Abstract
Carboxyl terminus of Hsc70-interacting protein (CHIP) is a 35-kDa cytoplasmic protein expressed in human striated muscle, brain, aortic smooth muscle, endothelial cells, and other tissues. Studies have confirmed that CHIP regulates cell growth, apoptosis, cell phenotype, metabolism, neurodegeneration, etc. However, whether CHIP is involved in pulmonary artery smooth muscle cell (PASMC) proliferation, a vital contributor to chronic hypoxia-induced pulmonary hypertension (CHPH), remains unknown. In this study, we first evaluated CHIP expression in the pulmonary arteries (PAs) of CHPH model rats. Subsequently, by silencing CHIP, we investigated the effect of CHIP on hypoxia-induced PASMC proliferation and the underlying mechanism. Our results showed that CHIP expression was upregulated in the PAs of CHPH model rats. Silencing CHIP significantly suppressed the hypoxia-triggered promotion of proliferation, [Ca2+]i, store-operated Ca2+ entry (SOCE), and some regulators of SOCE such as TRPC1 and TRPC6 in cultured PASMCs. These results indicate that CHIP likely contributes to hypoxia-induced PASMC proliferation by targeting the SOCE-[Ca2+]i pathway through the regulation of TRPC1 and TRPC6 in the PASMCs. In conclusion, the findings of the current study clarify the role of CHIP in hypoxia-induced PASMC proliferation.
Keywords
Introduction
Pulmonary hypertension (PH) caused by progressive vascular remodeling and vasoconstriction in the pulmonary artery, eventually leading to right heart failure, is a lethal complication.1,2 Chronic hypoxia-induced PH (CHPH) is a type of PH, which was classified as group 3 at the sixth World Symposium of Pulmonary Hypertension held in Nice, 2018. Hypoxia-induced pulmonary artery smooth muscle cell (PASMC) proliferation, a key contributor to structural remodeling of distal pulmonary arteries (PAs), plays a significant role in CHPH.3,4 So far, the mechanism of hypoxia-induced PASMC proliferation has remained poorly understood; however, it has been generally accepted that elevation of intracellular Ca2+ concentration ([Ca2+]i) triggers PASMC proliferation.5,6 Commonly, [Ca2+]i increases via the following three pathways: (1) Ca2+ release from the intracellular Ca2+ store localized on the endoplasmic/sarcoplasmic reticulum; (2) Ca2+ introverted transshipment via Ca2+-ATPases or Na+/Ca2+ exchanger situated in the plasma membrane, and (3) extracellular Ca2+ influx via plasma membrane Ca2+ channels.7,8 To date, three kinds of Ca2+ channels have been found: voltage-dependent Ca2+ channel (VDCC), receptor-operated Ca2+ channel (ROCC), and store-operated Ca2+ channel (SOCC).2,7,9,10 Previous reports indicated that extracellular Ca2+ influx via SOCC, named store-operated Ca2+ entry (SOCE), contributes greatly to hypoxia-induced elevation of [Ca2+]i, causing PASMC proliferation.11–13 Studies revealed that canonical transient receptor potential channel (TRPC) proteins, a member of the subgroup of the transient receptor potential (TRP) superfamily with seven isoforms (TRPC1-7), are the main components of SOCC.14–16 While only TRPC1 and TRPC6 expression is selectively upregulated by hypoxia, which causes promotion of SOCE and [Ca2+]i, and leads to PASMC proliferation.17–20
Carboxyl terminus of Hsc70-interacting protein (CHIP), a 35-kDa cytoplasmic protein, is highly expressed in human striated muscle, brain, aortic smooth muscle, endothelial cells, and other tissues. 21 CHIP contains three tandem tetratricopeptide repeat (TPR) motifs and a U-box domain. Through the TPR-motifs CHIP interacts with the chaperones Hsp70 and Hsp90 and negatively regulates their ATPase activity and chaperone efficiency, and through the U-box domain it ubiquitinates chaperone-bound substrates.21–23 Reports indicate that by targeting a number of Hsp70/90-associated substrates or aggregation prone proteins for degradation by proteasome, CHIP regulates cell growth, apoptosis, cell phenotype, metabolism, neurodegeneration, etc., and plays a crucial role in cancer, metabolic disorders, liver injury, and tauopathy.24–36
Although it has been shown that CHIP is involved in many disorders, whether it plays a role in PASMC proliferation, a vital contributor to CHPH, remains unknown. In this study, we sought to determine the influence of CHIP on hypoxia-induced PASMC proliferation and the mechanism behind it. We investigated CHIP expression in the PAs of CHPH model rats and the effects of silencing CHIP on PASMC proliferation, basal [Ca2+]i, SOCE, and some components of SOCE such as TRPC1 and TRPC6 in the cultured rat distal PASMCs.
Methods
Establishment of the rat CHPH model
Sprague-Dawley (SD) rats weighing approximately 150 g were bought from the Zhejiang Provincial Experimental Animal Center and were raised in an SPF grade animal room for 7 days firstly. After that, rats (218–252 g) were randomly divided into two groups: normoxia group and hypoxia group. Hypoxia group rats were housed in a hypoxia chamber (10% O2) placed in the SPF animal room for 21 days as described previously. 13
Primary culture of rat distal PASMCs
Distal (>4th generation) PAs of SD rats 6- to 8-weeks old were isolated from lungs and PASMCs were harvested from distal PAs and cultured as previously described. 37 Cellular purity was assessed based on the morphological appearance using phase-contrast microscopy and immunofluorescence staining for α-actin using fluorescence microscopy.2,37 The purity in all experiments was assessed to be >95%.
siRNA silencing and cell proliferation assay
All siRNA duplexes were synthesized by Shanghai GenePharma. Knockdown efficiency was determined by real-time quantitative PCR (RT-qPCR) and western blotting. Cell proliferation was assessed using the CytoSelectTM BrdU Cell Proliferation ELISA Kit (Roche) according to the manufacturer’s manual.
Measurement of [Ca2+]i and SOCE
[Ca2+]i and SOCE were measured using Fura-2-AM dye (Molecular Probes, Eugene, OR) and fluorescent microscopy as previously described.2,13 To obtain statistically convincing results, the fluorescence intensity was determined in at least 20 cells for each sample.
RNA extraction and measurement by RT-qPCR
Total RNA from distal PAs and PASMCs of rats was extracted using the TRIzol Reagent (Invitrogen, Carlsbad, CA) method as previously described. 2 Reverse transcription and RT-qPCR were also performed as described previously. 2 RT-qPCR analysis was performed on a CFX96™ Real-Time System (Bio-Rad C1000). Reactions were assembled following the manufacturer’s recommendations.
Protein analysis by western blotting
Distal PAs or PASMCs were homogenized in TPER lysis buffer (Pierce, Rockford, IL) containing 5% protease inhibitor cocktail (Sigma, St. Louis, MO), 1 mM EDTA, and 200M4 - (2-aminoethyl) benzenesulfonyl fluoride hydrochloride. Western blotting was conducted according to the method previously described. 2
Statistical analysis
Data are expressed as mean ± SEM, and n is the number of experiments, which equals to the number of rats providing PAs or cells. Statistical analyses were conducted using one-way ANOVA and Student’s t test. All data in this study were tested and found fit for normal distribution. Differences were considered to be significant when p < 0.05.
Results
CHPH rat model establishment
After exposure to hypoxia (10% O2 for 21d), rats developed typical CHPH indicated by a significant increase in the right ventricular systolic pressure (RVSP) from 24.60 ± 2.80 mmHg to 39.26 ± 2.80 mmHg, mean right ventricular pressure (mRVP) from 10.13 ± 2.16 mmHg to 20.78 ± 3.10 mmHg, weight ratio of right ventricle (RV) to left ventricle (LV) plus septum [RV/(LV + S)] from 0.31 ± 0.04 to 0.5 ± 0.05 and weight ratio of right ventricle to body weight (RV/BW) (g/kg) from 0.58 ± 0.11 to 1.06 ± 0.15 compared to the normoxia group (Fig. 1a–e) (n = 5, p < 0.01).
Assessment of CHPH rat model. Rats were exposed to normoxia or hypoxia. (a) Representative traces of right ventricular pressure (RVP) of two groups of rats. (b–e) Bar graphs showing RVSP, mRVP, RV/(LV + S) and RV/BW (g/kg), respectively. Bar values are mean ± SEM (n = 5 in each group, ##p < 0.01 versus normoxia group). (f) and (g) Bar graph showing the thickness of tunica media smooth muscle and the cross-sectional area percentage of arterial wall compared to artery in two groups (n = 5 in each group, ##p < 0.01 versus normoxia group). (h) Represents picture of each group.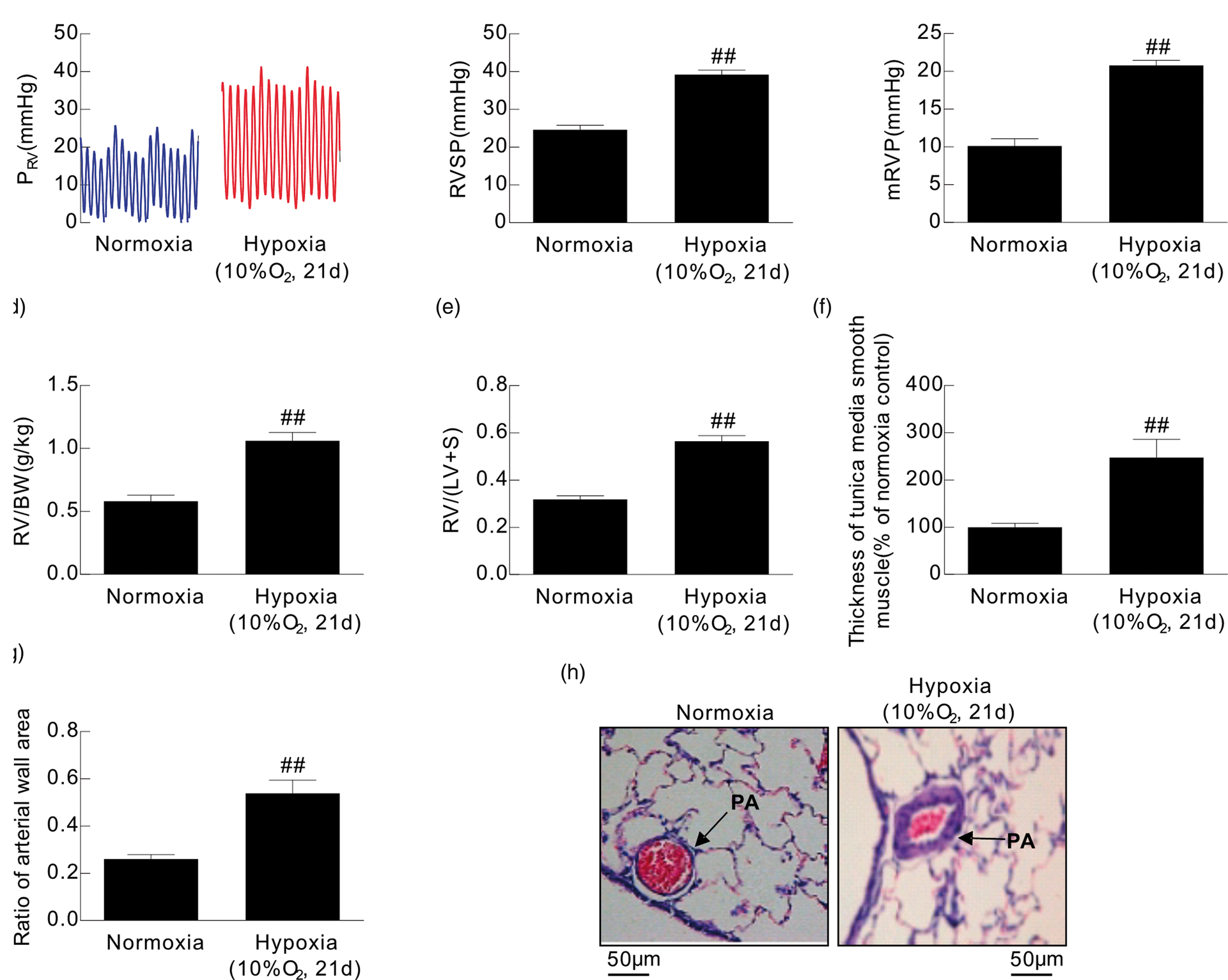
To evaluate pulmonary vascular remodeling, lung histology examination was performed in the two groups of rats. In accord with our previous results, PA wall thickening was significantly enhanced in the hypoxia group rats as demonstrated by the increase of the thickness of tunica media smooth muscle and ratio of arterial wall area, which further verified the establishment of the CHPH rat model (Fig. 1f–h) (n = 5, p < 0.01).
CHIP expression in distal PAs from CHPH model rats and cultured rat distal PASMCs exposed to hypoxia
Data showed that CHIP expression was upregulated by 95% and 180% at mRNA and protein levels respectively in the isolated distal PAs from lungs (Fig. 2a–c) (n = 4, p < 0.05, and p < 0.01 for mRNA and protein respectively). To determine whether the upregulated expression of CHIP was triggered directly by hypoxia in PAs, PASMCs isolated from normoxia rats were cultured in vitro, and exposed to hypoxia (4% O2, 60 h). Similar to the changes in PAs, CHIP was upregulated by 96% and 92% at mRNA and protein levels respectively (Fig. 2d–f) (n = 4, p < 0.01 for mRNA and protein respectively).
CHIP expression in PAs from CHPH rats and cultured rat distal PASMCs exposed hypoxia. CHIP expression in PAs and cultured rat distal PASMCs was assessed at mRNA and protein levels by RT-qPCR and western blot, respectively. (a) Bar graph showing the mRNA level of CHIP relative to 18S in PAs shown as percentage of normoxia group. (b) Representative blot of CHIP bands normalized to β-actin in PAs. (c) Bar graph showing the mean intensity of CHIP bands relative to β-actin in PAs. (d) Bar graph showing the mRNA level of CHIP relative to 18S in cultured rat distal PASMCs shown as percentage of normoxia group. (e) Representative blot of CHIP bands normalized to β-actin in cultured rat distal PASMCs. (f) Bar graph showing the mean intensity of CHIP bands relative to β-actin in cultured rat distal PASMCs. All data were presented as mean ± SEM (n = 4, #p < 0.05, ##p < 0.01 versus normoxia group).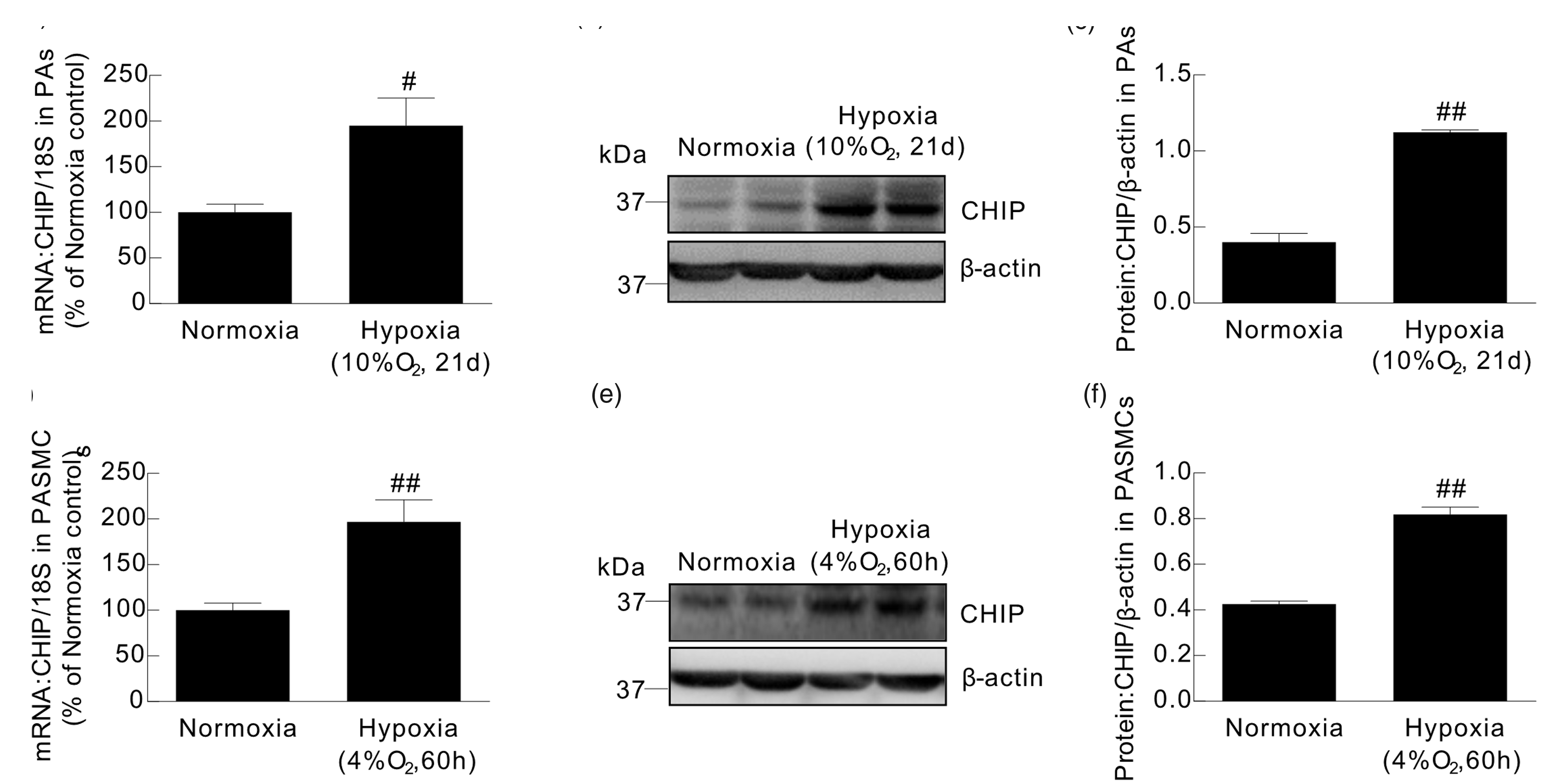
CHIP silencing effect on the hypoxia-triggered proliferation of cultured rat distal PASMCs
To investigate the effect of CHIP inactivation on the proliferation of cultured rat distal PASMCs, we silenced CHIP with siRNA. Knockdown efficiency was measured using RT-qPCR and western blotting, and was determined to be 85% and 59% at mRNA and protein levels respectively (Fig. 3a–c) (n = 4, p < 0.01 for mRNA and protein). After CHIP silencing, PASMCs were exposed to normoxia or hypoxia. Then, the proliferation rate of PASMCs was assessed by a BrdU incorporation assay. Data showed that silencing CHIP suppressed hypoxia-induced PASMC proliferation without an obvious effect on the proliferation of PASMCs exposed to normoxia (Fig. 3d) (n = 4, p < 0.01).
Assessment of CHIP knockdown efficiency and the effect of silencing CHIP on proliferation in cultured rat distal PASMCs. RT-qPCR and western blot were adopted to evaluate knockdown efficiency. (a) Bar graph showing the mRNA level of CHIP relative to 18S shown as percentage of normoxia control. (b) Representative blot of CHIP bands normalized to β-actin. (c) Bar graph showing the mean intensity of CHIP bands normalized to β-actin. Data were presented as mean ± SEM (n = 4, ##p < 0.01 versus normoxia control. (d) Bar graph showing PASMCs proliferation was assessed by BrdU assay, which is shown as percentage of normoxia control. Data were presented as mean ± SEM (n = 4, ##p < 0.01 versus normoxia control, **p < 0.01 versus hypoxia control).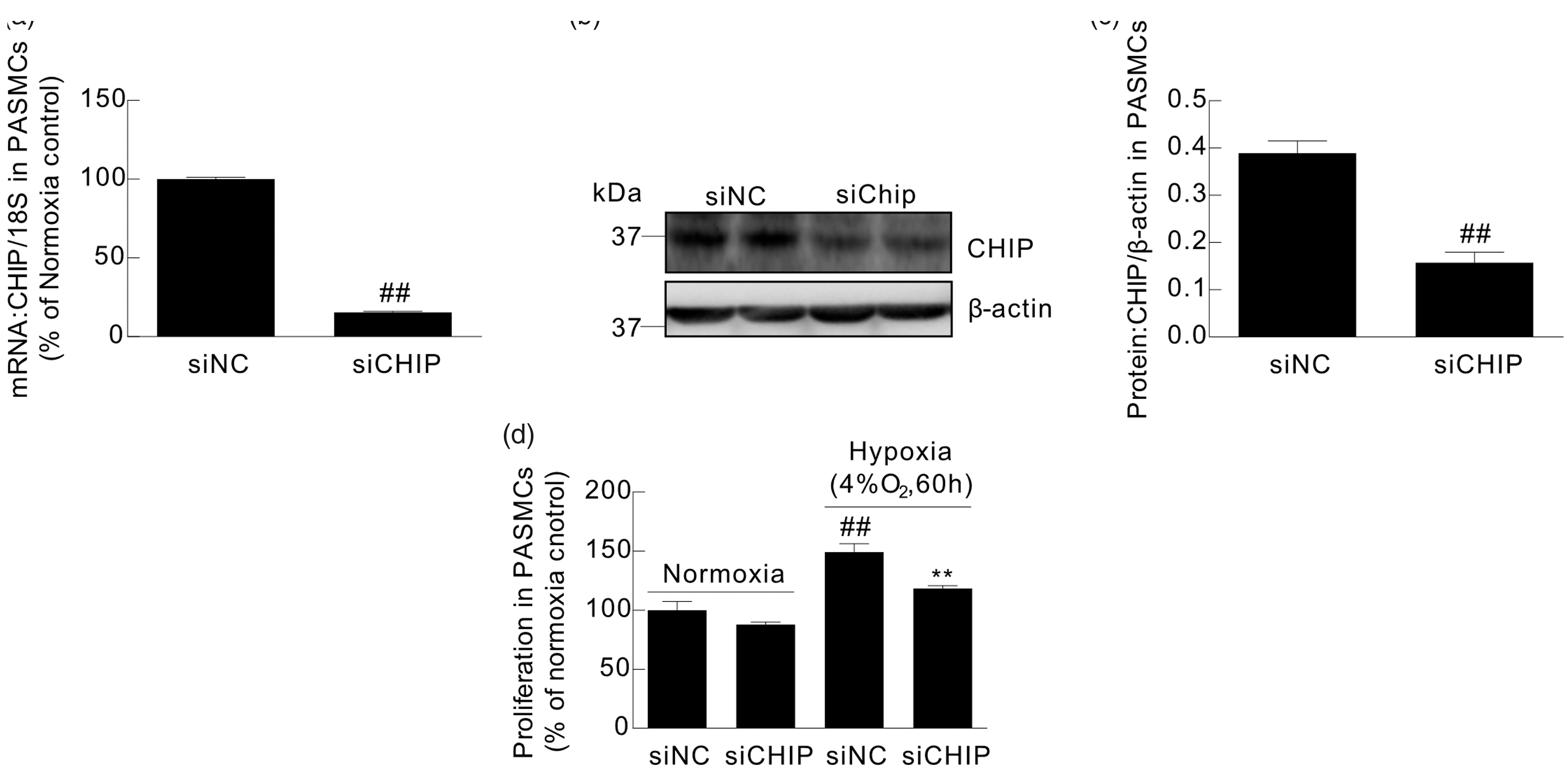
CHIP silencing effect on hypoxia-promoted basal [Ca2+]i and SOCE in cultured rat distal PASMCs
Results indicate that the ratio of F340/380 nm, an index indicating basal [Ca2+]i, increased to 1.34 ± 0.09 in PASMCs exposed to hypoxia compared with 1.02 ± 0.05 in PASMCs exposed to normoxia. (Fig. 4a and b) (n = 4, p < 0.01). However, silencing CHIP normalized the hypoxia-induced elevation of [Ca2+]i to 1.13 ± 0.06 (Fig. 4a and b) (n = 4, p < 0.01). SOCE evaluation by Mn2+ quenching at 360 nm and ΔF340/F380 nm during a 10 min perfusion with Ca2+-free PSS and a 10 min perfusion with Ca2+ PSS showed, in line with the change of basal [Ca2+]i, that CHIP silencing relieved hypoxia-induced SOCE in PASMCs. At the same time, we confirmed that silencing CHIP influenced neither basal [Ca2+]i nor SOCE in normoxic PASMCs (Fig. 4a and 4c–e) (n = 4, p < 0.01 for ΔF340/F380 nm and p < 0.05 for Mn2+ quenching at 360 nm).
Effect of silencing CHIP on SOCE and [Ca2+]i in cultured rat distal PASMCs. After silencing CHIP, PASMCs were exposed to normoxia or hypoxia. (a) Time course of F340/F380 nm during perfusion with KRBS in cultured PASMCs. (b) The bar graph shows ratio of F340/F380 nm in the first 5 min, which indicates [Ca2+]i in PASMCs. (c) Bar graph shows that, after restoration of extracellular [Ca2+], maximum increase of F340/F380 nm ratio indicating SOCE in PASMCs. (d) Time course of quenching of F360 nm by 200 μM Mn2+ after perfusion with Ca2+-free KRBS in PASMCs. (e) Bar graph indicates that quenching of F360 nm expressed as percentage at time 10 min in PASMCs. The bar values are the mean ± SEM (n = 4, ##p < 0.01 versus normoxia control, *p < 0.05, **p < 0.01 versus hypoxia control).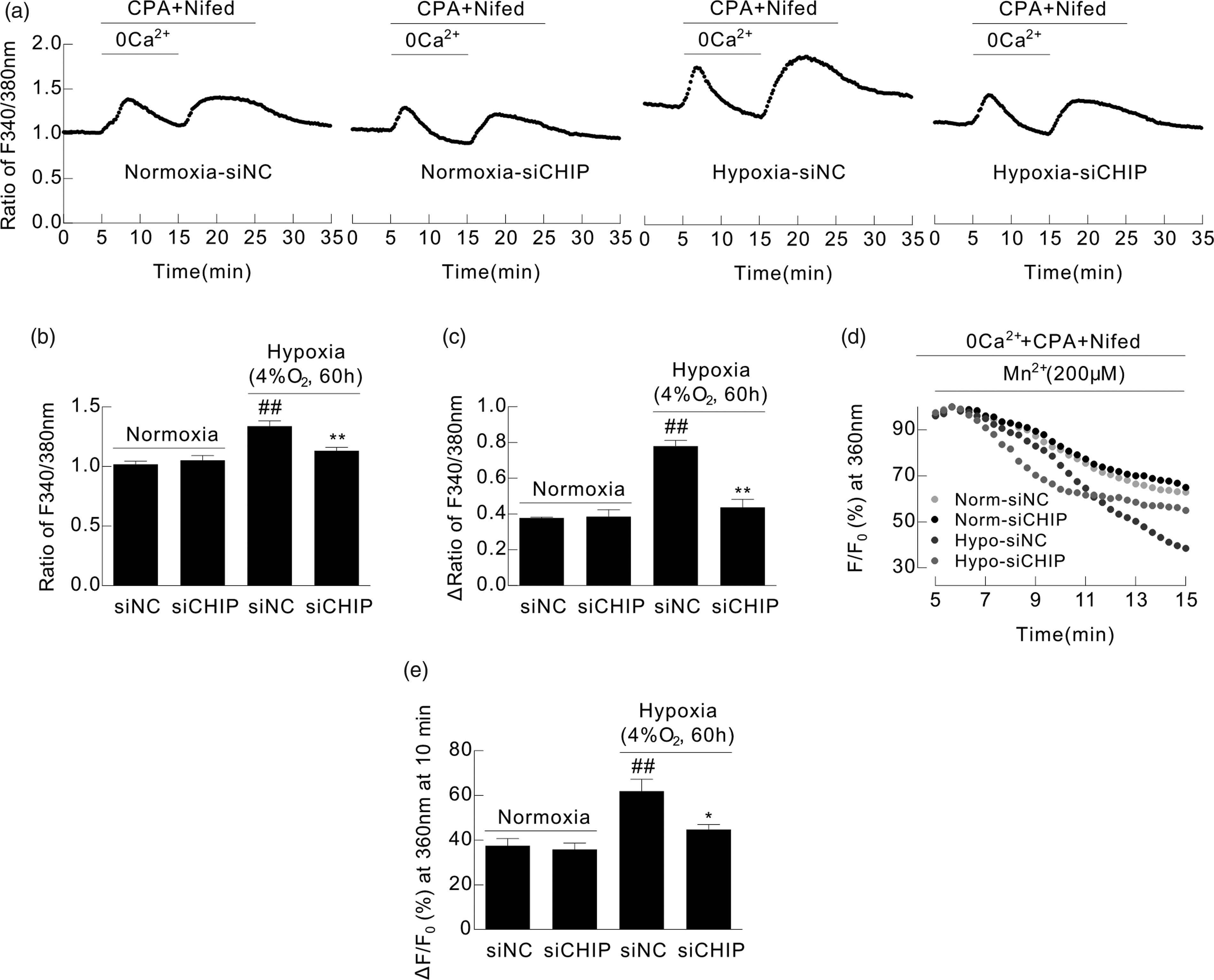
CHIP silencing effect on the hypoxia-upregulated TRPC1 and TRPC6 expression in cultured rat distal PASMCs
TRPC1 and TRPC6 expression analysis in PASMCs, as evaluated at both mRNA and protein levels, showed that hypoxia upregulated the expression of both TRPC1 and TRPC6. However, silencing CHIP inhibited this hypoxia-triggered upregulation without affecting TRPC1 and TRPC6 expression in PASMCs exposed to normoxia (Fig. 5a–g) (n = 4, p < 0.05 for mRNA and p < 0.01 for protein).
The effect of silencing CHIP on TRPC1 and TRPC6 expression in cultured rat distal PASMCs. The mRNA levels of CHIP, TRPC1, and TRPC6 relative to 18S were determined by RT-qPCR. And the results were shown as a percentage of the normoxia control (a–c). The protein levels of CHIP, TRPC1, and TRPC6 were assessed by western blot, and representative blots are shown (d). (e–g) The bar graphs show the mean intensity of each protein band relative to that of β-actin. Data are presented as the mean ± SEM (n = 4 in each group, #p < 0.05, ##p < 0.01 versus normoxia control, *p < 0.05, **p < 0.01 versus hypoxia control).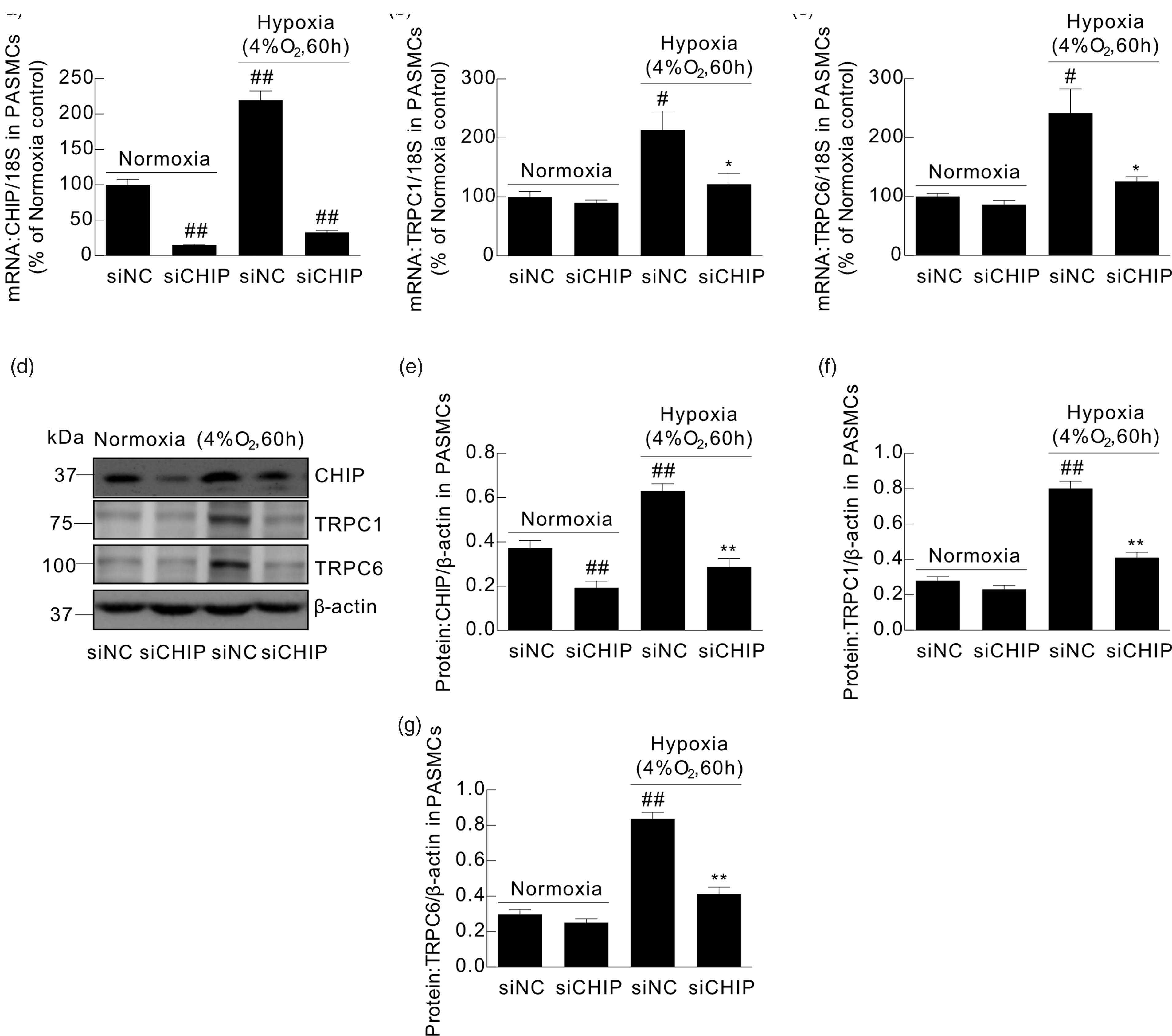
Discussion
This study demonstrated that silencing CHIP suppresses hypoxia-induced increase of proliferation, [Ca2+]i, SOCE, as well as TRPC1 and TRPC6 expression in cultured rat distal PASMCs.
Owing to its vital role in regulation of cell growth, apoptosis, cell phenotype, metabolism, neurodegeneration, etc., CHIP contributes greatly to many diseases such as cancer, metabolic disorders, live injury, and tauopathy.24–36 However, the precise role of CHIP in the pathogenesis of PH remains unclear. This study aimed to investigate whether CHIP is involved in CHPH and the underlying mechanism. To some extent, the expression change of CHIP is the precondition of its contribution to CHPH. If there is no change in CHIP expression in CHPH, we believe it is not likely that CHIP is involved in the pathogenesis of CHPH and there could be almost no value in researching this topic. Based on this view, we first investigated CHIP expression in PAs from CHPH rats. Results showed that CHIP expression was upregulated at both mRNA and protein levels. To determine whether the upregulation of CHIP expression in PAs resulted directly from the effect of hypoxia on PASMCs, CHIP expression was also detected in cultured PASMCs exposed to hypoxia in vitro. Consistent with the changes in PAs, the upregulation of CHIP expression was also detected. All these results indicate that hypoxia directly upregulates CHIP expression in PASMCs during the pathogenesis of CHPH suggesting that CHIP contributes to CHPH.
Previous studies showed that hypoxia-induced PASMC proliferation is a key contributor to the structural remodeling of distal PAs and plays a significant role in CHPH.3–5 Hence, it is rational to anticipate that CHIP contributes to CHPH by regulating PASMC proliferation in hypoxic conditions. To confirm this, we silenced CHIP with siRNA and evaluated its effect on PASMC proliferation. As anticipated, proliferation was suppressed in PASMCs exposed to hypoxia but not in PASMCs exposed to normoxia suggesting that CHIP regulates hypoxia-induced PASMC proliferation.
Next, we aimed to explore the mechanism underlying the regulatory effect of CHIP on hypoxia-induced PASMC proliferation. It is accepted that hypoxia-triggered elevation of [Ca2+]i is a crucial factor in PASMC proliferation and contributes greatly to CHPH.5,6,38 Previous studies demonstrated that Ca2+ entry via SOCC rather than VDCC stimulates hypoxia-enhanced [Ca2+]i in PASMCs.11–13 Therefore, we reasoned that the effect of CHIP on hypoxia- induced PASMC proliferation was probably mediated by SOCE-[Ca2+]i in PASMCs. To confirm this, we investigated the effect of CHIP on [Ca2+]i and SOCE in PASMCs by silencing CHIP. In order to exclude the possible influence of Ca2+ influx via VDCCs, an inclusion of VDCC specific inhibitor nifidepine in the perfusion buffer was designed during the measurement of SOCE. On condition of a CPA-induced calcium release from the cytoplasmic calcium store, we found that silencing CHIP decreased [Ca2+]i and SOCE in PASMCs exposed to hypoxia, but not in PASMCs exposed to normoxia. Thus, it is likely that silencing CHIP inhibited the hypoxia-induced PASMC proliferation by reducing [Ca2+]i via suppressing SOCE in PASMCs.
SOCE is operated by SOCC. We believe that the regulatory function of CHIP on SOCE is mediated by SOCC. TRPC1 and TRPC6 are the vital molecular components of SOCC, which are selectively upregulated in cultured rat distal PASMCs exposed to hypoxia and subsequently cause the promotion of SOCE, [Ca2+]i and proliferation of PASMCs, resulting in the structural remodeling of distal pulmonary arteries and stimulation of CHPH development.17–19 Consistent with our previous report, hypoxia upregulated TRPC1 and TRPC6 expression in the current study, however, silencing CHIP remarkably inhibited this hypoxia-triggered upregulation in PASMCs, indicating that CHIP probably regulated SOCE and [Ca2+]i by influencing the expression of TRPC1 and TRPC6 in PASMCs.
This study estimated the effect of CHIP inactivation on proliferation, [Ca2+]i, SOCE and TRPC1 and TRPC6 expression by silencing CHIP in PASMCs. Whether activating CHIP could cause their enhancement deserves further investigation, which might further reveal the influence of CHIP on these parameters. Moreover, further research is needed to understand whether CHIP regulates PASMC proliferation via the TRPC1,6-SOCE-[Ca2+]i pathway. Peroxisome proliferation-activated receptor gamma (PPARγ) is a ligand-activated transcription factor from the nuclear receptor family of peroxisome proliferator-activated receptors (PPARs), which was confirmed to attenuate PASMC proliferation by targeting the TRPC1,6-SOCE-[Ca2+]i pathway.39–42 Kim et al. 32 reported that CHIP could directly bind to and induce the ubiquitylation of PPARγ, leading to the degradation of PPARγ by a proteasome-dependent mechanism. Based on these reports, we assume that PPARγ likely mediates the CHIP associated regulation of the TRPC1,6-SOCE-[Ca2+]i pathway. However, further studies are required to confirm this.
The current study has few limitations. The mechanism by which CHIP causes [Ca2+]i alteration remains uninvestigated. Moreover, the function of CHIP in CHPH has not been demonstrated in genetically modified animals.
Nevertheless, our study demonstrates the important role of CHIP in hypoxia-induced PASMC proliferation. In addition, we report a potential correlation between this effect and the TRPC1,6-SOCE-[Ca2+]i pathway. As a result of its contribution to the hypoxia-induced PASMC proliferation, CHIP might be a novel target candidate for controlling CHPH.
Footnotes
Authors’ contribution
JZ initiated and designed the project, analyzed data, and wrote the paper. JZ and FD performed the animal, functional, and molecular experiments.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This study was funded by the PhD Research Startup Fund of Lishui University (QD1819).
Statement of ethics
All procedures were approved by the Animal Research Ethics Board of the Lishui University and all experiments were performed according to relevant guidelines and regulations.
