Abstract
The diagnosis of pulmonary hypertension (PH) requires a right heart catheterization (RHC) that reveals a mean pulmonary artery pressure ≥ 25 mmHg. The pulmonary artery catheter traverse the right atrium and ventricle on its way to the pulmonary artery. The presence of abnormal right heart structures, i.e. thrombus, vegetation, benign or malignant cardiac lesions, can lead to complications during this procedure. On the other hand, avoidance of RHC delays the diagnosis and treatment of PH, an approach that might be associated with worse outcomes. This paper discusses the impact of right heart lesions on the diagnosis of PH and suggests an approach on how to manage this association.
Introduction
Pulmonary hypertension (PH) is a condition characterized by an elevated pulmonary artery pressure (PAP) that could lead to right heart failure and death. 1 Echocardiography is an adequate screening test, 2 but given the limitations of non-invasive tests, the diagnosis of PH requires a right heart catheterization (RHC). PH is present when the mean pulmonary artery pressure (mPAP) is ≥ 25 mmHg. 2 Besides confirming the presence of PH, hemodynamic determinations obtained during RHC are essential to narrow possible etiologies. PH can be broadly divided into pre-and/post-capillary PH.2,3 Pre-capillary PH is characterized by a pulmonary artery wedge pressure (PAWP) ≤ 15 mmHg, whereas post-capillary PH is distinguished by a PAWP of > 15 mmHg. Post-capillary PH can be further divided into the isolated or combined (pre- and post-capillary) forms. 2
RHC is a relatively safe procedure 4 with very low risk of serious complications or death. In experienced centers, the overall risk of serious adverse events in patients with PH is around 1%. 4 Most frequent complications are related to venous access (local hematoma/pneumothorax), arrhythmias, and hypotensive episodes (vagal reactions). In a large multicenter series (n = 7,218 RHC), the RHC-related mortality was 0.055%. 4
On certain occasions, echocardiography (transthoracic or transesophageal) or other imaging modalities such as computed tomography (CT) or cardiac magnetic resonance imaging (CMRI) show abnormal structures on the right side of the heart. These abnormalities may include clots, right valve vegetations, benign or malignant tumors, or more frequently normal anatomical variants that could be mistaken for abnormal lesions. The presence of abnormal right heart structures may expose patients to higher risks during RHC, such as embolic events in the pulmonary or systemic (in the presence of a patent foramen ovale) circulations, arrhythmias, cardiac perforation, etc. Little is known on how to approach the diagnosis and subsequent management of PH in patients with abnormal structures in the right side of the heart. In this review, we present two challenging cases, examine the literature, and make recommendations on how to approach the diagnosis of PH in patients with right heart lesions.
Case presentation
Case 1
A 64-year-old woman presented with dyspnea on exertion and clinical suspicion of PH. She had been anticoagulated with apixaban given her recurrent lower extremity deep vein thrombosis in the setting of factor V Leiden mutation and protein S deficiency. An echocardiogram showed elevated right ventricular systolic pressure (RVSP) and severe dysfunction of the right ventricle. Unexpectedly, a pedunculated structure was found in the right ventricle. CT angiography (CTA) showed no filling defects in the pulmonary arteries and ventilation perfusion scan revealed normal pulmonary perfusion. Given concerns for an intraventricular clot, a RHC was not done; however, since the presumption of having idiopathic PAH was high, she was treated with PAH-specific therapies (riociguat 2.5 mg thrice daily and macitentan 10 mg daily). Three months after receiving apixaban, a transthoracic echocardiogram showed no variations in the size of the right ventricular mass (Fig 1a). CMRI described an ill-defined mass of 1.6 × 0.7 cm attached to a 4.0 × 0.6 cm stalk that originated from the right ventricular free wall. The right ventricle was hypertrophic with systolic and diastolic septal flattening and presented an ejection fraction of 31% (Fig. 1b). The cardiac lesion was attributed to a papillary fibroelastoma and RHC was performed. RHC revealed a mPAP of 52 mmHg, PAWP of 20 mmHg, and pulmonary vascular resistance (PVR) of 7.1 Wood units (WU). No complications occurred with anticoagulation or during RHC. Treatment with intravenous epoprostenol was subsequently initiated.
Case 2
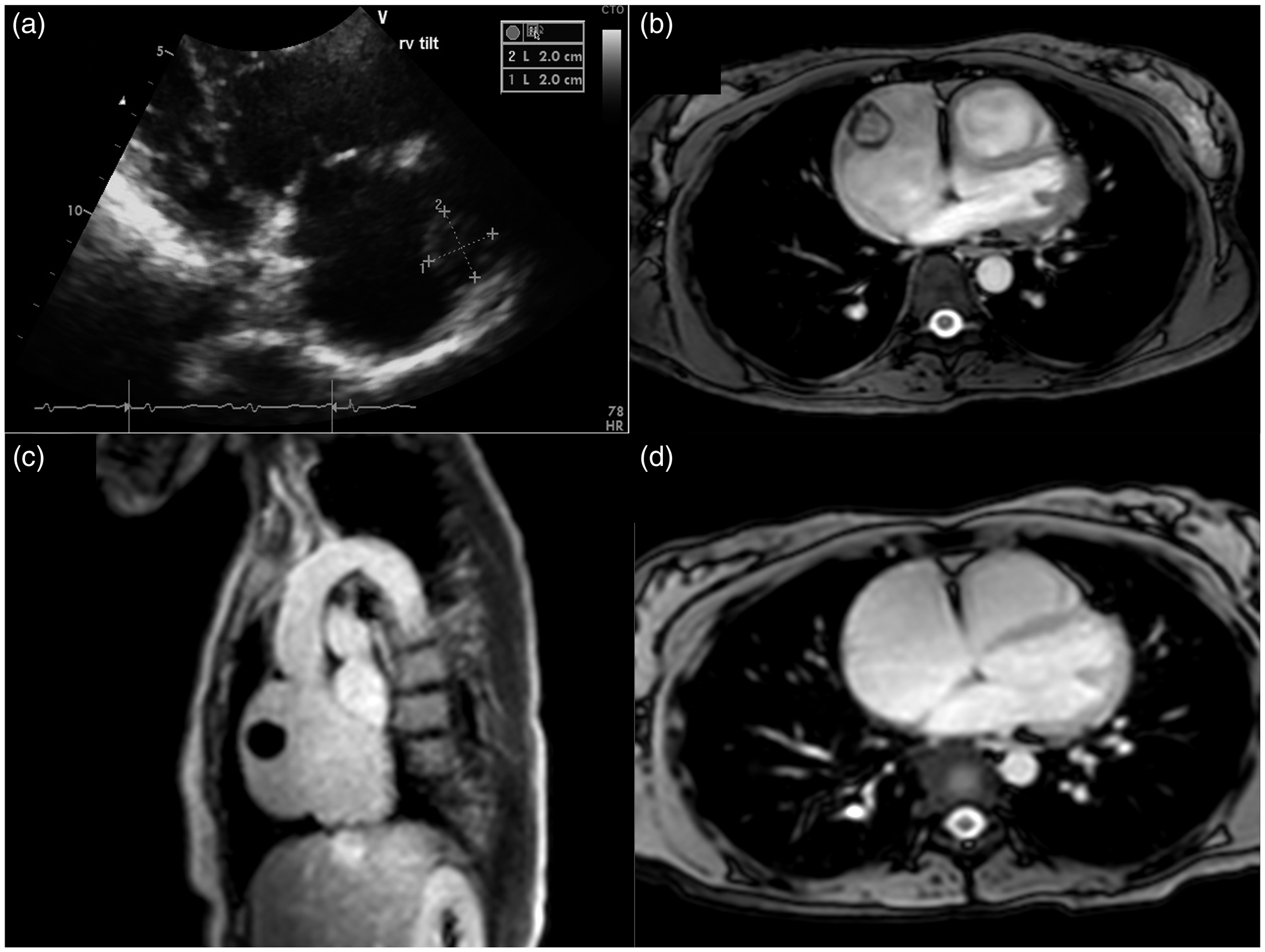
A 48-year-old woman presented with progressive dyspnea and echocardiographic findings suggestive of PH, including an elevated RVSP at 86 mmHg with severe dysfunction of the right ventricle. As an incidental finding she had a rounded fixed mass in the dome of the right atrium (Fig. 2a). CMRI revealed right ventricular hypertrophy with an ejection fraction of 29% and a 2.4 × 2.3 × 2.5 cm mass attached to the superior aspect of the anterolateral right atrial wall (Fig. 2b–c) with features consistent with a thrombus. Since a RHC could not be done, she received anticoagulation with warfarin and was started on PAH-specific therapies, i.e. tadalafil 40 mg and ambrisentan 10 mg daily. The international normalized ratio goal was between 2 and 3. No bleeding complications were observed during treatment. At three-month a follow-up CMRI showed a complete resolution of the right atrial mass (Fig. 2d). The right ventricle remained dilated with an ejection fraction of 31%. A subsequent RHC confirmed the presence of PAH with a mPAP of 38 mmHg, PAWP of 10 mmHg, cardiac index of 2.61 L/min/m2, and PVR of 7 WU. No complications occurred during or immediately after RHC. Subcutaneous treprostinil was added to her PH treatment.
Echocardiogram and CMR at 3-month. (a) A right ventricular mass (1.54 cm in length and 0.9 cm wide). (b) A pedunculated lesion in the right ventricle. Echocardiogram and CMR at baseline and CMR at three months. (a) Echocardiogram reveals a 2 cm mass in the right atrium. (b) Axial CMR section shows a circular structure in the right atrium. (c) Sagittal CMR view depicts a circular mass in the right atrium. (d) Coronal CMR section in a three-month follow-up study shows resolution of the right atrial mass.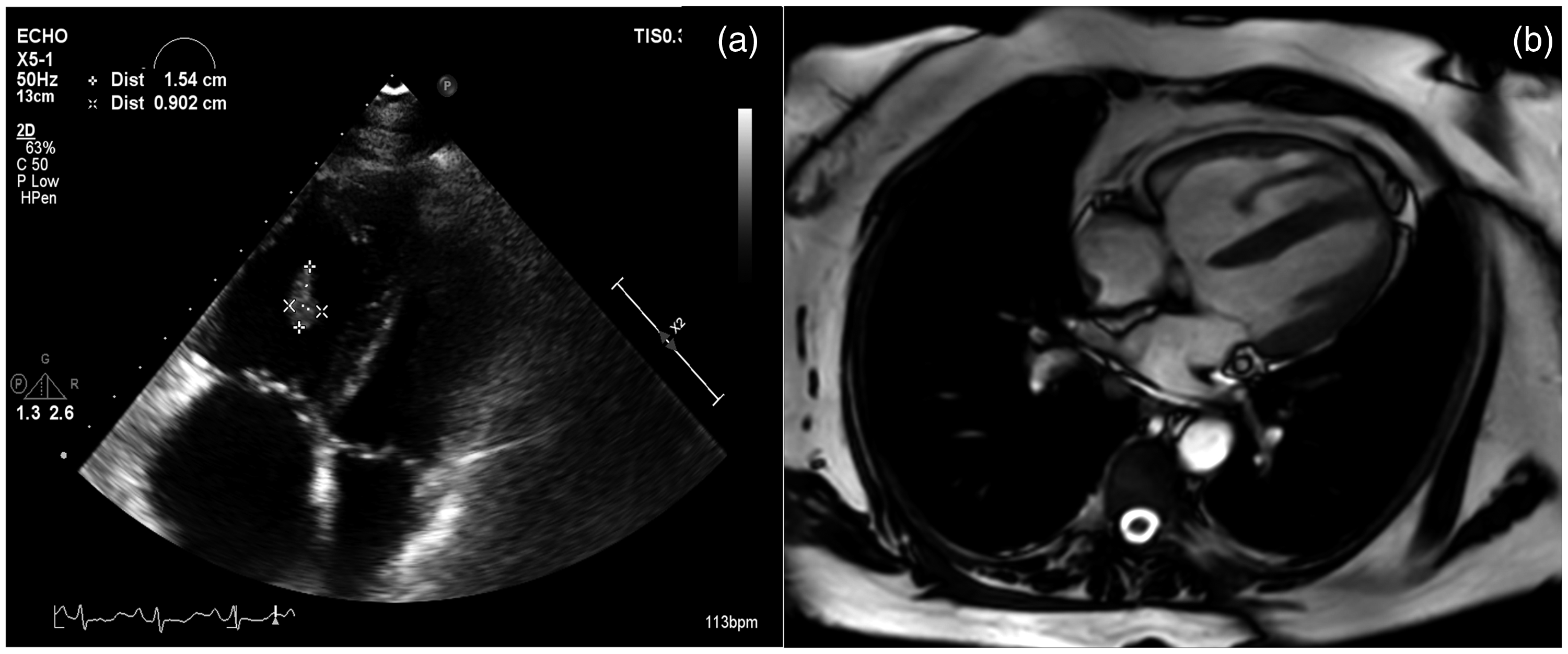
Abnormal right heart structures
Right cardiac lesions.
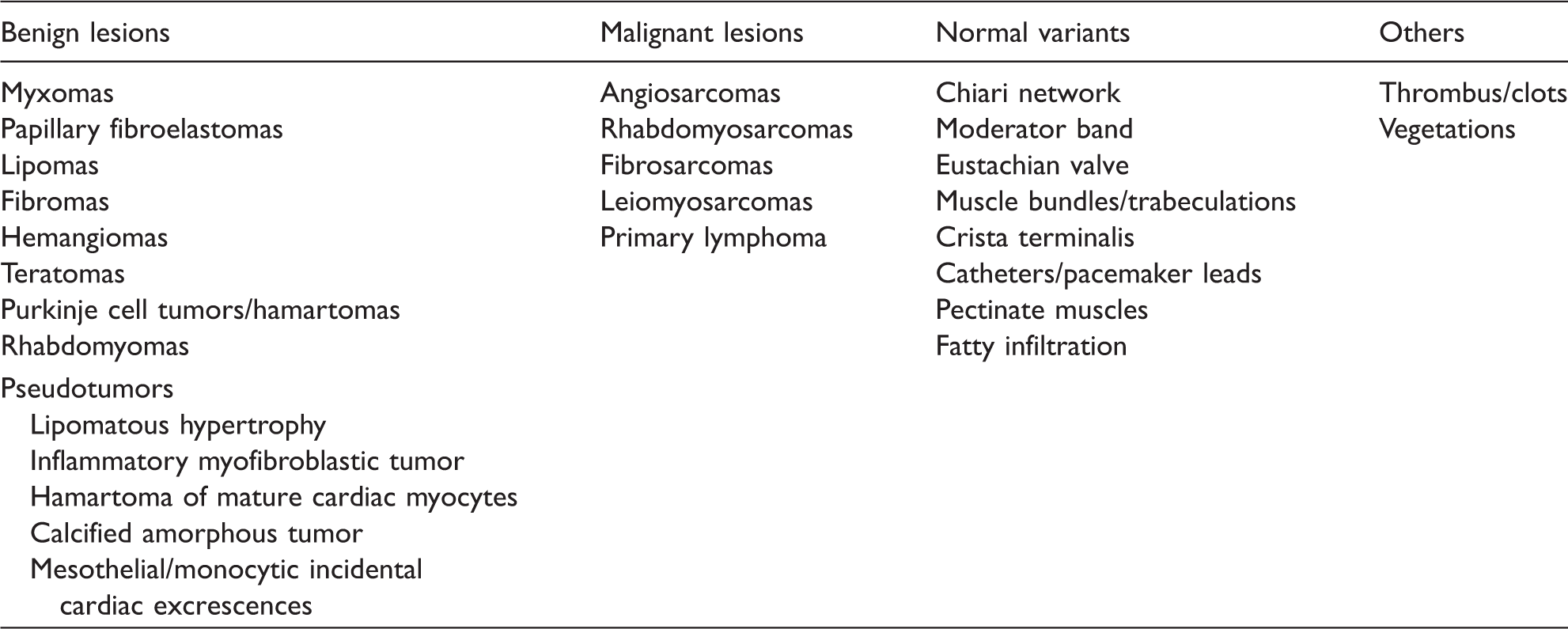
Right heart thrombus can be seen in the context of acute pulmonary embolism, 5 atrial fibrillation, wall motion abnormalities, or in association with pacemaker leads or prosthetic valves. Most cases of infectious endocarditis of the tricuspid or pulmonic valve are seen in injection drug users. 6 More than 90% of cardiac tumors are benign. 7 Malignant lesions could originate in the heart but are more commonly metastasis of extracardiac neoplasms. 7 It is imperative to recognize these conditions given that their treatments are markedly different.
Cardiac thrombus could appear in patients with hypercoagulable disorders, acute pulmonary embolism, 5 blood stagnation (myocardial wall motion abnormalities and atrial fibrillation), 8 or foreign bodies such as indwelling catheters, 9 pacemaker leads, prosthetic valves, and atrial or ventricular septal closure devices. 10 A right atrial thrombus has been observed in about 10% of patients with pulmonary thromboembolism 11 and in 7% of all autopsies (n = 23,796). 11 The right atrium is an unusual site for thrombus formation, even in patients with atrial fibrillation; nevertheless, thrombi could form in enlarged right atria with lower appendage emptying velocities.12,13 Patients with right atrial thrombus are treated with anticoagulants, thrombolytic agents, or surgical thrombectomy.14–16
Endocarditis of the right heart presents with vegetations in the tricuspid and/or pulmonic valves. Valvular endocarditis can have an infectious or non-infectious origin. Non-infectious endocarditis can occur in patients with an underlying malignancy or systemic inflammatory illness (e.g. systemic lupus erythematosus, rheumatoid arthritis). 17 Endocarditis of the right heart valves comprise 5–10% of all cases of infectious endocarditis 18 and is generally due to Staphylococcus aureus infection. 19 Risk factors include intravenous drug use, presence of foreign material, underlying heart disease, invasive dental, medical, or surgical procedures, and prior episodes of endocarditis. 19 Pulmonary events occur in 80% of these patients, including septic emboli and cavitation. 20
Cardiac myxomas are the most common benign cardiac neoplasms. The majority of cardiac myxomas (∼80%) originate in the left atrium while most of the remainder come from the right atrium.21–24 Myxomas are the most common cardiac tumors seen in the right atrium. 25 Patients with myxomas tend to have constitutional symptoms 26 and findings suggestive of tricuspid stenosis or recurrent embolism. Papillary fibroelastomas are the second most common benign cardiac tumors in adults. 27 About one-third of these patients are asymptomatic, while the rest may present with pulmonary embolism, syncope, myocardial infarction, or sudden death.27,28
Malignant cardiac tumors are significantly less common and can be either primary (originating in the heart) or secondary (originating in extracardiac structures). Primary cardiac tumors include cardiac sarcomas 29 and lymphomas. 30 Secondary tumors are significantly more common and derive from hematogenous metastases, direct invasion from nearby structures (e.g. bronchogenic carcinoma of the lung, breast cancer, lymphoma, melanoma, mesothelioma, and osteoscarcoma), 31 or intravascular extension across the superior or inferior vena cava into the right atrium (e.g. renal cell carcinoma 31 ).32,33
Clinical presentation and impact of abnormal right heart structures
In general, patients with right-sided cardiac lesions do not have characteristic clinical symptoms. One can suspect this diagnosis in patients with recurrent acute pulmonary embolism, caused by fragments dislodging from the right heart mass.34–37 A right atrial mass that extends to and obstructs the superior vena cava will present with superior vena cava syndrome which is characterized by edema of the face and arms, jugular vein distention, and facial cyanosis. If the atrial mass extends into the inferior vena cava it may cause Budd–Chiari syndrome, characterized by right upper quadrant pain, hepatomegaly, ascites, and acute liver failure. 38 If the right atrial mass causes a reduction of the right ventricular inflow area, the right atrial pressure may increase, leading to venous congestion. 22 In the presence of a patent foramen ovale, a higher right atrial pressure may expose a right-to-left shunt potentially leading to hypoxemia.39–41 Abnormal structures in the right ventricle can embolize to the pulmonary artery or interfere with the filling or outflow of the right ventricle, resulting in venous congestion or reduction in the cardiac output, respectively. 25 Cardiac lesions may disturb the electrical automaticity and conduction of the heart, leading to arrhythmias and/or heart block. Another potential complication of abnormal heart structures is pericardial effusion or myocardial rupture which might lead to pericardial tamponade.42,43
Use of imaging modalities in the diagnosis of cardiac masses
There is no imaging technique that can precisely identify whether a cardiac structure is a thrombus, tumor (benign or malignant), or normal variant/artifact. Nevertheless, echocardiography or CMRI provide important clues that can help secure the correct diagnosis (Fig. 3). Abnormal right heart structures are generally noted during echocardiography.
44
The echocardiographic examination provides essential information on the anatomic location and extent of the mass, severity of valve regurgitation or stenosis, presence of chamber obliteration, pericardial effusion, or cardiac tamponade.
45
Transthoracic echocardiography may not detect all thrombi or might show normal structures that resemble thrombi.
46
For instance, small non-protruding mural thrombi may resemble normal or mildly thickened cardiac wall; on the other hand, normal structures such as papillary muscles, anomalous bands, or trabeculae may mimic thrombi. These misdiagnoses are routed on certain echocardiographic limitations such as poor image quality (e.g. patients with parenchymal lung disease), inability to adequately identify the myocardium-thrombus interface and the limited near-field resolution of the right ventricular apex.
46
Echocardiography continues to be the gold standard imaging for confirming the presence valvular vegetations. Importantly, ample expertise in echocardiographic acquisition and interpretation is essential to identify and adequately characterize abnormal right heart structures.
45
Imaging approach to cardiac masses. CMR, cardiac magnetic resonance; TEE, transesophageal echocardiography.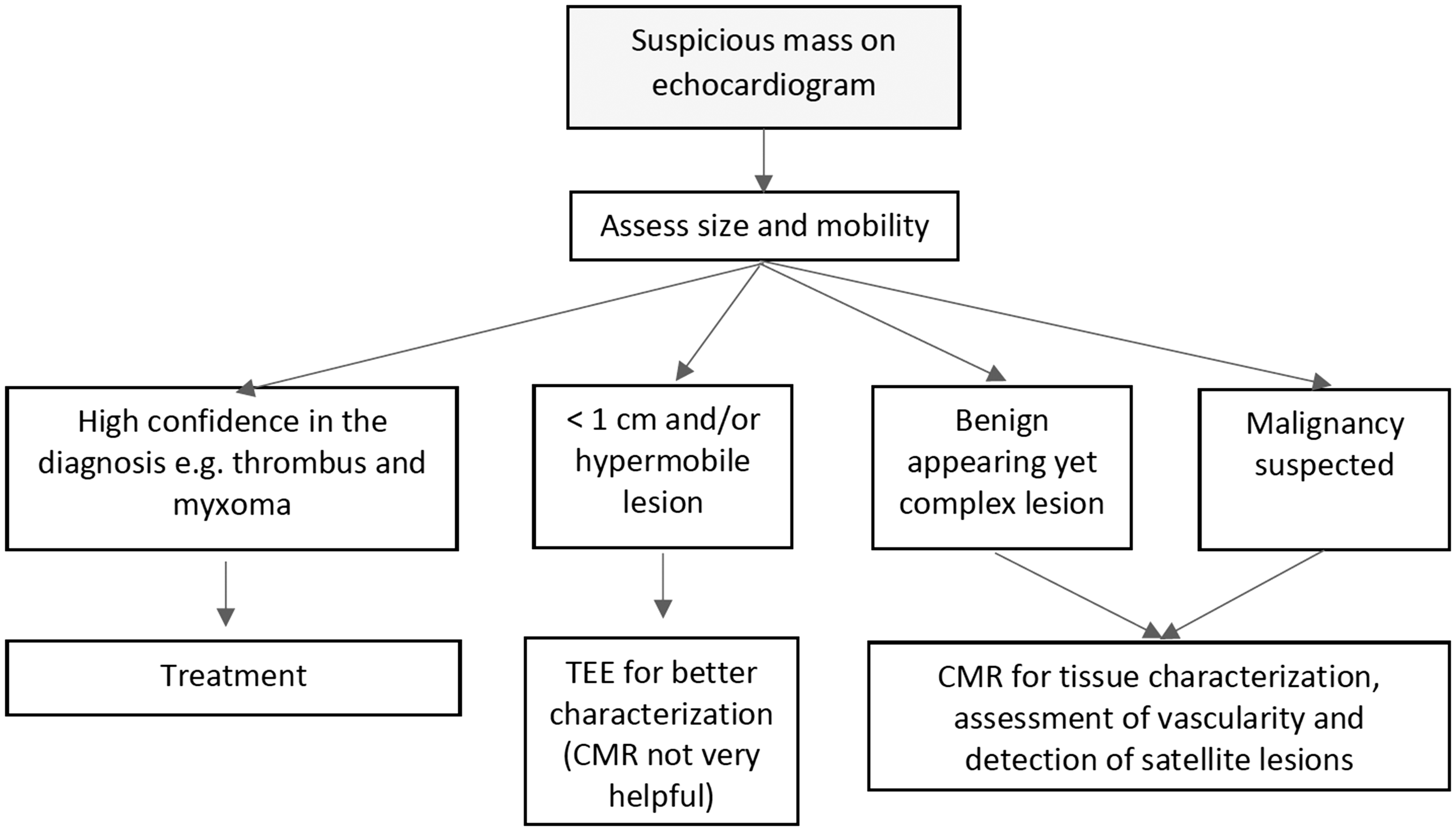
Transesophageal echocardiography has been shown to be superior to transthoracic echocardiography for detection of intracardiac thrombi, since it allows excellent visualization of cardiac chambers.47,48 Transesophageal echocardiography may offer clues that help narrow the differential diagnosis of cardiac lesions. For example, malignant lesions may have intra-myocardial extension and vena cava invasion. 45 Contrast echocardiography and three-dimensional echocardiography may enhance the traditional image acquisition. 49
CMRI is currently the best modality to discriminate among the different etiologies of abnormal right heart structures. 50 A good number of CMRI features help differentiate benign and malignant cardiac masses. 51 Certain characteristics such as mass surface, size, location, T1-weighted and T2-weighted response, presence of hemorrhage or calcifications, valvular involvement, and pericardial effusion provide diagnostic clues. 52 For instance, malignant cardiac tumors are usually larger, 53 have ill-defined and invasive borders52,54 and broad-based myocardial insertion, 53 invading the myocardium, pericardium, and extracardiac structures.
In fact, the presence of pericardial effusion in the setting of a cardiac mass increases the suspicion of malignancy. 53 Large foci of calcification are a characteristic hallmark of cardiac osteosarcomas, whereas small areas of calcification can be seen in cardiac fibromas. 53 Tumors arising from the right atrial wall should be considered suspicious for malignancy since this location is uncommon for benign tumors. 52 Lobulated masses that appear distinct from the myocardial wall, in the presence of severe wall motion abnormality, strongly suggest a thrombus rather than a tumor. 55 Thrombi are avascular and therefore show no contrast uptake; therefore the CMRI shows thrombi as a low-signal-intensity filling defect in the heart cavities. 56 Delayed enhanced sequences on CMRI are useful for detection myocardial scars, which are a risk factor for thrombosis.46,57 Gultai et al. concluded that CMRI performed better than a combination of transthoracic and transesophageal echocardiography in detecting intracardiac masses. The authors reported that CMRI is better suited for determining the cardiac mass etiology, extent, and relationship to adjacent structures, as well as the hemodynamic impact of the lesion. 58
Diagnosis of PH in patients with right heart abnormalities
The diagnosis of PH requires a RHC. Conditions in which RHC could be avoided include left heart and lung diseases (PH groups 2 and 3) without signs of severe PH or right ventricular dysfunction. 59 In the rest, RHC is essential to determine whether patients have pre- and/or post-capillary PH, assess the hemodynamic severity, and determine pulmonary vasoreactivity (e.g. vasodilatory challenge with nitric oxide). These data are essential, not only for diagnosis and hemodynamic characterization of the disease, but also for prognostication and treatment.
Unfortunately, in the presence of abnormal right cardiac structures, a RHC is largely contraindicated given potential complications. For instance, during RHC a thrombus, vegetation or piece of a tumor may dislodge and embolize to the lung or cause paradoxical embolism in the presence of patent foramen ovale with right-to-left flow. Other potential complications in this particular context include arrhythmias and cardiac perforation with or without tamponade.
The prevalence of abnormal right-sided lesions in patients suspected of having PH remains unknown. It is certainly possible that since PH patients tend to develop right ventricular hypertrophy, certain normal variants, i.e. muscle bundles or trabeculations, may appear more prominent and be wrongly characterized as a clot or other abnormal structures. In this setting, one needs to use the best clinical judgment to decide whether a particular patient is likely to have PH and employ complementary tools to determine the most likely etiology and establish disease severity and need for treatment. Helpful tools include New York Heart Association (NYHA) FC, history of syncope, clinical signs of right heart failure, rate of disease progression, 6-min walk distance, serum NT-pro brain natriuretic peptide, and certain echocardiographic determinations such as right atrial area, right ventricular function, and presence of pericardial effusion. If there is a high clinical probability of PAH, particularly when the risk of complications during RHC are moderate to high, PAH-specific treatments should be initiated as soon as possible, since delays may lead to worse outcomes. 60 The type of PAH-specific treatment is selected based on the FC and severity of disease. 61
There are possible alternatives to obtain pulmonary pressures, besides the traditional RHC. For instance, one could measure the left ventricular end-diastolic pressure, to assess whether the post-capillary component could explain their suspected PH. A novel approach was proposed by Suliman et al. 62 who measured the pulmonary pressures in a patient with a mechanical tricuspid valve by trans-septal puncture and retrograde catheterization with occlusion of a pulmonary vein, akin to a reverse wedge. This procedure allowed the measurement of the transpulmonary gradient (mPAP-left atrial pressure).
The best approach for diagnosis PH in the setting of an abnormal right heart structure is unknown. We proposed the algorithm presented in Fig. 4. It is essential that experienced physicians assess the clinical, echocardiographic, and CMRI characteristics of the cardiac mass to narrow the differential diagnosis. If the probability of a cardiac thrombus is high, one could consider anticoagulation for three months and then reassess. If the mass persists, further evaluation is needed to rule out other etiologies. Treatment of infectious endocarditis include adequate antibiotic therapy based on the bacterial isolation and antimicrobial susceptibility. Management of a certain benign tumors, i.e. myxoma, and malignant tumors may require biopsy and potentially surgery; which carry increased risks in patients with underlying PH. RHC could be performed when the clot or vegetations resolve. In cases of cardiac tumors, risk/benefits of RCH and alternative approaches need to be discussed with the patient.
Algorithm on to approach the diagnosis of PH in patients with a right cardiac mass. CMR, cardiac magnetic resonance; RA, right atrium; RHC, right heart catheterization; RV, right ventricle; TEE, transesophageal echocardiography; TTE, transthoracic echocardiography.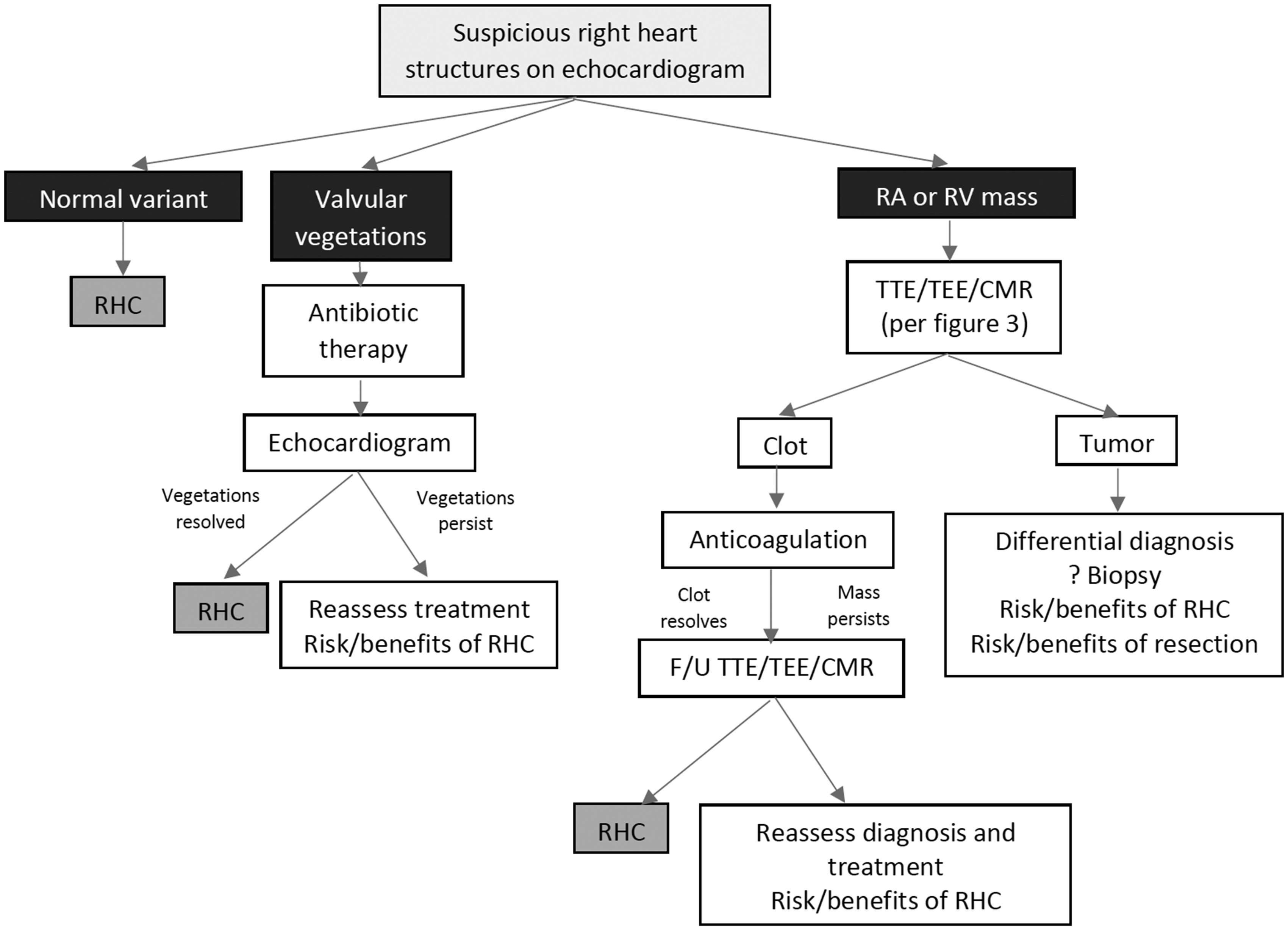
Conclusion
The presence of abnormal right heart structures presents a diagnostic challenge in PH, since RHC, the required diagnostic procedure, is contraindicated in this context. In these cases, it is essential to carefully evaluate and closely follow the right heart mass to identify the most likely etiology and assess response to treatments. In the appropriate patients, PAH-specific treatments may need to be initiated based on clinical suspicion and results of complementary testing. When safe, a RHC needs to be performed to confirm the diagnosis and determine hemodynamic severity.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
MYD is supported by the Haslam Family Endowed Chair in Cardiovascular Medicine. ART is supported by NIH grant no. R01HL130307.
