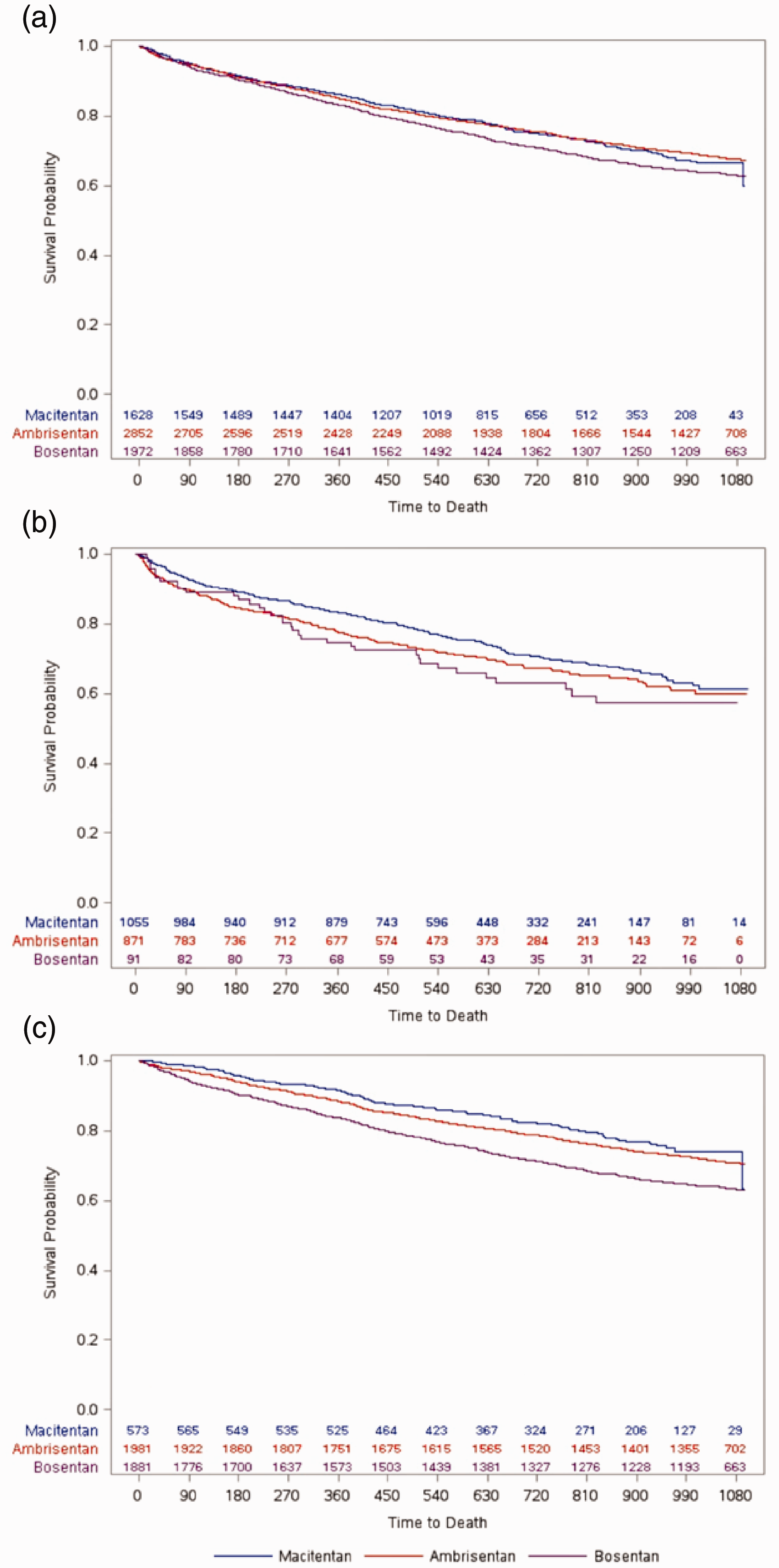
After adjusting for confounders, patients in the macitentan cohort had a significantly lower risk for mortality compared with patients in the ambrisentan and bosentan cohorts. In the overall patient population, macitentan was associated with an 18% lower risk for mortality than ambrisentan (HR: 0.82, 95% CI: 0.72–0.93; P = 0.0026) and a 39% lower risk than bosentan (HR: 0.61, 95% CI: 0.53–0.71; P < 0.0001). In the overall population, ambrisentan was associated with a 26% lower mortality risk than bosentan (HR: 0.74, 95% CI: 0.67–0.83; P < 0.0001). In ERA–naïve patients, macitentan was associated with a 22% lower mortality risk than ambrisentan (HR: 0.78, 95% CI: 0.65–0.92; P = 0.0035) and a 32% lower risk than bosentan (HR: 0.68, 95% CI: 0.47–0.97; P = 0.0316). In the ERA–naïve subgroup, there was no significant difference in mortality risk between ambrisentan and bosentan (HR: 0.87, 95% CI: 0.61–1.25; P = 0.4605).
Discussion
The demographics of patients who are treated for PAH are changing, and the mean age of those seen in clinical practice is increasing.3,24,31 The prevalence of comorbidities increases with advancing age, making drug treatment for this population more complicated. The paucity of evidence on treatment for the growing elderly subgroup, due to these patients typically being excluded from clinical trials, represents a challenge to prescribers. Our study in a predominantly elderly patient population being treated in routine clinical practice in the US helps to fill this information gap. A key finding was the increased risk of mortality associated with comorbidities, confirming the urgency of more research aimed at improving outcomes for these patients.
Using recent data for a Medicare population, we found that differences exist regarding the risk of mortality among patients prescribed ERAs after adjusting for demographic and clinical differences, including identified risk factors for increased risk of mortality (e.g., increasing age, CCI >4, baseline portal hypertension, and any inpatient hospitalization). Patients treated with macitentan experienced a lower risk for death compared with patients receiving ambrisentan or bosentan, even after adjusting for demographic and clinical differences between groups. Differences were seen in patients new to ERA therapy and in the overall patient population. A more rigorous comparison of ERA cohorts would adjust for patient risk level as calculated using PAH risk assessment tools,7,32–36 but the Medicare database does not record the clinical variables required to construct risk scores as clinical information is not recorded unless it is attached to a billing code.
However, comparisons with the ERA–naïve bosentan cohort should be viewed with caution given the small size of this cohort (n = 91). The small number of patients initiating ERA therapy with bosentan may indicate prescriber preference to start patients on newer ERAs that have demonstrated benefit on composite clinical worsening primary endpoints in long-term, event-driven clinical trials, namely the SERAPHIN trial for macitentan in monotherapy and added to background PDE-5 inhibitor or prostacyclin, 20 and the AMBITION trial for ambrisentan in initial combination therapy with the PDE-5 inhibitor tadalafil. 37
Our findings also highlight the importance of adjusting for baseline characteristics like prior therapy when comparing outcomes with different drugs using claims data. The unadjusted survival curves for the entire population obscured differences between ERAs that were evident in the separate ERA–naïve and ERA-experienced subpopulations, because the proportions of ERA–naïve and ERA-experienced patients differed for the different ERAs. For example, at two years, unadjusted survival was higher with macitentan than with ambrisentan within both the ERA–naïve population (71% vs. 67%) and the experienced subgroup (82% vs. 79%), but survival was higher in the ambrisentan-experienced subgroup (79%) than in the macitentan–naïve group (71%). Because the vast majority of ambrisentan patients were ERA-experienced whereas most macitentan patients were ERA–naïve, unadjusted survival for the overall population at two years appeared higher for ambrisentan than macitentan.
At three years, 60% or more of patients in the macitentan and ambrisentan cohorts were still alive in the all-patient and ERA–naïve populations. While this is substantially lower than the 84% three-year survival reported for patients randomized to macitentan 10 mg in SERAPHIN and continuing on macitentan 10 mg in its open-label extension, 38 the age of these Medicare patients needs to be taken into account. Reflecting the eligibility criteria for Medicare coverage, the mean age of these patients was nearly 70 years in all ERA cohorts, considerably higher than in the pivotal trials for ERAs: 46 years in SERAPHIN; 20 50 and 51 years in the ambrisentan trials ARIES-1 and ARIES-2, respectively; 19 51 and 48 years in the bosentan trials Study 351 17 and BREATHE-1, 18 respectively.
Comparison of survival estimates from this analysis with RCT results is also complicated by the fact that patients in the Medicare database may have received their PAH diagnosis at some unknown time prior to the index date (including prior to the start of the baseline period), unlike in a clinical trial in which time since diagnosis can be verified. Another consideration complicating comparisons is that ARIES-1 and -2 and SERAPHIN were not powered to detect a decrease in mortality alone as this was part of their composite endpoints, and patients experiencing a hospitalization were censored and thus may not have been assessed for mortality.
The most commonly used PAH medications during the 12-month baseline period were PDE-5 inhibitors (Fig. 4 and Supplemental Table 2). In the present study, the proportion of patients receiving PDE-5 inhibitor therapy at baseline was 44% for the three ERAs pooled, and 47% in the macitentan cohort. This was similar to the 43% prevalence of PDE-5 inhibitor usage seen in the OPUS registry for patients aged ≥65 years (the mean age of this subgroup was 73 years, comparable to the mean age in our Medicare cohort). 24 Unlike macitentan and ambrisentan, bosentan is an inducer of cytochrome P450 isoenzymes and carries the potential for significant drug–drug interactions with sildenafil, a CYP3A4 inducer. 11 Co-administration of multiple doses of 125 mg twice daily bosentan and 80 mg three times daily sildenafil resulted in a reduction of sildenafil plasma concentrations by 63% and increased bosentan plasma concentrations by 50%. 39 Although we did not analyze results separately for patients receiving sildenafil and tadalafil, it is likely that a high proportion received sildenafil and that results in the bosentan group could have been influenced by drug–drug interactions.
Assessment of adverse effects was not a goal of the present study, but would be valuable to undertake in future, given the different safety profiles of the three ERAs. 30 In particular, hepatotoxicity in clinical practice should be evaluated—unlike macitentan or ambrisentan, bosentan is associated not only with the potential for drug–drug interactions but also liver enzyme elevation and bile salt export pump inhibition, 30 and the bosentan prescribing information carries a boxed warning to draw attention to the risk of aminotransferase elevation and liver failure. 39
This analysis provides data from routine clinical practice regarding the clinical characteristics, treatment, and survival of predominantly elderly patients with PAH in the US Fee-for-service Medicare database, which allows linkage of patient-level healthcare and death records in almost 20% of the US population (56 million). However, our study is subject to a number of limitations. Because there is no unique ICD-9-CM or ICD-10-CM code that distinguishes all forms of PAH recognized in the modern clinical classification system from the World Symposium on Pulmonary Hypertension (WSPH)40–42 from other types of PH, 43 patients with PAH had to be identified based on an algorithm using diagnostic codes for PH. All ERAs assessed are indicated only for WSPH Group 1 PAH, not other forms of PH, so it is unclear what percentage of medication use in this population may have been off-label. The apparent rate of RHC was lower than would be expected in an exclusively PAH population, but an unknown proportion of patients missing a record of RHC might have undergone this procedure prior to the study period and thus not have been captured in our analysis.
Although patients with a medical claim for left-sided heart disease were excluded, other codes may be used to describe these patients. Also, patients may have COPD or left-sided heart failure as comorbidities, making it difficult to distinguish between off-label use and concomitant diagnoses. Comparability of the results of retrospective analyses like ours with findings from clinical trials could be enhanced in future studies by using more restrictive algorithms to identify patient populations that more closely match the eligibility criteria of key trials (e.g., by excluding patients with diagnosis codes for comorbidities that were trial exclusion criteria).
Despite covariate adjustment in the multivariable Cox proportional hazard models, unmeasured confounders such as World Health Organization Functional Class, date of market entry of each ERA, provider type (e.g., specialty pharmacy or pharmacy benefit manager), and additional sources of unrecognized bias may impact the study results. Furthermore, the comorbidity and PH diagnoses were obtained from administrative claims data that are not clinically validated. Claims data are collected for insurance payments not research and may be subject to coding error. Administrative claims also do not provide information on whether the prescriptions were taken as prescribed.
This study included US patients with Medicare coverage; therefore, results may not be generalizable to all patients. Finally, like all retrospective database studies, it is not possible to draw firm conclusions regarding causality of the associations found in this study.
Conclusion
This is the first study in the Fee-for-service Medicare dataset that assessed mortality in a predominantly elderly PAH population with high comorbidity rates treated with ERAs. Variables that independently increased the risk of mortality included higher comorbidity index, increasing age, and inpatient hospitalizations at baseline. We found that the risk of mortality among these patients prescribed ERAs differed, even after adjusting for demographic variables and comorbidities. Due to the limitations described above, our results should be considered as hypothesis-generating and should not be taken as the basis for influencing clinical practice for this patient population. Direct comparison of these agents in a prospective, controlled trial is the only way to definitively answer the question of whether one of the available ERAs confers a survival advantage over the others. Therefore, we advocate conducting an event-driven head-to-head RCT in an elderly and appropriately risk-enriched PAH population.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020954158 - Supplemental material for Comparative effectiveness of endothelin receptor antagonists on mortality in patients with pulmonary arterial hypertension in a US Medicare population: a retrospective database analysis
Supplemental material, sj-pdf-1-pul-10.1177_2045894020954158 for Comparative effectiveness of endothelin receptor antagonists on mortality in patients with pulmonary arterial hypertension in a US Medicare population: a retrospective database analysis by Raymond L. Benza, Cassandra A. Lickert, Lin Xie, William Drake Adesuwa Ogbomo, Huseyin Yuce and Michele R. Cole in Pulmonary Circulation
Footnotes
Acknowledgements
The authors would like to thank Huiqi Wang, MS, from STATinMED Research for performing the programming for these analyses. Medical writing and editorial support were provided by W. Mark Roberts, PhD, Montréal, Québec, Canada, funded by Actelion Pharmaceuticals US, Inc., a Janssen Pharmaceutical Company of Johnson & Johnson.
Contributorship
R. L. B., C. A. L., L. X., A. O., H. Y., and M. R. C. contributed to study design. L. X., A. O., and H. Y. acquired and analyzed the data. R. L. B., C. A. L., L. X., A. O., and M. R. C. interpreted the data. All authors were involved in study conception, participated in drafting the manuscript and/or revising it critically for important intellectual content, approved the final version for publication, and take responsibility for appropriate portions of the content.
Conflict of interest
R. L. B. reports no relevant conflicts of interest. C. A. L. and W. D. III were employees of Actelion Pharmaceuticals US, Inc. when this research was conducted and hold stock in Johnson & Johnson. L. X. and A. O. are employees of STATinMED Research, which received consulting fees from Actelion Pharmaceuticals US, Inc. for services related to the conduct of this research. H. Y. received consulting fees from Actelion Pharmaceuticals US, Inc. via STATinMED Research for services related to the conduct of this research. M. R. C. is an employee of Actelion Pharmaceuticals US, Inc. and holds stock in Johnson & Johnson.
Ethical approval
This study is a retrospective claims database analysis and thus does not contain any experiments on human or animal subjects for which ethical approval is required.
Funding
This research was sponsored by Actelion Pharmaceuticals US, Inc. The sponsor was involved in data analysis and interpretation, funded medical writing and editorial support to the authors, and was involved in the decision to publish the finished manuscript.
Guarantor
M. R. C. is the guarantor for this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
