Abstract
The biological mechanisms behind the development of pulmonary hypertension in the setting of left heart failure (HF-PH), including combined pre- and post-capillary pulmonary hypertension (Cpc-PH), remains unclear. This study aimed to use candidate polymorphisms in nitric oxide synthase (NOS) genes to explore the role of NOS in HF-PH. DNA samples from 118 patients with HF-PH were genotyped for the NOS3 rs1799983 and NOS2 rs3730017 polymorphisms. A multiple regression model was used to compare hemodynamic measurements between genotype groups. Patients with the T/T genotype at rs1799983 possessed a nearly 10 mmHg increased transpulmonary gradient (TPG) compared to those with other genotypes (P = 0.006). This finding was replicated in an independent cohort of 94 HF-PH patients (P = 0.005). However, when tested in a cohort of 162 pre-capillary pulmonary arterial hypertension patients, no association was observed. In a combined analysis of both HF-PH cohorts, mean pulmonary artery pressure (mPAP), diastolic pulmonary gradient (DPG), and CpcPH status were also associated with rs1799983 genotype (P = 0.005, P = 0.03, and P = 0.02, respectively). In patients with HF-PH, the NOS3 rs1799983 polymorphism is associated with TPG, and potentially mPAP and DPG as well. These findings suggest that endothelial NOS (encoded by NOS3) may be involved in the pulmonary vascular remodeling observed in Cpc-PH and warrants further study.
Pulmonary hypertension (PH) is estimated to occur in 50–80% of patients with left heart failure (HF).1,2 PH stemming from left heart disease, or World Health Organization (WHO) Group 2 PH, is defined as a mean pulmonary artery pressure (mPAP) ≥25 mmHg and a pulmonary artery wedge pressure (PAWP) ≥15 mmHg. The underlying mechanism for development of Group 2 PH is thought to be related to pulmonary venous hypertension – passive increases in pulmonary venous pressures transmitted from increased left atrial pressure. However, a significant number of patients have a mPAP disproportionate to that expected from their left HF, known as combined pre- and post-capillary PH (Cpc-PH). The addition of PH to a diagnosis of left heart failure (HF-PH) confers an increased risk of mortality, but whether Cpc-PH confers additional mortality risk is controversial.3–6
The development of Cpc-PH (or reactive PH), indicated by diastolic pulmonary gradient (DPG) ≥7 mmHg (or transpulmonary gradient [TPG] ≥12 mmHg), is associated with pulmonary vascular remodeling, likely from chronic pressure-induced injury. The biological mechanisms behind this remodeling are unclear, but are thought to include pulmonary vasoconstriction, fibrosis, smooth muscle cell proliferation, and endothelial dysfunction.7–9 Central to endothelial dysfunction in PH is an imbalance of the vasodilator nitric oxide (NO) and the vasoconstrictor endothelin-1. 10 In addition to its vasodilatory properties, NO also has antiproliferative and antioxidant effects, and decreases platelet aggregation.11,12 Thus, a decreased NO availability can affect multiple biological pathways in PH.
Our hypothesis was that genetic polymorphisms affecting baseline nitric oxide synthase (NOS) enzyme function would lead to increased PH severity in patients with left HF. The aim of this study was to use candidate polymorphisms in NOS genes to explore the role of NOS in PH severity associated with left HF.
Methods
Patient populations
All patients provided written, informed consent for participation and recruitment protocols were approved by the institutional review boards at each respective institution. The discovery cohort included heart failure with preserved ejection fraction (HFpEF) and heart failure with reduced ejection fraction (HFrEF) patients recruited from cardiology and pulmonology clinics at the University of Illinois at Chicago (UIC). HF-PH was confirmed in individuals by a documented mPAP ≥25 mmHg and PAWP ≥15 mmHg as measured by right heart catheterization (RHC), without echocardiographic evidence of moderate or severe valvular disease. To permit the potential replication of associations found in the discovery cohort, an independent HF-PH validation population was also included. This cohort comprised individuals from Northwestern University and Stanford University using identical criteria for HF-PH. To compare potential associations with other forms of PH, a cohort of patients with pulmonary arterial hypertension (pre-capillary or WHO Group 1 PH) recruited from UIC was also analyzed. Group 1 PH was defined as mPAP ≥25 mmHg and PAWP <15 mmHg as measured by RHC, with no history of pulmonary embolism or diseases that are associated with other forms of PH.
Genotyping
The two candidate enzymes included in this study were inducible nitric oxide synthase (iNOS) and endothelial nitric oxide synthase (eNOS). The single nucleotide polymorphism (SNP) most likely to affect gene expression was chosen for both NOS2 and NOS3, the genes that encode iNOS and eNOS, respectively. For NOS2, the non-synonymous SNP rs3730017 (Arg221Trp) was chosen because in silico analyses using SIFT 13 and Polyphen-214 predict that the variant is damaging to the protein structure. For NOS3, the well-studied non-synonymous SNP rs1799983 was chosen, which has been previously associated with alterations of endogenous nitric oxide (NO) production.15,16
DNA from individuals was isolated from either whole blood (UIC and Stanford) or buccal cells (Northwestern). Genotyping of rs1799983 and rs3730017 was completed using pyrosequencing on the HS96 platform (Qiagen, Valencia, CA, USA).
Statistical analysis
Comparisons of genotype frequencies between HF-PH patients and control HF patients were completed using a Chi-square test. To assess associations between severity of PH and candidate SNPs, mPAP, PAWP, TPG, DPG, cardiac output (CO), and pulmonary vascular resistance (PVR) were compared by genotype in the discovery cohort using a multiple regression model, adjusting for the following covariates: age; sex; race; systolic blood pressure; and diastolic blood pressure. Genotype effects were analyzed using a recessive genetic model (A/A + A/a vs. a/a) for rs1799983 and a dominant model (A/A vs. A/a + a/a) for rs3730017, due to the very small number of homozygous variant subjects. Identical regression analyses were also completed with rs1799983 in the HF-PH validation and PAH cohorts (except for DPG, which was not available), adjusting for the same covariates. Secondarily, a combined analysis (including both discovery and validation HF-PH cohorts) was completed in an attempt to leverage increased statistical power to detect associations with smaller effect sizes, using an identical regression as described above. In addition, the genotype frequencies of rs1799983 and rs3730017 were compared under the same genetic models as above in patients with and without CpcPH (defined as TPG ≥ 12 mmHg or DPG ≥ 7 mmHg), as well as between patients with HFpEF (EF ≥ 50%) and HFrEF (EF < 50%) using a two-sample unpaired t-test. A sensitivity analysis was also completed with rs1799983 in the combined cohort to test the same associations by race (patients of African descent and patients of European/Latino descent). All statistical analyses were performed using R statistical software version 3.4.0 (R Foundation for Statistical Computing).
Results
Characteristics of patient cohorts.
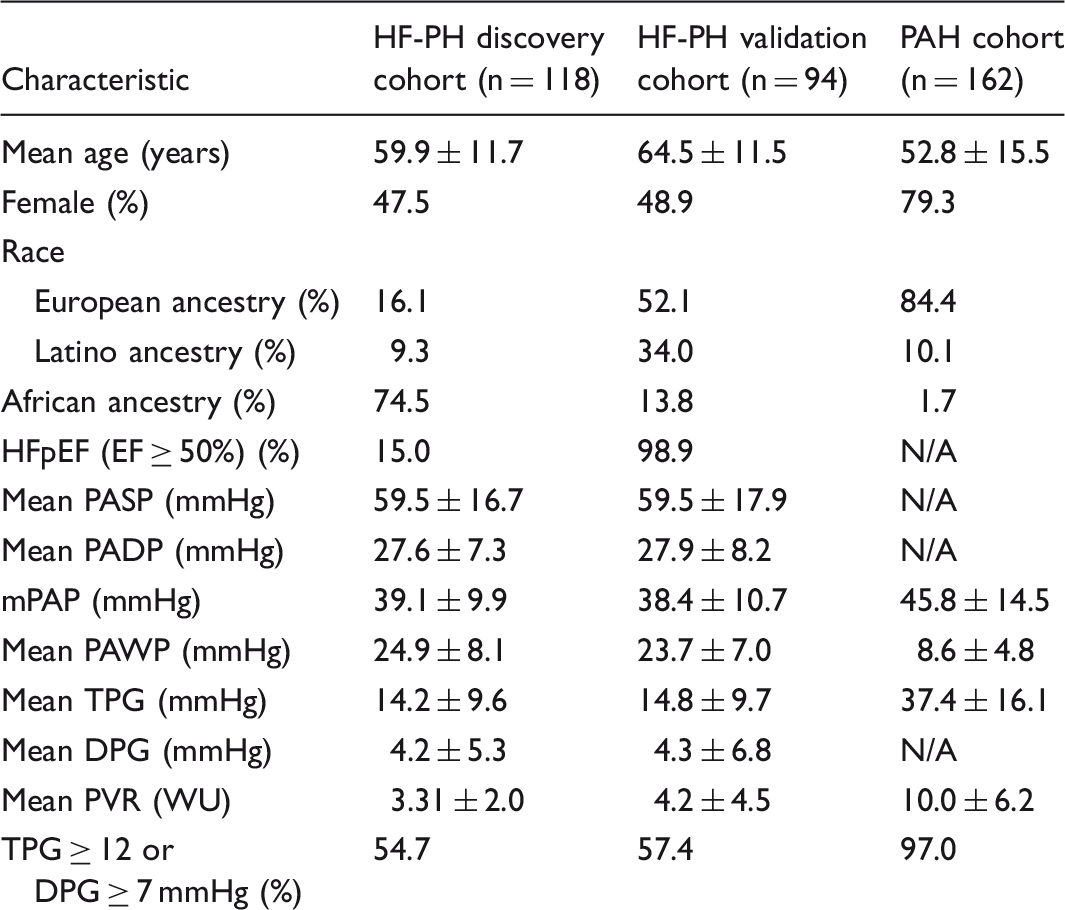
All values are presented as mean ± SD unless otherwise stated.
HFpEF, heart failure with preserved ejection fraction; PASP, pulmonary artery systolic pressure; PADP, pulmonary artery diastolic pressure; mPAP, mean pulmonary artery pressure; PAWP, pulmonary artery wedge pressure; PVR, pulmonary vascular resistance; TPG, transpulmonary gradient; DPG, diastolic pressure gradient; N/A, not available.
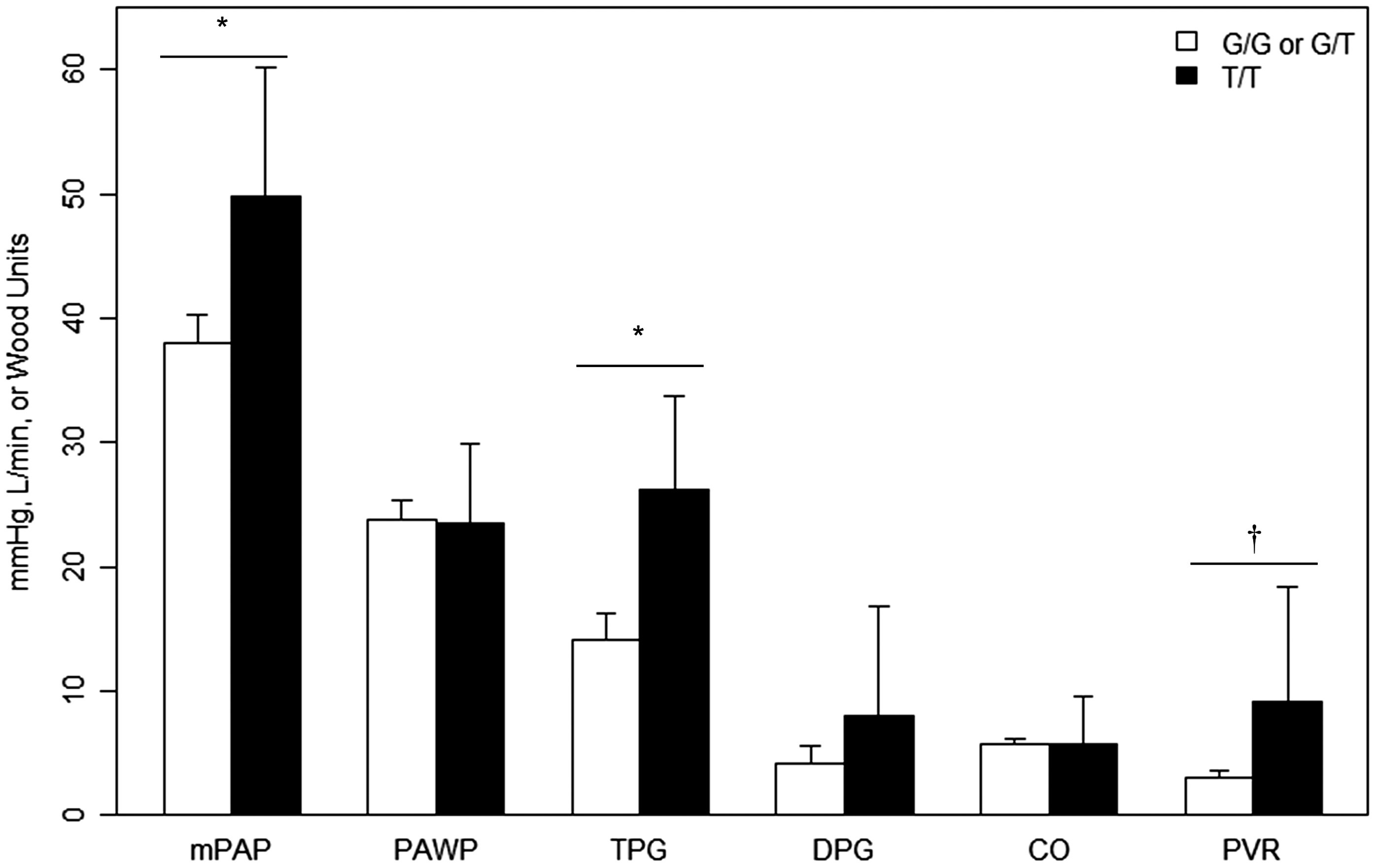
In the discovery cohort, patients with the variant T/T genotype at rs1799983 had a mean TPG that was 9.5 mmHg greater than patients with other genotypes (Padj = 0.0059; Fig. 1a). This association between TPG and rs1799983 was also observed in the validation cohort, with T homozygotes possessing a mean TPG that was 12 mmHg higher (Padj = 0.0047; Fig. 2). There was also a trend toward increased mean DPG values (3.5 mmHg higher) in the discovery cohort (Padj = 0.082), but no association in the validation cohort (4.3 mmHg higher, Padj = 0.29; Figs. 1a and 2). In the validation cohort, T/T genotype was associated with a 12.2 mmHg higher mPAP (Padj = 0.026) and a 5.9 Wood Unit (WU) increase in mean PVR (P = 0.0008; Fig. 2). No associations were observed in either cohort between rs1799983 and PAWP or CO (Figs. 1a and 2). Likewise, we found no associations between rs3730017 and any of the analyzed hemodynamic measures (Fig. 1b).
Mean hemodynamic measurements of HF-PH discovery cohort by (a) rs1799983 and (b) rs3730017 genotype. *Padj ≤ 0.05. Mean hemodynamic measurements of HF-PH validation cohort by rs1799983 genotype. *Padj ≤ 0.05; †Padj < 0.001.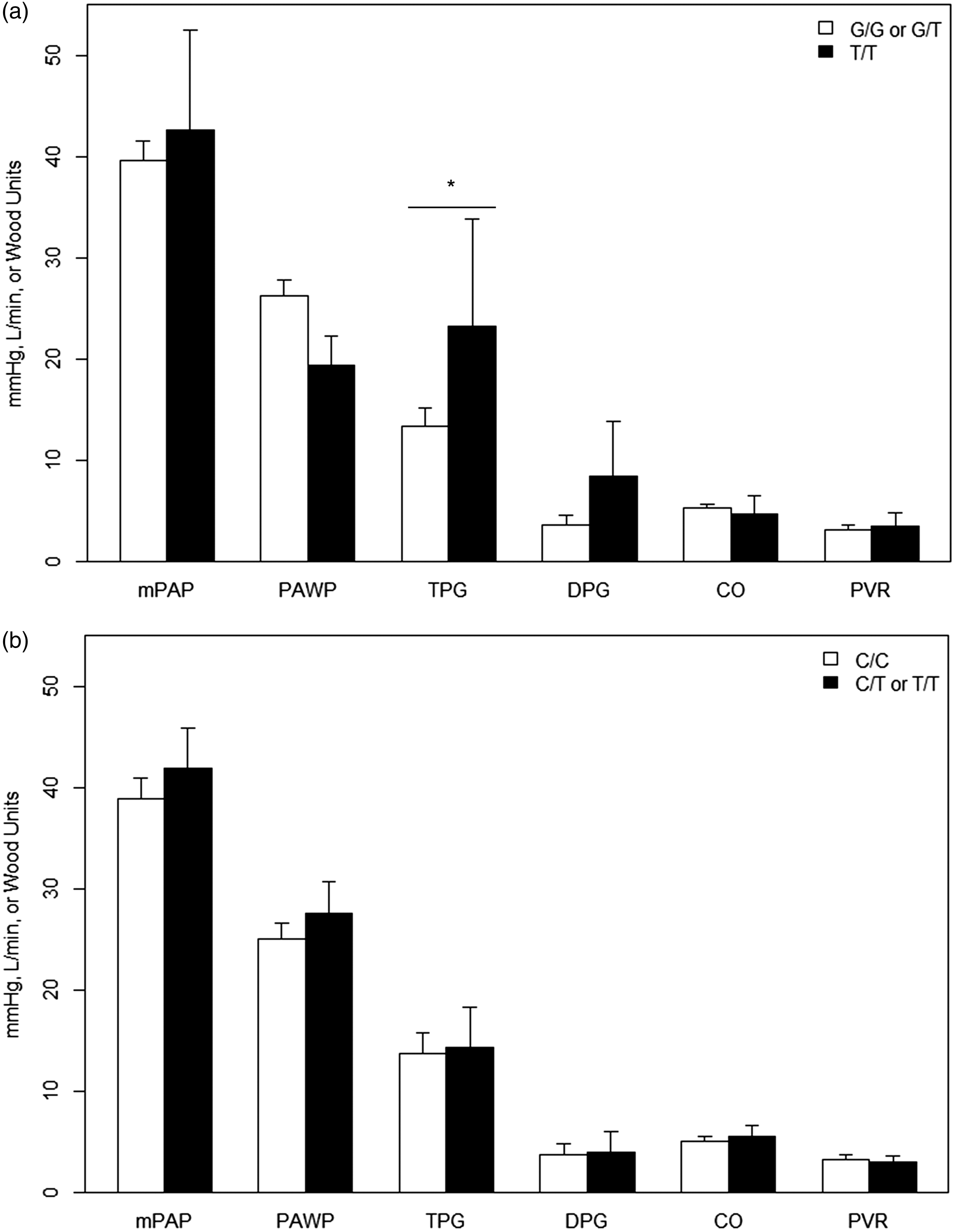
Second, we conducted a combined analysis including both the discovery and validation cohorts. In addition to a much stronger association with TPG (Padj = 3.0 × 10–5), we found that patients with the variant T/T genotype had an average mPAP that was 10.4 mmHg greater (Padj = 0.0054), a mean DPG that was 4.2 mmHg higher (Padj = 0.031), and a mean PVR 3.0 WU higher (Padj = 0.0023) than other genotypes. Again, no associations were observed with PAWP and CO (Fig. 3a). Consistent with previous findings in the individual cohorts, we found no association between rs3730017 and any measured hemodynamic values (Fig. 3b).
Mean hemodynamic measurements of the combined discovery and validation HF-PH cohorts by (a) rs1799983 and (b) rs3730017 genotype. *Padj ≤ 0.05; †Padj < 0.001.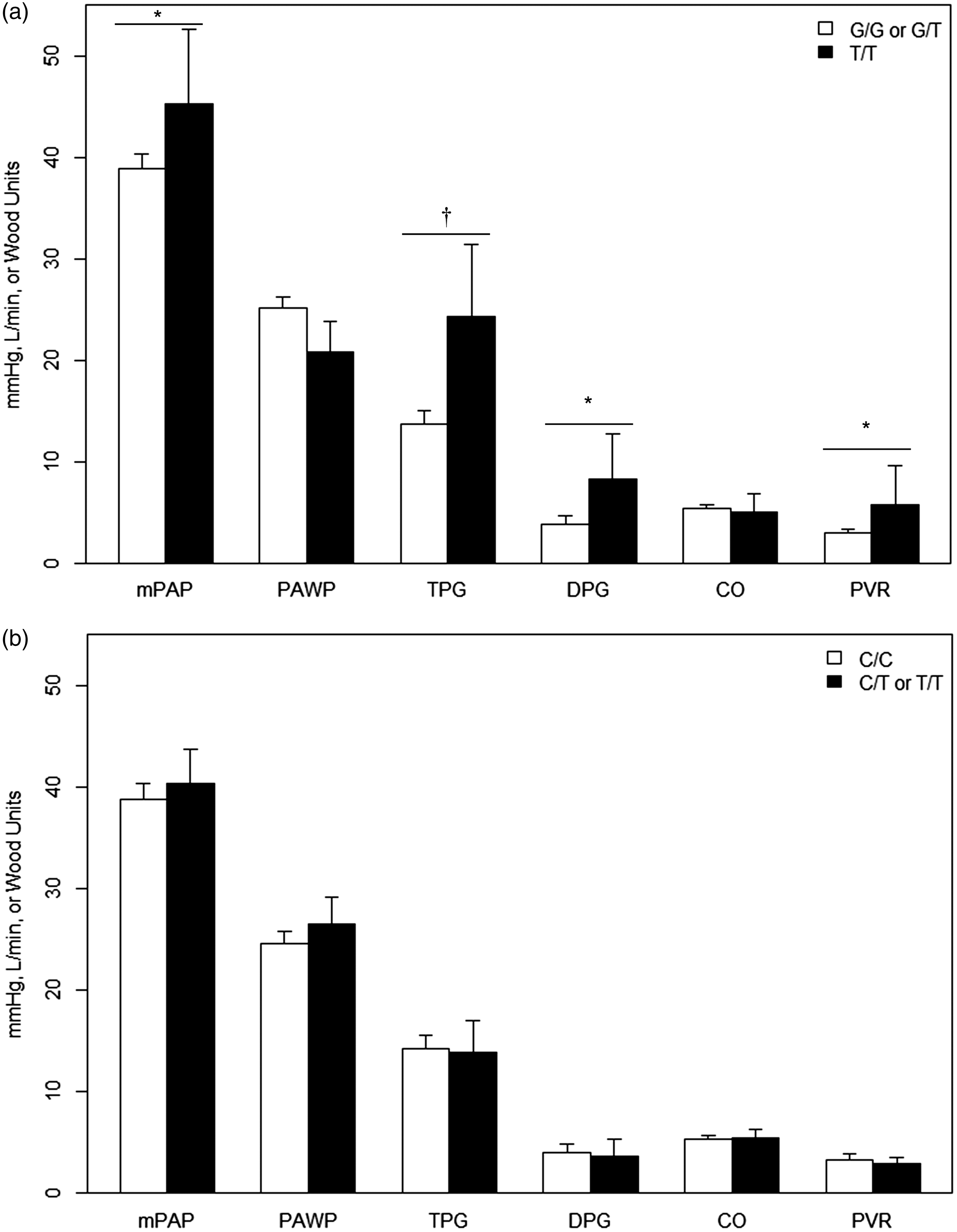
Characteristics of the combined patient cohorts by CpcPH status.
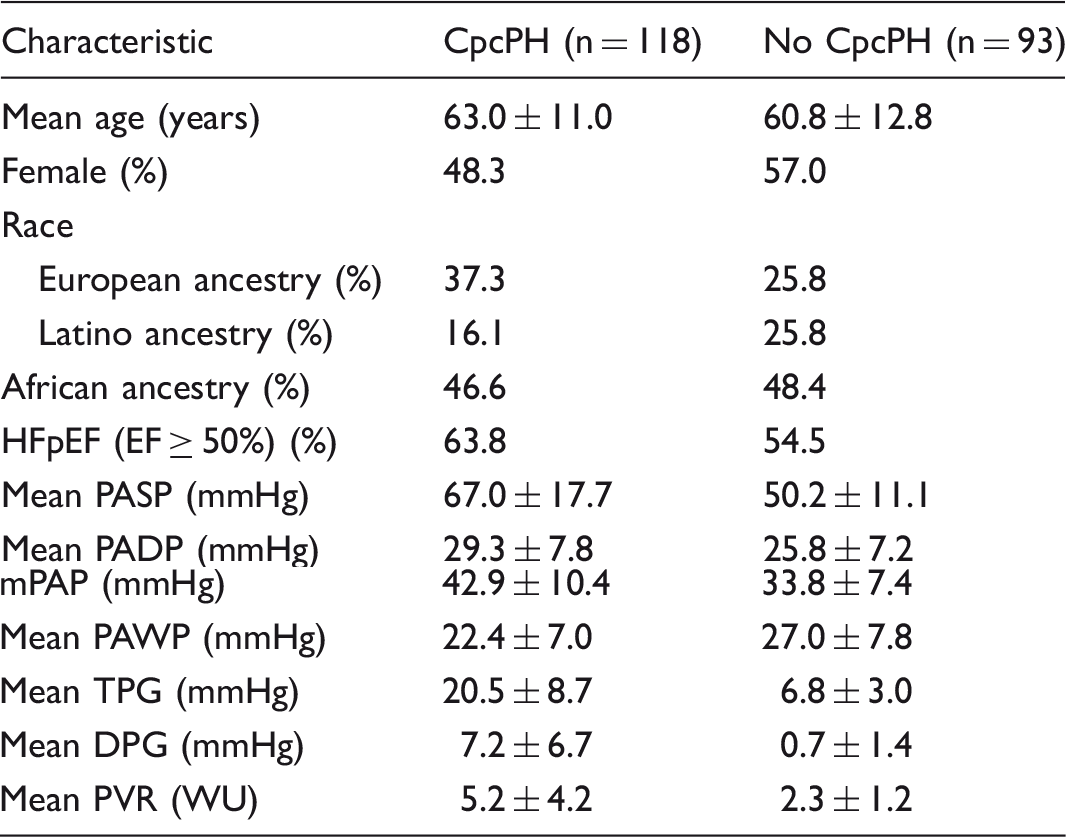
All values are presented as mean ± SD unless otherwise stated.
HFpEF, heart failure with preserved ejection fraction; PASP, pulmonary artery systolic pressure; PADP, pulmonary artery diastolic pressure; mPAP, mean pulmonary artery pressure; PAWP, pulmonary artery wedge pressure; PVR, pulmonary vascular resistance; TPG, transpulmonary gradient; DPG, diastolic pressure gradient; N/A, not available.
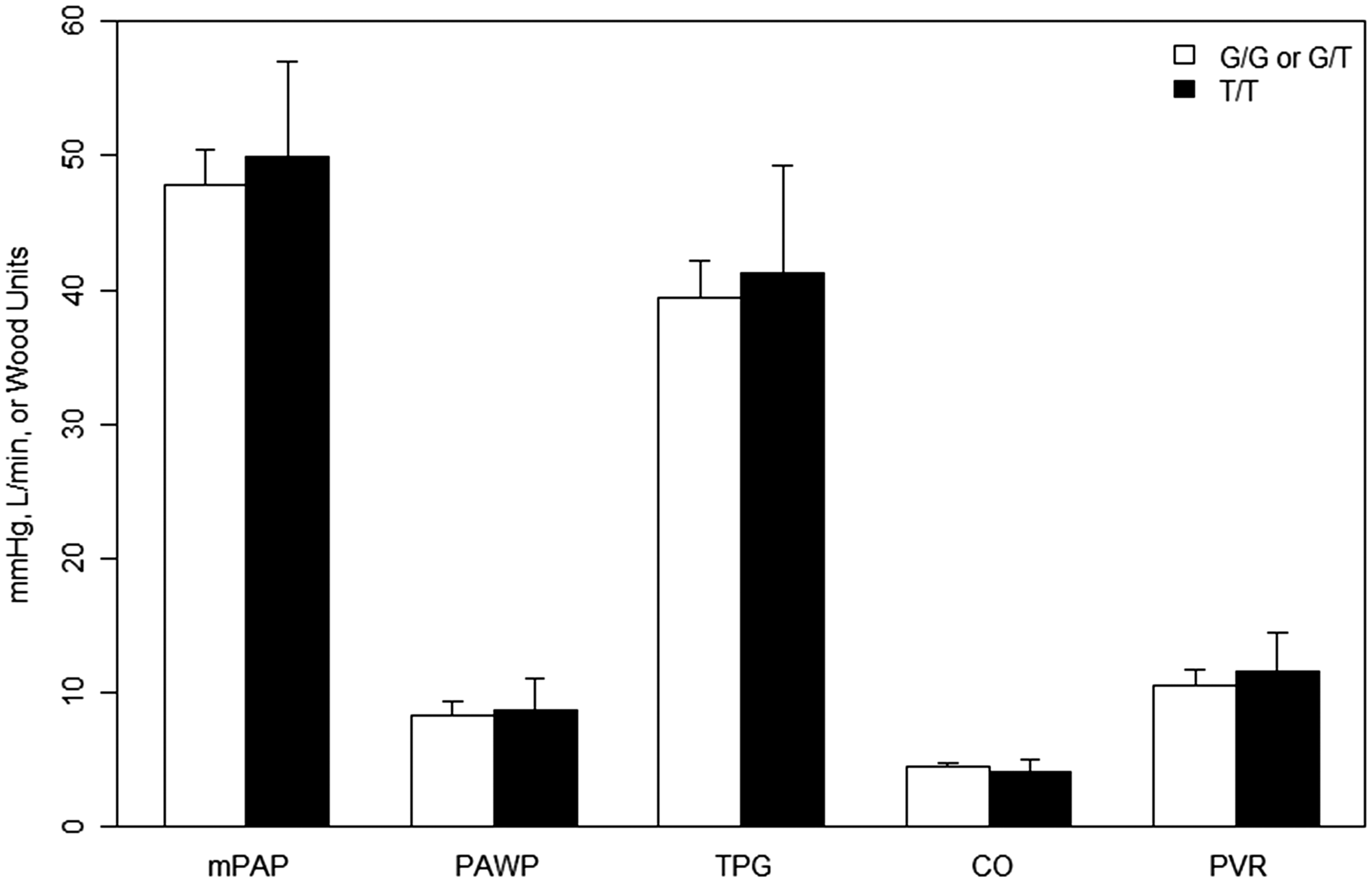
Mean hemodynamic measurements of pulmonary arterial hypertension (Group 1 PH) cohort by rs1799983 genotype. *Padj ≤ 0.05.
As a sensitivity analysis, rs1799983 was analyzed within the combined cohort by race. Associations with TPG (Padj = 5.7 × 10–5), DPG (Padj = 0.0057), and mPAP (Padj = 0.0046) were similar in those of European and Latino ancestry as in the overall combined cohort (Suppl. Fig. S1a). In African Americans, similar associations with TPG were also detected, but only trended toward significance (Padj = 0.077), but PVR remained significantly associated (Padj = 0.001). DPG and mPAP were not significantly associated in the African American subset (Suppl. Fig. S1b).
Discussion
The current study demonstrates that rs1799983, a putatively functional SNP within eNOS, is associated with TPG in HF-PH patients. The association is present in two independent patient cohorts with varying racial compositions. In addition, DPG, PVR, and mPAP may also be associated with rs1799983 in this patient population, based on associations observed in a combined analysis of both HF-PH cohorts. Further, we found that CpcPH patients were more likely to carry the rs1799983 variant T/T genotype. The lack of similar hemodynamic associations in patients with PAH suggests that these associations may be specific to HF-PH. This claim is further supported by previous research indicating that rs1799983 is also not associated with Group 3 PH (PH due to lung disease or hypoxia). 17
Rs1799983 is a non-synonymous SNP in NOS3 (the gene encoding eNOS) that leads to an amino acid substitution (Glu298Asp) and has been associated with decreased baseline production of NO in humans. 15 The aspartate substitution (resulting from the T allele) in eNOS is thought to cause cleavage of the enzyme, likely shortening its half-life, and may also effect the enzyme’s secondary structure. 16 The T allele at this SNP has been previously associated with increased risk of end-stage renal disease, dilated cardiomyopathy, and decreased survival in patients with HFrEF.18–20 While associations between the variant T allele and increased systemic blood pressure have previously been both reported and refuted,21–24 we did not find associations with systemic blood pressure in either cohort. However, to our knowledge, this is the first report of an association with rs1799983 and any type of PH.
Elevated mPAP, PVR, TPG, and DPG have all been associated with decreased survival in patients with left HF.3,4 This is likely because, to some extent, they are all linked to Cpc-PH. While the exact definition of Cpc-PH has evolved over the past few years (from TPG ≥ 12 to DPG ≥ 7 to DPG ≥ 7 and/or PVR > 3 WU) the definitions consistently include measurements that increase definitively with the development of pulmonary vascular remodeling. 7 This remodeling is thought to be the cause of the increased mortality risk some have observed in CpcPH patients.4,25 Because of its previous connection with many other cardiovascular diseases and the established role of NO in vascular remodeling, the observed connection between rs1799983 and markers of worsening vascular remodeling is biologically reasonable.
Vasoconstriction plays a key role in TPG changes within the pulmonary circuit. 7 Thus, it seems plausible that TPG would have one of the strongest association with rs1799983. The increased PVR observed with T/T patients in the combined cohort supports this concept. The potential U-shaped survival curve previously associated with DPG 4 could explain the weaker association we observed with DPG. The significantly larger sample size in the combined cohort likely provided the improved power to detect this weaker association. The fact that PAWP, an estimator of left atrial pressure, was not associated with rs1799983 genotype in any analysis seems to imply that the degree of pulmonary venous hypertension (stemming from left HF) is not affected by eNOS genetic variation. In addition, there was no difference in rs1799983 genotype frequency between HFpEF and HFrEF patients suggesting that the genetic effect is likely not driven by HF subtype or severity. These data further support the interpretation that patients with variation at rs1799983 (and likely with a baseline NO imbalance) may be at greater risk for developing pulmonary vascular remodeling, possibly via increased susceptibility to endothelial dysfunction.
Rs3730017 is a non-synonymous SNP in NOS2 (the gene encoding inducible NOS) that leads to an amino acid substitution (Arg298Trp), calculated in silico by Polyphen-2 (http://genetics.bwh.harvard.edu/pph2/) to be possibly damaging. This SNP is not well-studied, likely because its minor allele frequency (MAF) < 3% in Europeans. However, with a MAF = 20–24% in those of African descent, it was deemed a reasonable candidate to investigate in the predominantly African American discovery cohort. We found no association between rs3730017 and any hemodynamic measurement tested. While these findings indicate that this specific SNP is unlikely to be associated with PH severity in HF, it cannot rule out NOS2 as a potential contributor.
Importantly, associations between rs1799983 and TPG were observed in both a predominantly African American cohort (discovery) and a predominantly European American cohort (validation). It is not surprising that the association was stronger in the European American cohort because the MAF of rs1799983 is much higher those of European descent (MAF = 33–34%) than in those of African descent (MAF = 7–16%; per NCBI: https://www.ncbi.nlm.nih.gov/projects/SNP/snp_ref.cgi?rs=1799983). Although race was adjusted for in all regression analyses, an additional sensitivity analysis was conducted, re-analyzing hemodynamic associations by race to assess potential confounding with rs1799983 associations that could be due to population stratification. In both race groups, the mean TPG, mPAP, and DPG were higher in T/T homozygotes, with the associations strongly significant in European Americans and TPG trending toward significance in African Americans. However, with our observed TPG values, α = 0.05, and MAF = 16%, we would require a sample size of African American individuals slightly larger (n = 108) than were analyzed (n = 101) to achieve a power (1 -β) of 80%. In those of European ancestry (MAF = 33%), a much smaller sample size (n = 35) is needed. Thus, it is likely that we were slightly underpowered to detect an effect in TPG in the African American subset.
This study has several limitations. First, it used a small sample size of patients with multiple types of HF; thus, we are unable to assess whether the described eNOS associations are more prominent in a specific HF subtype. However, little is known regarding the etiology of CpcPH; thus, it is unknown at this point whether HF subtype is a potential confounder in these analyses. However, the fact that this association was detected in both a primarily HFrEF cohort (discovery) and a primarily HFpEF cohort (validation) suggests that this effect is independent of HF subtype. Second, comprehensive drug histories were not available on many of the patients (47%), so the potential confounding from drugs that could affect pulmonary vascular pressure could not be determined. However, from the data available, use of PH-specific therapy (phosphodiesterase-5 inhibitors, prostacyclins, or endothelin-1 receptor antagonists) was low (12%). Next, because these data were from cohorts assessed at a single point in time, we were unable to examine the role of rs1799983 in the development of PH. Further, biological validation by measuring NO generation would strengthen the proposed link between rs1799983 and NO signaling. However, biological samples appropriate for NO estimation were not available in either cohort. Lastly, functional validation of the reported SNP associations has yet to be tested and is necessary to confirm that rs1799983 plays a causative role in PH severity.
Patients with Cpc-PH are known to undergo pulmonary vascular remodeling, including endothelial dysfunction.8,10 Our results suggest that genetic variability affecting the NO signaling pathway, potentially by causing abnormal pulmonary endothelial dysfunction, may influence susceptibility to CpcPH. Further study of NO signaling in HF-PH may provide insight into PH pathogenesis in patients with left HF and could potentially lead to new therapeutic targets for the disease.
Footnotes
Acknowledgments
The authors would thank Shitalben Patel for her genotyping assistance.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research was supported by: NIH/NIGMS K23 GM112014 (JDD), NIH/NHLBI R01 HL141281 (AAD), NIH/NHLBI R01 HL111656, R01 HL127342, and R01 HL133951 (RFM). The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
