Abstract
Venous malformations have static venous lakes that predispose to spontaneous venous thrombosis within the malformation due to its low-flow static state. Thrombi of varying sizes can then embolize continually into the pulmonary arterial circulation, and occlude and narrow elastic pulmonary arteries causing chronic thromboembolic pulmonary hypertension (CTEPH). Pulmonary thromboendarterectomy (PTE) is potentially curative in CTEPH, but has not been previously reported in the setting of mediastinal and chest wall venous malformations. We report the case of a 21-year-old female with such a large malformation treated successfully with PTE. The patient underwent complete endovascular reconstruction of her subclavian vein system from the axillary vein to the innominate vein stump with covered stent grafts to exclude the malformations from causing recurrent pulmonary emboli. This was followed by embolization of the malformation to allow for the surgical approach. The series of events in this case serves as a novel approach in managing such rare patients.
Introduction
Venous malformations (VMs) predispose to deep venous thrombosis that may cause pulmonary emboli (PE). Recurrent thromboembolism can occlude and narrow elastic pulmonary arteries, resulting in potential complications such as chronic thromboembolic pulmonary hypertension (CTEPH). The treatment of choice for CTEPH is pulmonary thromboendarterectomy (PTE), the surgical removal of fibrotic thromboembolic lesions. PTE improves symptoms, right heart hemodynamics, and survival.1,2 PTE has been performed in patients with VMs involving the extremities, most notably in the setting of Klippel-Trenaunay syndrome.3–5 We report the case of a 21-year-old woman with CTEPH due to an extensive mediastinal–left chest wall VM and, unusually, congenital absence of the ipsilateral left subclavian vein. She underwent endovascular reconstruction of her left subclavian vein system thereby excluding the VMs from any possibility for causing further PE, then underwent a multi-staged endovascular isolation and embolization of the VMs problematic for surgical access, successfully followed by PTE.
Case report
A 21-year-old woman with a massive mediastinal–left chest wall VM, atresia of the left subclavian vein, and CTEPH was referred for evaluation primarily for PTE and secondarily for heart–lung transplantation. The patient was born with a compressible mass extending from the left neck, left chest, and left axillary areas. Six months prior to referral, she was diagnosed with an acute PE and treated with warfarin. She had not had an acute venous thromboembolic event prior to this and therefore was not previously on anticoagulation. She then reported progressive exercise intolerance progressing to severe dyspnea on exertion, ultimately becoming wheelchair-bound. Her pulmonary artery pressure was elevated and oral tadalafil therapy was started. Contrast-enhanced chest computed tomography (CT) revealed an extensive VM in the left anterior mediastinum immediately behind the sternum, extending across the midline anterior to the ascending thoracic aorta and through the thoracic outlet into left axilla and left chest wall. The vascular mass abutted the right ventricular outflow tract and the main pulmonary artery (Fig. 1).
Pre-treatment chest computed tomography. Axial contrast enhanced computed tomography images demonstrate a large venous malformation with mediastinal component extending via the thoracic outlet into the chest wall (*, panel a). The malformation abuts critical vascular structures in the mediastinum and lies deep to the sternum (*, panel b). Figure 1c demonstrates chronic intraluminal pulmonary artery thrombus in the left upper lobe (arrow, panel c).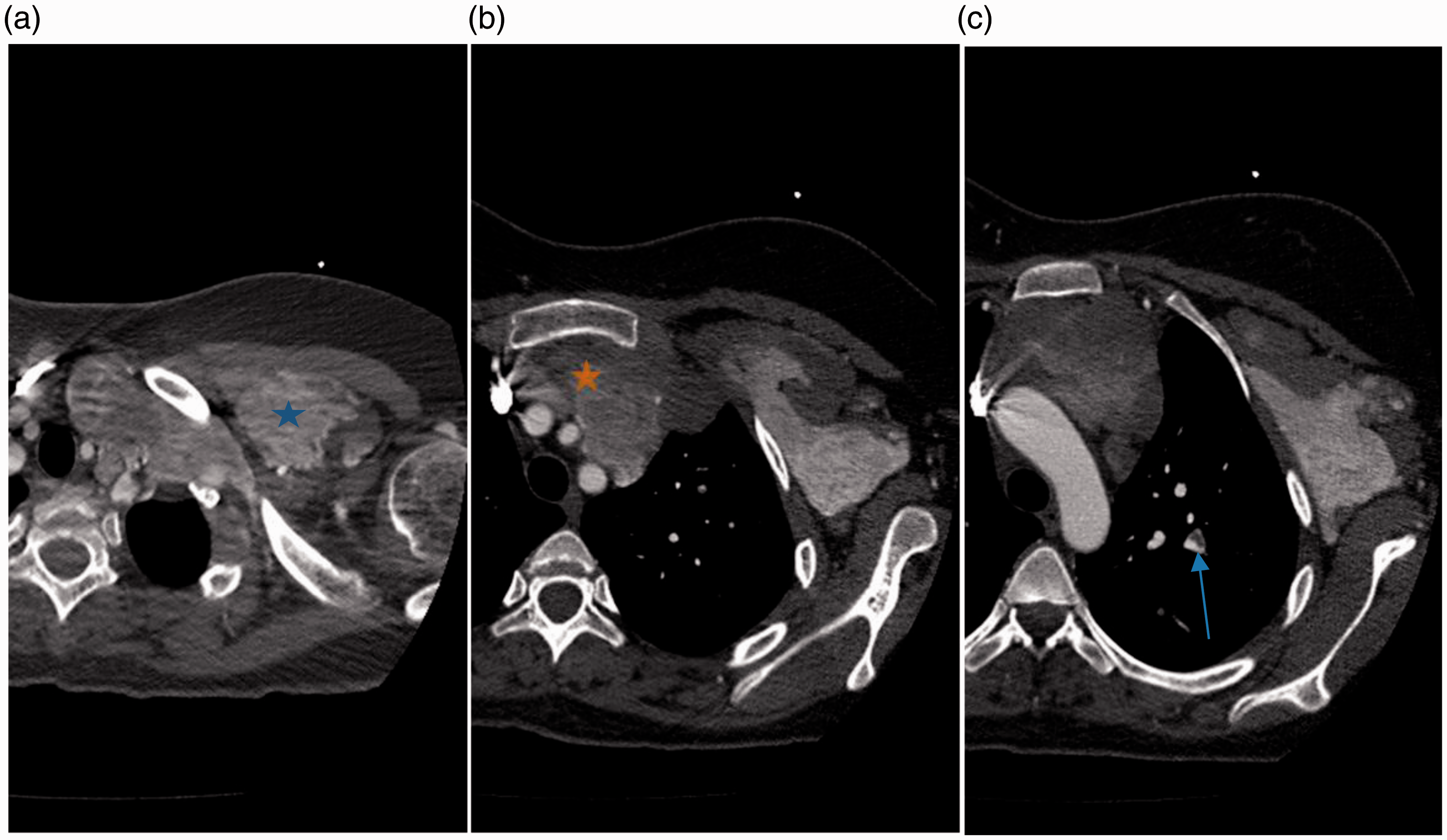
Left upper extremity venography demonstrated complete interruption in the continuity between the left axillary vein and innominate vein due to congenital absence of the left subclavian vein. The left chest VM was directly opacified from the left axillary vein on the contrast injection. The only venous egress from the massive left chest VM was through the left innominate vein stump, which connected into the superior vena cava (SVC), then to the right atrium. Thus, spontaneous thrombi in the left chest VM intermittently detached and caused recurrent PE. Lifelong anticoagulation alone was not considered to be a viable solution given it would have to be interrupted perioperatively for the PTE leaving her at high-risk for recurrent PE postoperatively. Additionally, should she develop any type of bleeding complication, there was no mechanical method (an “IVC filter equivalent”) to prevent PE if future transient interruptions of anticoagulation became necessary.
Reconstruction of the left subclavian vein was then required to exclude the VM from the venous circulation, thus obviating any further possibility for PE. Right femoral vein access allowed catheterization into the SVC, then via the left innominate vein stump into the left chest wall VM cavernous spaces. The left arm percutaneous basilic vein access allowed catheterization through the left axillary vein (which ended in the VM) into the left chest VM cavernous spaces. This dual venous access then allowed snaring of an exchange-length guide wire, thus bridging the congenital left subclavian vein atresia with through-and-through endovascular control from the right groin to the left arm. A series of covered stent-grafts were deployed (Fig. 2) establishing continuous flow from the left upper extremity into the SVC excluding the left chest wall VM from causing any further PE. In order to maintain long-term patency of these stent-grafts, a small arterio-venous fistula was created between a small branch of the left brachial artery and basilic vein.
Reconstruction of the left axillary-subclavian-innominate veins. Figure 2(a) demonstrates a measuring wire snared across the congenitally absent left subclavian vein and extensive contrast opacification of the venous malformation on venography. Figure 2(b) was obtained following placement of series of covered stent-grafts from the left axillary vein to the stump of the left innominate vein bridging the vein interruption and excluding the venous malformation as a source of pulmonary embolism.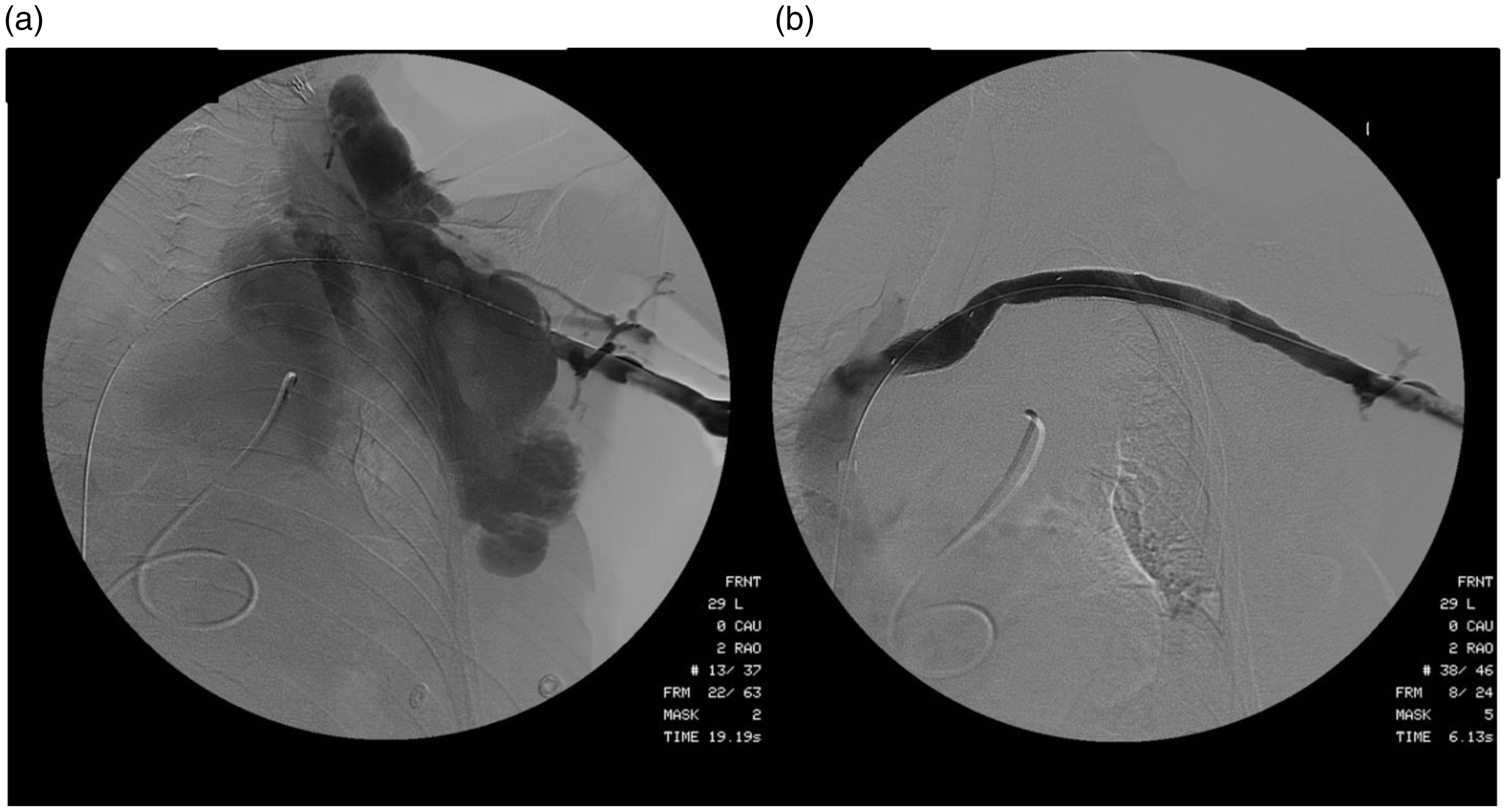
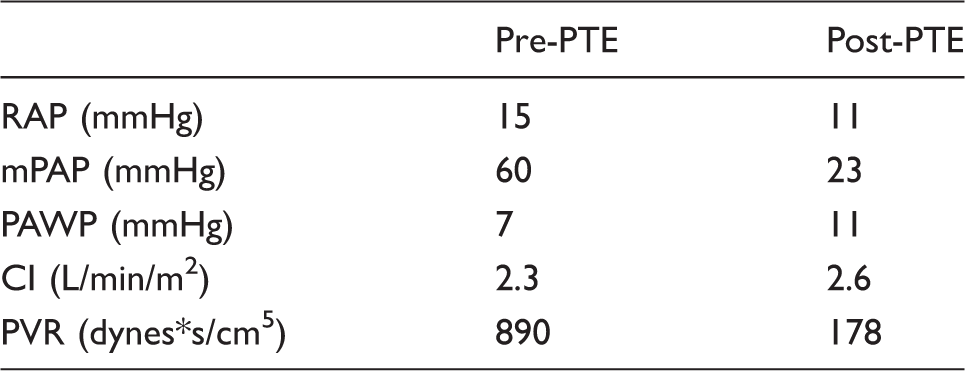
Digital subtraction pulmonary angiography documented central and peripheral chronic thromboembolic disease (Fig. 3). Large cavernous compartments of the left chest–mediastinal VM were defined by direct puncture angiograms. Right heart catheterization demonstrated severe pulmonary hypertension (Table 1).
Digital subtraction pulmonary angiography. Right and left pulmonary angiograms demonstrating markedly dilated central pulmonary arteries with tapering, marked tortuosity of the segmental branches, lobar and segmental areas of decreased perfusion (blue arrow), vascular amputations (red arrow), vascular webs (black arrow), foci of stenosis, all consistent with chronic thromboembolic pulmonary hypertension. Right heart catheterization measurements before and immediately after pulmonary thromboendarterectomy. CI, cardiac index; mPAP, mean pulmonary artery pressure; PAWP, pulmonary artery wedged pressure; PTE, pulmonary thromboendarterectomy; PVR, pulmonary vascular resistance; RAP, right atrial pressure.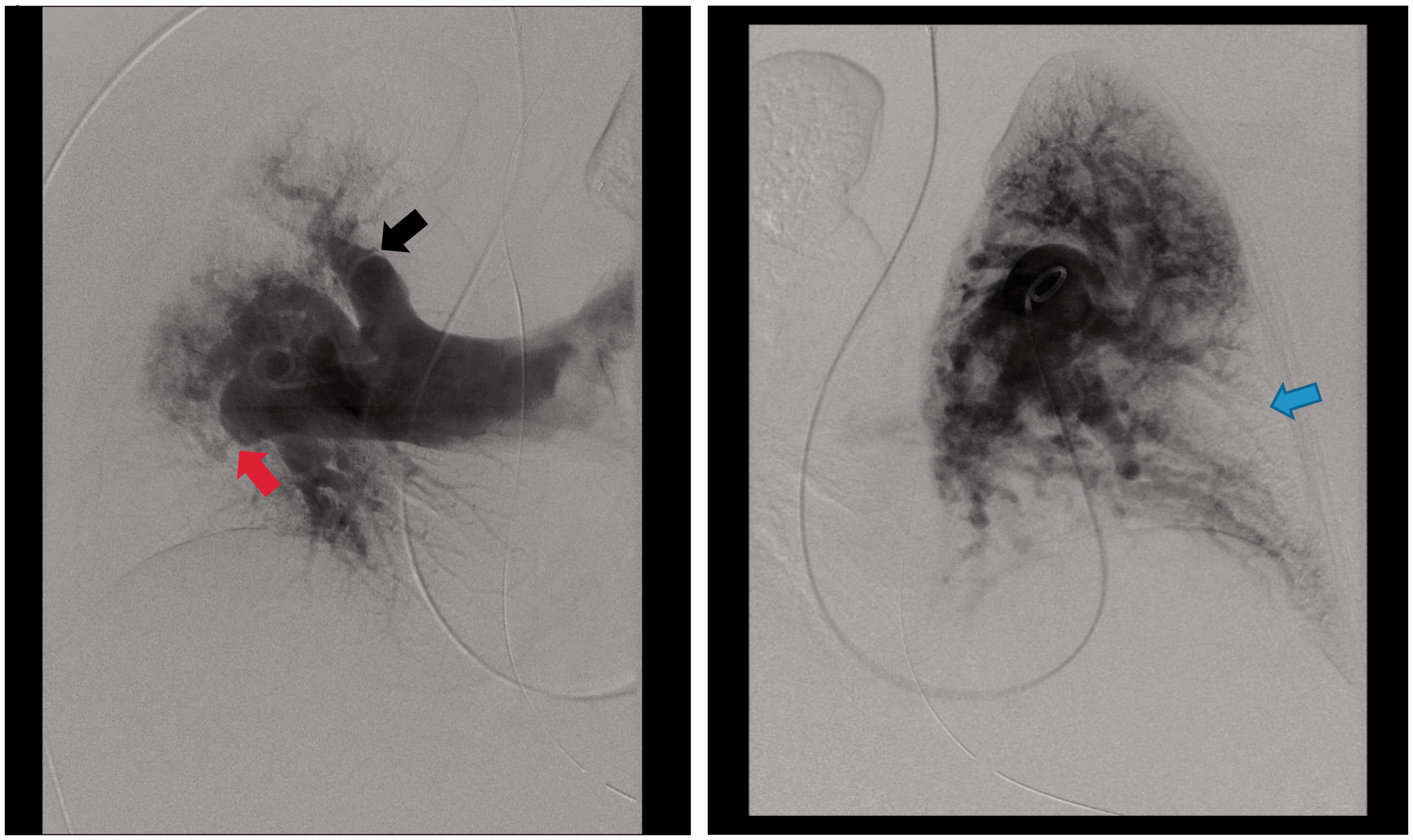
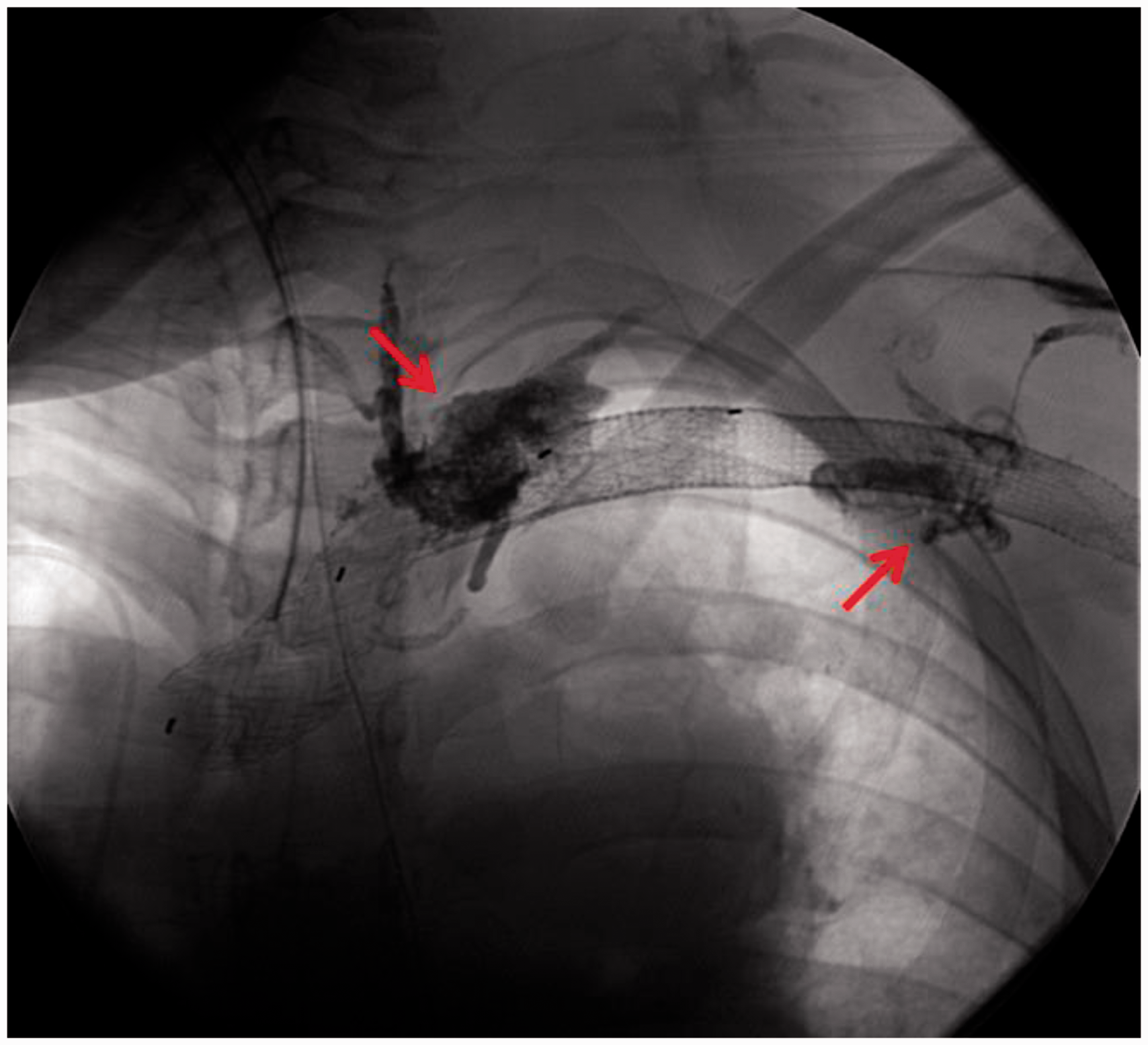
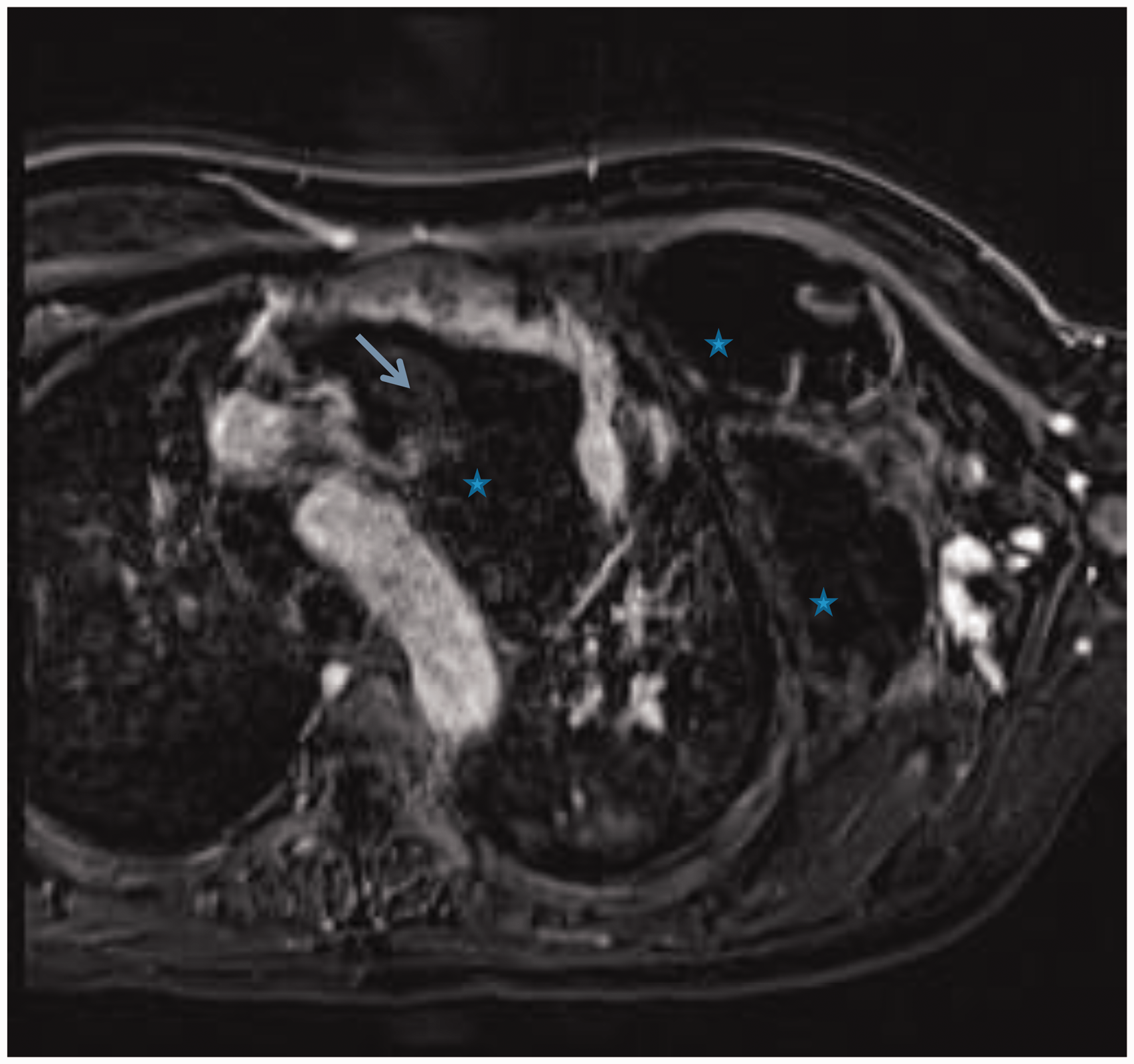
To identify the vein tributaries that also connected to the left chest VM, ultrasound-guided access was obtained into an outflow vein of the arterio-venous fistula and a time-resolved gadolinium magnetic resonance (MR) venography was performed. This identified several venous connections to the VM from the left axillary vein (Video 1, Fig. 4). Endovascularly, each of these vein connections were successfully occluded, and residual venous lakes in the left chest wall component of the malformation were occluded by direct puncture (Fig. 5). Follow-up gadolinium MR venography demonstrated significant thrombosis of the VM (Fig. 6) and exclusion of the malformation from the systemic circulation (Video 2). Embolization of the VM allowed safe entry into the mediastinum. The VM was partially thrombosed and had to be dissected off the innominate vein, aorta, and SVC. An incision was made in the VM, the clot was extracted, and a basket sucker was used to maintain it in a decompressed state. Once the VM was mobilized, cannulation for cardiopulmonary bypass was performed in the usual fashion and the PTE was accomplished during periods of circulatory arrest. A complete PTE was performed using 23 min of circulatory arrest for the left side and 10 min for the right side. The patient had an uneventful postoperative course with significant hemodynamic (Table 1) and clinical improvement.
Time resolved magnetic resonance angiogram (TWIST sequence). This was performed while injection of contrast via the outflow vein of the arterio-venous fistula and it demonstrates multiple venous feeding pathways (red arrows) that briskly fill both the mediastinal and chest wall component of the malformation (*). The stent-grafts (blue arrow) in the axillary, subclavian and innominate veins are patent. Venous malformation embolization. Embolization of multiple feeding pathways (red arrows) identified on the magnetic resonance angiography using NBCA glue and sodium tetradecyl sulfate solution using selective catheter access of each one of the venous pathways and direct puncture some of the venous malformation cavernous spaces. Post-embolization magnetic resonance venography. Post-gadolinium axial image demonstrates areas of absent flow (*) indicative of thrombosis within the venous malformation, and the residual partially thrombosed venous malformation compartments (blue arrows).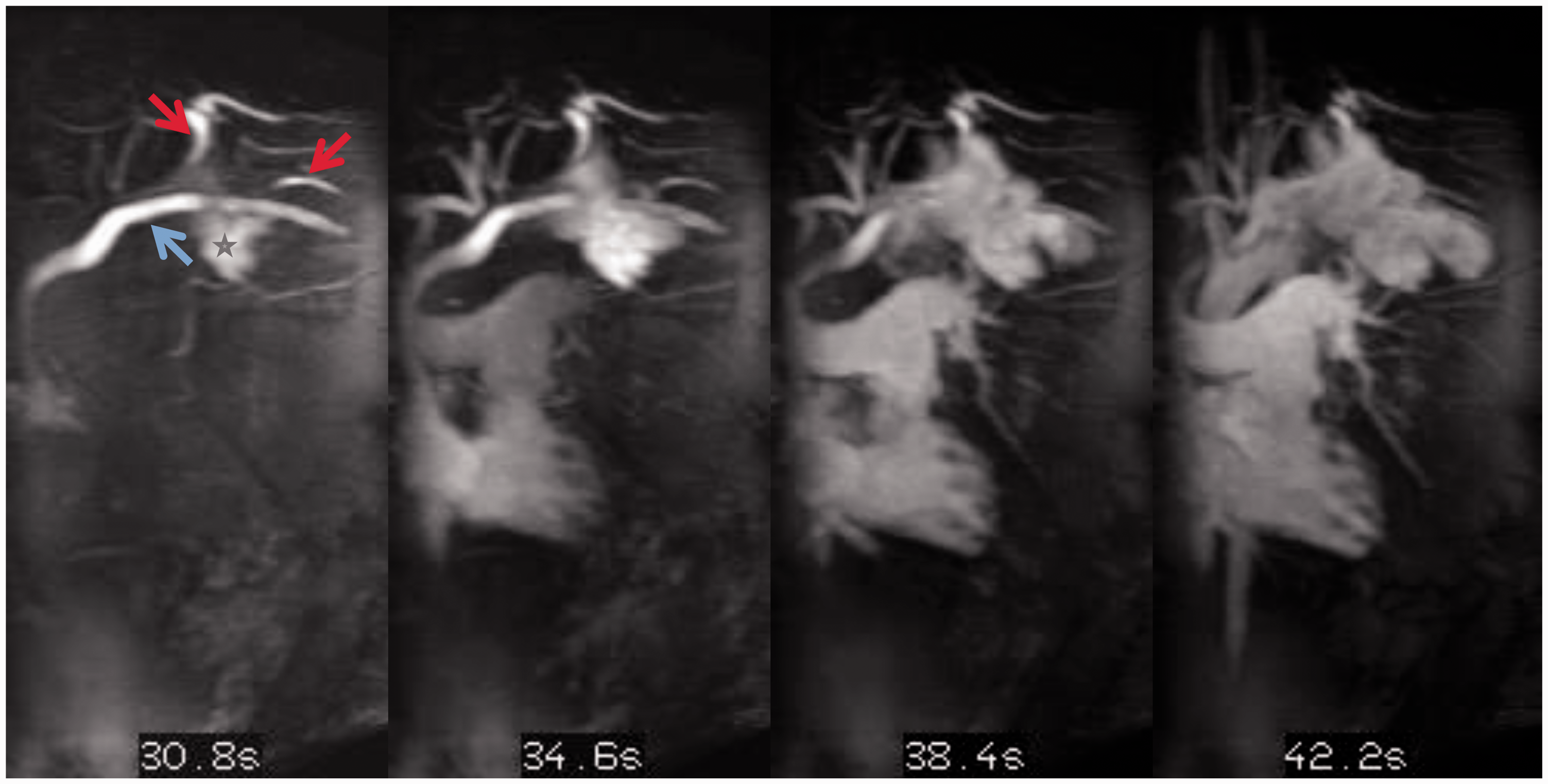
Discussion
CTEPH secondary to recurrent PE from the cavernous spaces of a mediastinal–left chest wall VM with congenital absence of the ipsilateral left subclavian vein has not been reported in the world’s literature. A collaborative effort among multiple specialists was critical in the successful management of this complex patient. PTE was feasible only after innovative solutions that led to successful thrombosis of the malformation. Reconstruction of the left axillary to innominate vein with covered stent-grafts was essential to obviate any further PE phenomena. Otherwise, PTE alone would have only been a temporizing measure. Utilizing MR imaging, identification of smaller vein connections to the cavernous VM enabled selective endovascular embolization, and then the partial VM percutaneous thrombosis allowed surgical access into the mediastinum.
Following PTE, this patient’s hemodynamics (Table 1) and symptoms improved greatly. The estimated 5-year survival rates for post-PTE are 85–90%1,2 compared to the estimated 50% 5-year survival post-lung transplant. 7 Accordingly, performing PTE significantly prolonged the estimated life expectancy in this young 21-year-old severely debilitated patient. To our knowledge, this represents the first PTE performed in the setting of a mediastinal–chest wall VM, the first complete construction of a congenitally absent left subclavian vein, and direct puncture endovascular occlusion of the chest and mediastinal compartments of the VM, to allow PTE. This case exemplifies a pioneering team approach in the management of a unique, complex patient.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
