Abstract
Nitric oxide (NO) is a ubiquitous gas with free radical groups that is soluble in water, and which is involved in numerous physiological functions including inflammatory and immune responses. However, the role of NO in tumor biology is controversial and misunderstood. NO has been shown to have both anti-cancer and carcinogenic effects, which are dependent on the time, location, and concentration of NO. This duality presents a double challenge to determine the net impact of NO on cancer and to define the therapeutic role of NO-centered anti-cancer strategies. Nevertheless, it is believed that a comprehensive and dynamic understanding of the cascade of molecular and cellular events underlying tumor biology that are affected by NO will allow researchers to exploit the potential anti-tumor properties of drugs that interfere with NO metabolism.
Introduction
Nitric oxide (NO) is a transient endogenous gas that acts as a signal transduction molecule in vivo and as an endothelium-derived relaxing factor. Almost all cells produce NO with the involvement of NO synthases (NOS). 1 Mammalian cells contain three subtypes of NOS encoded by three genes, NOS1, NOS2 and NOS3, which share around 50% homology in their location, regulation, catalysis, and inhibitors. 1 The activity of NOS1 and NOS3 is dependent on Ca2+ concentrations, and they only produce transient NO at low concentrations of Ca2+. In contrast, the NOS2 subtype is inducible and independent of Ca2+ concentrations, so is also known as iNOS. 2 All isoforms require L-arginine as a substrate for the synthesis of NO.
NO characteristics
NO exerts its function via a series of chemical reactions, which depend on the concentration of NO and subtle intracellular and extracellular changes. Small amounts of NO are produced to regulate anti-inflammatory and anti-oxidative responses under normal physiological conditions, while high concentrations of NO within tissues up-regulate the function of iNOS and promote oxidation. 3 NO and O2– interact rapidly to produce cytotoxic nitroperoxide, which is a powerful oxidant in tumorigenesis that triggers lipid peroxidation and DNA cleavage.
Dual effects of NO in tumors and underlying mechanisms
NO regulates tumor-related processes, including angiogenesis, apoptosis, cell cycle progression, invasion, and metastasis. 4 iNOS-derived NO has been associated with many tumors and their progression into metastasis, while iNOS positivity has been consistently reported in human cancers at a variety of sites, including the lungs, breast, oral cavity, esophagus, and colon. NO can directly damage DNA chains by being transformed into toxic substances and inducing mutagenesis, and its oxidative and deaminating effects can inhibit the repair of DNA damage and lead to genetic toxicity. 5 NO can also promote p53 mutagenesis, leading to the loss of its inhibitory effect, and its up-regulation of Bcl-2 expression exerts anti-apoptotic effects. 6
Typical mechanisms of NO-mediated apoptosis include caspase activation, chromatin condensation, and DNA fragmentation. 7 NO can inhibit DNA synthesis by hypoxia or the removal of iron metabolites, and regulate the expression of p53 and other apoptosis-related proteins by activating proteins in the caspase family, which inhibits tumor growth or kills tumor cells via cytotoxicity. Accumulating evidence has indicated that endothelial cell-derived NO mediates the elimination of disseminated tumor cells. For example, it was shown that endothelial cell-derived NO exerts a cytotoxic effect on disseminating tumor cells. 8 A bystander signaling mechanism that depends on reactive oxygen species (ROS)- or iNOS-derived NO has also been determined to induce cell killing effects. 9 Clarifying the role of NO in tumor biology will be helpful in the study of NO-based anti-cancer therapy, resulting in the prevention and treatment of more human cancers.
NO in various human cancers
NO plays diverse roles in various human tumors. 10 Understanding its different actions within tumors at the molecular level will aid the development of NO-based diagnostic or prognostic markers and provide potential strategies for cancer prevention and treatment. Cancer types with high NO production or NOS activity are listed in Table 1.11–19
Cancer types with high NO production or NOS activity.
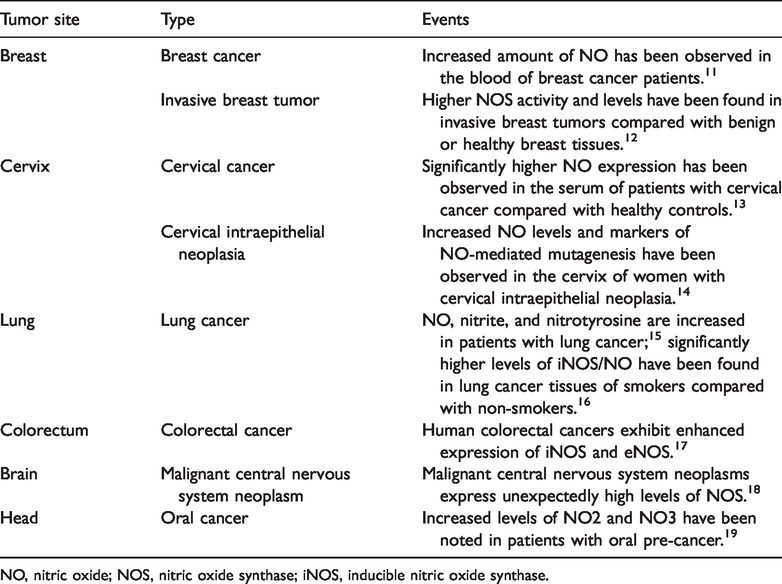
NO, nitric oxide; NOS, nitric oxide synthase; iNOS, inducible nitric oxide synthase.
NO and breast cancer
Breast cancer is the most common female cancer worldwide, with more than 1.3 million cases and 450,000 deaths each year. NO has been studied as a potential promoter of breast cancer, and an increase in NO concentration was detected in the blood of breast cancer patients. 20 Compared with benign breast tumors and healthy breast cells, invasive breast cancer cells and progressive breast cancers show increased NOS activity. NOS2 was identified as a biomarker of breast cancer disease progression and patient survival. 20 NOS2-derived NO can alter the redox state of cells, induce DNA, lipid, and protein modifications, promote an immunosuppressive microenvironment, and mediate angiogenesis and wound healing, which are all key events in cancer disease progression. 21 Additionally, breast tissue estrogen can stimulate the release of eNOS, and the expression of eNOS and estrogen receptor can also be detected in breast cancer cell lines, suggesting that eNOS expression may be the root of breast cancer carcinogenesis. 22
NO and cervical cancer
Cervical cancer is one of the most common malignant gynecologic tumors and it has a high incidence and mortality worldwide. Compared with healthy controls, NO levels were reported to be much higher in the serum of cervical cancer patients. 23 Moreover, the concentrations of NO and NO-mediated mutant markers were increased in the cervical tissues of patients with cervical intraepithelial neoplasia. High-risk human papillomavirus infection is a major risk factor of cervical cancer that can increase NO concentrations in the cervical microenvironment, 24 which further decreases pRb and p53 gene expression.
NO and ovarian cancer
Accumulating evidence suggests that NO signaling is involved in multiple aspects of ovarian cancer, including growth, apoptosis, cancer-stromal cell interaction, angiogenesis, and response to chemotherapy. 25 iNOS/NO plays a dual role in tumor glycolysis and progression via the epidermal growth factor receptor/extracellular signal‑regulated kinase (ERK)2/pyruvate kinase isozyme 2 pathway, and the interference with glycolysis may be a valuable new therapeutic approach for the treatment of ovarian cancer. 26
NO and lung cancer
Patients with lung cancer have high levels of exhaled NO and its metabolites nitrite and nitrotyrosine, as well as high levels of serum nitrite/nitrate, which have been linked to advanced stage and poor survival. 27 Smoking produces high levels of ROS and NO, 28 and a higher ratio of iNOS/NO was found in patients with lung cancer who were smokers compared with non-smokers. 29 The expression of iNOS and eNOS in atypical hyperplasia of the lung is strongly immunoreactive. Long-term exposure to chromate particles can induce chronic inflammation in the lung, increase NO production, and eventually induce lung cancer. NO may also promote the occurrence of lung cancer by protein nitrification. NO and its metabolites interact with ROS to produce an effective nitrifier, which leads to the formation of 3-nitrotyrosine in protein and chemical modifications during oxidative/nitrification stress. High levels of NO and nitrification can reduce the activity of p53, resulting lung cancer, and most patients show p53 defects. The effects of NO on angiogenesis, extensive glycolysis, p53 activity, and the antioxidant capacity of the lungs promote tumor heterogeneity and lead to metastasis. 30
NO and gastrointestinal (GI) carcinoma
The relationship between GI cancer and NO was first proposed in the 1970s. 31 p53, iNOS, and vascular endothelial growth factor positive expression significantly increases in gastric cancer. 32 NOS can induce DNA deamination and mutations in tumor suppressor genes, and genetic changes in the gastric epithelium, leading to the occurrence of gastric cancer. Excessive NO is also one of the underlying mechanisms of the Helicobacter pylori-induced development of gastric cancer through the promotion of gastric epithelial cell apoptosis. 33 iNOS was reported to be involved in the occurrence, development, invasiveness, and metastasis of esophageal carcinoma, 34 while high expression of iNOS, eNOS, and NOS mRNA was found in human colorectal cancer tissues. NO was found to induce p53 accumulation and phosphorylation, leading to cell cycle arrest, and iNOS and p53-p-Ser15 levels were positively correlated with the degree of inflammation. Consistent with nitrosative stress and the deamination of 5-methylcytosine, p53 mutations were also detected in sporadic colon cancer tissue and were associated with iNOS activity in these tissues. 35
NO and brain tumors
NO is an important neurotoxic medium in central nervous system disorders. 36 In the process of differentiation and the development of malignancy in brain tumors, neuronal tissue NOS is generally regarded as a useful marker. High expression of NOS is observed in malignant tumors of the central nervous system, and the production of NO has been shown to be related to important pathophysiological processes of these tumors. 18 However, in glioblastoma, NO generation by tumor cells may facilitate a pro-growth environment for tumor cell proliferation and neovascularization. NO may alter redox balances by virtue of preconditioning tumor subpopulations to pro-oxidant stresses. Alternatively, the direct antioxidant role of NO may predominate in particular stages. 37
NO concentration dependence and tumor therapy
NO may exert a biphasic response, such that when NO levels are beyond a critical concentration that would be suitable for tumor growth and survival, growth arrest and/or apoptotic pathways are initiated. It is worth noting that tumor cells have quite different NO concentration requirements that cause either a pro-tumor or an anti-tumor phenotype. Therefore, identifying the steady state of NO is particularly important. Experimentally, the activation of specific signaling pathways occurred in MCF7 breast cancer cells in response to distinct fluxes of NO. 38 Additionally, a low NO level (<50 nM) was associated with increased cGMP-mediated ERK phosphorylation, while an intermediate NO level (>100 nM) led to hypoxia-inducible factor-1α stabilization. However, the pro-survival effect of NO was significantly compromised at high concentrations (>400 nM) and was accompanied by an increase in the phosphorylation of p53. 39 The treatment of breast cancer cells with 30 to 60 µM NO donor DETA/NO increased pAkt and pERK levels and interrupted ERK/Akt phosphorylation-inhibited breast cancer cell proliferation. 40 Moreover, prolonged exposure of breast cancer cells to 1 mM DETA/NO resulted in mitogen-activated protein kinase phosphatase 1-mediated pERK and pAkt dephosphorylation together with the subsequent induction of apoptosis. Although NO expression is transient, the sustained NO flux generated by iNOS varies in duration from seconds to days, so determines the anti- or pro-apoptotic effects of NO. In melanoma patients, elevated NO expression often prognosticates a poor outcome because NO promotes the genesis and proliferation of melanoma by interacting with inhibitory immune cells, inhibiting apoptosis, stimulating pro-tumorigenic cytokines and tumor-associated macrophages, altering angiogenic processes, and stimulating melanoma formation itself. 41 However, in some processes, the relative concentration of NO in different environments may also serve to limit melanoma proliferation. 41
Gene therapy involving the transfer of an NOS-encoded cDNA sequence into tumor cells is considered one mechanism through which NO can be delivered. 42 An alternative is the use of NO-releasing drugs or NO donors, which have a longer half-life; additionally, their dosage is measurable and they exhibit anti-tumor activities such as the promotion of apoptosis, inhibition of metastasis and angiogenesis, and resistance to hypoxia. NO donors have dual effects on tumor treatment: to increase the sensitivity of chemotherapy and immunotherapy and to inhibit tumor metastasis. However, it is necessary to identify an optimal NO donor with the strongest anti-differentiation properties and minimal side effects to produce a novel NO hybrid that can provide a unique and favorable micro-environment for anti-tumor medicine-based therapy. 43 The advantage of combining NO with certain anti-tumor drugs, such as non-opioid anti-inflammatory drugs and statins, is that this combination can enhance anti-tumor effects.
The NO donor can also be incorporated into or associated with certain chemicals to form biopolymers, which simulate the generation of endogenous NO for targeted therapy. 44 The nano-carrier for NO can improve NO targeting to the tumor and the sensitivity of its circulation. 45 The local delivery of NO using biopolymers has also been reported. For example, polyethylene glycol gel supplied exogenous NO to tumor cells exposed to light to induce NO-mediated anti-tumor effects. 46 However, NO-releasing hydrogels or glass nanoparticles are more desirable in tumor treatment than simple NO-releasing compounds because they are dependent on the hydration rate rather than on chemical or enzymatic decomposition. 47 The former are also highly stable in the skin and can improve the release of NO under visible light, so are potential therapeutic strategies for skin cancer treatment. 48 Thermosensitive liposomes can also be used as a nano-carrier to store, transfer, and activate the release of NO in a heat-mediated manner, which is a novel tumor treatment method. 49 Fluorescent nanoparticles can be combined with an NO donor to provide an effective means for the photodynamic therapy of giant tumors. 50 The application of light and the excitation of quantum dots through ROS and NOS induce the generation of nitroso compounds, causing further tumor cell death. 50
Discussion and prospects
NO plays important roles in tumor initiation, growth, and metastasis. It can be incorporated into a novel therapeutic strategy for tumor treatment that enhances the sensitivity of refractory tumors to radiotherapy, chemotherapy, and immunotherapy. The precise reactions of NO depend on the concentration reached and on subtle variations in the composition of intracellular and extracellular milieu. NO has been suggested to have a cytostatic and/or cytotoxic effect on tumor cells; however, once the cancer has developed, NO seems to play a pro-tumor rather than an anti-tumor role. The tumoricidal properties of NO are currently being used in cancer treatment, but further validation and experimental/clinical trials are required to develop NO-based strategies for cancer prevention and treatment.
Of the numerous purported NO signaling mechanisms, epigenetic regulation has gained considerable interest in recent years. NO has been shown to influence key aspects of epigenetic regulation including histone posttranslational modifications, DNA methylation, and microRNA levels. 51 Studies across disease states have found NO-mediated regulation of epigenetic protein expression and enzymatic activity, resulting in remodeling of the epigenetic landscape, which ultimately influences gene expression. In addition to the well-established pathways of NO signaling, epigenetic mechanisms may provide much-needed explanations for poorly understood context-specific effects of NO. These findings will provide greater insights into the molecular mechanisms of NO signaling and increase our ability to dissect its functional roles in specific micro-environments in health and disease. However, more in-depth research is needed to reveal NO signaling mechanisms, and clinical trials are also warranted to verify NO-based prevention and treatment of tumors.
