Abstract
This analysis aimed to characterize the pharmacokinetics (PK) and PK/pharmacodynamic (PK/PD) relationship of riociguat and its metabolite M1 in patients with chronic thromboembolic pulmonary hypertension (CTEPH) or pulmonary arterial hypertension (PAH). Blood samples were collected in two phase 3 studies—PATENT-1 (Pulmonary Arterial Hypertension Soluble Guanylate Cyclase-Stimulator Trial 1; 12 weeks; PAH) and CHEST-1 (Chronic Thromboembolic Pulmonary Hypertension Soluble Guanylate Cyclase–Stimulator Trial 1; 16 weeks; CTEPH)—and long-term extensions. Patients were initially randomized to receive placebo or riociguat, and they received riociguat in the extensions. Nonlinear mixed-effects modeling was used to develop a population PK model describing riociguat PK. PK/PD relationships were investigated by comparing derived PK parameters with changes in PD parameters. Covariate analyses included smoking status, bosentan comedication, bilirubin levels, and baseline creatinine clearance. The PK of riociguat/M1 was described by a one-compartment model. Mean population estimates for riociguat absorption rate constant, clearance, and volume of distribution were 2.17/h, 1.81 L/h, and 32.3 L, respectively; for M1 they were 0.258/h, 3.16 L/h, and 124 L. Interindividual variability was moderate for riociguat and moderate to high for M1. There was no evidence of time- or dose-dependent changes in riociguat/M1 PK. Riociguat clearance was higher in smokers (120% increase) and bosentan-treated patients (36% increase) than in nonsmokers and those not receiving bosentan. There was an inverse correlation between bilirubin and riociguat clearance. In PK/PD analyses, 6-minute walk distance was related to hemodynamic parameters, particularly pulmonary vascular resistance. Riociguat PK were described by a one-compartment model. Effects of covariates on riociguat and M1 PK were established, and a PK/PD relationship was demonstrated. (ClinicalTrials.gov identifiers: PATENT-1, NCT00810693; PATENT-2, NCT00863681; CHEST-1, NCT00855465; CHEST-2, NCT00910429.)
Impairment of nitric oxide (NO) synthesis and signaling through the NO—soluble guanylate cyclase (sGC)—cyclic guanosine monophosphate (cGMP) pathway plays a key role in the pathogenesis of pulmonary hypertension.1,2 Riociguat (Fig. 1) is an oral stimulator of sGC with a dual mode of action.2,3 First, riociguat stimulates sGC directly through an NO-independent mechanism; second, it increases the sensitivity of sGC to NO. Studies in animal models of pulmonary hypertension have shown that riociguat increases cGMP levels, resulting in vasorelaxation, and also has antiproliferative and antifibrotic effects, especially in the pulmonary vasculature. 4
Molecular structure of riociguat.
Riociguat is the first drug to be approved for the treatment of two separate pulmonary hypertension indications: pulmonary arterial hypertension (PAH) and inoperable or persistent/recurrent chronic thromboembolic pulmonary hypertension (CTEPH) after surgical treatment.5–7 The efficacy and tolerability of riociguat have been demonstrated in a phase 3 trial program involving two randomized, double-blind, placebo-controlled studies of riociguat 0.5–2.5 mg three times daily (tid). PATENT-1 (Pulmonary Arterial Hypertension Soluble Guanylate Cyclase—Stimulator Trial 1) was a 12-week study in patients with PAH, while CHEST-1 (Chronic Thromboembolic Pulmonary Hypertension Soluble Guanylate Cyclase—Stimulator Trial 1) was a 16-week study in patients with CTEPH.8,9 Both studies were followed by long-term, open-label extensions (PATENT-2 and CHEST-2), in which all patients received riociguat (maximum dose: 2.5 mg tid), confirming the long-term efficacy and tolerability of riociguat.10–13
The aim of the present study was to characterize the pharmacokinetics (PK) of riociguat and its main active metabolite, M1, and establish the PK/pharmacodynamic (PD) relationship in patients with PAH or CTEPH, on the basis of data derived from PATENT-1/2 and CHEST-1/2, as well as to examine covariates that influence PK.
METHODS
Study designs
PATENT-1 and CHEST-1 were multicenter, randomized, double-blind, placebo-controlled trials to investigate the efficacy and tolerability of chronic riociguat therapy in patients with symptomatic PAH (PATENT-1) or CTEPH (CHEST-1) in an individual dose-titration scheme.8,9 PATENT-2 and CHEST-2 were open-label, single-arm extensions to PATENT-1 and CHEST-1, respectively, in which all patients received riociguat.10–13
In all four studies, riociguat was administered orally tid as immediate-release tablets. During the initial 8 weeks of PATENT-1 and CHEST-1, the dose of riociguat was adjusted every 2 weeks according to an individual dose-adjustment plan based on the patient's systolic blood pressure (SBP) and signs or symptoms of hypotension. From a starting dose of 1 mg tid, riociguat dose was increased (by 0.5 mg tid to a maximum of 2.5 mg tid) if trough SBP was ≥95 mmHg, maintained if SBP was 90–94 mmHg, and decreased (by 0.5 mg tid) if SBP was <90 mmHg without symptomatic hypotension. In PATENT-1, an exploratory arm included patients whose riociguat dose was capped at 1.5 mg tid. Patients from the placebo arms of PATENT-1 and CHEST-1 who entered the long-term extension studies received riociguat and underwent the same dose-adjustment regimen. Blinding was maintained in all studies through the use of sham dose adjustments.
The study designs, patient populations, ethical approval, inclusion and exclusion criteria, randomization procedure, efficacy, and tolerability in the four studies have been described elsewhere.8–13
PD end points
PD parameters collected were 6-minute walk distance (6MWD), N-terminal pro—brain natriuretic peptide (NT-proBNP) level, pulmonary vascular resistance (PVR), systemic vascular resistance (SVR), cardiac output (CO), and mean pulmonary arterial pressure (PAPmean) measured by Swan-Ganz right heart catheterization. Diastolic blood pressure, SBP, heart rate, and World Health Organization (WHO) functional class (FC) were also recorded.
PK samples
To describe the PK profile of riociguat and its main active metabolite, M1, protocol-specified blood samples were obtained from all patients at the first visit (day 0), at each subsequent every-other-week study visit during the double-blind studies (PATENT-1 and CHEST-1), at the termination visit, on day 1 of the extension studies (PATENT-2 and CHEST-2), and again after 8 weeks. Samples were obtained approximately 1 hour before administration of study drug in the main and extension studies of PATENT and CHEST. In the PATENT-1 study, blood samples were also obtained 2–3 hours after the first and second dose of study drug at the first visit.
Plasma concentrations of riociguat and M1 were measured by a validated high-pressure liquid chromatography/tandem mass spectrometry method; quality control and calibration samples were analyzed concurrently with study samples. Previous studies had established that the calibration range of the procedure was from 2.0 μg/L (the lower limit of quantification) to 500 μg/L for both riociguat and M1. Quality control samples in the concentration range 6.0–400 μg/L indicated an accuracy of 96.8%–104% and a precision of 5.02%–8.49% for riociguat and an accuracy of 96.9%–100% and a precision of 3.35%–7.56% for M1. The method validation and analysis of the study samples were performed in accordance with the 2001 Food and Drug Administration guideline on bioanalytical method validation. 14 All samples were stored at −15°C and analyzed within 18 months after sampling. Stability data indicated that the analytes were stable for this time period.
Population PK/PD modeling
All analyses were performed using a population PK modeling approach with a first-order conditional estimation method (NONMEM software, ver. 7.2.0; ICON Development Solutions, Dublin, Ireland). Interindividual variability was modeled using exponential random-effect models. Covariates such as demographics and measurable physiological information, which could explain some of the observed between-subject variability, were tested in the model.
For the current studies, a PK model was derived that accounted for random effects, interindividual and interoccasion variability, and patient covariates. Covariates investigated for relationships between the NONMEM estimates of individual PK parameters were age, body weight, race, primary diagnosis, smoking status, comedication, creatinine clearance, and total bilirubin. Covariates were incorporated into the model if linear regression testing showed significance at the P = 0.01 level and were retained if after backward stripping from the model the likelihood ratio showed significance at P = 0.001.
To determine the validity of the model, a visual predictive check was carried out. Individual empirical Bayesian (NONMEM post hoc) estimates for the PK parameters were estimated using the population means and individual patient information by simulating 100 subproblems. The 90th percentile of the predictions was calculated and compared with the actual observations at each visit day. Ideally, 90% of the observations should fall within the 90% prediction interval, and the observations should be symmetrically distributed around the simulated median concentration.
The relationship between the exposure of riociguat and relevant PD parameters was investigated by comparing derived PK parameters—area under the plasma concentration-time curve (AUC), maximum concentration (Cmax), and trough concentration (Ctrough)—and absolute changes in the relevant PD parameters (6MWD, NT-proBNP, SVR, PVR, and PAPmean) with baseline values.
RESULTS
Patient population
In total, plasma concentrations from 642 patients were included in the population PK analysis (Fig. 2). Clinical characteristics of the patients have been presented elsewhere.8–11 Baseline and ongoing values for continuous and categorical covariates investigated are shown in Table 1.
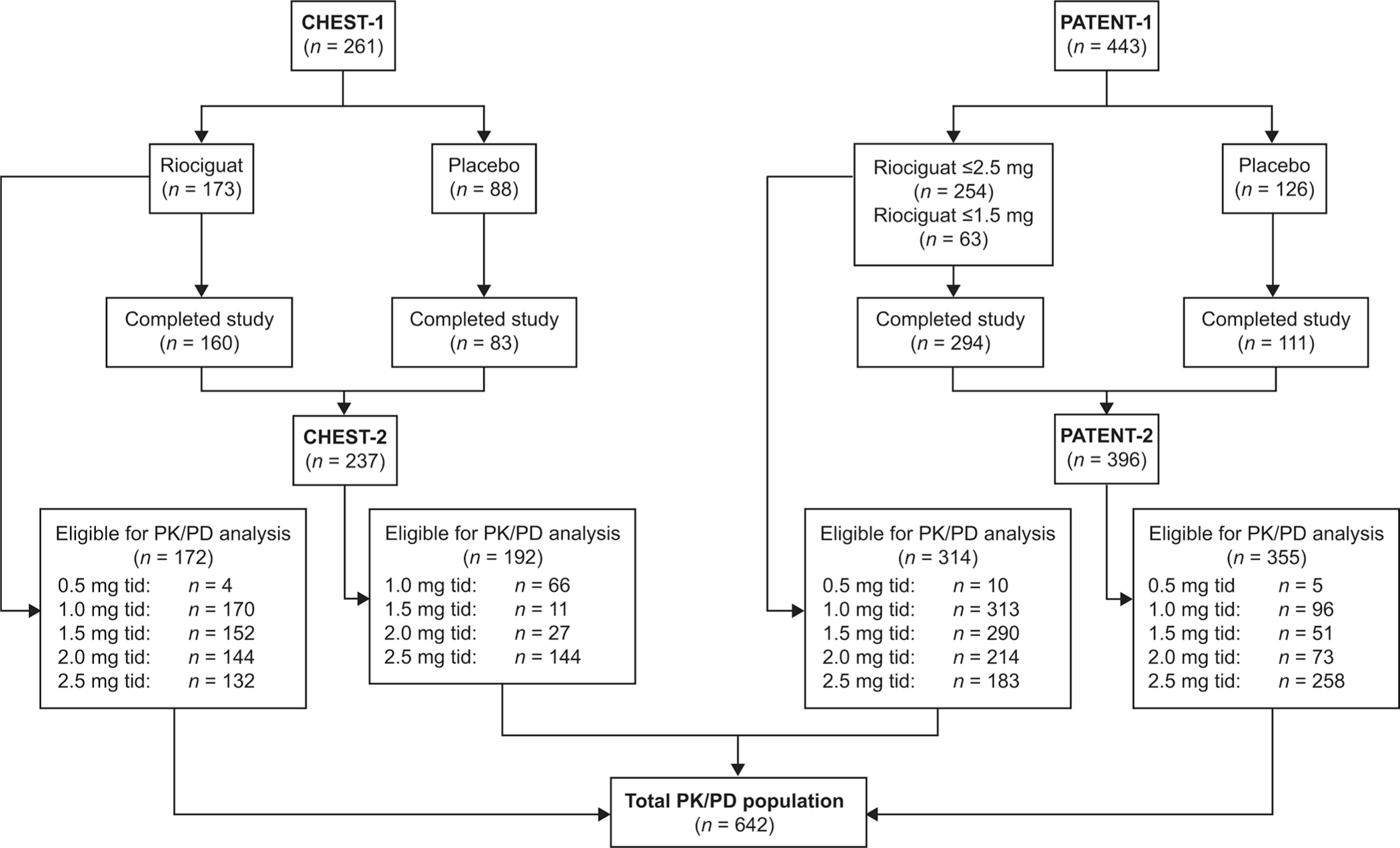
Patient disposition. PD: pharmacodynamic; PK: pharmacokinetic; tid: three times daily.
Baseline and ongoing values for continuous and categorical covariates in the PATENT-1/2 and CHEST-1/2 studies
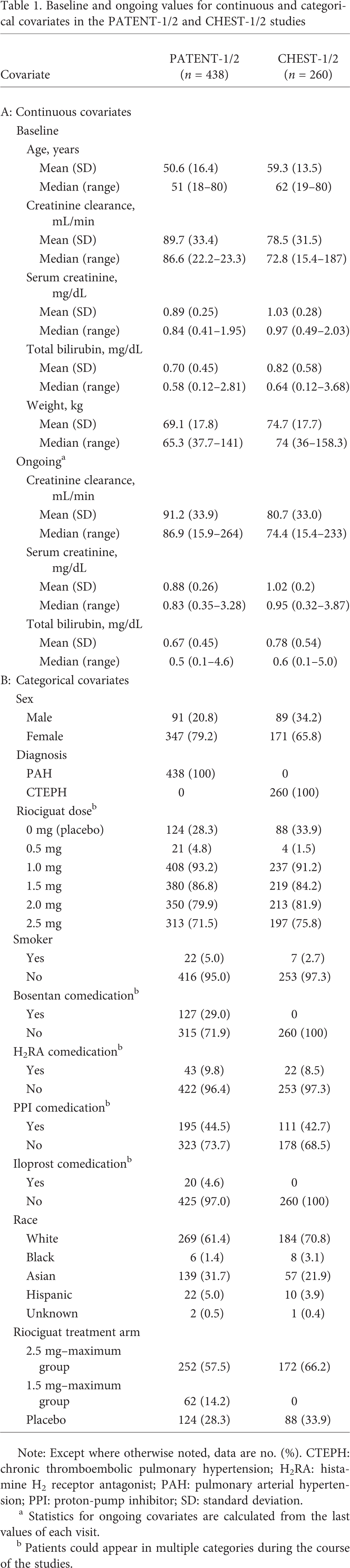
Note: Except where otherwise noted, data are no. (%). CTEPH: chronic thromboembolic pulmonary hypertension; H2RA: histamine H2 receptor antagonist; PAH: pulmonary arterial hypertension; PPI: proton-pump inhibitor; SD: standard deviation.
Statistics for ongoing covariates are calculated from the last values of each visit.
Patients could appear in multiple categories during the course of the studies.
Efficacy
Efficacy data for the four studies have been presented elsewhere.8–11 In brief, patients in PATENT-1 had a mean increase in 6MWD at week 12 of +30 m in the riociguat arm and a mean decrease of −6 m in the placebo arm (least squares mean difference: +36 m [95% confidence interval (CI): 20–52 m]; P < 0.001). 9 There were also significant benefits with riociguat over placebo in terms of PVR (P < 0.001), NT-proBNP levels (P < 0.001), WHO FC (P = 0.003), time to clinical worsening (P = 0.005), and Borg dyspnea score (P = 0.002). In CHEST-1, 6MWD had increased by a mean of +39 m in the riociguat arm at week 16, compared with a decrease of −6 m in the placebo arm (least squares mean difference: +46 m [95% CI: 25–67 m]; P < 0.001). 8 Riociguat was also associated with significant improvements over placebo in PVR (P < 0.001), NT-proBNP level (P <0.001), and WHO FC (P = 0.003). After 1 year of treatment, 6MWD had increased by +51 ± 74 and +51 ± 62 m in PATENT-2 and CHEST-2, respectively.10,11
PK results
In total, 5,245 riociguat plasma measurements and 5,246 M1 plasma measurements were valid for PK analysis. Plasma concentrations of riociguat and M1 were well described by a linear one-compartment model. Because of the limited number of measured plasma concentrations in the absorption phase, the absorption rate was fixed to the estimated value from previous evaluations (data not shown). For riociguat, covariate effects (creatinine clearance, bilirubin concentration, comedication, and smoking) reduced the unexplained interindividual variability of clearance from 48.3% to 41.2%. The covariate body weight reduced the unexplained interindividual variability of volume of distribution from 27.1% to 25%. A post hoc estimate of riociguat plasma concentrations in PATENT and CHEST showed minimal fluctuation in peak to trough concentrations following administration (Fig. 3). The model estimates of the population PK parameters are listed in Table 2, part A.
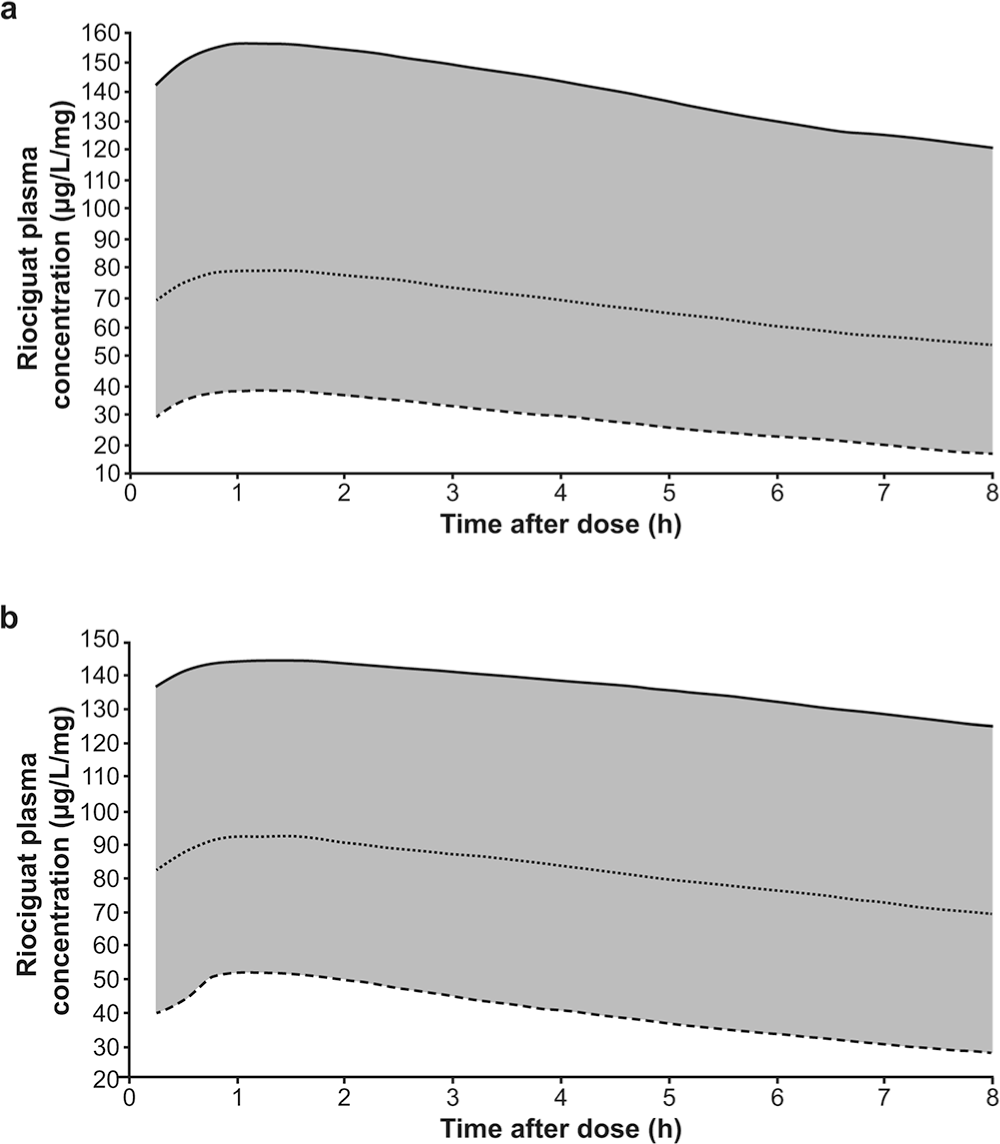
Estimated riociguat plasma concentrations at steady state on the last day of the PATENT-1 (a; day 84) and CHEST-1 (b; day 112) studies.
Population estimates for the final pharmacokinetic model for riociguat and Ml
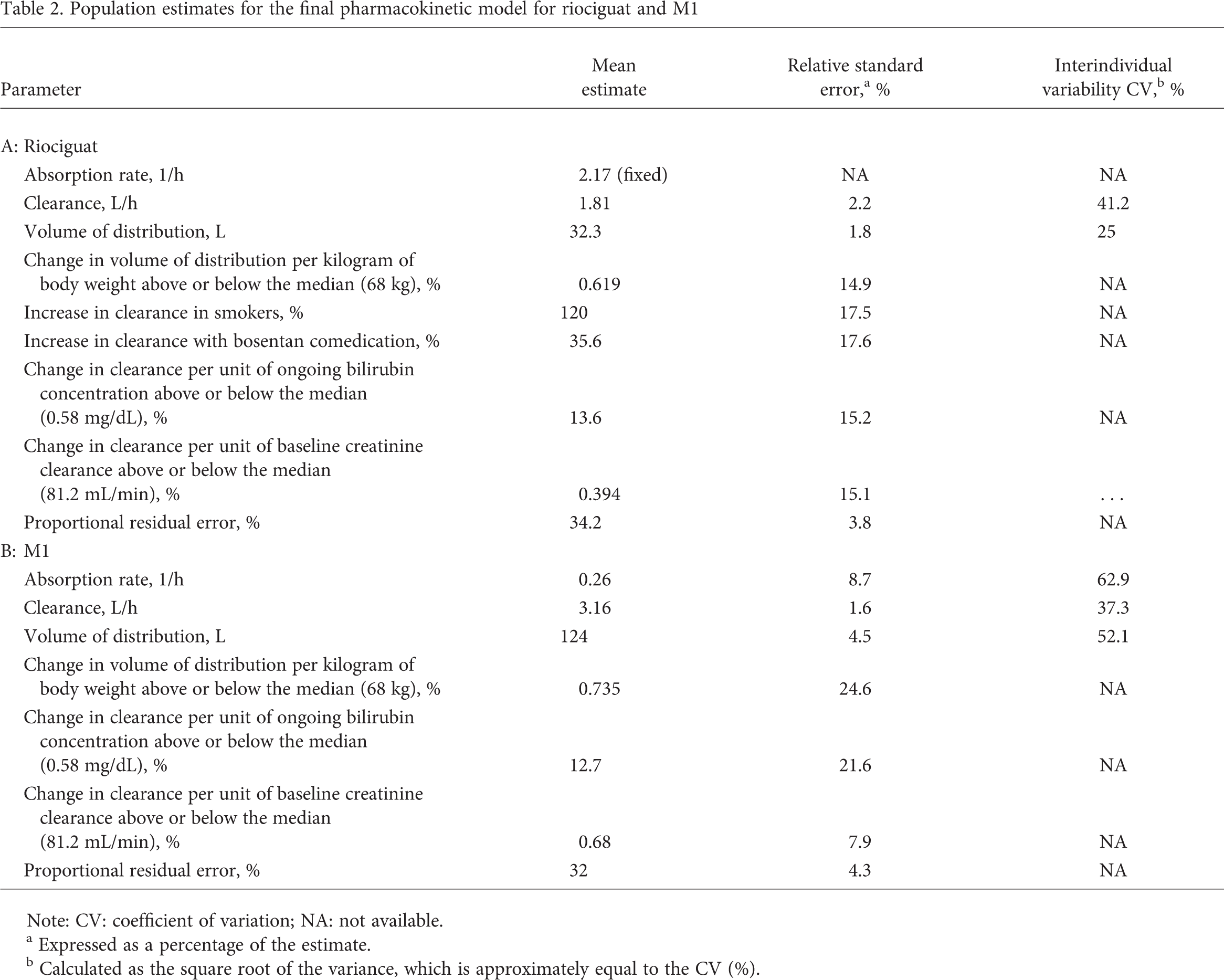
Note: CV: coefficient of variation; NA: not available.
Expressed as a percentage of the estimate.
Calculated as the square root of the variance, which is approximately equal to the CV (%).
A prediction-corrected visual predictive check (pcVPC) was conducted on the final models for riociguat and M1. pcVPC confirmed the validity of the final PK models for both riociguat and M1, although the pcVPC showed a tendency to underestimate the concentrations at Cmax for both riociguat and M1. This may be explained by the limited availability of samples at Cmax (Fig. 4).
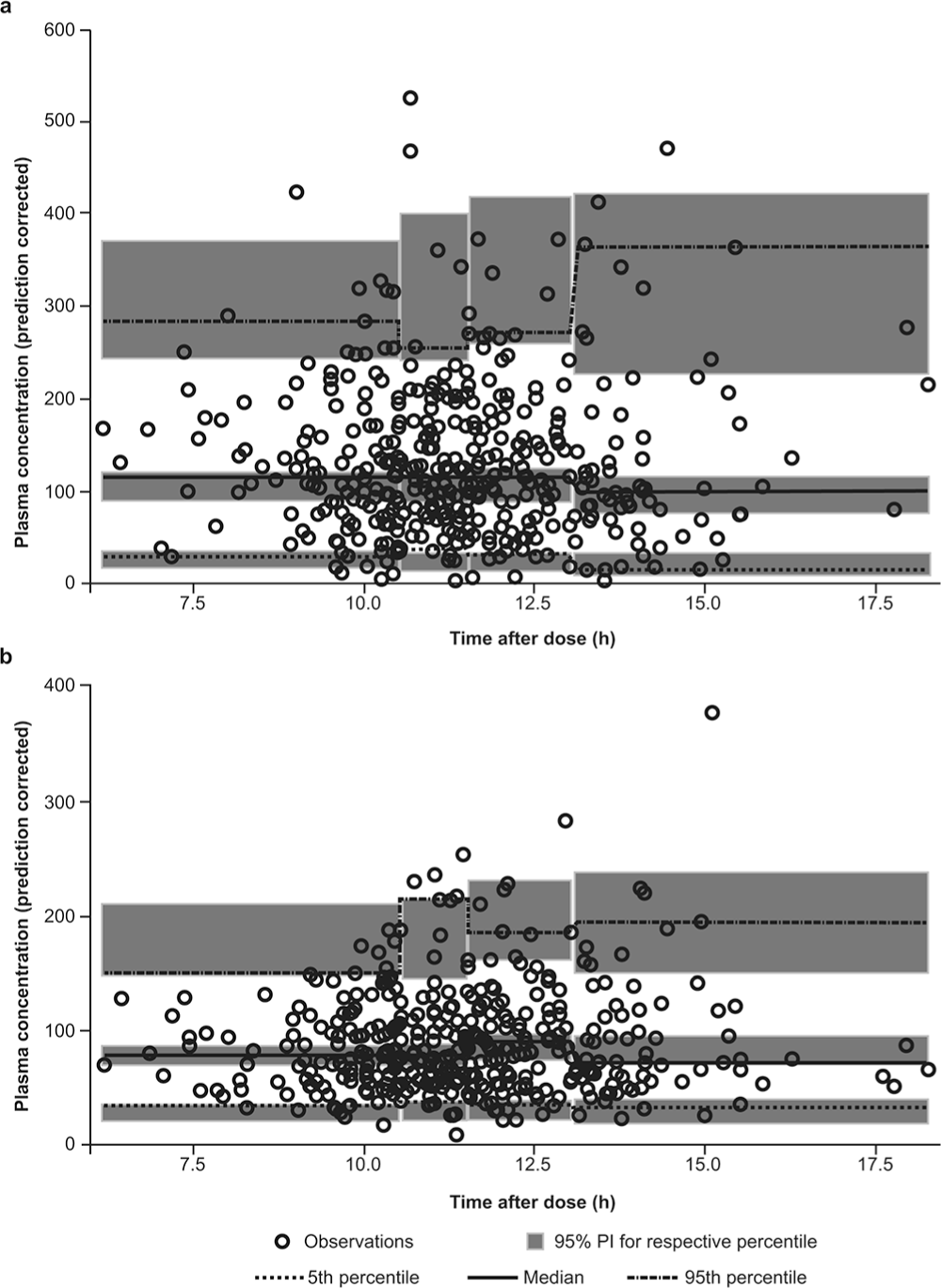
Prediction-corrected visual predictive check of the final pharmacokinetic models for riociguat (a) and M1 (b). Gray areas (from top to bottom) show the 95% prediction intervals for the 95th, 50th, and 5th percentiles of the simulated prediction-corrected values. Lines (from top to bottom) show the 95th, 50th, and 5th percentiles of the observed prediction-corrected values. Circles show the individual prediction-corrected observations. PI: prediction interval.
For M1, the covariate creatinine clearance reduced the unexplained interindividual variability of clearance from 42% to 37%, while the covariate body weight reduced the interindividual variability of volume of distribution from 51.8% to 50%. Although the influence of the covariate bilirubin on the volume of distribution was substantial, there was no reduction in interindividual variability. The model estimates of the population PK parameters for M1 are listed in Table 2, part B.
PK parameters of riociguat calculated at steady state are shown in Table 3. Exposure to riociguat (AUC) showed a numerical increase with increasing dose (Fig. 5), and there was no evidence of time- or dose-dependent alterations in riociguat PK over the time course of the studies (Fig. 6). In the PATENT studies, smokers showed higher riociguat clearance than nonsmokers (4.2 vs. 1.8 L/h; Fig. 7a), and riociguat exposure (AUC) was decreased by 56% (90% CI: 0.38%–0.51%). The effect of smoking on riociguat clearance was also observed in the CHEST studies (Fig. 7b). Patients receiving bosentan as comedication showed higher riociguat clearance than patients not receiving bosentan (2.3 vs. 1.8 L/h; Fig. 7a), and riociguat exposure was decreased by 27% (90% CI: 0.68%–0.78%).
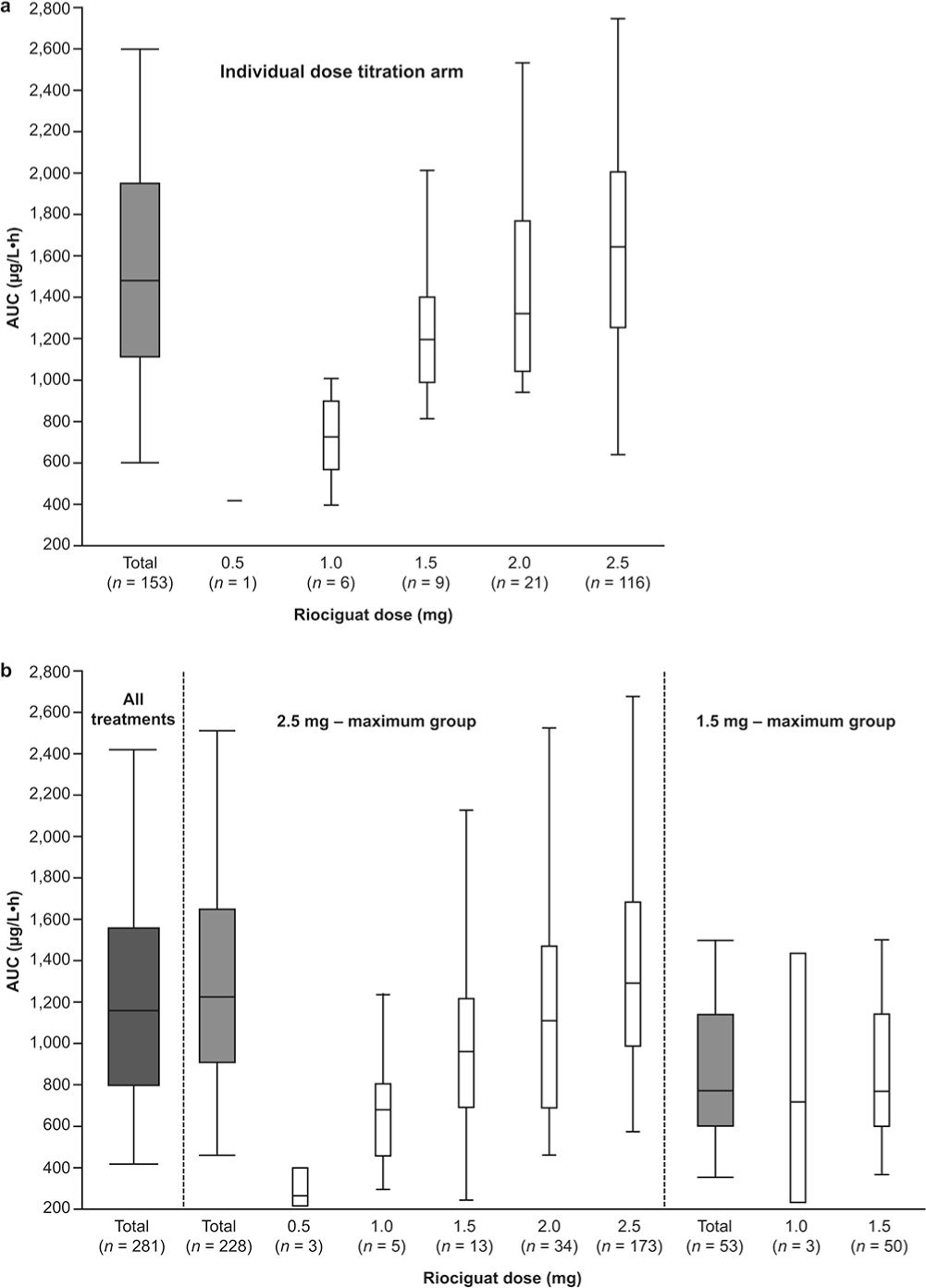
Riociguat exposure (area under the plasma concentration-time curve [AUC]) per dose in the CHEST-1 (a) and PATENT-1 (b) studies. Boxes show 25th–75th percentiles, whiskers show 5th-95th percentiles, and horizontal lines show the median.
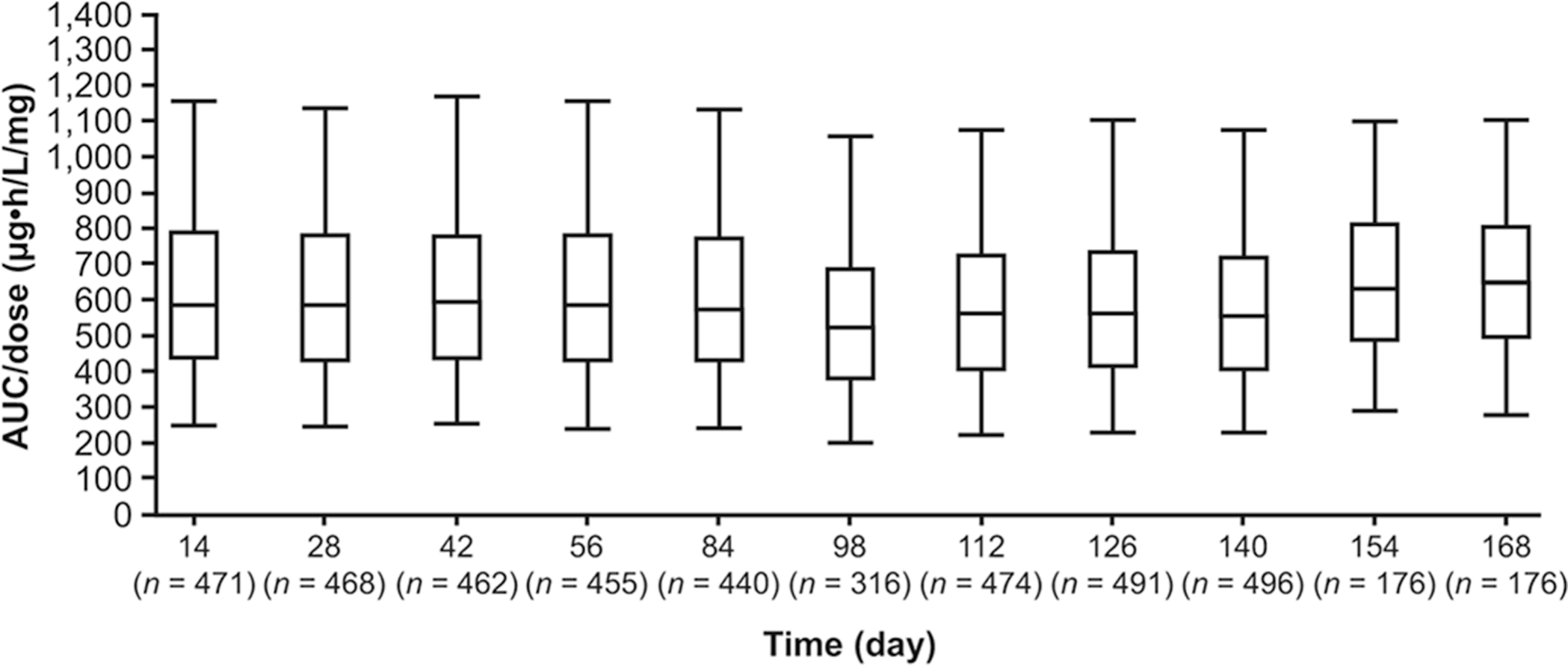
Box plots of riociguat dose-normalized area under the plasma concentration–time curve (AUC) by visits across the four studies.
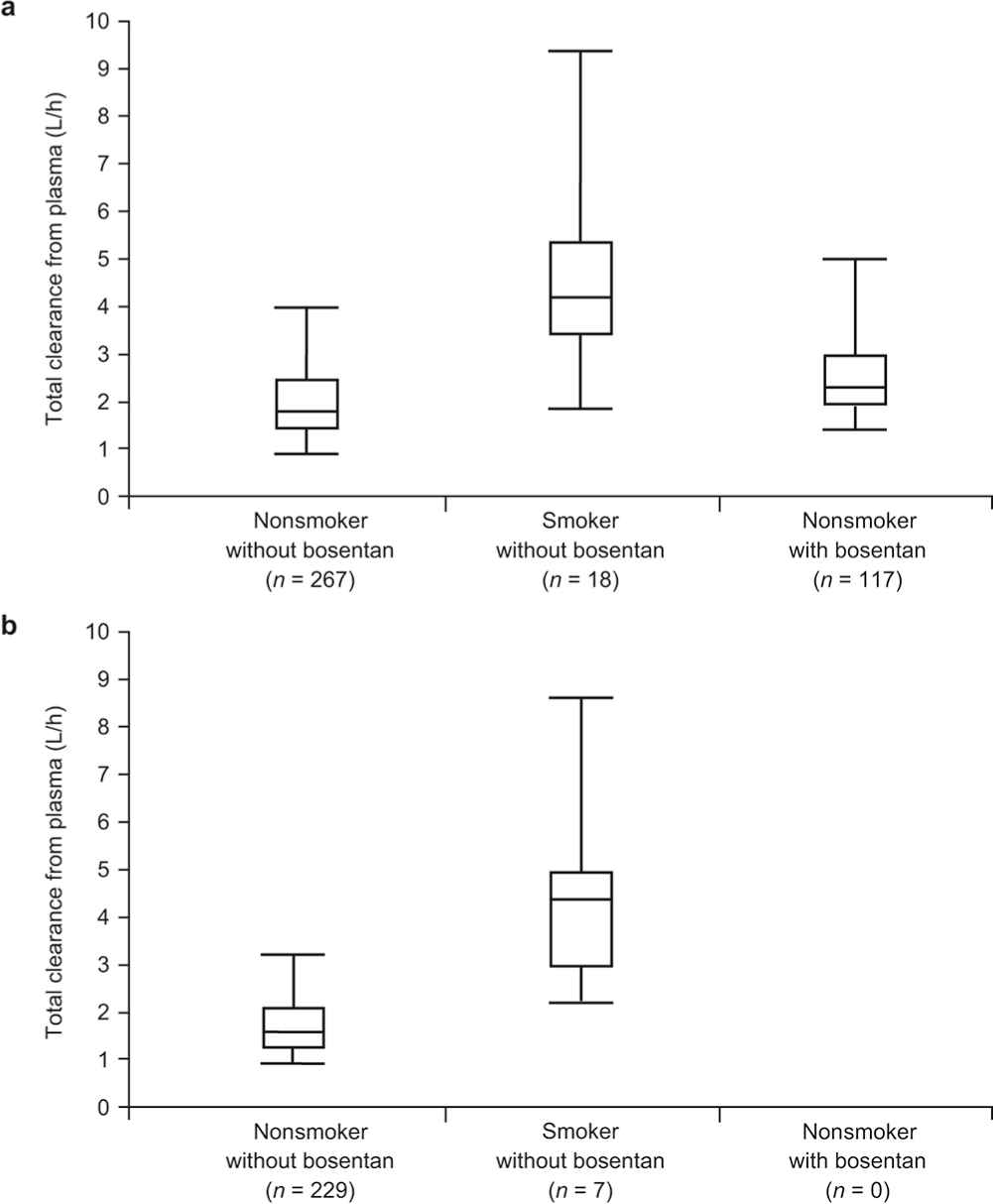
Box plots of individual post hoc estimates of the apparent clearance of riociguat versus smoking status and bosentan comedication in the PATENT-1/2 (a) and CHEST-1/2 (b) studies. In PATENT-1/2, median values of riociguat clearance taking into account covariates effects (bilirubin and creatinine clearance) were 1.8 L/h in nonsmokers not receiving bosentan, 4.2 L/h in smokers not receiving bosentan, and 2.3 L/h in nonsmokers receiving bosentan as comedication. In CHEST-1/2, median values of riociguat clearance taking into account covariates effects (bilirubin and creatinine clearance) were 1.6 L/h in nonsmokers and 4.2 L/h in smokers.
Riociguat exposure data in patients after multiple doses of riociguat under steady-state conditions
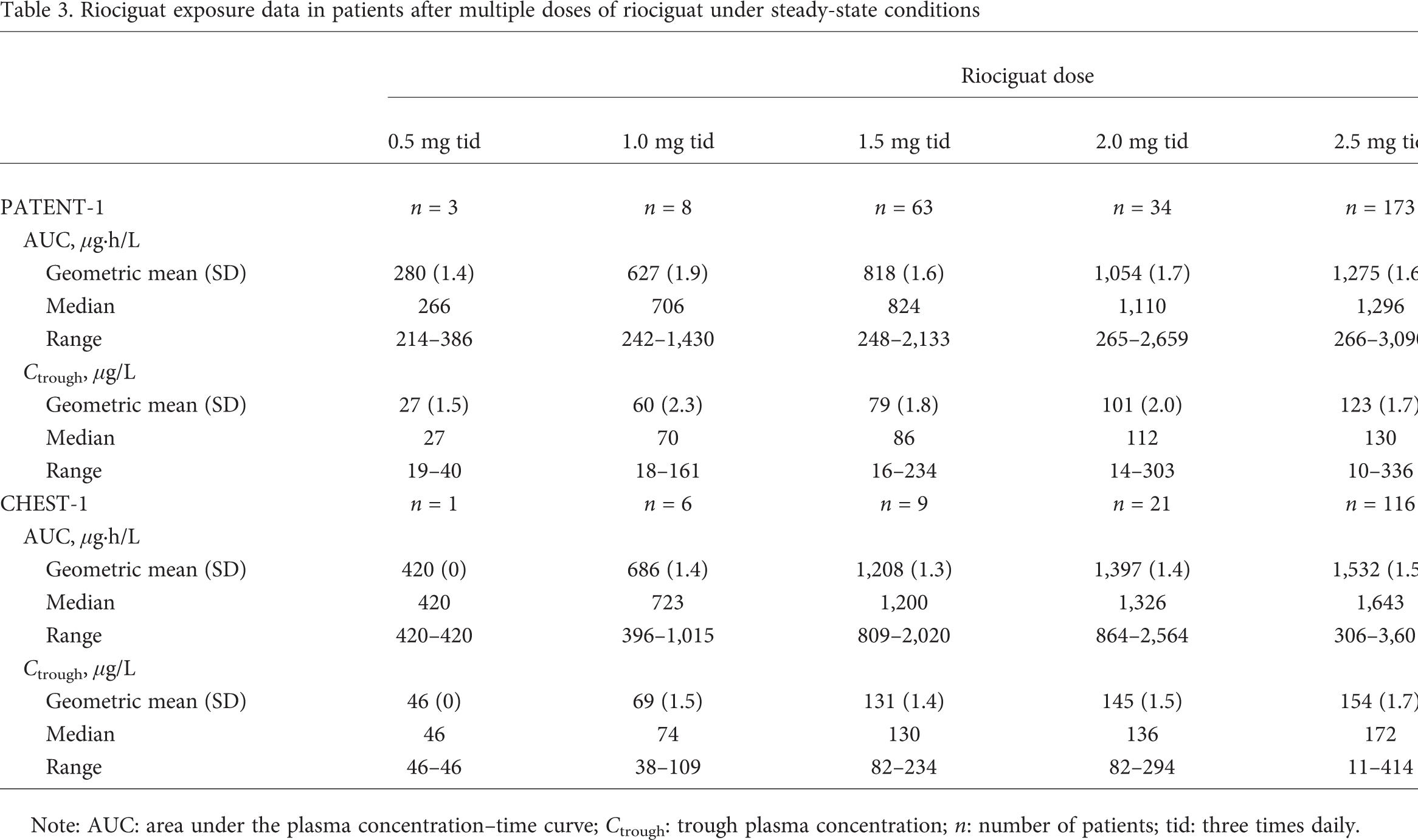
Note: AUC: area under the plasma concentration-time curve; Ctrough: trough plasma concentration; n: number of patients; tid: three times daily.
PK/PD relationship
PK/PD relationships were investigated by comparing derived PK parameters (AUC, Cmax, and Ctrough) and absolute changes in the relevant PD parameters (e.g., 6MWD, NT-proBNP, CO, PVR, and SVR) with their baseline values. The relationship between different PD parameters was also investigated. These relationships were analyzed in an exploratory manner using graphics showing individual or grouped data.
In PATENT-1 and CHEST-1, change from baseline in the PD parameter 6MWD was found to be related to hemodynamic PD parameters, most notably change from baseline in PVR (Fig. 8). Other exploratory graphical analyses showed that trough plasma riociguat concentrations were correlated with changes in PVR (Fig. 9), SBP (Fig. 10), and CO (Fig. 11); however, 6MWD did not directly correlate with riociguat exposure in both PAH and CTEPH patients (data not shown).
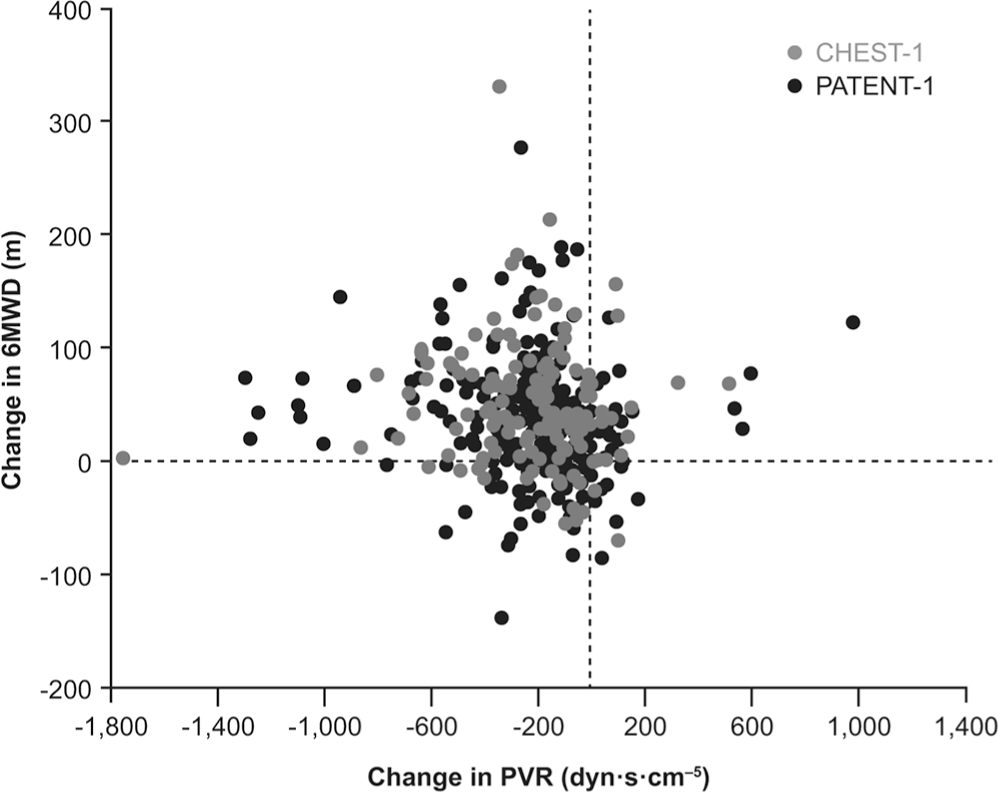
Relationship between 6-minute walk distance (6MWD) and pulmonary vascular resistance (PVR) at the end of the PATENT-1 (day 84) and CHEST-1 (day 112) studies.
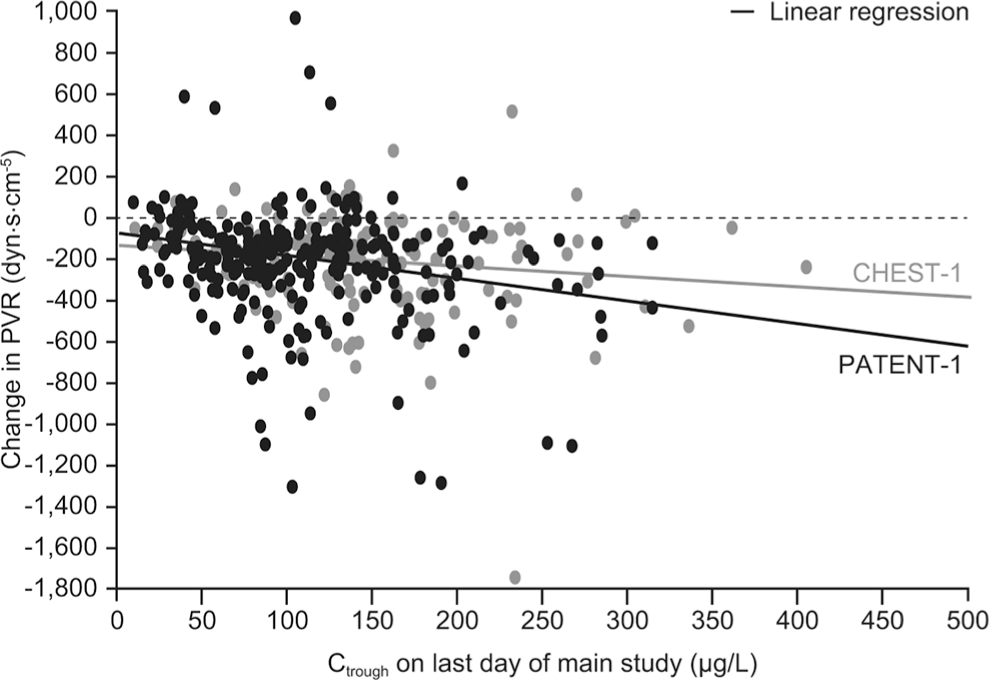
Exposure-response relationship between riociguat plasma trough concentration (Ctrough) and changes in pulmonary vascular resistance (PVR) in the PATENT-1 and CHEST-1 studies.
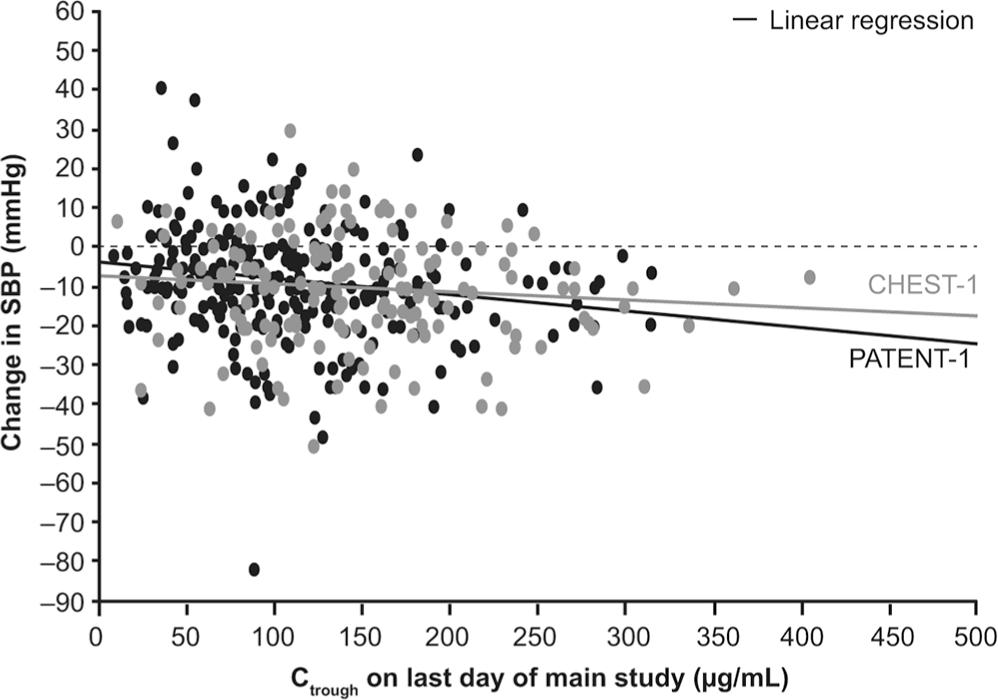
Exposure-response relationship between riociguat plasma trough concentration (Ctrough) and changes in systolic blood pressure (SBP) in the PATENT-1 and CHEST-1 studies.
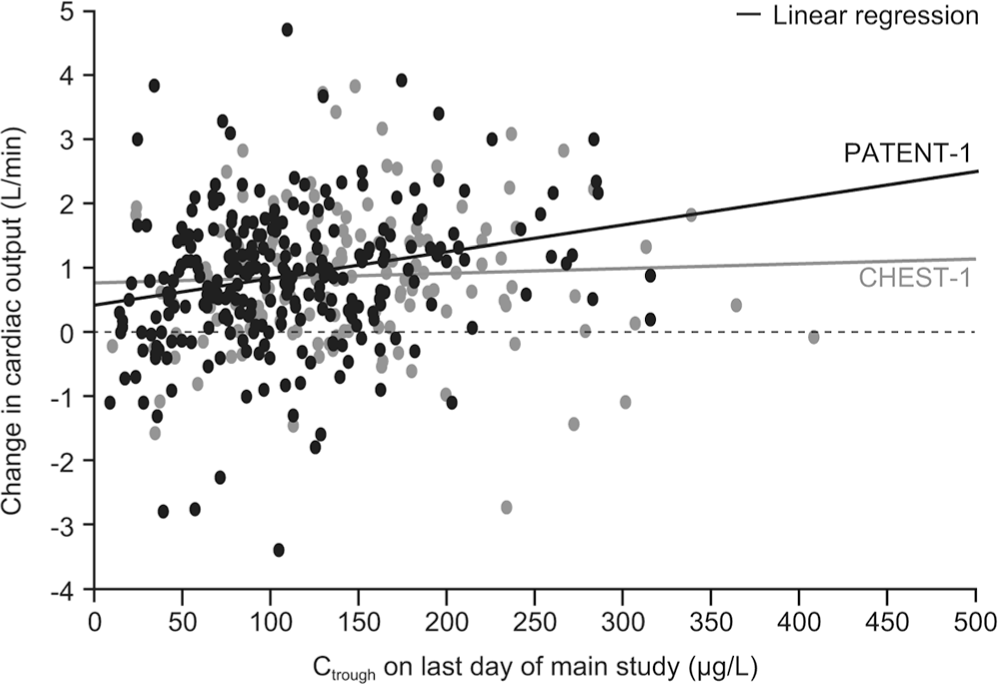
Exposure-response relationship between riociguat plasma trough concentration (Ctrough) and changes in cardiac output in the PATENT-1 and CHEST-1 studies.
DISCUSSION
The phase 3 PATENT-1 and CHEST-1 studies and their long-term extensions demonstrated the efficacy and tolerability of riociguat for the treatment of PAH and CTEPH, respectively.8–11 On the basis of the results of the present PK analysis, riociguat and M1 concentrations were well described by a simple linear one-compartment model. This is in accordance with the results of the phase 1 study in healthy subjects, where overall riociguat plasma concentrations and AUC showed dose-dependent increases with no obvious deviation from dose proportionality or linear PK. 15 In the current study, the interindividual variability of PK parameters was moderate for riociguat but moderate to high for the metabolite M1. This is consistent with PK interindividual variability previously seen in healthy male subjects 15 and is comparable with PK data from the first acute hemodynamic study of riociguat in patients with pulmonary hypertension. 3
Smokers showed higher riociguat clearance than nonsmokers, which can be explained by the difference in enzymatic activity of cytochrome P450 (CYP) 1A1 in these patient subgroups, as CYP1A1 is the decisive enzyme of riociguat metabolism5,6 and is induced by smoking.16,17 In patients receiving comedication with bosentan, an inducer of CYP3A and CYP2C9, 18 riociguat clearance was increased, although the effect was small and no dose adjustment is necessary in patients receiving endothelin receptor antagonists.
The correlation between riociguat trough concentrations and PVR demonstrates a good correspondence between exposure and hemodynamic response in the studied patient populations. No direct correlation between plasma concentration of riociguat and 6MWD was observed, which could be explained by the absence of a simple mechanistic link between plasma drug exposure and the improvement of exercise capacity in patients with PAH or CTEPH. In general, the correlations between 6MWD and hemodynamic parameters are consistent with other studies19–22 and represent improvements in CO or skeletal muscle oxygen delivery.
One possible weakness of these studies is the sparse plasma sampling regimen, as specified in the study protocols. Such a regimen, however, reduced the burden on patients and study centers and enabled a larger patient population to be included than is usual in PK studies.
In conclusion, plasma concentrations of riociguat and its main active metabolite, M1, were well described by a simple linear one-compartment model, with no evidence for time- or dose-dependent alterations in riociguat or M1 PK. Smokers and patients receiving bosentan as a comedication showed higher riociguat clearance than nonsmokers and those not receiving bosentan. Concomitant administration of endothelin receptor antagonists should not necessitate dose adjustment of riociguat. A good correlation between riociguat exposure and improvement in hemodynamic parameters (e.g., PVR) was shown. However, no correlation was observed between 6MWD and riociguat exposure in both PAH and CTEPH patients.
Footnotes
ACKNOWLEDGMENTS
We gratefully acknowledge Dagmar Klein and Matthias Frede (Clinical Sciences, Pharmacometrics, Bayer HealthCare Pharmaceuticals, Wuppertal, Germany) for the skillful data analysis and their contributions to the study.
