Abstract
Several new medications for pulmonary arterial hypertension (PAH) have recently been introduced; however, current real-world data regarding US patients with PAH are limited. We conducted a retrospective administrative claims study to examine PAH treatment patterns and summarize healthcare utilization and costs among patients with newly diagnosed PAH treated in US clinical practice. Patients newly treated for PAH from 1 January 2010 to 31 March 2015 were followed for ≥12 months. Patient characteristics, treatment patterns, healthcare resource utilization, and costs were described. Adherence (proportion of days covered), persistence (months until therapy discontinuation/modification), and the probability of continuing the index regimen were analyzed by index regimen cohort (monotherapy versus combination therapy). Of 1637 eligible patients, 93.8% initiated treatment with monotherapy and 6.2% with combination therapy. The most common index regimen was phosphodiesterase type 5 inhibitor (PDE-5I) monotherapy (70.0% of patients). A total of 581 patients (35.5%) modified their index regimen during the study. Most patients (55.4%) who began combination therapy did so on or within six months of the index date. Endothelin receptor agonists (ERAs) and combination therapies were associated with higher adherence than PDE-5Is and monotherapies, respectively. Healthcare utilization was substantial across the study population, with costs in the combination therapy cohort more than doubling from baseline to follow-up. The majority of patients were treated with monotherapies (most often, PDE-5Is), despite combination therapies and ERAs being associated with higher medication adherence. Index regimen adjustments occurred early and in a substantial proportion of patients, suggesting that inadequate clinical response to monotherapies may not be uncommon.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a rare, chronic, progressive disease characterized by high blood pressure in the pulmonary arteries, often leading to right heart failure and premature mortality.1–3 Patients with PAH generally carry a substantial co-morbidity burden4–8 and have a high mortality rate—for newly diagnosed patients, approximately 39% after five years7—as well as high healthcare costs, on average >4 times those of controls.6,9
Until recently, the pathophysiology of PAH was not well understood and therapeutic advancements came slowly; however, elucidation of several of the physiological pathways that mediate PAH has led to development of a burgeoning arsenal of targeted therapies shown to reduce morbidity and improve functional status among patients with the condition.10,11 Several PAH medications have been approved by the U.S. Food and Drug Administration (FDA) since 2013, including the soluble guanylate cyclase stimulator (sGCS) riociguat, the oral prostacyclin analogue treprostinil, the oral endothelin receptor antagonist (ERA) macitentan, and the non-prostanoid IP agonist selexipag. Despite the availability of new treatment options, the long-term prognosis for PAH remains poor—in part because of the non-specific nature of the symptomology, which often leads to delayed diagnosis. Given that multiple pathways are involved in PAH, combination therapy with medications from two or more classes has emerged as a strategy for improving outcomes among patients with advanced disease or an inadequate clinical response on monotherapy. 12 Already in use for treatment of various chronic conditions, including heart failure and systemic hypertension, combination therapy is an appealing therapeutic approach for PAH because three distinct signaling pathways can be targeted with different drugs: the prostacyclin pathway (prostacyclins and a non-prostanoid IP agonist); the endothelin pathway (ERAs); and the nitric oxide pathway (phosphodiesterase type 5 inhibitors [PDE-5Is] and sGCSs). 12
Evidence from clinical trials indicates that combination therapy reduces the risk of clinical worsening and improves functional class among patients with PAH.13,14 Although combination therapy with the ERA bosentan plus the PDE-5I sildenafil was not shown to reduce long-term morbidity and mortality, 15 newer agents have demonstrated positive results in event-driven trials. The ERA macitentan plus a PDE-5I or prostacyclin 16 and the non-prostanoid IP agonist selexipag plus an ERA and/or PDE-5I 17 were shown to effectively reduce long-term morbidity/mortality in the SERAPHIN and GRIPHON trials, respectively. Accordingly, current guidelines recommend sequential combination therapy for patients with inadequate clinical response to monotherapy, but also note the potential benefit of initial combination therapy as a pre-emptive measure. 18 In 2015, initial treatment with a combination of the ERA ambrisentan and the PDE-5I tadalafil was approved by the FDA for use in treatment-naïve patients with PAH on the basis of findings from the AMBITION trial.18,19
As new medications and drug combinations are introduced, stakeholders advocating for cost-effective treatment are particularly interested in understanding treatment patterns and healthcare use outcomes among patients with PAH who are prescribed combination therapy compared with monotherapy. However, real-world U.S. data regarding PAH treatment patterns are limited, and information regarding patient characteristics in the U.S. population with PAH has come largely from registries such as REVEAL. While REVEAL has been a valuable data source, it does not contain data for patients using the most recently approved PAH therapies and lacks information regarding healthcare resource utilization or costs. Moreover, REVEAL enrollees were from pulmonary hypertension (PH) centers, where patients are more likely to receive guideline-recommended diagnostic testing and treatment compared with those from the general population. 20 The present study used administrative claims data to examine treatment patterns for PAH-specific therapies, describe patient demographic and clinical characteristics, and summarize healthcare resource utilization and costs among patients with newly diagnosed PAH treated with monotherapy or combination therapy in U.S. clinical practice.
Methods
Study design and data source
This was a retrospective administrative claims study conducted from 1 July 2009 through 31 March 2016 using the Optum Research Database, a large, population-representative database containing medical and pharmacy data from individuals enrolled in U.S. commercial and Medicare Advantage with Part D (MAPD) health plans. Medical claims included diagnosis and procedure codes from the 9th and 10th revisions of the International Classification of Diseases, Clinical Modification (ICD-9-CM and ICD-10-CM), and Current Procedural Terminology or Healthcare Common Procedure Coding System codes. Outpatient pharmacy claims included National Drug Codes (NDCs) for dispensed medications, quantity dispensed, dose, and number of days’ supply. Both medical and pharmacy claims included cost information.
Study sample selection and cohort assignment
The study included commercial and MAPD health plan members with evidence of medication treatment for PAH from 1 January 2010 through 31 March 2015 (patient identification period). Included patients were required to have at least one pharmacy claim for a PAH-specific medication (ERAs [ambrisentan, bosentan, macitentan]; PDE-5Is [sildenafil, tadalafil]; prostacyclins and a non-prostanoid IP agonist [epoprostenol, iloprost, treprostinil, and selexipag]; and sGCS [riociguat]; identified using NDCs to ensure that prescriptions were intended for treatment of PAH) during the patient identification period. The index date was defined as the date of the first claim for a PAH-specific medication. Additional inclusion criteria were: at least one claim with a diagnosis code for PH (ICD-9-CM 416.0, 416.8, or 416.9) in any position during the six-month period before and including the index date; no pharmacy claims for PAH-specific medication therapy during the six-month period before the index date (baseline period); and continuous health plan enrollment with medical and pharmacy benefits during the six-month period before the index date and for at least 12 months after and including the index date (follow-up period). The follow-up period ended with the earlier of health plan disenrollment or study end (31 March 2016). Study cohort assignment was based on the number of different classes of PAH-specific medications filled within 30 days, starting with the index date. Patients with pharmacy claims for a PAH-specific medication from only one medication class were assigned to the index monotherapy cohort, and those with pharmacy claims for more than one class of PAH-specific medication were assigned to the index combination therapy cohort. The index regimen was defined as the PAH-specific medication(s) filled during the 30 days starting with the index date. Patients whose index regimens changed during the study (i.e., addition, switching, or discontinuation of a medication) were considered as starting a second regimen.
Study measures
Patient demographic and clinical characteristics (age, sex, insurance type, and co-morbidities) were assessed during the baseline period. Treatment patterns for PAH-specific therapies (medication classes, adherence, persistence, and discontinuation) were assessed during the first 12 months of the follow-up period. Adherence to the index regimen was assessed using proportion of days covered (PDC), which was calculated by dividing the number of days on which the index regimen was available (based on filled prescriptions) by the number of days in the first 12 months of follow-up. PDC for index combination therapy was based on possession of any medication in the regimen, as adherence measures for individual components of a regimen of related medications tend to reflect each other closely. 21 Persistence with the index regimen was measured using claims for the index class(es), inclusive of fills on the index date. 22 Persistence was defined as the number of months from the index date until therapy discontinuation (defined as a gap in therapy of at least 90 days) or modification (defined as a fill for a new PAH-related therapy, due to either discontinuation or augmentation). Kaplan–Meier analysis was conducted to estimate the probability of continuation of the index regimen.
Healthcare resource utilization and healthcare costs were assessed during the baseline and follow-up periods and stratified by whether patients were enrolled in commercial or MAPD plans (to account for differences in cost structure). Healthcare resource utilization (all-cause) was calculated as the number and percentage of patients with at least one encounter for ambulatory visits, emergency room visits, and inpatient admissions. Total healthcare costs (all-cause and PAH-related) were calculated as the combined health plan- and patient-paid amounts per patient per month (PPPM) to account for varying length of follow-up and adjusted to 2015 US dollars using the annual medical care component of the Consumer Price Index. 23 Costs were defined as PAH-related if the claim had a diagnosis for PH in the first or second position (ICD-9-CM 416.0, 416.8, or 416.9; and during dates for which ICD-10-CM was in use, ICD-10-CM I27.0, I27.2, I27.81, I27.89, or I27.9).
Statistical analysis
Results were stratified by treatment cohort; numbers and percentages were provided for categorical variables and means and standard deviations were provided for continuous variables. Between-cohort differences in baseline patient characteristics were analyzed using Student’s
Results
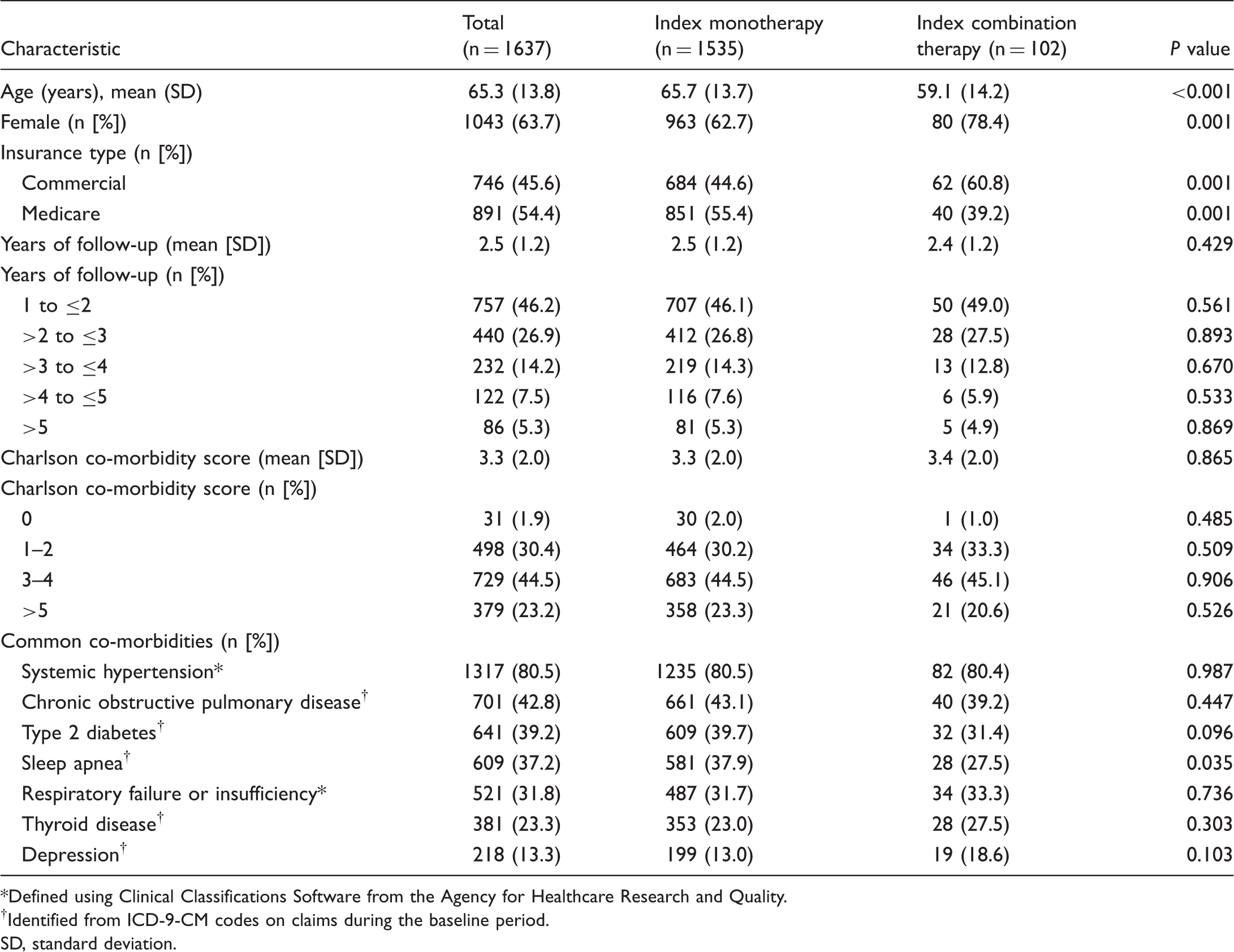
Study sample
Of 6925 patients with at least one pharmacy claim for a PAH-specific therapy during the identification period, 1637 met all remaining study criteria (Fig. 1). The index monotherapy cohort and index combination therapy cohort contained 1535 patients and 102 patients, respectively. The study population was predominantly female (63.7%) and the mean age was 65.3 years (SD = 13.8 years; Table 1). Slightly more patients were enrolled in MAPD versus commercial insurance plans (54.4% vs. 45.6%). Mean follow-up duration for the full study population was 2.5 years (SD = 1.2 years), with 53.8% followed for at least two years and 12.7% followed for at least four years. Common co-morbidities among the total patient group included systemic hypertension (80.5%), chronic obstructive pulmonary disease (42.8%), type 2 diabetes (39.2%), and sleep apnea (37.2%). Although the co-morbidity profiles of the two cohorts were similar, the prevalence of sleep apnea was significantly higher in the index monotherapy cohort versus the index combination therapy cohort (37.9% vs. 27.5%, Patient selection and attrition. PAH, pulmonary arterial hypertension; PH, pulmonary hypertension. Baseline patient characteristics. Defined using Clinical Classifications Software from the Agency for Healthcare Research and Quality. Identified from ICD-9-CM codes on claims during the baseline period. SD, standard deviation.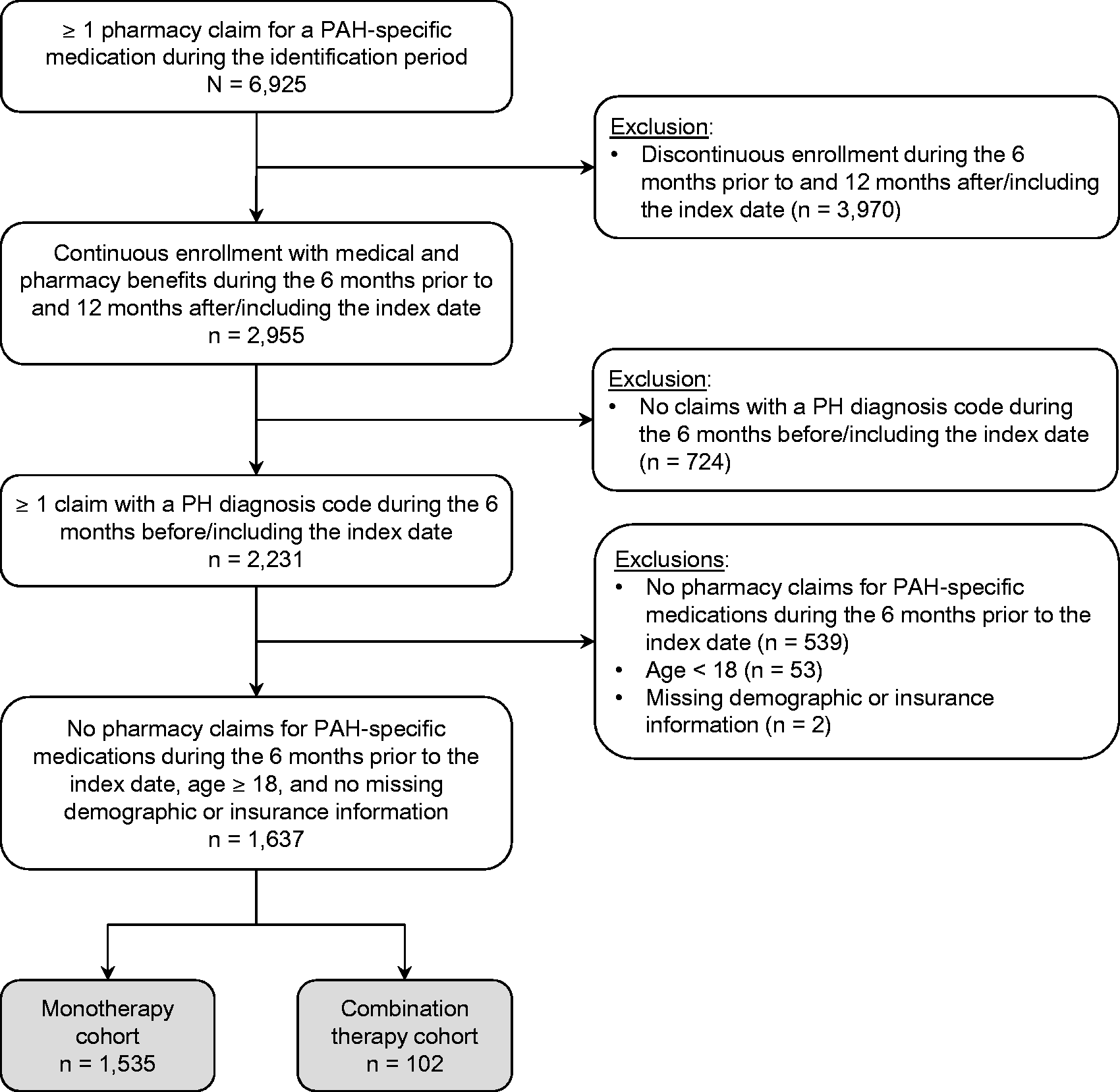
Medication regimens
Most patients (1535/1637, or 93.8%) initiated treatment with monotherapy; only 6.2% (102/1637) initiated treatment with combination therapy. A total of 581/1637 patients (35.5%) modified their index regimen to a second regimen during the study period, including 284 who switched from monotherapy to combination therapy (18.5% of the index monotherapy cohort). Of patients with a regimen modification, the proportion treated with combination therapy increased to 42.7% (248/581) in the second regimen. The majority of patients who began combination therapy during the study, including those with index combination therapy as well as those who switched to combination therapy at a subsequent point, did so on or within six months of the index date (214/386, or 55.4%).
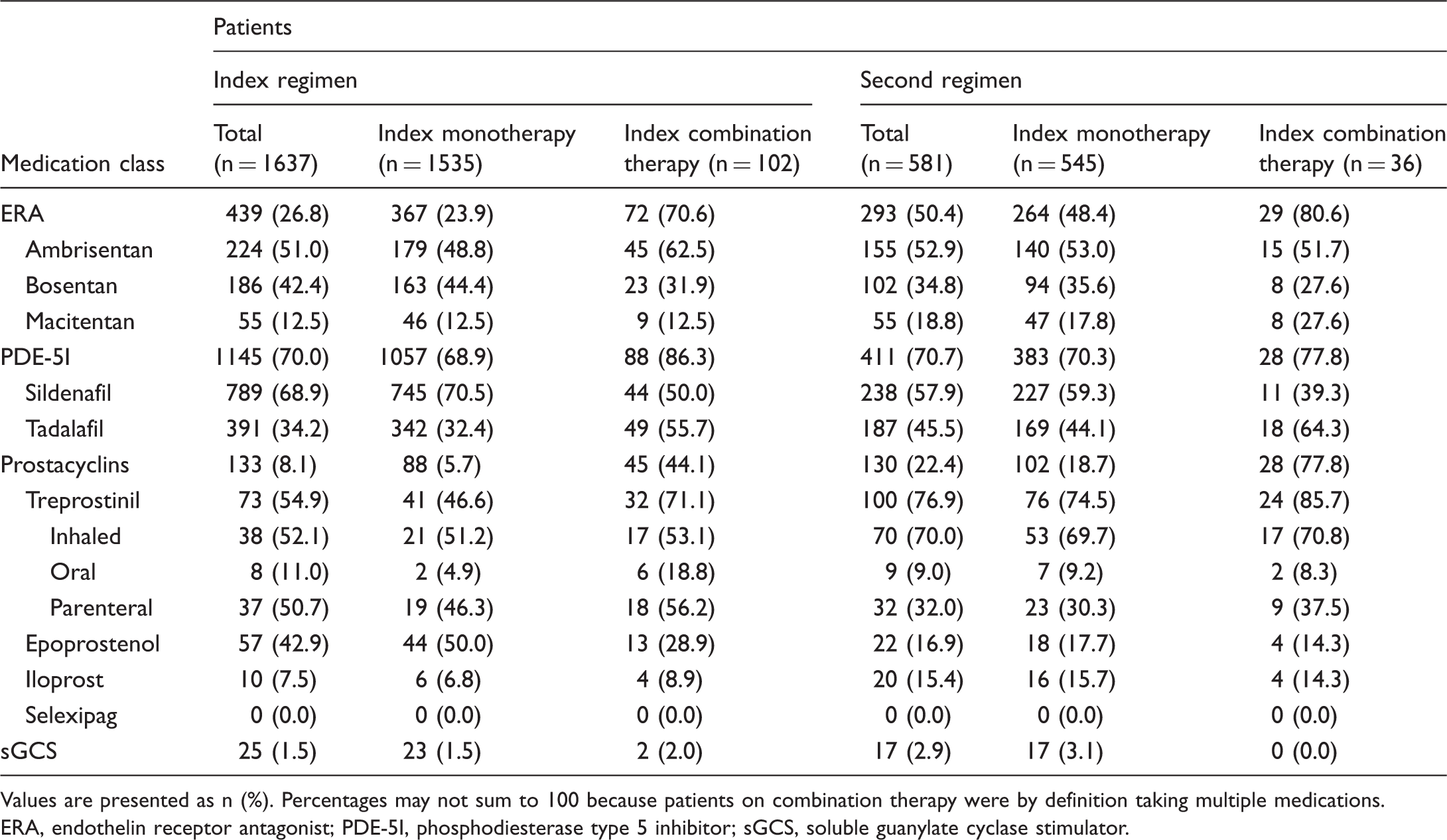
Medication classes used in index and second-regimen therapies are shown in Table 2. PDE-5Is, ERAs, and prostacyclins were used in 70.0%, 26.8%, and 8.1% of index treatment regimens, respectively, across all patients. The proportion of patients using ERAs (index regimen, 26.8% vs. second regimen, 50.4%) and prostacyclins (index regimen, 8.1% vs. second regimen, 22.4%) increased from the index regimen to the second regimen but remained similar for PDE-5Is (index regimen, 70.0% vs. second regimen, 70.7%). PDE-5Is were the most common monotherapy overall in both index regimens and second regimens, while the most common combination therapy was ERA plus PDE-5I (index regimen, 53.9%; second regimen, 58.5%; Fig. 2). Among patients on combination therapy, the percentage on triple therapy was 2.9% in the first regimen and 8.9% in the second regimen (Fig. 2); the remaining patients were on dual therapy.
Medication classes used in combination therapies, by regimen. Percentages may not sum to 100 because of rounding. ERA, endothelin receptor antagonist; PDE-5I, phosphodiesterase type 5 inhibitor; sGCS, soluble guanylate cyclase stimulator. Prevalence of medication classes in index regimens and second regimens, by cohort. Values are presented as n (%). Percentages may not sum to 100 because patients on combination therapy were by definition taking multiple medications. ERA, endothelin receptor antagonist; PDE-5I, phosphodiesterase type 5 inhibitor; sGCS, soluble guanylate cyclase stimulator.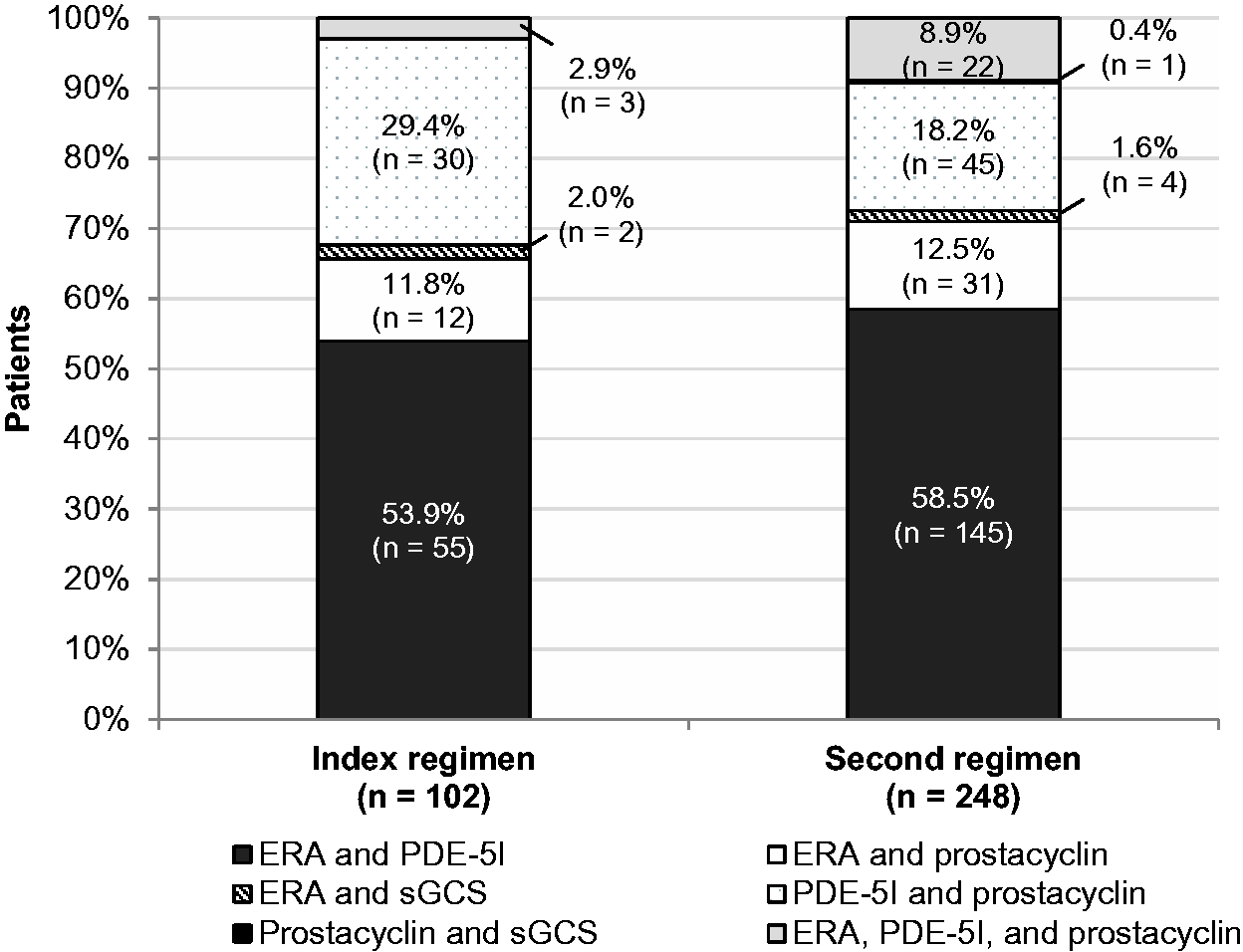
Medication adherence, persistence, and discontinuation
Overall, index regimens containing ERAs compared with PDE-5Is were associated with higher adherence (mean [SD] PDC = 0.8 [0.4] vs. 0.6 [0.4]; Adherence to the index regimen (proportion of days covered, PDC) by cohort. ERA, endothelin receptor antagonist; PDE-5I, phosphodiesterase type 5 inhibitor; SD, standard deviation; sGCS, soluble guanylate cyclase stimulator.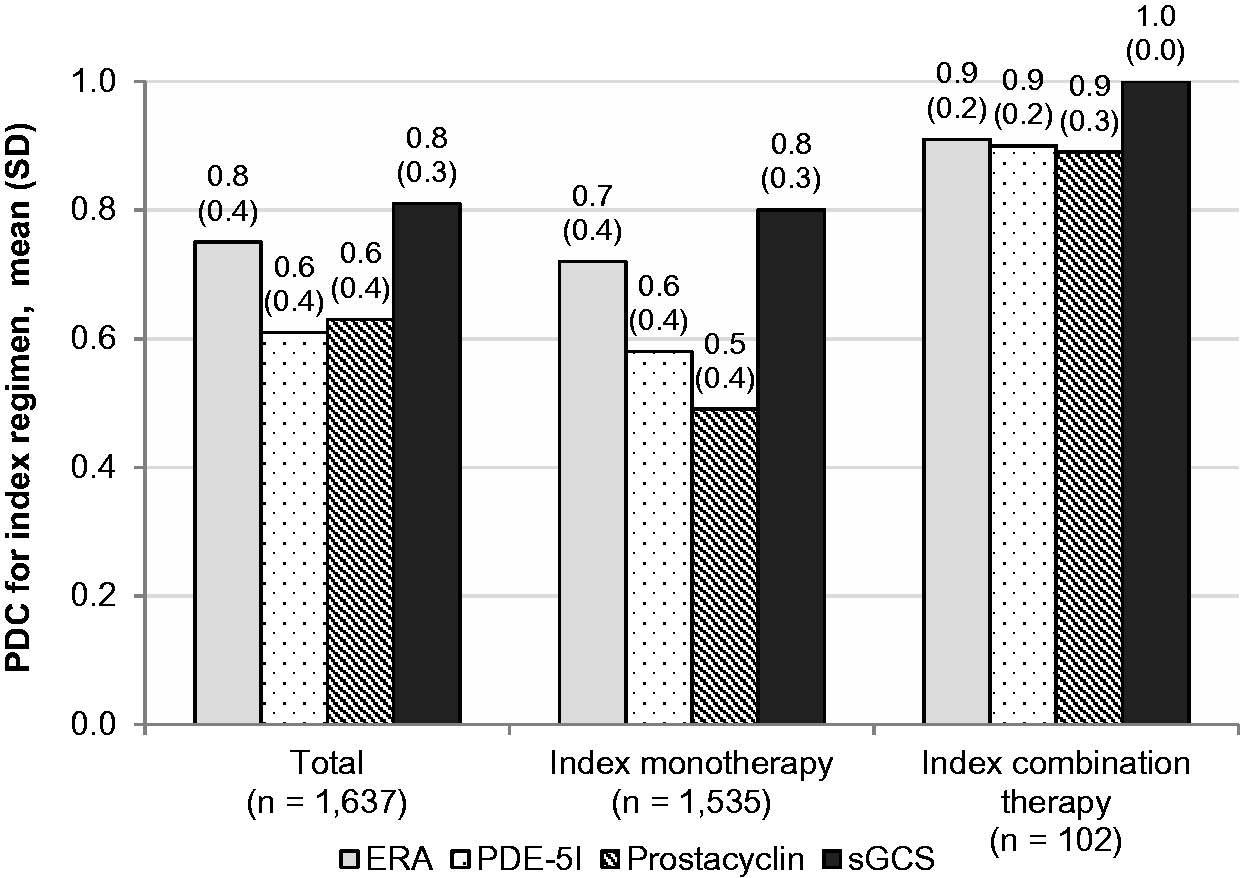
Of the 1637 study patients, 613 (37.4%) remained on their index regimen without a discontinuation or modification until the end of the study, 171 (10.4%) discontinued and then resumed their index medication, and 443 (27.1%) discontinued their index regimen without starting a second regimen. Of the 1024 patients who discontinued or modified their index regimen, 78.9% did so within one year (mean [SD] persistence = 7.6 [9.0] months, median = 4.0 months, interquartile range = 1.4–10.4 months). Among patients who eventually discontinued the index regimen, the proportion of patients who were still on their index regimen at one year was 21.1% overall, 20.5% for monotherapy, and 33.3% for combination therapy. Kaplan–Meier analysis revealed that the probability of continuing the index regimen at one year was 50.3% overall; however, patients who initiated monotherapy were less likely to remain on their index regimen than those who initiated combination therapy (49.2% vs. 68.3%; Fig. 4). More than half of patients who had combination therapy as their index regimen (52.9%) remained on the initial combination therapy until the end of the study, compared with 36.4% of patients who had index monotherapy.
Kaplan–Meier analysis of time to discontinuation or modification of the index regimen.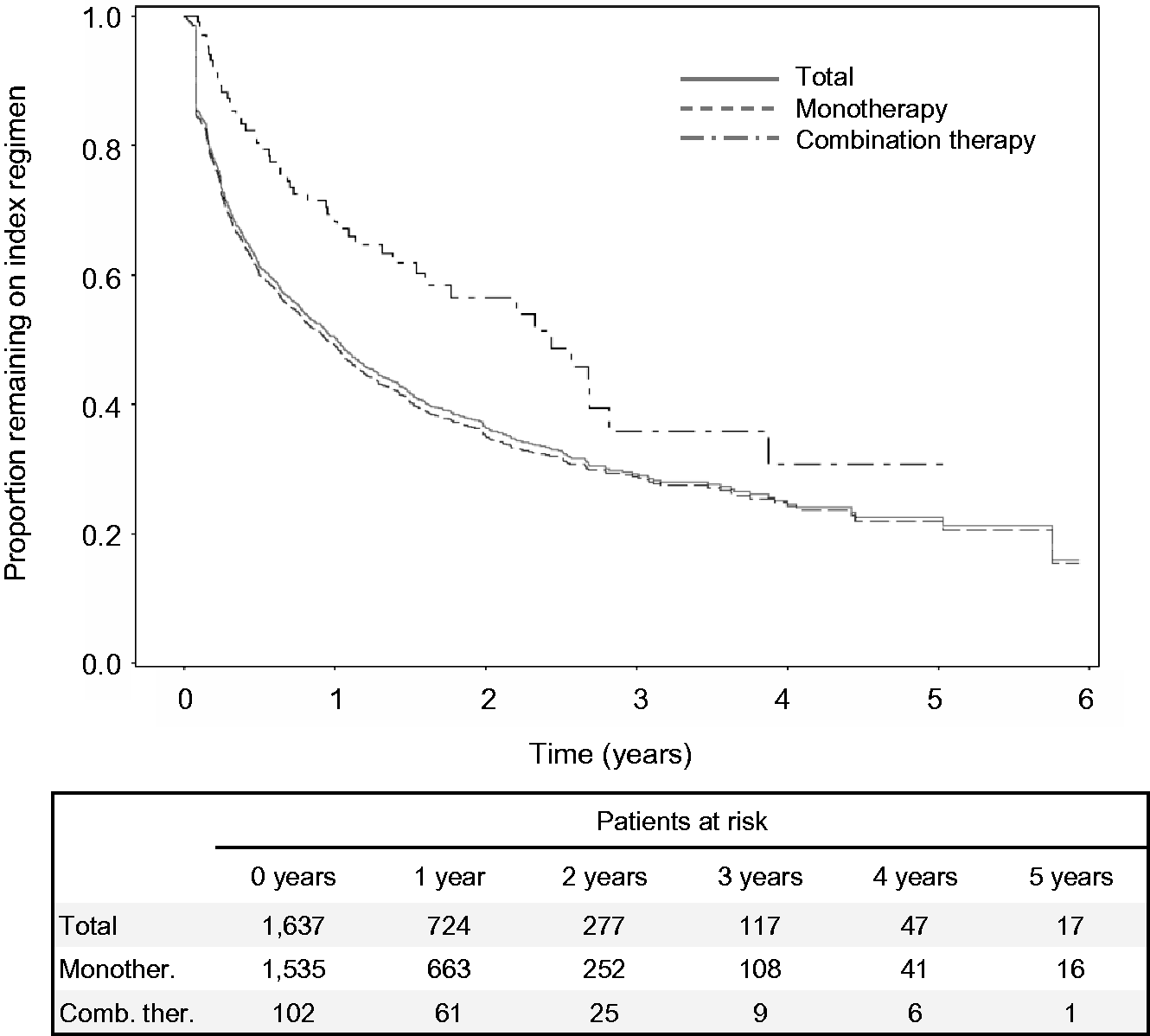
Healthcare resource utilization
All-cause healthcare resource utilization.
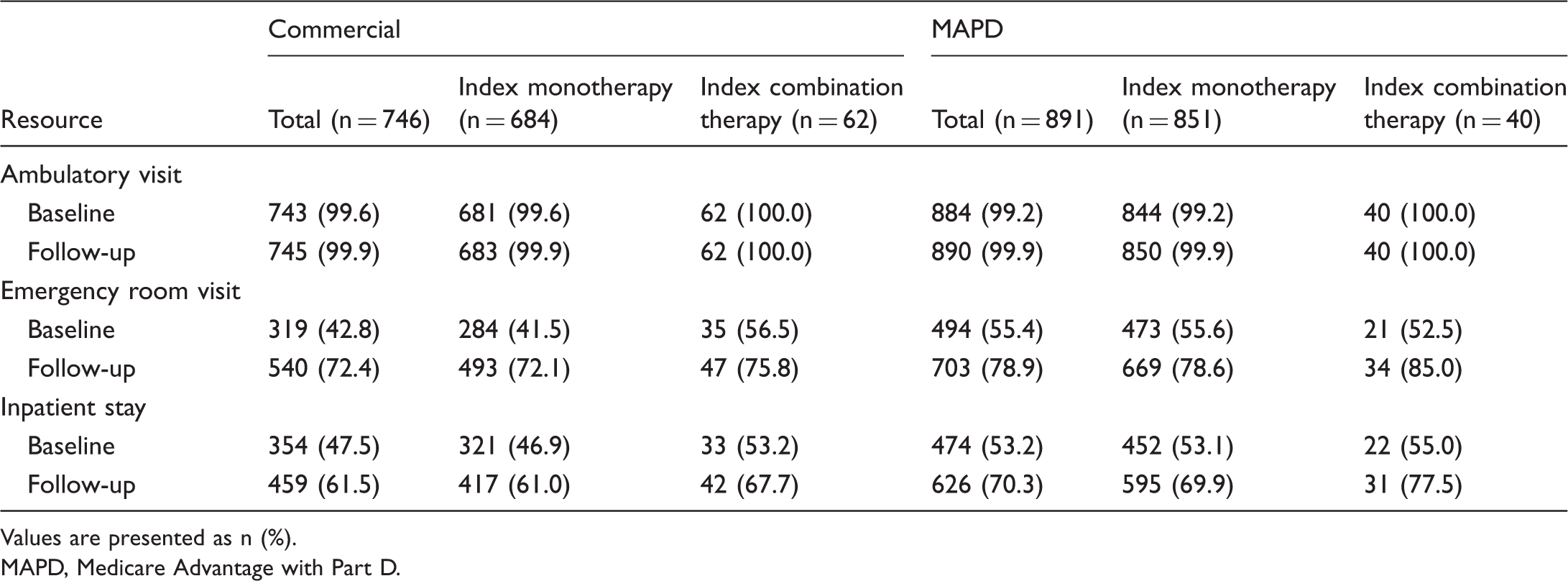
Values are presented as n (%).
MAPD, Medicare Advantage with Part D.
Healthcare costs
PPPM total healthcare costs were higher among commercial enrollees than MAPD enrollees. Costs were similar between baseline and follow-up for the index monotherapy cohort (commercial, $11,167 [$25,028] vs. $9503 [$24,408]; MAPD, $5506 [$11,478] vs. $6271 [$5530]; Fig. 5), but increased more than twofold from baseline to follow-up in the index combination therapy cohort for both insurance types (commercial, $7222 [$9140] vs. $16,240 [$11,536]; MAPD, $5504 [$10,458] vs. $14,340 [$8545]; Fig. 5). In particular, follow-up pharmacy costs were substantially higher for patients whose index regimen was combination therapy versus monotherapy (commercial, $8183 [$6009] vs. $3221 [$3556]; MAPD, $7213 [$4056] vs. $3148 [$3493]). Total follow-up PAH-related healthcare costs also were higher for index combination therapy versus index monotherapy (commercial, $6107 [$9479] vs. $2218 [$6927]; MAPD, $5310 [$6432] vs. $1410 [$3408]).
Per-patient-per-month (PPPM) total all-cause healthcare costs. Bars represent standard deviations. MAPD, Medicare Advantage with Part D.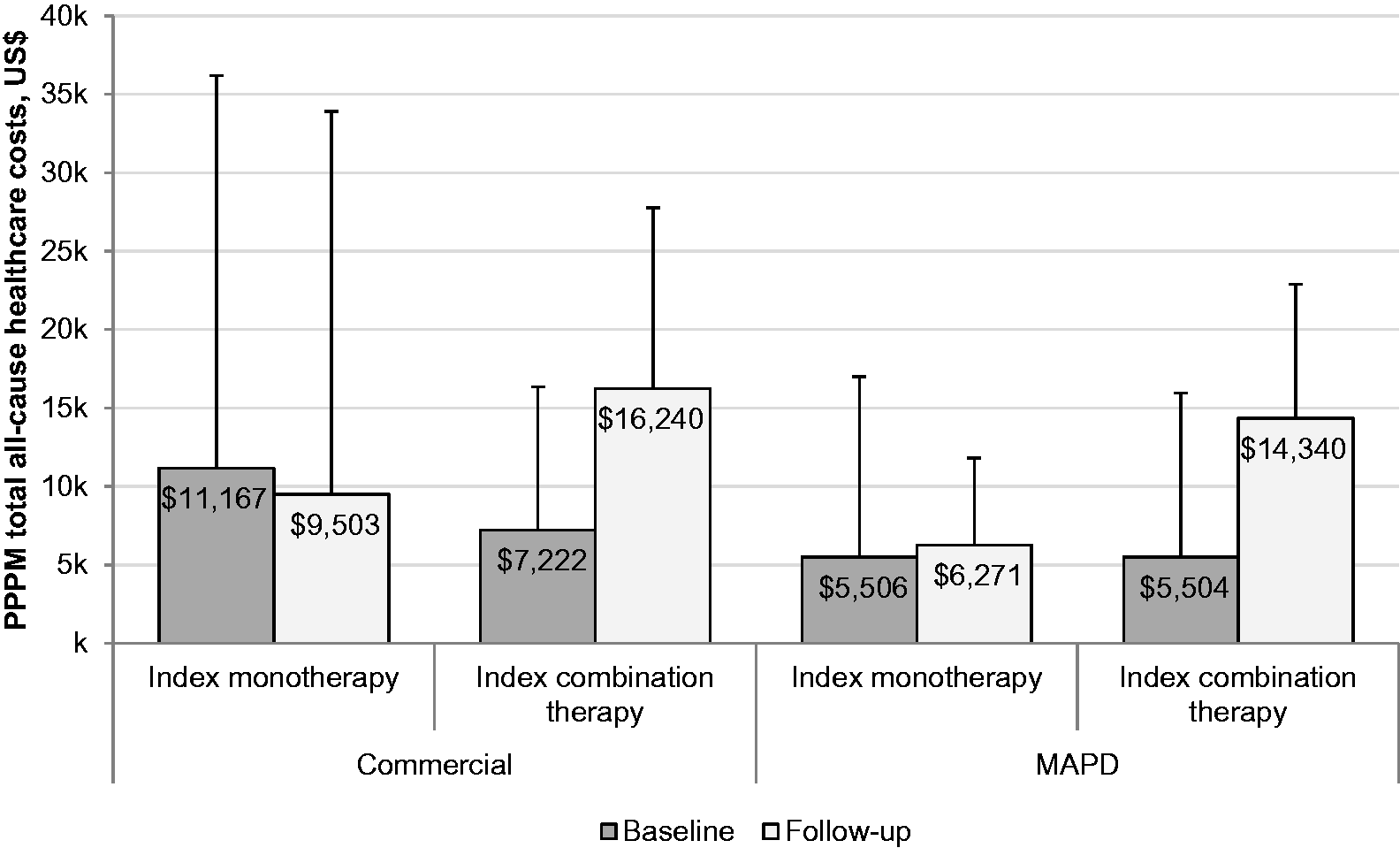
Discussion
In this real-world analysis of U.S. patients newly diagnosed with PAH, the vast majority—nearly 94%—initiated treatment with monotherapy. Although this figure is substantially higher than the 55% observed in the REVEAL registry, 8 the discrepancy may be explained by the starkly different patient populations: more than half of REVEAL registrants, who were enrolled from PH specialty centers, were at World Health Organization functional class (WHO-FC) III or IV, and only 14% were newly diagnosed. 8 It is possible that REVEAL enrollees comprised a higher proportion of patients with more severe disease compared with our newly diagnosed, general population sample, or that specialty centers tend to provide more aggressive treatment. On the other hand, our findings are similar to those of previous administrative claims analyses of PAH patient populations, in which the proportion receiving monotherapy has been found to be in the approximate range of 80–94%.4,24–26 Notably, mean follow-up in the present study was relatively long, with 54% of patients followed for two years or more; this suggests that many patients in the study population could be considered prevalent (i.e., diagnosed >6 months previously 27 ) during the overall study period.
According to guidelines for treatment of PAH from the European Society of Cardiology and the European Respiratory Society, initial monotherapy is appropriate for patients who are treatment naïve and whose clinical signs and symptoms suggest low or intermediate mortality risk. 12 However, the guidelines also indicate that initial combination therapy may be considered at this risk level, in light of increasing evidence for the effectiveness of combination therapy as a treatment strategy.18,28,29 In the multicenter, placebo-controlled AMBITION trial, initial combination therapy with tadalafil and ambrisentan escalated to therapeutic dosages over eight weeks was found to reduce clinical failure (defined as death, hospitalization for worsening PAH, disease progression, or unsatisfactory clinical response) among patients with incident PAH by 50% compared with monotherapy, while improving exercise capacity and the rate of satisfactory clinical response. 18 The patient identification period of the present study ended before the U.S. market approval of initial combination therapy with ambrisentan/tadalafil; thus, the proportion of patients receiving initial combination therapy would likely be higher than the observed 6.2% if the identification period were extended further.
Given that the guidelines recommend intensification of therapy until a satisfactory clinical response is observed (e.g., WHO-FC I/II), 12 it is noteworthy that the index regimen was modified for more than one-third of patients in our sample, and that those who began combination therapy tended to do so early—within six months of the index date. This may be related to the progressive nature of PAH, which suggests that patients will need additional therapies as they move through the disease continuum and that monotherapy with currently available medications may not produce an adequate clinical response for many patients. While evidence continues to mount that certain combination therapies can significantly delay disease progression compared with monotherapy,10,13,14 only about half of currently available drug combinations have been studied in randomized controlled trials; 30 moreover, only the aforementioned SERAPHIN, GRIPHON, and AMBITION trials were long-term, event-driven trials with robust composite endpoints.16–18 Clinical research in PAH has recently undergone a paradigm shift from short-term trials with functional endpoints to long-term trials assessing morbidity/mortality; however, further research is still required to solidify recommendations for the most effective combination therapy treatment strategies.
In our patient sample, PDE-5I monotherapy and ERA monotherapy were the most common index regimens (64.6% and 22.4%, respectively), while PDE-5I plus ERA was the most common combination therapy (53.9% of index combination regimens, 58.5% of subsequent combination regimens). These findings are consistent with those of other claims analyses of PAH4,31,32 and reflect the substantial evidence that monotherapy with PDE-5I or ERA as well as combination therapy with PDE-5I plus ERA are associated with significantly less clinical worsening and more improvement in functional class compared with placebo. 10 Nevertheless, it should be noted that because monotherapy with PDE-5Is and most other agents currently approved for treating PAH (with the exceptions of macitentan, 16 selexipag, 17 and ambrisentan/tadalafil combination therapy 18 ) were evaluated in short-term trials assessing improvements in exercise capacity, 33 there is little evidence for their effectiveness on long-term morbidity/mortality endpoints. The prevalence of prostacyclin use was relatively low in our study (8.1% of index regimens; 22.4% of second regimens), comparable with an earlier claims analysis that found prostacyclin use among 22.0% of patients with identified PAH. 32 This finding may be related to the burden of administration and side effects of parenteral and inhaled prostanoids. With the availability of newer oral agents, the use of medications targeting the prostacyclin pathway may increase.
Despite the higher proportion of PDE-5I use compared with ERA use in our study, medication adherence and persistence were considerably higher for index regimens containing ERAs compared with PDE-5Is; PDC by drug class ranged from 0.6 (for PDE-5I and prostacyclins) to 0.8 (for ERA and sGCS). These values are somewhat lower than those from previous claims analyses, in which PAH-specific therapy adherence as assessed by medication possession ratio (MPR) was in the range of 0.86–0.96.24,25 However, PDC has become increasingly preferred over MPR as an adherence metric, as MPR-based adherence may appear inflated for multidrug regimens and among patients who refill their medications early. 34 The overall proportion of patients with PDC ≥ 0.90 was 49.4%, congruent with the 48.6% adherence found in an earlier administrative claims analysis of PAH-specific therapies 35 and within the 40–60% range estimated for adherence among patients with chronic diseases. 36 Treatment discontinuation was surprisingly common overall: 10.5% of patients had a gap of 90 days or more in their index regimen, while 27.1% discontinued their index regimen without starting another treatment during the course of the study. Although the reasons for treatment discontinuation cannot be discerned from claims data, these findings raise the possibility that misdiagnosis, intolerable side effects, or high medication costs were an issue for some patients. Interestingly, adherence in this study was considerably higher for patients using combination therapy versus monotherapy (90.4% vs. 46.6%), and patients who initiated combination therapy were more likely to remain on their index regimen at one year than those who initiated monotherapy (68.3% vs. 49.2%). While sequential combination therapy remains a valid and guideline-recommended approach, 12 it is possible that patients who were on initial combination therapy were less likely to discontinue or switch regimens because they had better clinical responses to treatment. This is congruent with the findings of recent meta-analyses indicating that combination therapy significantly reduces clinical worsening compared with monotherapy.13,14
Our study sample exhibited a varied and substantial comorbidity profile, which is consistent with previous analyses4–8 and reflects the known complexity, fragility, and high mortality of this patient population. 7 Conditions that have been shown to increase morbidity and/or risk of death among patients with PAH—including hypertension, obesity, and diabetes37—were widespread. Unsurprisingly, these patients were also frequent users of the healthcare system across both treatment cohorts, with nearly all having at least one ambulatory visit, 72% (commercial) to 79% (MAPD) having at least one emergency room visit, and 62% (commercial) to 70% (MAPD) having at least one hospitalization during the 12-month follow-up period. Heavy utilization of healthcare resources is a common finding among patients with PAH4,24,31,38,39 and congruent with the ongoing follow-up and regular assessment these individuals require. 12
Several co-morbidities were notably more prevalent in the present study compared with REVEAL, including systemic hypertension (81% vs. 40%), obstructive airway disease (43% vs. 22%), and sleep apnea (37% vs. 21%). While the higher prevalence of hypertension, diabetes, and sleep apnea may reflect the older age profile (mean age 65 years in our study vs. 53 years for REVEAL), the higher percentage of obstructive airway disease and the substantial proportion with respiratory failure or insufficiency (32%) suggest that some patients with group 2/3 PH or PH with mixed clinical phenotypes (e.g., disease with both left- and right-heart components 40 ) may have been included in our study cohort. The presence of patients who had group 2/3 PH despite being prescribed PAH-specific medications would not be surprising, as PH is often misdiagnosed and/or treated inappropriately, including as PAH, particularly outside of specialty centers.20,41,42 For example, in a 2016 analysis of patients referred to PH specialty centers after being diagnosed with PAH in community hospitals, 33% were found to have been misdiagnosed and 37% had not undergone cardiac catheterization, which is required for a definitive PAH diagnosis. 43 The widespread misdiagnosis of PAH may be attributable to its non-specific symptoms but also to its rarity, as non-specialists encounter few patients with PAH in clinical practice and may be unfamiliar with the proper diagnosis and referral pathways.
Our findings also underscore the heavy economic toll of PAH, with PPPM all-cause follow-up healthcare costs ranging from $6271 (MAPD) to $9503 (commercial) for the monotherapy cohort and $14,340 (MAPD) to $16,240 (commercial) for the combination therapy cohort. These figures are in line with other claims-based assessments of PPPM healthcare costs in this patient population, which have been in the range of $2576–11,875 in 2014 US dollars.4,6,9,24,25,31,44,45 However, the range of results from previous studies is somewhat wide, owing to differences in study methodology and patient selection (e.g., the proportion of patients with commercial insurance versus Medicare, the level of disease severity in the study group, and the criteria used for identification of PAH). Notably, PPPM total healthcare costs in the present study were similar between the baseline and follow-up periods among index monotherapy patients for commercial and MAPD enrollees, but more than doubled among index combination therapy patients for both insurance types. It is possible that the cost difference reflects a more advanced disease state among patients who initiated treatment with combination therapy. Higher pharmacy costs among patients treated with combination therapy also contributed to the cost difference, which may have been exacerbated by differences in disease progression between cohorts during follow-up. In the future, cost-effectiveness studies may help clarify the relative benefits of monotherapy versus combination therapy for PAH with regard to clinical outcomes.
Limitations
Our findings should be interpreted in light of certain limitations. Patients with index combination therapy constituted a small proportion of the overall study population; thus, direct comparison of the monotherapy versus combination therapy cohorts may have limited utility and was not a study objective. This study was conducted in a U.S. managed care population among individuals fulfilling the study criteria and may not be generalizable to other populations. The presence of a pharmacy claim does not indicate that the medication was consumed or that it was taken as prescribed; furthermore, medications provided through assistance programs or as samples from a physician are not observed in pharmacy data. In addition, the presence of a diagnosis code on a medical claim may not prove the presence of disease (as diagnosis codes may be incorrectly coded or included as rule-out criteria), and treatment patterns were not examined by PAH severity because FC cannot be discerned from retrospective claims data. This study did not incorporate the amounts estimated to be paid by other payers, resulting in more conservative cost estimates. Finally, ICD-9-CM, which was in use during the patient identification period of this study, contained diagnosis codes for PH but not specifically for PAH; however, the use of NDC pharmacy codes for PAH-specific drugs to identify patients in addition to ICD-9-CM diagnosis codes ensured that patients had been prescribed drugs with the intention of treating PAH.
Conclusion
Despite the robust clinical data in support of combination therapy for PAH, the majority of patients initiated medication treatment with monotherapies—most often PDE-5Is—and remained treated with monotherapies throughout the study, which had a mean follow-up time of >2 years. Adjustments to the index regimen occurred early and in a substantial proportion of patients, suggesting that inadequate clinical response may not be uncommon among patients treated with current monotherapies. ERAs and combination therapies were associated with higher medication adherence compared with PDE-5Is and monotherapies, respectively. Healthcare resource utilization was frequent, and costs were substantial across the study population, but more than doubled between the baseline and follow-up periods in the combination therapy cohort. Our findings highlight the potential value of future real-world studies examining the effect of various therapies on downstream clinical and cost outcomes among patients with PAH.
Conflict of interest
Authors J.P., Y.T., and W.D. are employees of Actelion Pharmaceuticals US, Inc. (South San Francisco, CA), which funded this study. Author S.S. provided consulting services to Actelion. Authors M.H. and E.K. are employees of Optum (Eden Prairie, MN), which was contracted by Actelion to perform this study. Medical writing services were provided by Yvette M. Edmonds, an employee of Optum.
Funding
This research was funded by Actelion Pharmaceuticals US, Inc.
