Abstract
Currently, five classes of drug are approved for the treatment of pulmonary arterial hypertension (PAH): phosphodiesterase 5 inhibitors (PDE5i); endothelin receptor antagonists; prostacyclin analogs; the IP receptor agonist selexipag; and the soluble guanylate cyclase (sGC) stimulator riociguat. For patients with inoperable or persistent/recurrent chronic thromboembolic pulmonary hypertension (CTEPH), riociguat is currently the only approved pharmacotherapy. Despite the development of evidence-based guidelines on appropriate use of specific drugs, in clinical practice patients are often prescribed PAH-targeted therapies off label or at inadequate doses.
PDE5i are the most often prescribed class of drugs as initial therapy, either alone or in combination with other drug classes. However, a proportion of patients receiving PAH therapies do not reach or maintain treatment goals. As PDE5i and riociguat target different molecules in the nitric oxide-sGC-cyclic guanosine monophosphate (NO-sGC-cGMP) signaling pathway, for patients with PAH without an initial or sustained response to PDE5i, there is a biological rationale for switching to riociguat. However, robust data from randomized controlled trials on the safety and efficacy of switching are lacking, as is formal guidance for clinicians. Here we review studies of sequential combination therapy, and trial data and case studies that have investigated switching between PAH-approved therapies, particularly from PDE5i to riociguat in patients with PAH with an insufficient response to PDE5i, and in patients with CTEPH who were receiving off-label treatment. These studies summarize the current evidence and practical real-life experience on the concept of switching treatments.
Keywords
Introduction
Pulmonary hypertension (PH) is a progressive disease that leads to pulmonary arteriolar vasoconstriction, right-ventricular overload, and ultimately death. Pulmonary arterial hypertension (PAH) is characterized by reduced levels of the vasodilators nitric oxide (NO), cyclic guanosine monophosphate (cGMP), and prostacyclin, and overexpression of the vasoconstrictor endothelin. This results in increased vascular tone, and promotes vascular remodeling and the progressive destruction of the pulmonary vascular bed. Chronic thromboembolic pulmonary hypertension (CTEPH) is caused by obstruction of the pulmonary vasculature by residual organized thrombi, leading to increased pulmonary vascular resistance (PVR), progressive PH, and ultimately right ventricular failure.1–3
Five classes of drug are approved for the treatment of PAH, targeting three signaling pathways (Fig. 1):4–7 prostacyclin analogs (PCAs) epoprostenol, iloprost, treprostinil, and beraprost; the IP receptor agonist selexipag; the endothelin receptor antagonists (ERAs) bosentan, ambrisentan, and macitentan; the phosphodiesterase (PDE5) inhibitors (PDE5i) sildenafil and tadalafil; and the soluble guanylate cyclase (sGC) stimulator riociguat, also approved for patients with inoperable CTEPH or persistent/recurrent CTEPH after surgery.
Impairment of the NO sGC-cGMP signaling pathway (Fig. 2) is central to the pathogenesis of PAH and CTEPH,8–10 where endothelial dysfunction leads to impaired NO synthesis.10–14 The progression of PAH and CTEPH also correlates with low NO production.
Mechanisms of action of approved classes of PAH-targeted therapies.6 cAMP, cyclic adenosine monophosphate; cGMP, cyclic guanosine monophosphate; endothelin receptor antagonists (ERA), endothelin receptor A; ETRB, endothelin receptor B; IP, prostacyclin; NO, nitric oxide; PDE5, phosphodiesterase type 5; PH phosphodiesterase 5 inhibitors (PDE5i); pulmonary hypertension; PKA, phosphate kinase A; PKG, cGMP-dependent protein kinase; RV, right ventricular; sGC, soluble guanylate cyclase.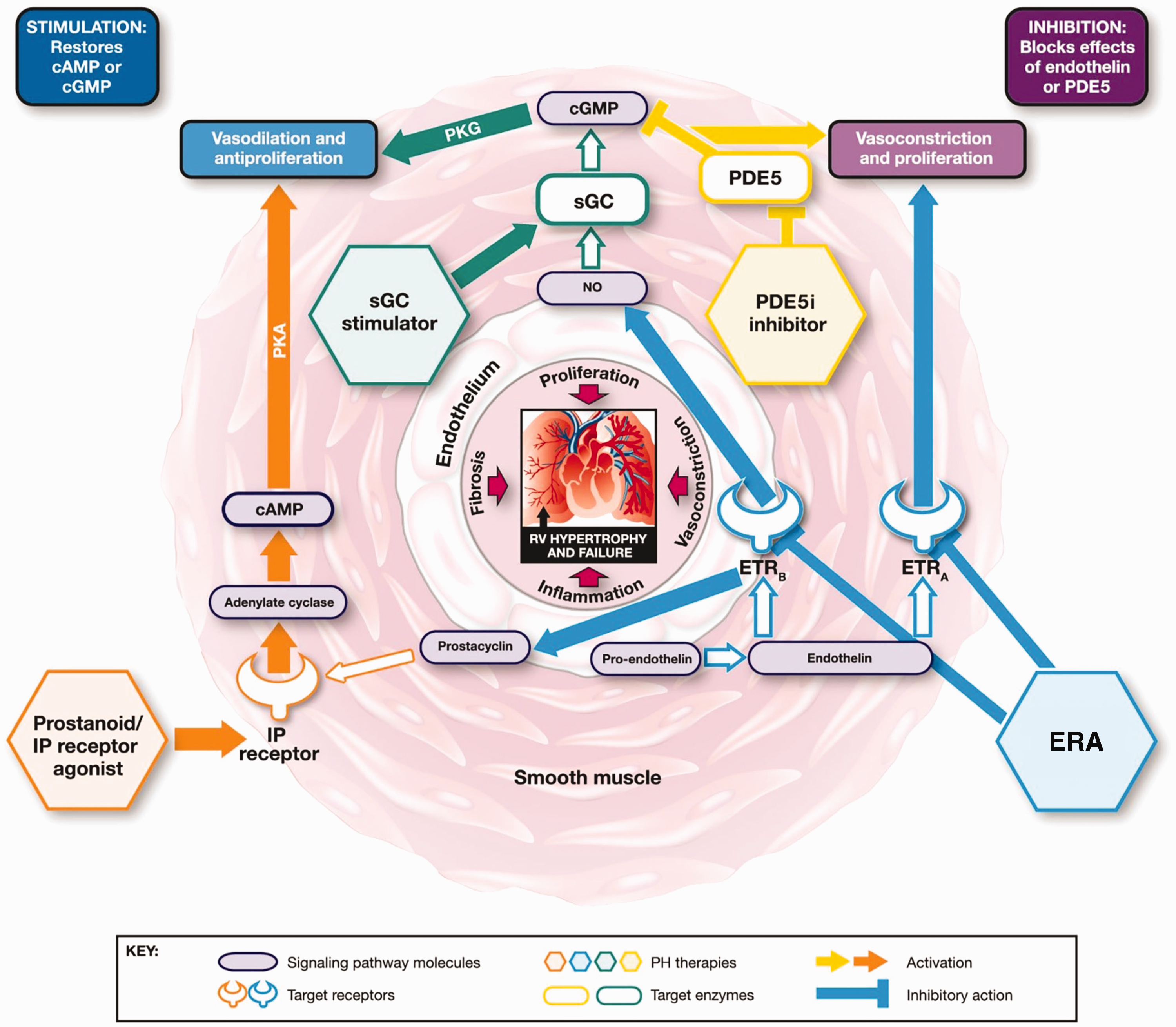
The purpose of this review is to provide a summary of published experience of trials and case studies that have investigated switching between approved PAH therapies, particularly switching within the NO pathway in patients with PAH and switching from off-label therapies to riociguat in patients with CTEPH, and an overview of the options for sequential combination therapy. Given that switching patients from PDE5i to riociguat is already taking place in clinical practice despite a lack of guideline recommendations, we also provide some cautionary notes on best practice.
Methods
To summarize a broad review of trials and case studies, a PubMed literature search was performed using the following search terms: “pulmonary arterial hypertension,” “pulmonary arterial hypertension” AND “transition,” and “pulmonary arterial hypertension” AND “switch.” To identify studies of combination therapy, we searched for the following drug names: riociguat, sildenafil, tadalafil, bosentan, ambrisentan, macitentan, selexipag, epoprostenol, treprostinil, iloprost, and beraprost.
Current treatment strategies for PAH
PDE5i and riociguat both target the NO-sGC-cGMP signaling pathway to promote vasodilation with different mechanisms of action (MoAs) (Fig. 1).
6
PDE5 deactivates and degrades cGMP, is abundantly expressed in pulmonary vasculature, and is upregulated in PAH. PDE5i occupy the catalytic site on PDE5, blocking degradation of cGMP (Fig. 1).6,15 However, the MoA of PDE5i is dependent on endogenous NO bioavailability, and evidence suggests that NO and intracellular levels of cGMP are depleted during the progression of PAH, which could render PDE5i less effective. This may explain why some patients do not have a sufficient sustained response to PDE5i. Riociguat has a dual MoA; it sensitizes sGC to endogenous NO and directly stimulates sGC via a second binding site, independent of NO, and has been shown to increase sGC activity regardless of NO and cGMP levels, resulting in increased cGMP. ERAs, PCAs, and selexipag target different pathways. ERAs prevent endothelin-1 (ET-1)- mediated vasoconstriction by blocking the binding of ET-1 to ET-1 receptors (Fig. 1), which are upregulated in PAH. PCAs are synthetic analogs of the pulmonary vasodilator prostacyclin (also known as prostaglandin I2) and selexipag is a high-affinity agonist of the human IP receptor. In PAH, prostacyclin synthase is downregulated and thus, prostacyclin levels are decreased (Fig. 1).
The role of the NO signaling pathway in PAH and CTEPH pathogenesis16 (amended from Buglioni and Burnett. Annu Rev Med 2016; 67: 229–243). cGMP, cyclic guanosine monophosphate; GC-A, particulate guanylate cyclase A; GTP, guanosine triphosphate; NO, nitric oxide; NOS, nitric oxide synthase; PDE, phosphodiesterase; PKG, cGMP-dependent protein kinase; sGC, soluble guanylate cyclase.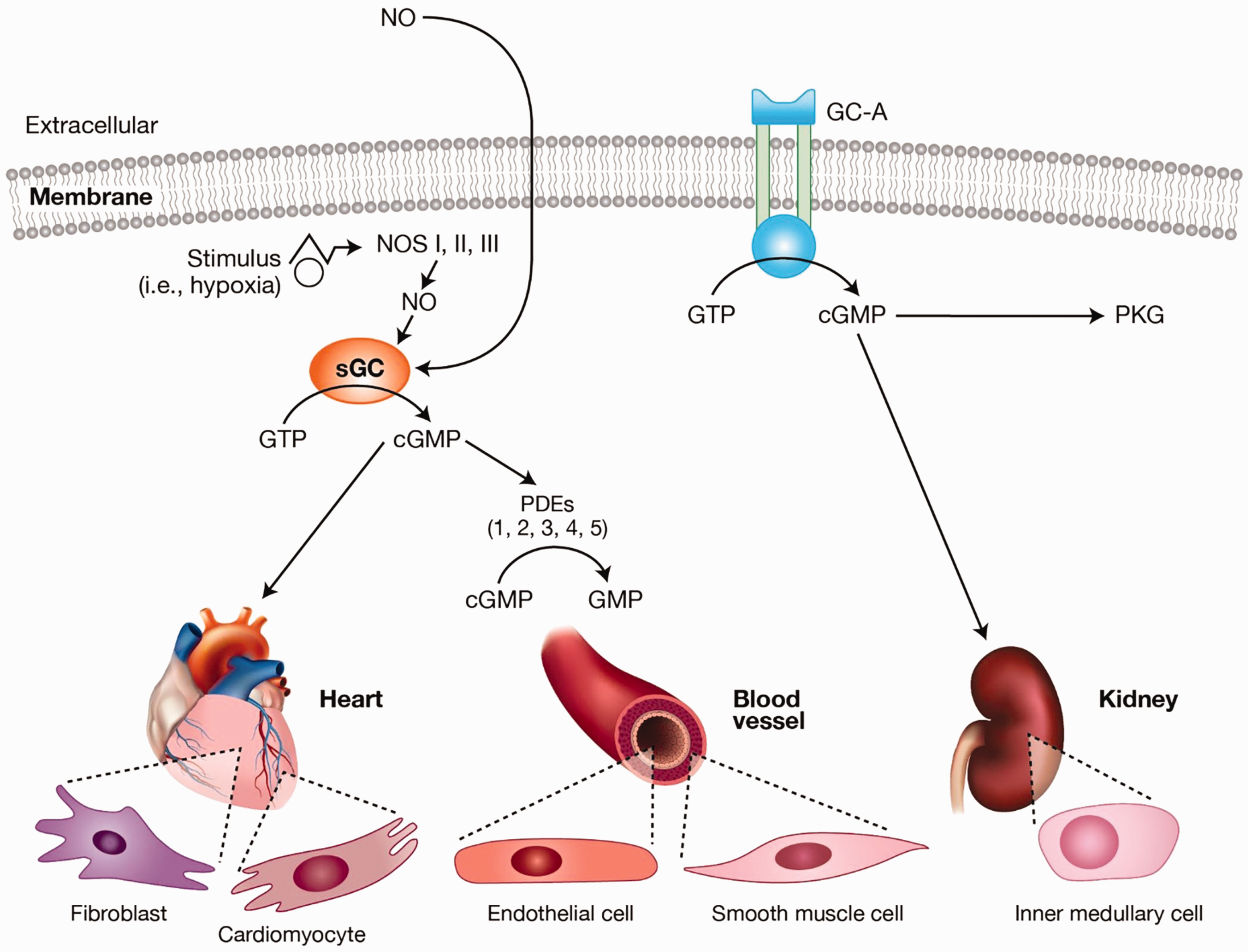
Sequential combination therapy strategies tested in clinical randomized controlled trials.
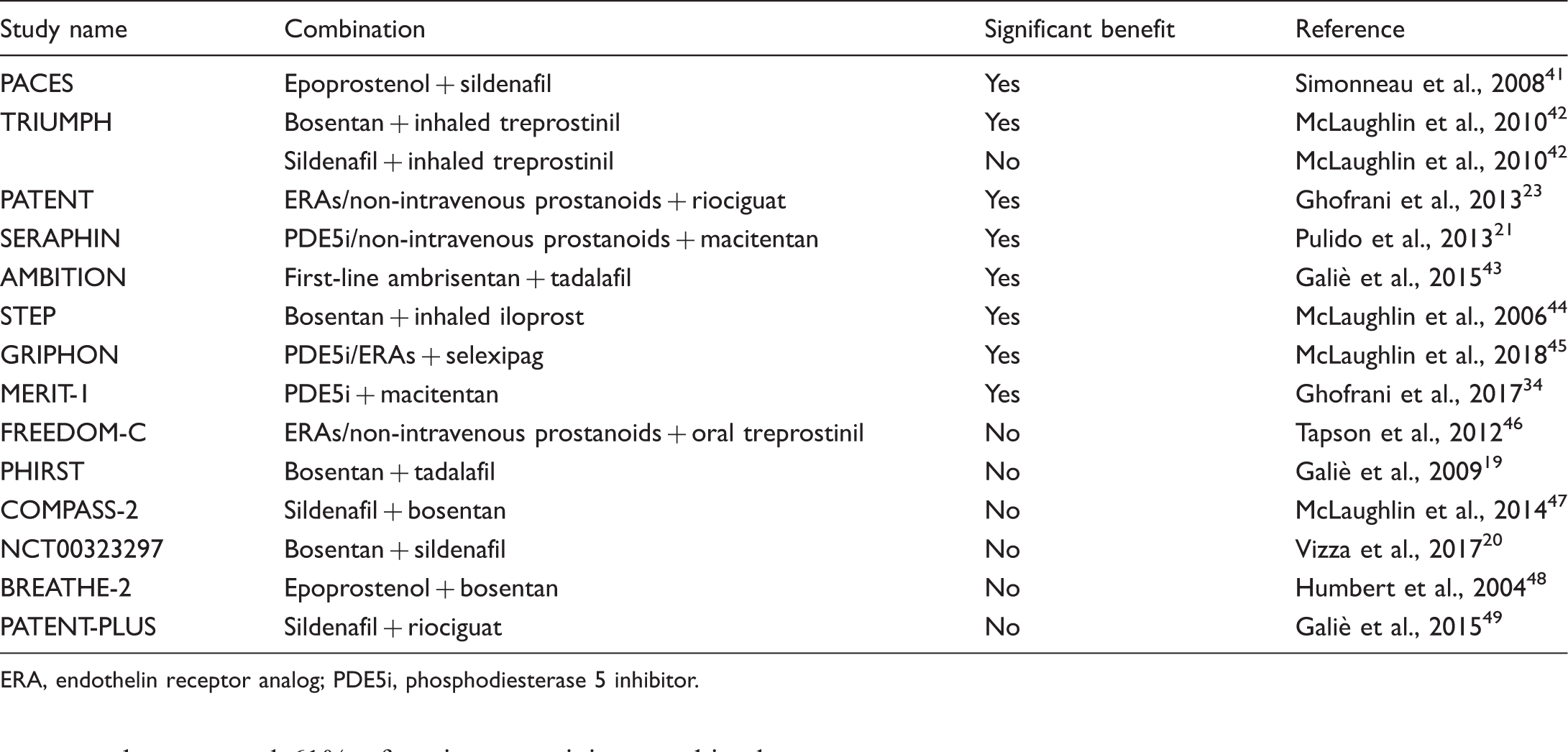
ERA, endothelin receptor analog; PDE5i, phosphodiesterase 5 inhibitor.
The concomitant administration of PDE5i and riociguat is contraindicated due to the risk of systemic hypotension. For patients already receiving an ERA and either a PDE5i or riociguat, a PCA or IP receptor agonist may be administered. Although triple combination therapy has the advantage of acting on three different molecular targets, it also increases pharmacologic complexity and the potential risk of adverse events (AEs), and may lead to increased drug-related healthcare costs. The addition of PCAs or selexipag to an existing treatment regimen may cause additional side effects such as headache, flushing, and jaw pain, and the intravenous administration of PCAs carries the risk of sepsis and embolic phenomena.
PDE5i are the most frequently prescribed treatment for PAH, either as monotherapy or combination therapy. However, clinical data indicate that 20–60% of patients receiving PDE5i do not reach or maintain treatment goals as set out in the 2015 ESC/ERS guidelines. Furthermore, in AMBITION, 73% of patients with PAH receiving tadalafil as monotherapy, and 61% of patients receiving combined ambrisentan and tadalafil, did not achieve the secondary composite endpoint of satisfactory clinical response at week 24. In the SERAPHIN study of macitentan in PAH, the morbidity/mortality event-free survival of patients receiving PDE5i monotherapy was approximately 50% at three years.
The 2015 ESC/ERS treatment guidelines advise that the treatment objective for patients with PAH is to achieve a low-risk status, indicated by stable, non-progressive disease, an approach supported by analyses of registry data. Patients who do not reach or maintain a low-risk status are described as having an insufficient response to treatment; the current treatment guidelines recommend sequential addition of oral therapies and then the addition of a prostanoid. However, as a direct stimulator of sGC, riociguat targets a different molecule in the same signaling pathway as PDE5i. For patients without a sustained response to PDE5i, there is therefore a biological rationale for switching to riociguat in this setting to augment pharmacological effects and lower risk. However, data are scarce on switching between drug classes in PAH, including riociguat data, and the majority of studies have focused on switching within a drug class, e.g. from parenteral to inhaled prostanoids. The concept of switching to riociguat from a PDE5i is an attractive approach, particularly in patients at low/intermediate risk and not rapidly deteriorating, to avoid therapy complexity, mitigate overlapping side effects, and minimize costs. Furthermore, in adults, switching to riociguat does not preclude subsequent escalation to triple therapy. However, further evidence to support this concept is still required and it is important to remember that switching between classes is not included in the 2015 ESC/ERS guidelines.
Studies of switching between PAH-approved therapies
Multiple studies have evaluated switching to PAH therapies other than riociguat, switching within a drug class, and from one drug class to another (Table 2). However, switching between drug classes is not recommended by the guidelines due to the lack of randomized controlled trial (RCT)-based evidence. 4 PCAs are the longest-established therapy for PAH, and switching between PCAs has been shown to be well tolerated, prevent clinical deterioration, and maintain functional status.25–27 In a retrospective assessment of switching from intravenous epoprostenol to iloprost, after one year 78% of patients remained on iloprost and 81% were free from clinical worsening. Patients in these studies were switched either because of drug-related AEs, because clinical improvement during PCA therapy allowed a switch to a non-intravenous formulation, or to achieve a more convenient administration schedule when switching between inhaled PCAs.25–27 Other studies have assessed switching from a prostanoid to an ERA such as bosentan 28 or a PDE5i such as sildenafil. 29 These studies demonstrated a prolonged and stable functional class, and improved 6-minute walking distance (6MWD) and quality of life. 28 After switching to sildenafil, approximately 70% of patients maintained improved quality of life and improved 6MWD for up to 3 months. 29 Reasons for switching from a PCA to an ERA included having met specific disease stability criteria while receiving a PCA, 28 and for those patients who switched to a PDE5i, concerns over the availability of PCA therapy in the study region. 29 Switching within other drug classes such as ERAs 30 and PDE5i31–33 has also been evaluated and, although data are limited, exercise capacity and World Health Organization functional class (WHO FC) were sustained. The patients who switched from sitaxsentan to ambrisentan were US patients enroled in an open-label study of sitaxsentan in PAH and switched at study closure because sitaxsentan did not receive Food and Drug Administration (FDA) approval. 30 Patients in studies of switching between PDE5i did so due to drug-related AEs, 31 the desire for greater adherence and convenience,32,33 as well as potentially lower costs and higher quality of life. 33 The SITAR study assessed safety and patient satisfaction after switching from sildenafil to tadalafil, and demonstrated that tadalafil was well tolerated with improved convenience for patients. 32 Other studies have also supported these findings demonstrating tolerance and reduced deterioration.31,33
Current treatment strategies in CTEPH
Pulmonary endarterectomy (PEA) is the recommended treatment for CTEPH as it is potentially curative and can result in the near normalization of pulmonary hemodynamics. However, up to 40% of patients are ineligible for surgery, and although balloon pulmonary angioplasty (BPA) may be an option for some of these patients, the remainder will require pharmacologic therapy, as will the 51% of patients undergoing PEA who experience persistent/recurrent PH after surgery. Moreover, patients or physicians may be reluctant to consider surgery, despite the recommendations of treatment guidelines. Before the approval of riociguat for inoperable and persistent/recurrent CTEPH, patients with CTEPH were often prescribed drugs approved for PAH off label.
At the time of writing, riociguat is the only approved drug for the treatment of inoperable or persistent/recurrent CTEPH, based on the phase 3 CHEST-1 study and its open-label, long-term extension CHEST-2. However, the recent phase 2 MERIT study of macitentan in patients with inoperable CTEPH demonstrated encouraging results, including in patients who were receiving background PDE5i therapy (46%). 34 In patients who are inoperable or have persistent/recurrent CTEPH after PEA, and are not reaching treatment goals on off-label medical therapy, switching to riociguat is a logical treatment choice. 35 WHO FC III and 6MWD of 165–440 m were part of the definition of unsatisfactory response to previous therapy in the RESPITE and REPLACE clinical studies, and may also be useful as criteria for patients with CTEPH who are not reaching treatment goals and could benefit from switching to riociguat.
BPA
Studies of BPA combined with background medical therapy.
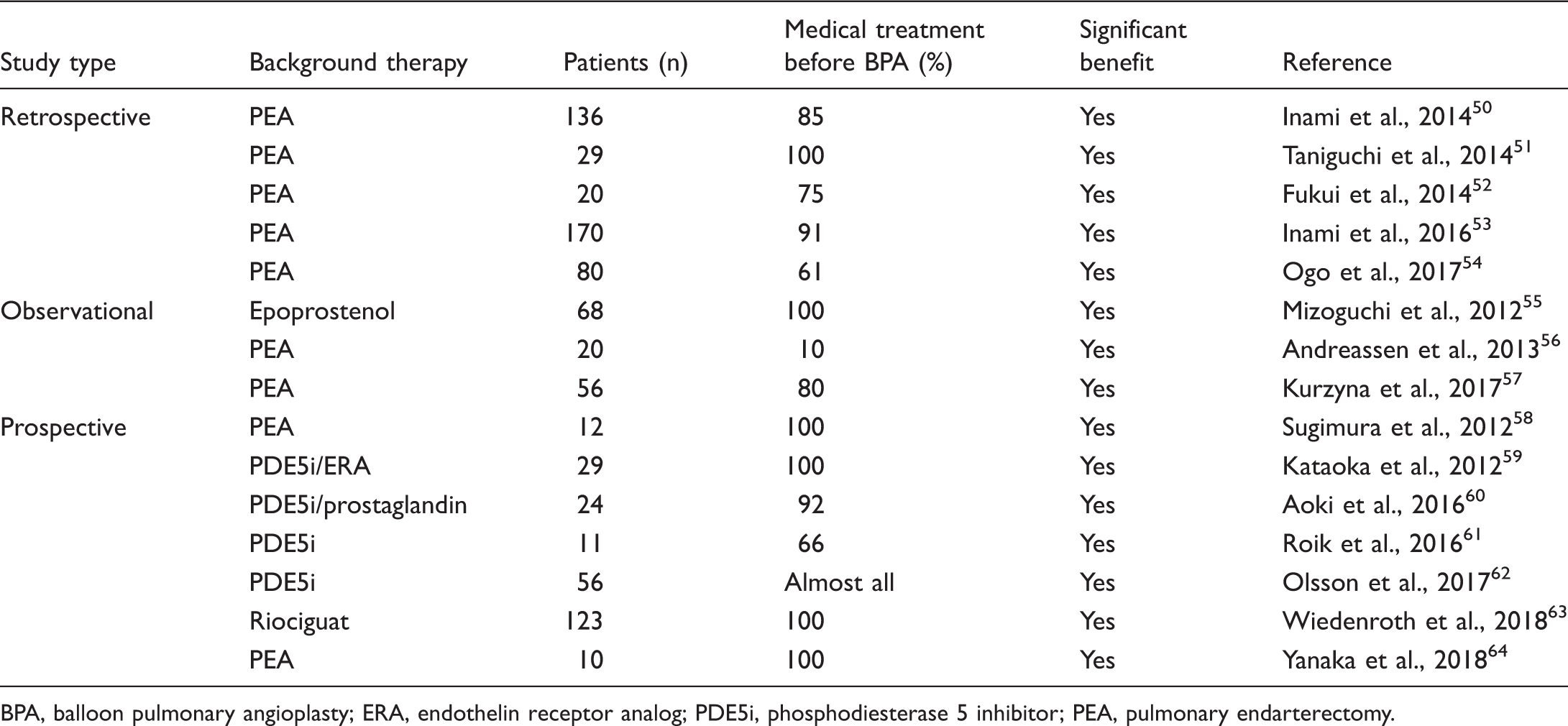
BPA, balloon pulmonary angioplasty; ERA, endothelin receptor analog; PDE5i, phosphodiesterase 5 inhibitor; PEA, pulmonary endarterectomy.
Studies evaluating switching between other PAH-approved therapies.
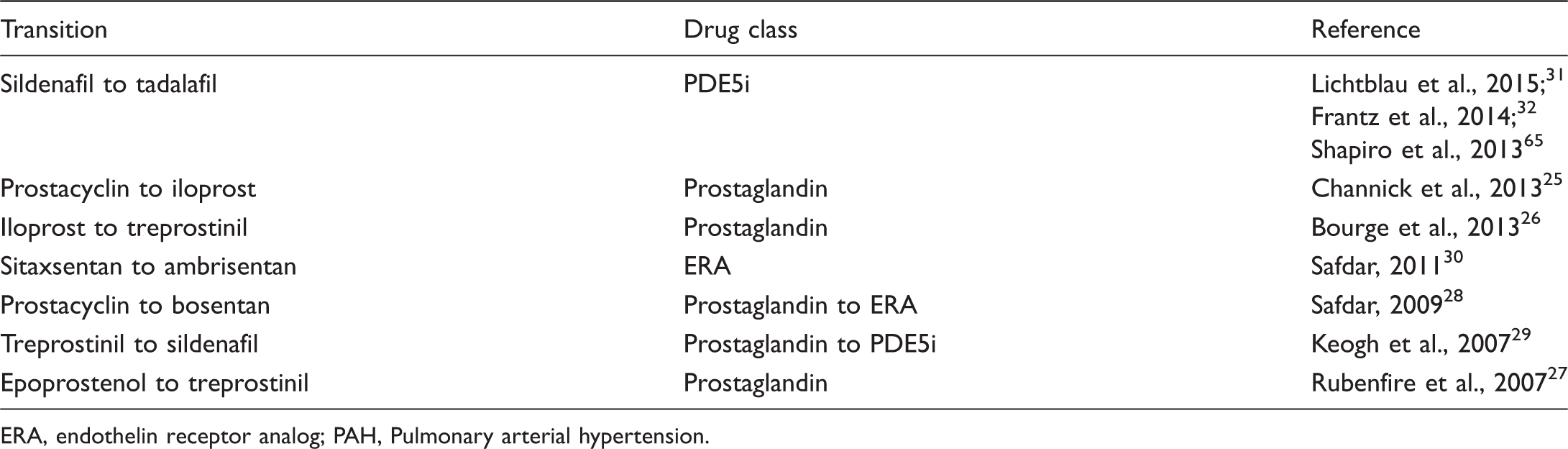
ERA, endothelin receptor analog; PAH, Pulmonary arterial hypertension.
Clinical trials of switching to riociguat
RESPITE
PDE5i were stopped 1–3 days before the first dose of riociguat to allow a PDE5i treatment-free period of at least 24 h for sildenafil and at least 72 h for tadalafil. The exploratory endpoints included 6MWD, N-terminal prohormone of brain natriuretic peptide (NT-proBNP), WHO FC, pulmonary hemodynamics, EuroQol 5 Dimensions (EQ-5D) quality of life score, clinical worsening, and safety.
Of the 61 patients enrolled in RESPITE, 51 completed 24 weeks of treatment; at baseline, 50 patients (82%) were receiving combination therapy with ERAs. The majority of patients in RESPITE reached the maximum dose of riociguat after switching from PDE5i. In those patients completing the study, mean ± standard deviation (SD) 6MWD increased by +31 ± 63 m (95% confidence interval [CI] = 13–49 m; P = 0.0010), mean ± SD NT-proBNP decreased by −347 ± 1235 pg/mL, a relative reduction of 22% (95% CI = 5–37; P = 0.0170), and 28 patients (54%) had improved WHO FC (52% to WHO FC II and 2% to WHO FC I; P < 0.001). Hemodynamic data, available for 49 patients at baseline and week 24, showed that mean ± SD PVR was reduced by −103 ± 296 dyn·s·cm–5 (95% CI = −188–−18; P = 0.0184), mean ± SD mPAP was reduced by −2.8 ± 8.8 mm Hg, and right atrial pressure was reduced by −0.8 ± 4.2 mmHg. Mean ± SD cardiac index and mixed venous oxygen saturation increased by +0.3 ± 0.5 L/min/m2 (95% CI = 0.2–0.5 L/min/m2; P = 0.0001) and +1.0 ± 6.3%, respectively. A post hoc analysis indicated that at week 24, 41% of the overall population achieved a low-risk status according to the ESC/ERS treatment guidelines risk assessment (where an overall low-risk status was assumed when > 50% of the available variables met the low-risk thresholds). 37
The switch to riociguat was tolerated by the majority of patients. Fifty-eight patients (95%) experienced an AE, the most common of which were dyspepsia (n = 14 [23%]), headache (n = 12 [20%]), and diarrhea (n = 11 [18%]). No serious adverse events (SAEs) or clinical worsening events were reported during the PDE5i treatment-free period; two patients (3%) experienced SAEs during the first two weeks of riociguat treatment. Two patients (3%) died (one subdural hematoma and one pneumonia, both not related to riociguat) during the study. Overall, the safety profile of riociguat was consistent with the results of the PATENT-1 and PATENT-2 studies.
The RESPITE study was limited by the lack of a control group, lack of blinding, and the drop-out rate (n = 10 [16%]), which may have biased the analysis. To address this potential bias, a sensitivity analysis was performed using worst-possible values for 6MWD, WHO FC, and EQ-5D, and the last observation carried forward for hemodynamics to provide the most conservative view of the data. Improvement was still in line with the overall results of the study, but no longer statistically significant for 6MWD and NT-proBNP; the change for WHO FC remained significant. 37
RESPITE was the first study to investigate the clinical safety and efficacy outcomes of switching from PDE5i to riociguat in PAH, and has provided preliminary evidence that switching may be safe, tolerable, and beneficial in patients with PAH with an insufficient response to PDE5i. The findings also offer preliminary support to the hypothesis that the NO-sGC-cGMP pathway might be optimized by riociguat. However, given the limitations of RESPITE noted above, larger controlled studies are needed to confirm this.
REPLACE
Following the preliminary signals of improvement observed in RESPITE, the phase 4, prospective, randomized, controlled, open-label study REPLACE (
CTEPH Early Access Study
The CTEPH Early Access Study assessed the safety and tolerability of riociguat in 300 patients in real-world clinical practice. This open-label, uncontrolled, phase 3b, long-term surveillance study included patients with inoperable or persistent/recurrent CTEPH after PEA, who were not satisfactorily treated, or treatment-naïve and could not participate in another CTEPH trial. The study provided early access to riociguat before market launch and consisted of three phases: an eight-week dose-adjustment phase; a maintenance phase until riociguat was available in a patient’s respective country; and a safety follow-up phase. All patients underwent a PDE5i/ERA/PCA treatment-free period of at least 72 h before initiating riociguat. Concomitant treatment with an ERA or PCA was permitted after the dose-adjustment phase.
Of the 300 patients enrolled in the study, 84 (28%) switched to riociguat from other single or combination PAH-targeted therapies, with a median treatment-free period of four days (range = 3–74 days). Fifty-eight patients (19%) previously received PDE5i (most frequently sildenafil [14%]), 44 patients (15%) ERAs (most commonly bosentan [12%]), and seven patients PCAs (most commonly iloprost [2%]). Twenty-four patients (8%) received dual (n = 23 [8%]) or triple (n = 1 [<1%]) combination therapy before switching.
The safety and tolerability of riociguat was similar between patients who switched from other PAH-targeted therapies and those who were treatment-naïve, and was comparable with that of the CHEST-1 and CHEST-2 studies. During the PDE5i/ERA/PCA treatment-free period, AEs were experienced by 11 patients (13%), eight of which were mild in severity. Two patients experienced SAEs during the treatment-free period (syncope and hospitalization due to septicemia), both of which resolved.
Overall, other PAH-targeted therapies were initiated or restarted by 42 patients (14%), the majority due to worsening CTEPH. In patients who switched from prior PAH-targeted therapy, 17 patients (20%) started an ERA and one patient (1%) started a PCA during the maintenance phase. Two patients (2%) restarted PDE5i treatment on the same day as discontinuing riociguat. Overall, 38 patients (13%) discontinued riociguat during the study, with AEs being the most frequent reason (n = 14), and four patients discontinued riociguat treatment during the safety follow-up phase.
Assessment of 6MWD was optional during the study; therefore, data were not available for all patients. In switched patients, mean ± SD baseline 6MWD was 389 ± 87 m. At week 12, mean ± SD change from baseline was + 28 ± 39 m (n = 32). The percentage of switched patients in WHO FC I/II/III/IV at baseline was 2% (n = 84). After 12 weeks of treatment, WHO FC had improved/stabilized/worsened in 21/76/3% (n = 70).
Case studies of switching to riociguat
Case studies of patients with PAH and CTEPH switching from PDE5i therapy to riociguat.
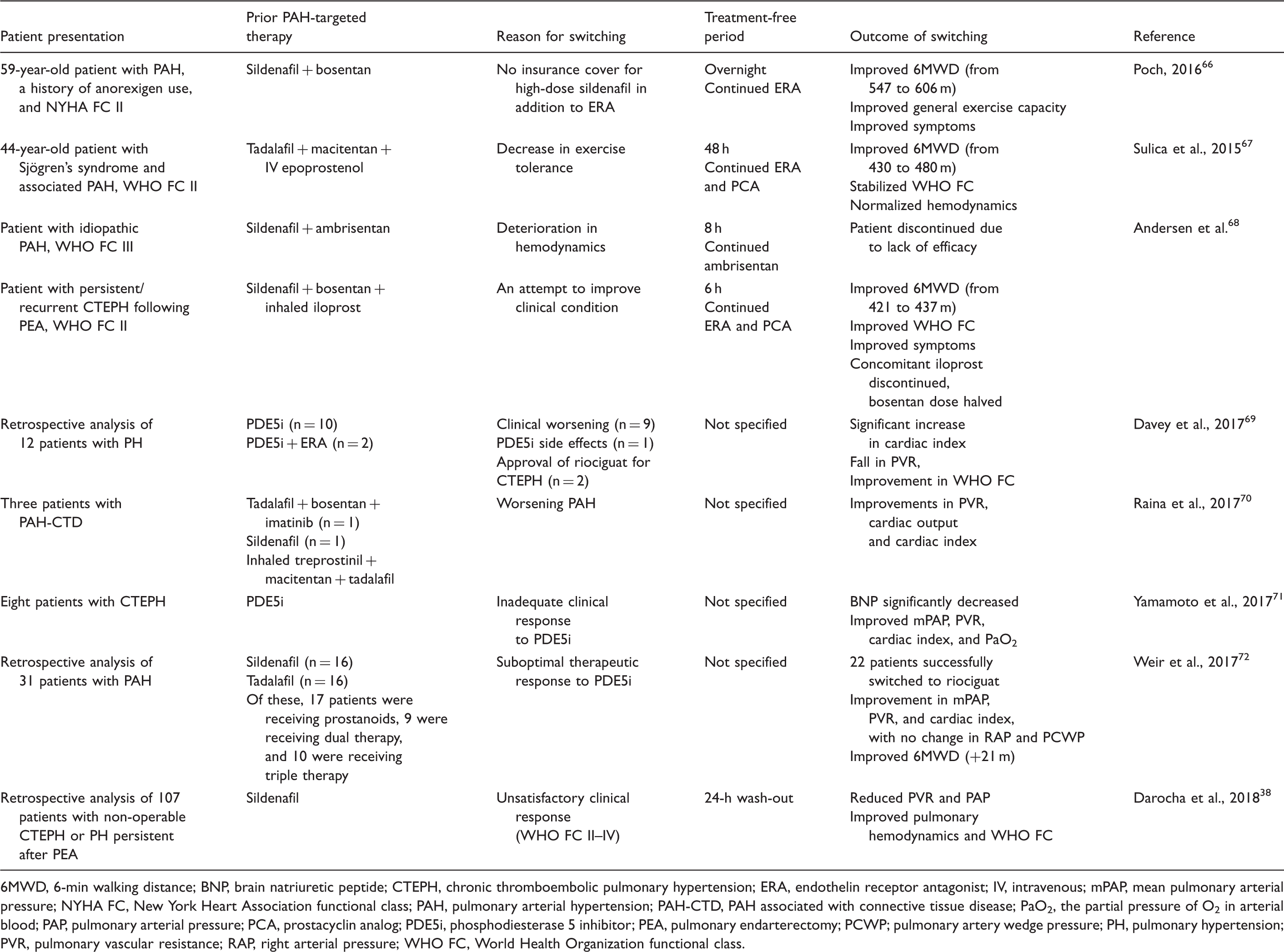
6MWD, 6-min walking distance; BNP, brain natriuretic peptide; CTEPH, chronic thromboembolic pulmonary hypertension; ERA, endothelin receptor antagonist; IV, intravenous; mPAP, mean pulmonary arterial pressure; NYHA FC, New York Heart Association functional class; PAH, pulmonary arterial hypertension; PAH-CTD, PAH associated with connective tissue disease; PaO2, the partial pressure of O2 in arterial blood; PAP, pulmonary arterial pressure; PCA, prostacyclin analog; PDE5i, phosphodiesterase 5 inhibitor; PEA, pulmonary endarterectomy; PCWP; pulmonary artery wedge pressure; PH, pulmonary hypertension; PVR, pulmonary vascular resistance; RAP, right arterial pressure; WHO FC, World Health Organization functional class.
The data from these case studies suggest that switching to riociguat has been considered in the clinical setting, with the suggestion of therapeutic efficacy, although the current role of this therapeutic modality in the treatment algorithm is not yet defined.
Non-interventional studies
The EXPERT registry (NCT02092818) has collected evidence from real-world clinical practice on patients with PAH or CTEPH receiving riociguat. Safety data from EXPERT on patients with PAH or CTEPH who switched from other PAH-targeted therapies indicated that tolerability was similar between switched and treatment-naïve patients, and that AEs and SAEs in switched patients were consistent with the known safety profile of riociguat. 39 The retrospective chart review study CAPTURE enrolled 125 patients with PAH, or inoperable or persistent/recurrent CTEPH, who switched to riociguat from another PH-targeted medical therapy. The aim of the study was to understand real-world procedures for switching to riociguat, and the results suggest that switching to riociguat from other PH-targeted therapies appears to be feasible in the context of current label recommendations.
How to switch within the NO pathway
Given the limited existing evidence and the lack of a recommendation for switching in published guidelines, we would not encourage switching until randomized data supporting this strategy are available. As noted above, the rationale for switching from PDE5i to riociguat is to optimize the NO-sGC-cGMP signaling pathway. Concomitant administration of riociguat and PDE5i exerts an additive effect on systemic blood pressure, and clinically significant hypotension can occur. Concomitant use of all PDE5i, or non-specific PDE5i and riociguat, is contraindicated and a PDE5i treatment-free period must be initiated before switching to riociguat. The recommended PDE5i treatment-free periods, as used in the RESPITE and REPLACE studies, are based on the pharmacokinetics of sildenafil and tadalafil, and data from real-world clinical practice. Sildenafil should be discontinued for at least 24 h and tadalafil should be discontinued for at least 48 h before administering riociguat. In addition, it is recommended to monitor for signs and symptoms of hypotension on initiation. Should a patient need to be switched from riociguat to a PDE5i, riociguat must be discontinued at least 24 h before initiation of the PDE5i, and the patient should be monitored for signs and symptoms of hypotension upon initiation of the new therapy. 40
If individual clinicians wish to attempt a switch to riociguat based on their medical judgment, patient enrolment in the ongoing REPLACE study should be considered. We suggest that possible candidates for a switch include: patients who have not reached a low-risk profile (e.g. one or two intermediate-risk characteristics) but who do not have any high-risk characteristics by ESC/ERS criteria, or patients with a REVEAL risk score of 6–8 who are stable/not declining rapidly for several months. Indeed, preliminary data from RESPITE suggest that patients with lower PVR and right arterial pressure, and higher 6MWD at baseline, were more likely to respond to a switch. Any decision regarding a change in therapy should be based on individual clinical status and/or the AEs of any previously administered therapy. If switching to riociguat from other PAH-specific drugs is considered appropriate or necessary, the decision regarding the execution of such a switch can only be made at the discretion of the experienced treating PH physician after discussion with the patient. REPLACE has the potential to provide further data on when switching might be considered over sequential combination therapy in patients not meeting treatment goals.
Expert opinion/clinical perspectives
Early, limited clinical evidence from RESPITE and case series data indicate that switching to riociguat may be a viable treatment option in carefully selected patients with PH who are not achieving treatment goals with their current regimen. There are, however, no RCTs comparing switching with either continued therapy or sequential combination therapy. Further trials are taking place that may determine whether switching will become a goal-oriented treatment strategy for minimizing risk in patients with PAH, and this may identify where switching could be placed in the PAH treatment algorithm. Although switching to riociguat may be of benefit to some patients, it is important to note that it is not possible to identify responders upfront. Previous data indicated that switching would be less appropriate in patients with deteriorating and advanced symptoms, and these patients are candidates for treatment with sequential combination therapy and/or parenteral prostanoids, as recommended in the ERS/ESC guidelines. We recommend that a patient’s risk is assessed by either REVEAL score or ERS/ESC risk criteria when making treatment decisions.
Conclusion
Data on switching between PAH-approved therapies in patients with PAH or CTEPH are still limited. The studies reviewed here summarize the available evidence and practical experience in a situation where there is no formal protocol or recommendation available for switching. In line with the recommendation of goal-oriented therapy to keep patients at a low-risk status, switching may present a potential additional strategy to sequential combination therapy in low- to intermediate-risk patients, keeping the option of adding a third drug class for a later stage in the disease course. There are no data to support switching between therapies for patients undergoing triple drug therapy.
The studies discussed here suggest that switching to riociguat may be safe, tolerable, and beneficial in patients with PAH who have an insufficient response to PDE5i, and in patients with CTEPH who are receiving off-label treatment. However, additional data from RCTs are needed to determine if this strategy is an effective approach to the management of patients with PAH and CTEPH.
Footnotes
Acknowledgments
Editorial assistance was provided by Adelphi Communications Ltd (Bollington, UK), supported by Bayer AG (Berlin, Germany).
Conflict of interest
The authors declare the following conflicts of interest: RLB reports grants from Bayer AG, Actelion, EIGER, United Therapeutics, and Gilead paid to his institution, and honoraria from Gilead, Bayer, and Actelion; PAC reports grant support from Bayer (University Research Fund), and fees for advisory boards and talks from Bayer, Actelion, and GSK; H-AG reports grants from DFG (German Research Foundation), honoraria from Actelion, Bayer, Ergonex, Gilead, GSK, Novartis, and Pfizer, consultancy fees from AbbVie, Actelion, Bayer, Bellerophon Pulse Technologies, Ergonex, Gilead, GSK, Medscape, MSD Sharpe & Dohme, Novartis, OMT, Pfizer, and Web MD Global, and speaker’s bureau fees from Actelion, Bayer, Ergonex, Gilead, GSK, Novartis, and Pfizer; GS reports grants, personal fees, and non-financial support from Bayer, Actelion, GSK, Lilly, Pfizer, and Novartis; VVM reports grants, personal fees, and non-financial support from Actelion, Bayer, Gilead, United Therapeutics, and Ikaria, and grants from Novartis, EIGER and SoniVie; AR reports honoraria from Bayer, research support from United Therapeutic, and consulting fees from St. Jude. MK has no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
