Abstract
Pulmonary arterial compliance is a measure of the pulsatile afterload of the right ventricle. Lower pulmonary arterial compliance is associated with reduced right ventricular function and worse prognosis in pulmonary hypertension. The effect of pulmonary vasodilators on pulmonary arterial compliance has not been evaluated in detail in pulmonary arterial hypertension or chronic thromboembolic pulmonary hypertension. In this post hoc analysis of patients with pulmonary arterial hypertension and chronic thromboembolic pulmonary hypertension in the PATENT and CHEST studies, we evaluated the change in pulmonary arterial compliance with riociguat versus placebo. Association of pulmonary arterial compliance with clinical outcomes was assessed using Kaplan–Meier and Cox proportional hazards analyses. Compared with placebo, riociguat significantly improved pulmonary arterial compliance in patients with pulmonary arterial hypertension or chronic thromboembolic pulmonary hypertension. Pulmonary arterial compliance at baseline was associated with survival and clinical worsening-free survival in pulmonary arterial hypertension but only with clinical worsening-free survival in chronic thromboembolic pulmonary hypertension. In patients with pulmonary arterial hypertension, pulmonary arterial compliance at follow-up ≥1.6 mL/mmHg was associated with better outcomes than pulmonary arterial compliance <1.6 mL/mmHg. In patients with chronic thromboembolic pulmonary hypertension, pulmonary arterial compliance at follow-up did not predict outcomes. Cox proportional hazards analyses showed no association between change in pulmonary arterial compliance and outcomes in patients with pulmonary arterial hypertension or chronic thromboembolic pulmonary hypertension. In conclusion, riociguat improved pulmonary arterial compliance in patients with pulmonary arterial hypertension or chronic thromboembolic pulmonary hypertension. Pulmonary arterial compliance at baseline or follow-up, rather than change in pulmonary arterial compliance, is of prognostic importance for outcomes.
Keywords
Pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH) are life-threatening diseases characterized by increased right ventricular (RV) afterload leading to RV failure, and ultimately death. In patients with PAH, the increased RV afterload is caused by progressive remodeling of the distal pulmonary vasculature, while in patients with CTEPH, it is caused by obstruction of the proximal pulmonary vasculature by organized thromboembolic material, as well as by distal proliferative vasculopathy.1–5 Initially, the right ventricle compensates for the increased afterload by increasing wall thickness and contractility, but these adaptations are eventually insufficient, resulting in RV failure. 6
In clinical trials and practice, pulmonary vascular resistance (PVR) is routinely used to represent RV afterload. PVR represents the static afterload but does not account for the pulsatile afterload. 7 Pulmonary arterial compliance (PAC) is a measure of the elasticity of the pulmonary vasculature and therefore provides insight into the pulsatile RV afterload.6–8 A combination of PVR and PAC is a more complete way to assess RV afterload,8–10 and their relationship has been shown to be inverse hyperbolic. 9 In support of this, a prospective study of 24 patients with PAH associated with systemic sclerosis showed a significant improvement in PAC following upfront combination therapy with ambrisentan and tadalafil. 10 Other prospective studies have found that lower PAC predicts worse survival in patients with PAH or PAH associated with systemic sclerosis.11,12 In contrast, a retrospective registry analysis of patients with PAH found that PAC did not independently predict outcomes at baseline or follow-up. 13 In patients with CTEPH undergoing pulmonary endarterectomy, postoperative PAC is a predictor of functional capacity and a stronger predictor of poor functional recovery than postoperative PVR.14,15 Despite its prognostic significance, the effect of pulmonary vasodilatory medical therapy on PAC has not been assessed in detail in patients with PAH or CTEPH.
PATENT-1 (NCT00810693) and CHEST-1 (NCT00855465) were phase 3 trials that assessed the efficacy and safety of riociguat in patients with PAH and those with inoperable or persistent/recurrent CTEPH, respectively.16,17 In both trials, riociguat significantly improved 6-minute walking distance, World Health Organization functional class, N-terminal prohormone of brain natriuretic peptide, PVR, cardiac output, mean pulmonary arterial pressure, and time to clinical worsening.16–19 Improvements in 6-minute walking distance, World Health Organization functional class, and N-terminal prohormone of brain natriuretic peptide were sustained at two years of treatment in the PATENT-2 (NCT00863681) and CHEST-2 (NCT00910429) long-term extensions.20,21 PATENT-1 and CHEST-1 represent one of the largest populations of patients with PAH or CTEPH with invasive hemodynamics measured before and after treatment. These studies provide the opportunity to assess the effect of riociguat on PAC in patients with PAH or CTEPH and identify the clinical significance of PAC in relation to long-term outcomes.
Accordingly, the primary aim of this exploratory post hoc analysis is to investigate the change in PAC with riociguat in patients with PAH or CTEPH. In addition, the change in PAC in patients categorized as low, average, or high risk according to their baseline REVEAL risk score (RRS) 22 is assessed. Finally, the associations between PAC at baseline, follow-up, change in PAC, and long-term clinical outcomes are evaluated.
Methods
Study design, procedures, and patients
The PATENT-1 and CHEST-1 study designs and inclusion criteria have been published previously.16,17 In brief, PATENT-1 was a 12 week, phase 3 study in which patients with PAH were randomized to placebo, riociguat individually adjusted up to 2.5 mg three times daily (tid), or riociguat individually adjusted up to 1.5 mg tid (exploratory; only included in overall PATENT-1 population analyses). 17 CHEST-1 was a 16 week, phase 3 trial in which patients with inoperable or persistent/recurrent CTEPH were randomized to placebo or riociguat up to 2.5 mg tid. 16
Patients completing PATENT-1 and CHEST-1 without ongoing study drug-related serious adverse events were invited to participate in the long-term extensions, PATENT-2 and CHEST-2.20,21 The long-term extensions comprised an eight-week, dose-adjustment phase for former placebo patients transitioning to riociguat 2.5 mg–maximum, followed by an open-label phase that will continue until all patients have transitioned to the commercially available drug. Informed consent was universally obtained, and all study procedures conformed to the ethical standards of the Declaration of Helsinki.
Outcome measures
Full details of the endpoints assessed in PATENT-1 and CHEST-1 have been published previously.20,21 Right heart catheterization was performed at baseline and follow-up (Week 12 in PATENT-1, Week 16 in CHEST-1). PAC was calculated post hoc as the ratio of stroke volume (SV) to pulmonary pulse pressure, i.e. SV/(systolic pulmonary artery pressure − diastolic pulmonary artery pressure), where SV = cardiac output/heart rate. RRS was calculated post hoc, as published previously.23,24 Based on their RRS at baseline, patients were stratified into one of three survival risk strata: low (score = 1–7), average (score = 8), and high (score ≥ 9).
Clinical worsening was defined as the first occurrence of any of the following events: death, lung or heart/lung transplantation, hospitalization due to worsening of pulmonary hypertension, start of new pulmonary hypertension-approved treatment, persistent decrease in 6-minute walking distance of > 15% from baseline, persistent worsening of World Health Organization functional class, atrial septostomy (PATENT-1/2 only), and the need for rescue pulmonary endarterectomy (CHEST-1/2 only).
Statistical analysis
Changes in PAC from baseline to follow-up in PATENT-1 and CHEST-1 were assessed by analysis of covariance, followed by a test for normality of the residuals and a nonparametric stratified Wilcoxon test upon rejection. Missing values were imputed at last visit according to the last observed postbaseline value.
The association of PAC and PVR with overall survival and clinical worsening-free survival was determined post hoc by univariate (baseline and posttreatment PAC) and bivariate (baseline and change from baseline to end of study visit) Cox proportional hazards regression analyses. Kaplan–Meier survival analyses were used to investigate the relationship between PAC and both survival and clinical worsening-free survival, stratified by median PAC values at baseline, end of study, and change throughout the study. P values < 0.05 were considered statistically significant
In the long-term analyses, for patients treated with riociguat in PATENT-1/CHEST-1, baseline was defined as PATENT-1/CHEST-1 baseline. For patients treated with placebo in PATENT-1/CHEST-1, baseline was defined as PATENT-1 Week 12/CHEST-1 Week 16 (the time immediately before the first riociguat dose). Patients who received placebo in PATENT-1/CHEST-1 were not included in assessments of the association of PAC and PVR at Week 12/Week 16 or change from baseline to Week 12/Week 16 and long-term outcomes.
Results
In total, 443 patients with PAH enrolled in PATENT-1, 254 received riociguat 2.5 mg–maximum tid, 63 received 1.5 mg–maximum tid, and 126 received placebo. Of the 261 patients with CTEPH enrolled in CHEST-1, 173 received riociguat 2.5 mg–maximum tid and 88 received placebo. Baseline characteristics of the PATENT-1 and CHEST-1 populations have been reported previously; those relevant to this study are shown in Supplementary Table S1.11,12 At PATENT-1 and CHEST-1 baseline, most patients were female (79% in PATENT-1 and 66% in CHEST-1) and were in World Health Organization functional class III (53% in PATENT-1 and 64% in CHEST-1). In PATENT-1, 50% of patients were treatment-naïve and 50% were pretreated with an endothelin receptor antagonist or prostacyclin. In CHEST-1, 72% had inoperable CTEPH and 28% had persistent/recurrent CTEPH.
Effect of riociguat treatment on PAC in PATENT-1 and CHEST-1
At PATENT-1 baseline, mean (standard deviation (SD)) PAC was 1.51 (0.82) mL/mmHg (median (interquartile range (IQR)), 1.33 (0.87–1.95) mL/mmHg) in the riociguat group (n = 227) and 1.45 (0.96) mL/mmHg (median (IQR), 1.25 (0.84–1.84) mL/mmHg) in the placebo group (n = 108) (Table S2, Fig. 1a). Patients with PAH associated with connective tissue disease had higher baseline PAC than those with idiopathic PAH (Table S2). At baseline, PAC showed a negative correlation with logPVR in PATENT-1 (riociguat: r = −0.747, P < 0.001; placebo: r = −0.681, P < 0.001; Fig. S1a).
Boxplots of PAC (a) at baseline and Week 12 of PATENT-1 and (b) at baseline and Week 16 of CHEST-1. Each box shows the median and IQR, with the mean indicated by a cross. The vertical lines extend to 1.5 IQR, with outliers plotted separately. PAC was defined as stroke volume/(systolic pulmonary artery pressure – diastolic pulmonary artery pressure). An outlier of PAC 25.20 mL/mmHg was not included in (b) for improved visualization of the figure.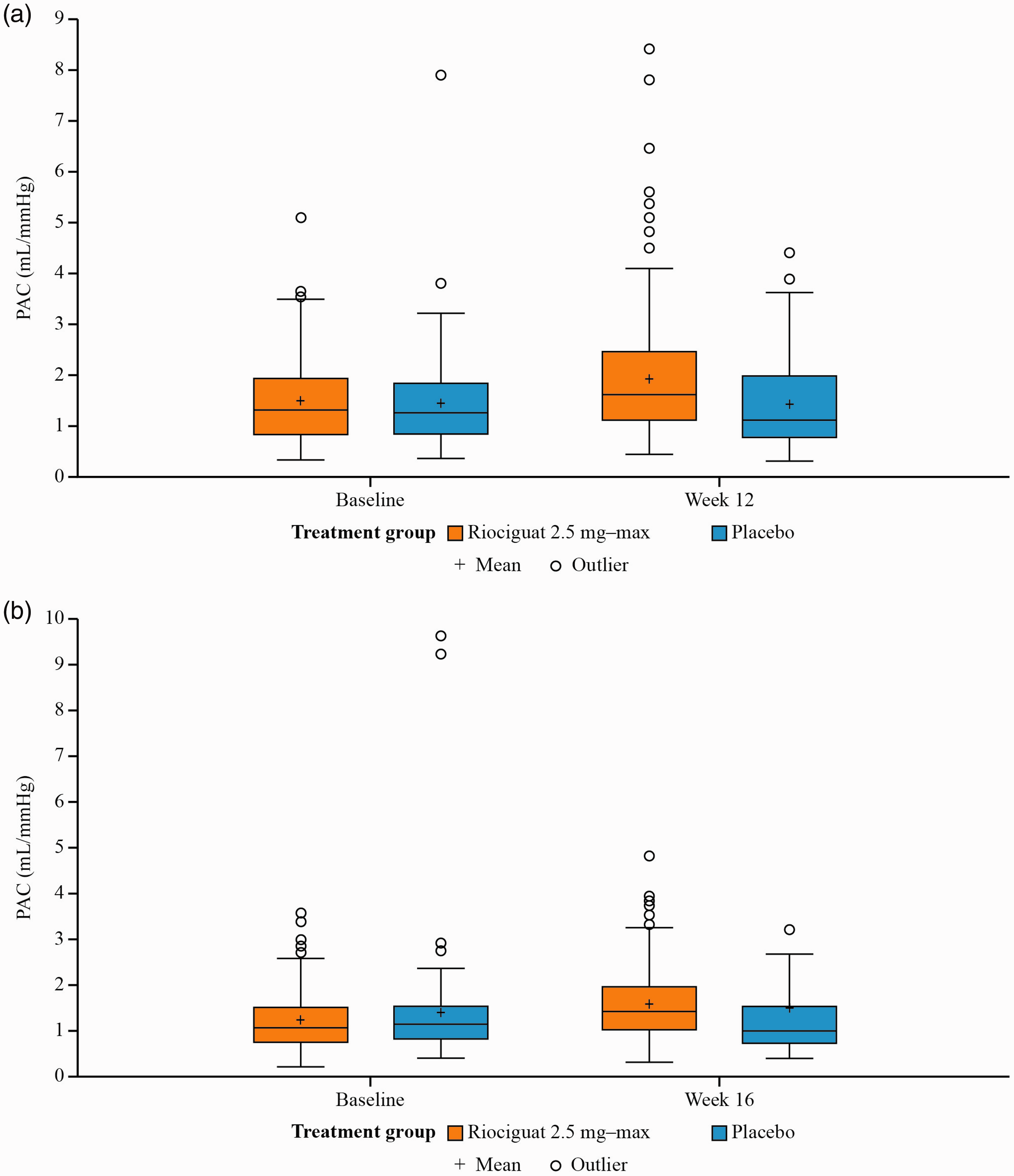
Significant improvements in PAC were observed from baseline to Week 12 with riociguat versus placebo (Fig. 1a), with a least-squares mean treatment difference of +0.44 mL/mmHg (95% confidence interval 0.26–0.62, P < 0.0001 using stratified Wilcoxon testing) (median (IQR) change from baseline, 0.27 (−0.02 to 0.73) mL/mmHg with riociguat, −0.02 (−0.31 to −0.21) mL/mmHg with placebo) (Fig. 1a). Similar improvements in PAC from baseline to Week 12 with riociguat were observed in the idiopathic PAH and PAH associated with connective tissue disease subgroups (Table S2). Treatment-naïve and pretreated patients showed similar increases in PAC with a mean (SD) change in PAC of +0.42 (0.93) mL/mmHg and +0.42 (0.87) mL/mmHg, respectively (Table S2).
At CHEST-1 baseline, mean (SD) PAC was 1.22 (0.65) mL/mmHg (median (IQR), 1.06 (0.74–1.48) mL/mmHg) in the riociguat group (n = 154) and 1.39 (1.40) mL/mmHg (median (IQR), 1.13 (0.81–1.53) mL/mmHg) in the placebo group (n = 81) (Table S3, Fig. 1b). As in PATENT-1, PAC showed a negative correlation with logPVR in CHEST-1 at baseline (riociguat: r = −0.761, P < 0.001; placebo: r = −0.534, P < 0.001; Fig. S1b). Similar to patients with PAH in PATENT-1, significant improvements in PAC were observed in patients with CTEPH in CHEST-1 from baseline to Week 16 with riociguat versus placebo (Fig. 1b; least-squares mean difference: +0.13 mL/mmHg (95% CI −0.34 to 0.59, P < 0.0001)) (median (IQR) change from baseline, 0.27 (0.01 to 0.57) mL/mmHg with riociguat and −0.05 (−0.29 to 0.14) mL/mmHg with placebo). Patients with inoperable CTEPH and persistent/recurrent CTEPH had similar PAC levels at baseline and showed similar changes in PAC under riociguat treatment with a mean (SD) change of +0.34 (0.48) mL/mmHg and of + 0.31 (0.65) mL/mmHg, respectively (Table S3).
In PATENT-1, the increase in PAC was accompanied by a reduction in mean ± SD PVR of −223 ± 260 dyn·sec·cm
−
5 (Fig. 2).
17
In CHEST-1, the increase in PAC was smaller than in PATENT-1 but was accompanied by a similar reduction in mean ± SD PVR to PATENT-1 of −226 ± 248 dyn·sec·cm
−
5 (Fig. 2).
16
Mean PAC–PVR relationship (a) at baseline and Week 12 of PATENT-1 and (b) at baseline and Week 16 of CHEST-1. Data are mean ± standard error of the mean.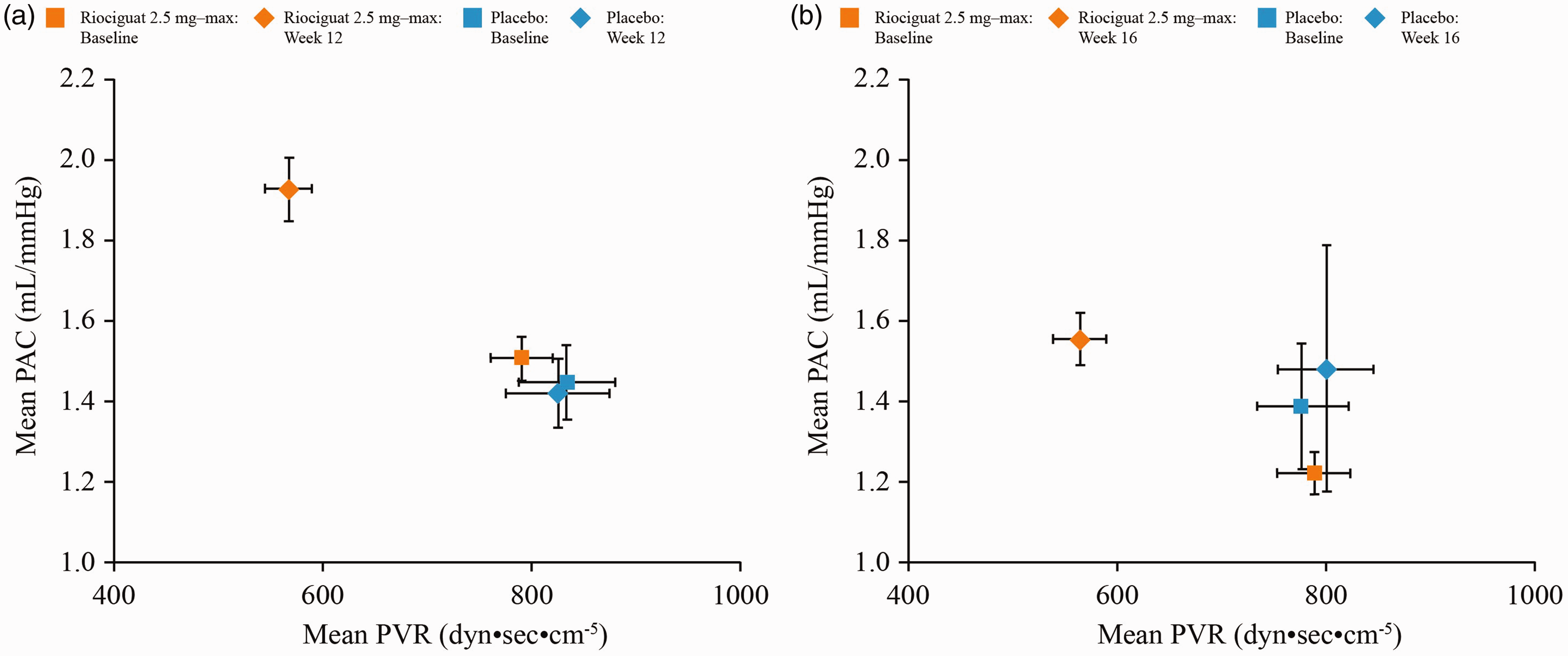
When stratified by RRS, patients in the low-, medium-, and high-risk REVEAL strata showed similar improvements in PAC from baseline to last visit with riociguat in PATENT-1 and CHEST-1 (Fig. 3). However, there was some variability according to REVEAL risk stratum in the change in PAC for patients receiving placebo (Fig. 3).
Change in PAC: in PATENT-1 and CHEST-1 for patients in low-, medium-, or high-risk REVEAL strata. Data are mean ± standard error of the mean.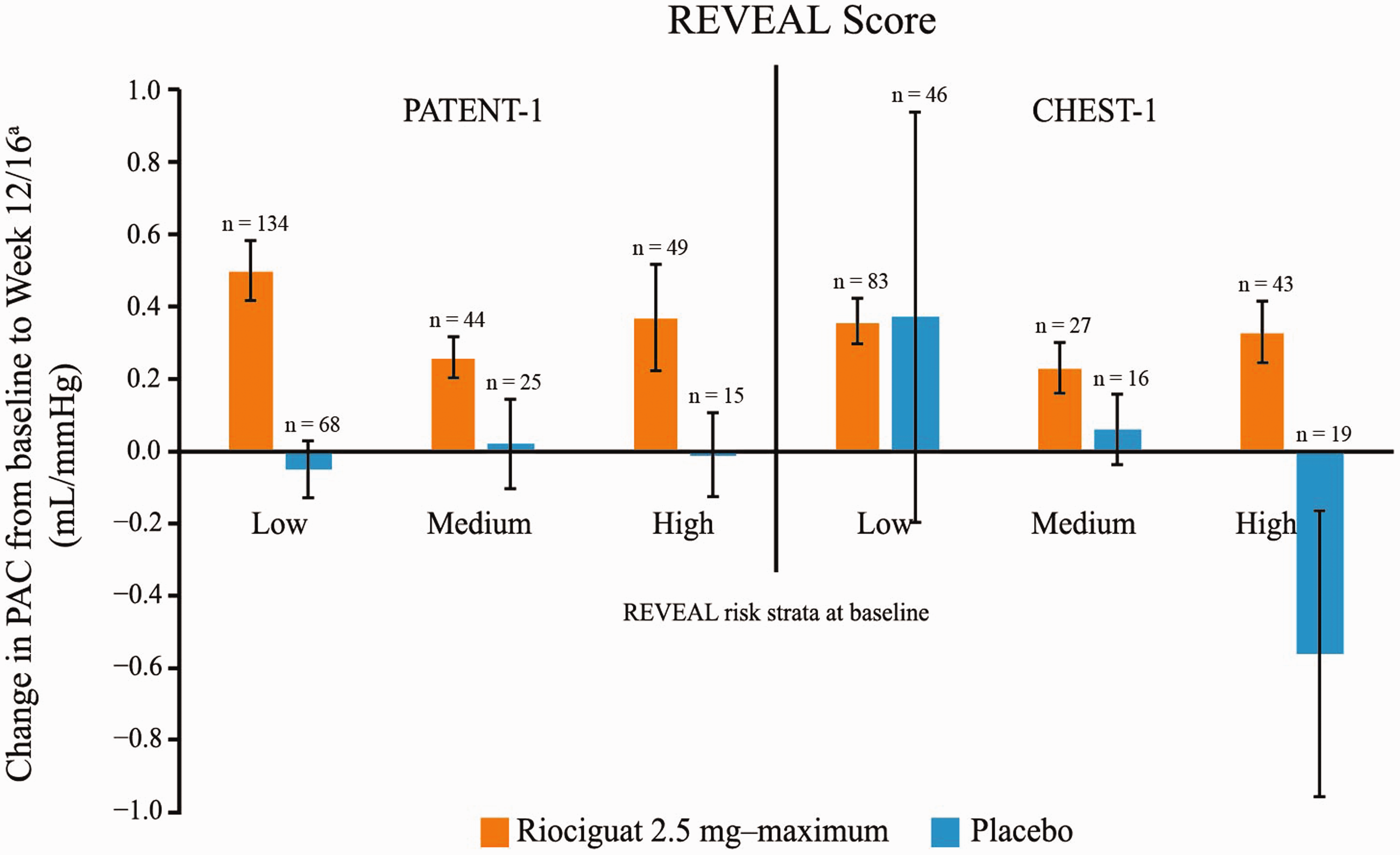
Association between PAC and long-term outcomes
PAC at baseline
In PATENT, Cox proportional hazards analyses showed that PAC at baseline was significantly associated with survival and clinical worsening-free survival in patients with PAH. For every 0.5 mL/mmHg increase in baseline PAC, patients had a 20% reduction in risk of death and a 15% reduction in the risk of experiencing a clinical worsening event (Table 1). Additional assessment of survival with Kaplan–Meier analyses found that patients with PAC above the median at baseline (1.23 mL/mmHg) had a significantly better chance of survival (Fig. 4a) and clinical worsening-free survival (Fig. 4b) versus those with PAC below the median at baseline.
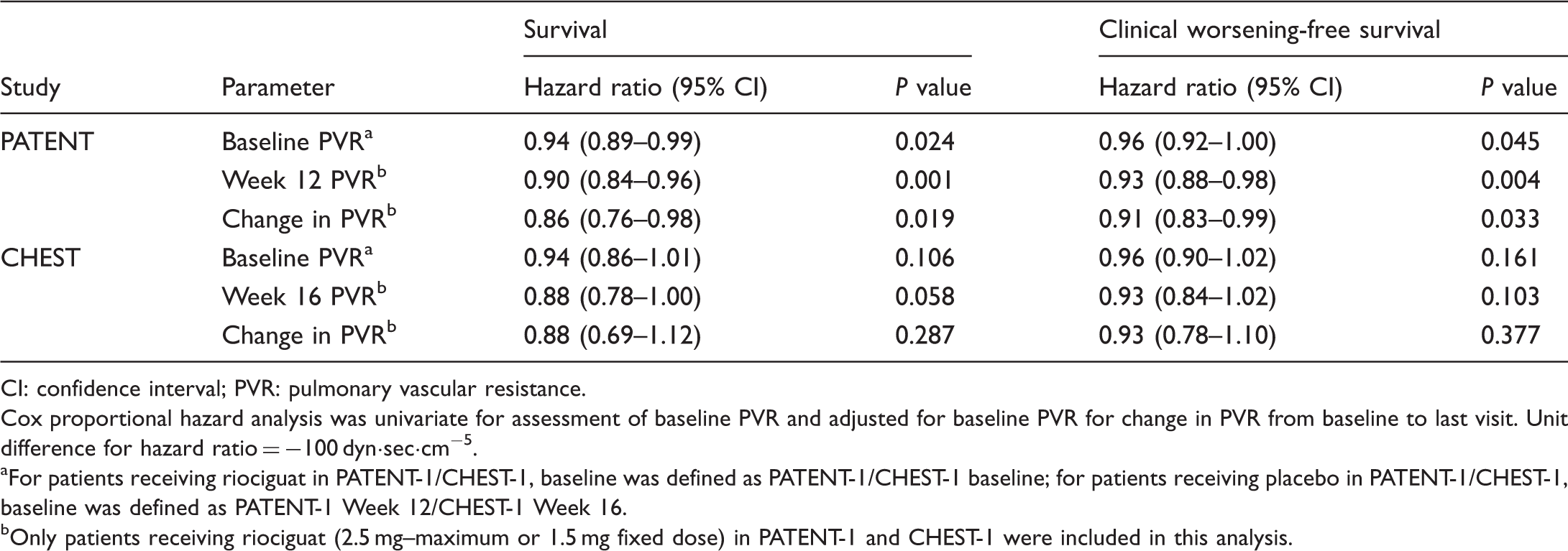
Kaplan–Meier analysis for (a) survival and (b) clinical worsening-free survival in PATENT-2 and (c) survival and (d) clinical worsening-free survival in CHEST-2, separated by patients with PAC ≥ or < than the median at baseline. Log-rank tests were used to assess significance. For patients treated with riociguat in PATENT-1/CHEST-1, baseline was defined as PATENT-1/CHEST-1 baseline. For patients treated with placebo in PATENT-1/CHEST-1, baseline was defined as PATENT-1 Week 12/CHEST-1 Week 16 (the time immediately before the first riociguat dose). Day 0 was defined as the first day of the long-term extension phase in patients who received riociguat in PATENT-1 or CHEST-1, and as PATENT-2 or CHEST-2 Week 12 in patients who received placebo in PATENT-1 or CHEST-1 (after patients had received four weeks of therapy at their optimum riociguat dose). Cox proportional hazards analyses of the association of baseline pulmonary vascular resistance with survival and clinical worsening-free survival in PATENT and CHEST. CI: confidence interval; PVR: pulmonary vascular resistance. Cox proportional hazard analysis was univariate for assessment of baseline PVR and adjusted for baseline PVR for change in PVR from baseline to last visit. Unit difference for hazard ratio = −100 dyn·sec·cm−5. For patients receiving riociguat in PATENT-1/CHEST-1, baseline was defined as PATENT-1/CHEST-1 baseline; for patients receiving placebo in PATENT-1/CHEST-1, baseline was defined as PATENT-1 Week 12/CHEST-1 Week 16. Only patients receiving riociguat (2.5 mg–maximum or 1.5 mg fixed dose) in PATENT-1 and CHEST-1 were included in this analysis.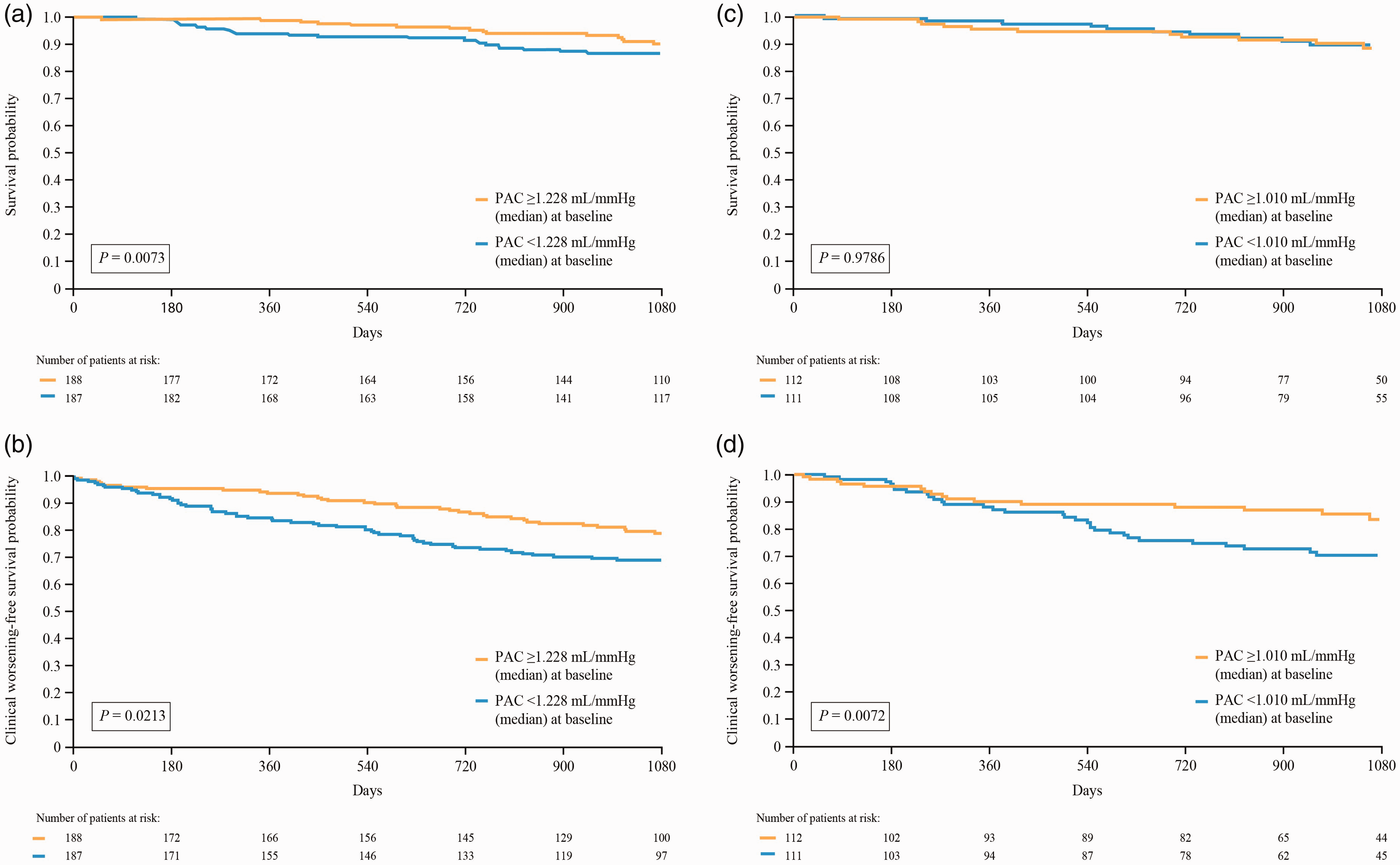
Cox proportional hazards analyses of the association of baseline pulmonary arterial compliance with survival and clinical worsening-free survival in PATENT and CHEST.
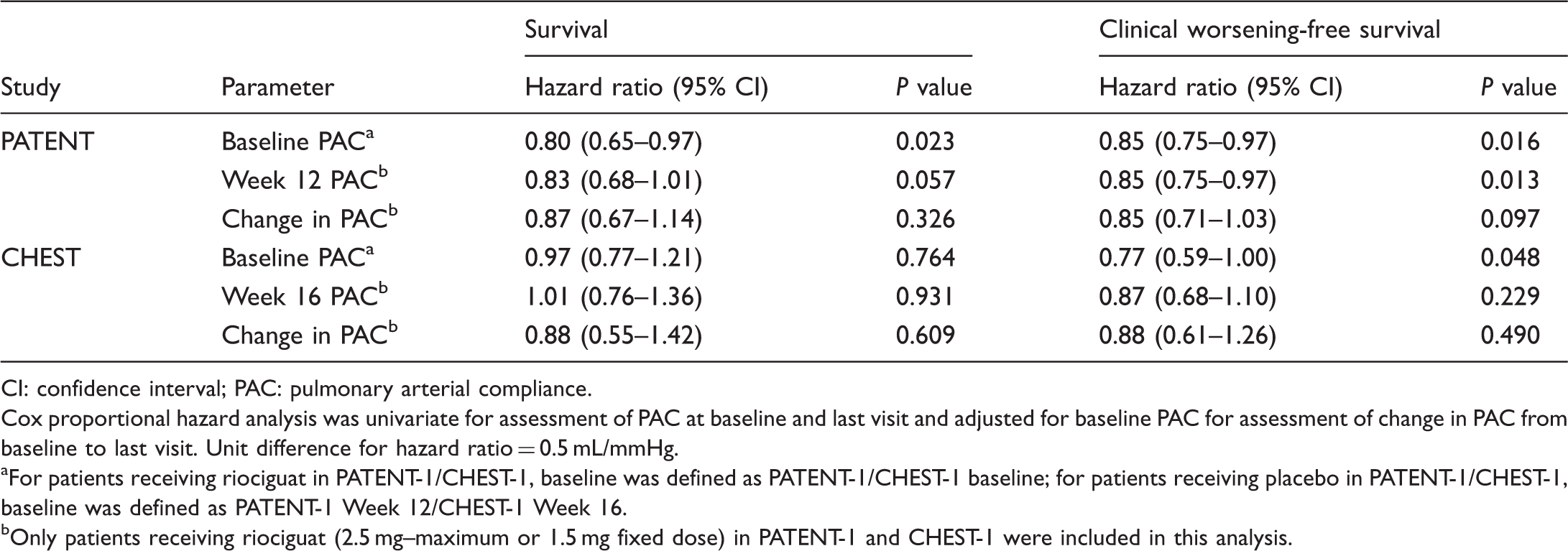
CI: confidence interval; PAC: pulmonary arterial compliance.
Cox proportional hazard analysis was univariate for assessment of PAC at baseline and last visit and adjusted for baseline PAC for assessment of change in PAC from baseline to last visit. Unit difference for hazard ratio = 0.5 mL/mmHg.
For patients receiving riociguat in PATENT-1/CHEST-1, baseline was defined as PATENT-1/CHEST-1 baseline; for patients receiving placebo in PATENT-1/CHEST-1, baseline was defined as PATENT-1 Week 12/CHEST-1 Week 16.
Only patients receiving riociguat (2.5 mg–maximum or 1.5 mg fixed dose) in PATENT-1 and CHEST-1 were included in this analysis.
PAC at follow-up
In PATENT, Cox proportional hazards analyses showed that PAC at follow-up was not associated with survival (Table 1) but was significantly associated with clinical worsening-free survival in patients with PAH. For every 0.5 mL/mmHg increase in PAC at follow-up, patients had a 15% reduction in risk of experiencing a clinical worsening event (Table 1). Kaplan–Meier analyses showed that patients with PAC greater than the median at follow-up (1.60 mL/mmHg) had a significantly better chance of survival (Fig. 5a) and clinical worsening-free survival (Fig. 5b) than those with PAC below the median at follow-up.
Kaplan–Meier analysis for (a) survival and (b) clinical worsening-free survival in PATENT-2 and (c) survival and (d) clinical worsening-free survival in CHEST-2, separated by patients with PAC ≥ or < than the median at Week 12/16. Log-rank tests were used to assess significance. Only patients receiving riociguat (2.5 mg–maximum or 1.5 mg fixed dose) in PATENT-1 and CHEST-1 were included in this analysis. Day 0 was defined as the first day of the long-term extension phase.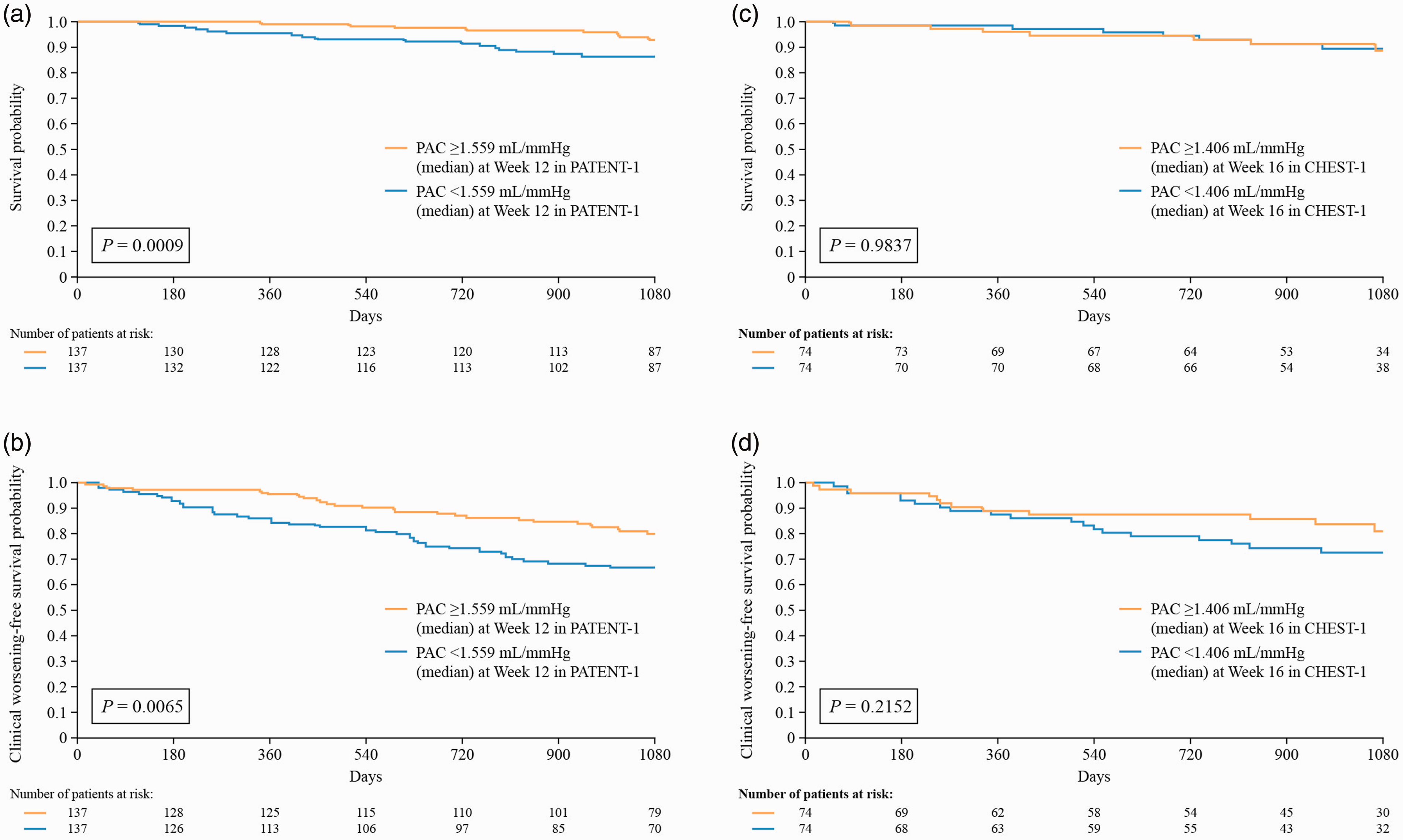
In CHEST, Cox proportional hazards analyses showed that PAC at follow-up was not associated with survival or clinical worsening-free survival in patients with CTEPH (Table 1). This was also supported by Kaplan–Meier analyses, which showed that when patients were stratified by the median posttreatment PAC value at follow-up (1.41 mL/mmHg), PAC values above or below the median were not associated with survival (Fig. 5c) or clinical worsening-free survival (Fig. 5d).
Change in PAC with treatment
Cox proportional hazards analyses showed no association between change in PAC with riociguat therapy and survival or clinical worsening-free survival (Table 1) in PATENT or CHEST. However, Kaplan–Meier analyses showed that in PATENT, patients with PAH who had a change in PAC from baseline to Week 12 above the median (0.254 mL/mmHg) had significantly better survival (Fig. 6a) and clinical worsening-free survival (Fig. 6b) than those with a change in PAC below the median. In CHEST, a change in PAC above the median (0.281 mL/mmHg) was not associated with survival in patients with CTEPH; however, it was significantly associated with increased clinical worsening-free survival (Fig. 6d).
Kaplan–Meier analysis for (a) survival and (b) clinical worsening-free survival in PATENT-2 and (c) survival and (d) clinical worsening-free survival in CHEST-2, separated by patients with a change in PAC from baseline to Week 12/16 ≥ or < than the median. Log-rank tests were used to assess significance. Only patients receiving riociguat (2.5 mg–maximum or 1.5 mg fixed dose) in PATENT-1 and CHEST-1 were included in this analysis. Day 0 was defined as the first day of the long-term extension phase.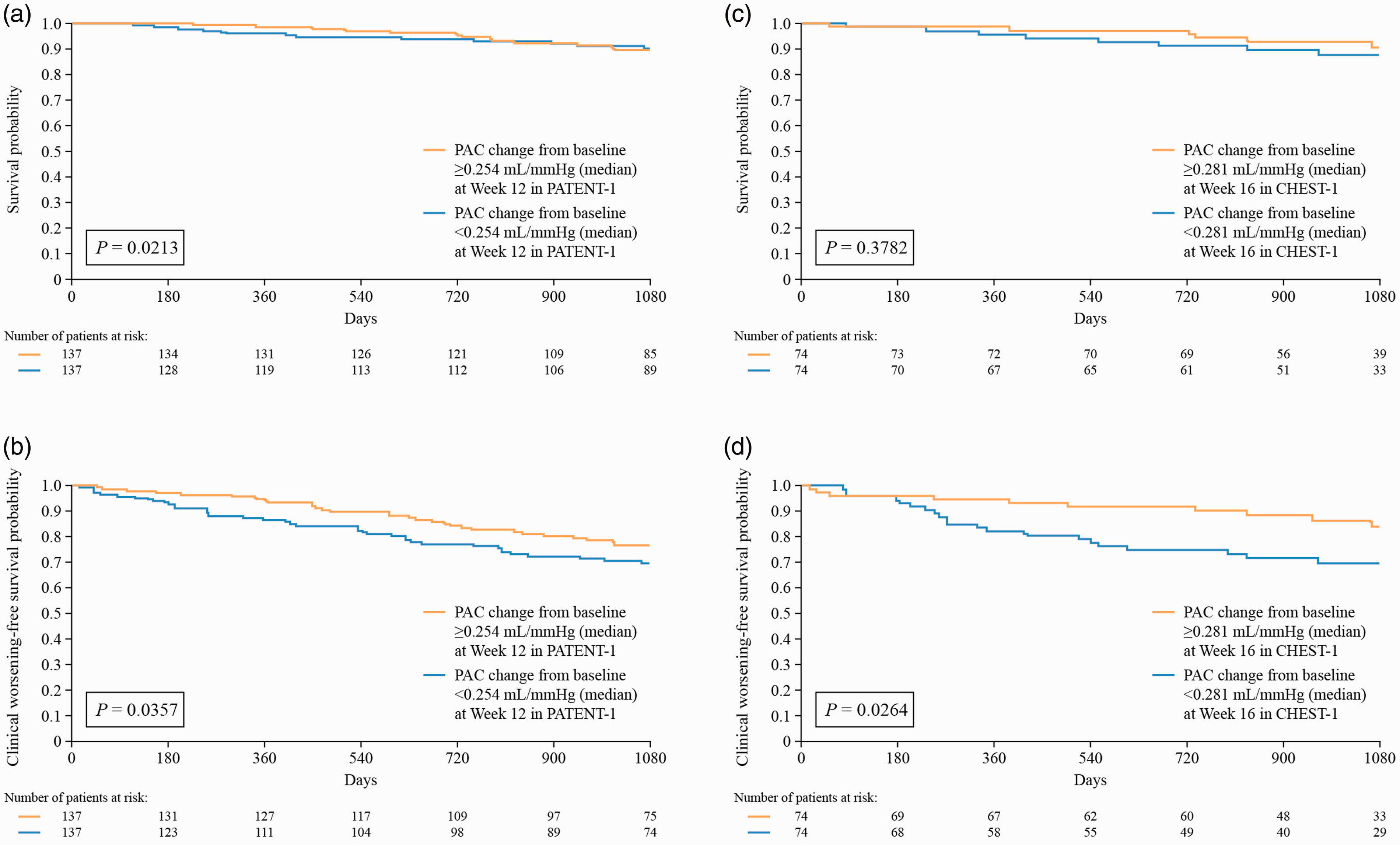
Association between PVR and long-term outcomes
PVR values at baseline and follow-up in PATENT and CHEST have been reported previously.19,25 In both studies, PVR was significantly improved with riociguat treatment compared with placebo. Cox proportional hazards analyses showed a significant association between PVR at baseline, follow-up, or change in PVR with survival and clinical worsening-free survival (Table 2) in patients with PAH in PATENT; however, no association was found between PVR at baseline, follow-up, or change from baseline with either survival or clinical worsening-free survival in patients with CTEPH in CHEST.
Discussion
We show that riociguat significantly improved PAC after 12 and 16 weeks of therapy in patients with PAH or CTEPH, respectively. The improvement in PAC with riociguat was irrespective of baseline RRS in both studies. PAC at baseline was prognostic for survival and clinical worsening-free survival in patients with PAH but only for clinical worsening-free survival in patients with CTEPH. PAC at follow-up was also associated with reduced risk of clinical worsening in patients with PAH, but not in those with CTEPH. Finally, change in PAC with riociguat therapy was not associated with survival or clinical worsening in patients with PAH or CTEPH. The potential mechanism by which riociguat improves PAC is unclear. The antifibrotic effects of riociguat may help to reverse the remodeling of distal pulmonary arteries observed in PAH and CTEPH. 26 Supporting this hypothesis, riociguat reduced RV collagen levels and reversed pulmonary artery remodeling in rodent models of pulmonary hypertension.27–29
In patients with pulmonary hypertension, reduced PAC and increased PVR contribute to RV dysfunction. 6 Previous results indicated that riociguat improved RV function in patients with PAH or CTEPH in PATENT-1 and CHEST-116,17 by significantly decreasing PVR, i.e. the static afterload of the right ventricle. Building on this, we show here that riociguat also increased PAC, which represents the pulsatile afterload of the right ventricle. This is significant as RV function is an important prognostic marker in patients with PAH or CTEPH. However, it should be noted that PAC and PVR are mathematically and pathophysiologically linked, with an inverse-hyperbolic relationship.9,30 The constant product of PVR and PAC, resistance-compliance time, is unaffected by therapy, and due to the inverse-hyperbolic relationship between the two parameters, posttreatment PAC levels could be a better hemodynamic target than PVR as an increase in PVR leads to a larger decrease in PAC. 30
It is critical to identify clinical parameters that correlate with long-term outcomes, thereby allowing physicians to assess disease severity and adjust treatment accordingly. 1 Corresponding with previous studies in patients with PAH or PAH associated with systemic sclerosis,10–12 in PATENT, every 0.5 mL/mmHg increase in PAC at baseline was associated with a 20% decrease in the risk of death and a 15% decrease in the risk of clinical worsening in patients with PAH. Higher PAC at follow-up was also associated with reduced risk of clinical worsening. Furthermore, patients with PAC ≥ 1.6 mL/mmHg at follow-up had improved survival and lower clinical worsening versus those with PAC < 1.6 mL/mmHg at follow-up. This is consistent with a recent observation by Ghio et al. 31 that patients with PAC ≥ 1.4 mL/mmHg at either treatment initiation or escalation had better overall survival compared with patients with PAC < 1.4 mL/mmHg with therapy. These results indicate that PAC is a relevant clinical parameter for predicting long-term outcomes and assessing treatment response in patients with PAH.
In CHEST, the association between PAC and long-term outcomes was not as robust as in PATENT. Only PAC at baseline was prognostic of clinical worsening, and there was no association between PAC at baseline or at follow-up with survival in these patients. The reason for the difference in the association between PAC and long-term outcomes between patients with PAH or CTEPH is unclear. However, the smaller number of patients with CTEPH in the CHEST study compared with the number with PAH in the PATENT study, and the different disease entities need to be taken into account. The event rate for mortality was low overall in both the PATENT and CHEST study cohorts (Fig. 4a and c).
Although there was an association between absolute PAC at baseline and follow-up with long-term outcomes in PATENT, the relationship between change in PAC and outcomes with riociguat was more ambiguous. Using Cox proportional hazards regression analysis, a change of 0.5 mL/mmHg was not significantly associated with a change in risk of mortality or clinical worsening. However, Kaplan–Meier assessment of patients grouped by PAC ≥ or < the median at follow-up in PATENT-1 or CHEST-1 found that those patients with a higher PAC had a significantly increased likelihood of survival and clinical worsening-free survival in PATENT-2 and a significantly increased likelihood of clinical worsening-free survival in CHEST-2. In a post hoc analysis of the hemodynamic effect of macitentan in patients with PAH, Galiè et al. 32 reported that, while the absolute values of cardiac index and right atrial pressure at baseline and at six months after treatment were associated with the combined morbidity/mortality endpoint, there was no relationship between the change in these parameters and morbidity and mortality. Similarly, in a study by Mazurek et al., 33 absolute values of tricuspid plane annular systolic excursion (a measure of RV function) at baseline and follow-up—but not the change from baseline—were associated with long-term survival. These results indicate that absolute cutoff values of indices of RV function are more important than the change in these parameters with therapy. Therefore, it may be that change in PAC with treatment is less important for assessing patient outcomes than absolute PAC values at baseline or follow-up for patients with PAH or CTEPH.
In our analysis, patients with idiopathic PAH had lower PAC than those with PAH associated with connective tissue disease. We believe this is because patients with idiopathic PAH had relatively severe pulmonary vascular disease with higher PVR compared with patients with PAH associated with connective tissue disease (mean PVR was 844 ± 471 vs. 647 ± 350 dyn·sec·cm − 5, respectively). Similar findings have been observed by others previously. 34
Limitations of this study include its exploratory post hoc nature. Due to the nature of the analysis, no adjustment for multiple statistical testing was conducted. In addition, PAC was estimated from the ratio of SV to pulmonary pulse pressure, which has several inherent limitations. This method overestimates compliance, as it does not account for blood flow from the pulmonary circulation into the capillary bed during systole. 35 Moreover, the method is based on two important assumptions that may not be true in all patients with PAH. First, it assumes that there is an exponential decay of the pulmonary artery diastolic pressure. Second, it assumes that the pressure–volume relationship in the pulmonary artery is linear. 35 Nevertheless, the ratio of SV to pulmonary pulse pressure correlates well with PAC estimation methods and is attractive for clinical application as it can be calculated from standard hemodynamic measurements.7,8 We also acknowledge that prospective studies are warranted to confirm and validate the present data. Further analyses that could provide interesting results include an investigation of a similar PAC cutoff in patients with PAH and CTEPH, including assessment of differences in specificity and sensitivity between the study populations, and evaluation of the potential impact of riociguat on the inverse-hyperbolic relationship between PAC and PVR.
In conclusion, this study demonstrates the beneficial effects of riociguat on PAC in patients with PAH and those with inoperable or persistent/recurrent CTEPH. The assessment of change in PAC with treatment is less important for assessing patient outcomes than absolute PAC values at baseline or follow-up for patients with PAH or CTEPH.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020963836 - Supplemental material for Effect of riociguat on pulmonary arterial compliance in the PATENT and CHEST studies
Supplemental material, sj-pdf-1-pul-10.1177_2045894020963836 for Effect of riociguat on pulmonary arterial compliance in the PATENT and CHEST studies by Thenappan Thenappan, Nadine Al-Naamani, Stefano Ghio, Hossein-Ardeschir Ghofrani, Paul M. Hassoun, Marc Pritzker, Adam Torbicki, Sylvia Nikkho, Dennis Busse and Ioana R. Preston in Pulmonary Circulation
Footnotes
Acknowledgments
The authors wish to thank the patients and investigators who participated in the PATENT and CHEST studies.
Author contributions
All authors had access to the study data. Bayer AG, T. Thenappan, and I. Preston were responsible for the design of the post hoc analysis and conceptualization of the manuscript. D. Busse undertook all statistical analyses. All authors contributed to the interpretation, drafting, and revision of the manuscript. All authors approved the manuscript for submission.
Conflict of interest
TT has received personal fees from Actelion Pharmaceuticals Ltd. and Gilead Sciences Inc. NA-N has received grants from Entelligence Young Investigator Program, ATS/PHA Aldrighetti Research Award for Young Investigators, and K23HL141584. SG, PMH, and MP do not have a financial relationship with a commercial entity that has an interest in the subject of the presented manuscript or other conflicts of interest to disclose. H-AG has received grants and personal fees from Actelion Pharmaceuticals Ltd., Bayer AG, ErgoNex Pharma GmbH, and Pfizer and personal fees from Gilead Sciences Inc., GSK, Merck, and Novartis. AT has received personal fees (speaker’s honoraria) from Bayer AG, Bristol-Myers Squibb, and Actelion Pharmaceuticals Ltd. and reports nonfinancial support from Pfizer for congress participation. SN is an employee of Bayer AG. DB was an employee of Chrestos Concept GmbH & Co. KG, Essen, Germany during the writing of this manuscript. IRP has received grants and personal fees from Actelion Pharmaceuticals Ltd., Arena Pharmaceuticals, Bayer AG, Gilead Sciences Inc., and United Therapeutics; grants from Liquidia Technologies; and personal fees from Pfizer and Reata Pharmaceuticals.
Ethical approval
Ethical approval was obtained for the PATENT-1 and -2 studies.
Funding
The PATENT-1, PATENT-2, CHEST-1, and CHEST-2 studies were supported by Bayer AG (Berlin, Germany) and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc. (Kenilworth, NJ, USA). Medical writing services provided by Robyn Bradbury, PhD, of Adelphi Communications Ltd (Macclesfield, UK) were funded by Bayer AG (Berlin, Germany) in accordance with Good Publications Practice (GPP3) guidelines.
Guarantor
T. Thenappan is the guarantor of this article and takes responsibility for the content of the manuscript, including the data and analysis.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
