Abstract
Although female predominance in the development of all types of pulmonary hypertension (PH) is well established, many clinical studies have confirmed that females have better prognosis and higher survival rate than males. There is no clear explanation of why sex influences the pathogenesis and progression of PH. Using a rat angioproliferative model of PH, which closely resembles the primary pathological changes observed in humans, we evaluated the role of sex in the development and progression of PH. Female rats had a more pronounced increase in medial thickness in the small pulmonary arteries. However, the infiltration of small pulmonary arteries by inflammatory cells was found only in male rats, and this corresponded to increased myeloperoxidase activity and abundant adventitial and medial fibrosis that were not present in female rats. Although the level of right ventricle (RV) peak systolic pressure was similar in both groups, the survival rate in male rats was significantly lower. Moreover, male rats presented with a more pronounced increase in RV thickness that correlated with diffuse RV fibrosis and significantly impaired right cardiac function. The reduction in fibrosis in female rats correlated with increased expression of caveolin-1 and reduced endothelial nitric oxide synthase–derived superoxide. We conclude that, in the pathogenesis of PH, female sex is associated with greater remodeling of the pulmonary arteries but greater survival. Conversely, in males, the development of pulmonary and cardiac fibrosis leads to early and severe RV failure, and this may be an important reason for the lower survival rate among males.
Pulmonary hypertension (PH) is characterized by the progressive elevation of pulmonary pressure, remodeling of the pulmonary arteries (intimal and medial hypertrophy, plexiform lesion formation, and fibrosis), and the eventual development of right ventricle (RV) failure. Despite the expansion of pharmacological therapies in past decade, PH leads to more than 15,000 deaths and more than 250,000 hospital visits per year.1,2 There is a strong sexual dimorphism in all types of PH, with the female-to-male ratio varying between 2:1 and 4:1.3,4 Female sex hormones and their metabolites have been postulated to be responsible for the development of PH in females.5,6 However, the available animal data suggest that estrogens exert a protective effect on the pulmonary vasculature, in contradiction with the well-documented female predominance in clinical studies, known as the “estrogen paradox.”7,8 Estrogens exert their effects through both genomic and nongenomic signaling pathways and have been shown to suppress endothelin 1 production, 9 increase the production of nitric oxide (NO) and prostacyclin, and attenuate both vasoconstriction and vascular remodeling.10–12 At the same time, estrogens have been shown to promote pulmonary vascular remodeling through increased expression of the angiogenic factor, vascular endothelial growth factor (VEGF);13,14 induce the proliferation of vascular cells;15,16 and possess prothrombotic properties,17,18 especially in conditions where the vascular endothelium is damaged. 19 Thus, overall, the available data regarding the role of sex hormones in the development of PH are conflicting.
In spite of the fact that females are more prone than males to the development of PH, they have a better survival prognosis than males. Indeed, male sex has been reported to be one of the primary independent predictors of death. 20 Recent analysis of the French, Spanish, and UK registries and of US patients enrolled in the Registry to Evaluate Early and Long-Term Pulmonary Arterial Hypertension Disease Management (REVEAL) identified a negative prognosis associated with male sex.20–23 However, there is no clear explanation of this observation. Because RV function is the primary determinant of survival in PH, 24 it is possible that the cardioprotective properties of female sex hormones play a role in the increased survival rate of females with PH. Indeed, sexual dimorphism in various types of heart disease is well established, 25 although the molecular mechanisms are considered to be complex and go beyond the estrogen alone.25–28 Nevertheless, the determination of these mechanisms is critical to allow the development of differential approaches to PH therapy in males and females. Therefore, in this study, we used a rat angioproliferative model of severe PH, originally developed by Voelkel and colleagues, 29 the most representative model of human PH, to study the role of sex in the development and progression of PH.
MATERIAL AND METHODS
Animals
All experimental procedures were approved by Georgia Regents University. Sprague Dawley rats (200–250 g) were used in this study. All animals were obtained from Charles River (Wilmington, MA) and were housed in the Georgia Regents University animal care facility for at least 1 week before being used in this experiment. Animals were kept in a 12-hour light: dark cycle at an ambient temperature of 22°C and received standard rodent food and water ad lib.
Rat model of PH
We used the rat model of occlusive-angioproliferative PH initially described by Voelkel and colleagues. 29 As previously described, this model mimics many of the clinical and histopathological changes seen in human PH, such as occlusive remodeling of the small pulmonary arteries with prominent medial hypertrophy (“concentric lesions”) and formation of complex glomeruloid-like vascular structures (“plexiform lesions”), and it exhibits a progressive disease character even after reexposure to normoxia.29,30 Before beginning the study, male and female rats were randomly allocated to either the control group, which received the vehicle for SU5614, or the SU group, which was injected with a single dose of the VEGF receptor 2 antagonist, SU5416 (SU; 20 mg/kg SC). Rats were then placed in Plexiglas chambers (BioSpherix, Redfield, NJ) for 4 weeks. Levels of O2 were maintained at 10% ± 0.5%, and the chambers were continuously flushed with nitrogen and oxygen to maintain low CO2 concentrations (<0.5%). The O2 and CO2 concentrations were continuously monitored (PROOX 110 BioSpherix oxygen controller and LB-2 CO2 analyzer, Sensormedics). After 4 weeks, the animals were returned to normoxic conditions for an additional 10 weeks. The control rats were kept under normoxic conditions for the duration of the study (14 weeks).
Echocardiographic measurements
The Vevo 2100 imaging system (VisualSonics, Toronto, Canada) was used to perform Doppler echocardiography. Baseline parameters were determined on day 0 and then every 4 weeks thereafter. For the Doppler echocardiography analyses, rats were anesthesitized using an isoflurane chamber and laid supine with all legs taped to electrocardiogram electrodes for monitoring heart rate. The rat's chest was treated with depilatory cream (Nair; Carter-Horner, Mississauga, Ontario, Canada), and a warm ultrasound gel was spread over the chest. Transthoracic two-dimensional, M-mode, and Doppler imaging were performed using 25-MHz transducer. RV free wall (RVFW) thickness was measured during end diastole using right parasternal long-axis view. These images were obtained from the right side of the rat, with the ultrasonic beam positioned perpendicularly to the wall of the mid-third of the RV. Pulsed-wave Doppler was used to measure pulmonary artery acceleration time (PAAT). The Doppler sample volume was centrally positioned within the main pulmonary artery, just distal from the pulmonary valve with the beam oriented parallel to the flow. The echocardiographic parameters were measured offline using tool section of the VisualSonic Vevo 2100 System by a reader blinded to the sex or treatment of the rat. Measurements were averaged on 15 cardiac cycles.
Invasive hemodynamic measurements
Fourteen weeks after experiment setup, subsets of animals were anesthetized (Inactin, 100 mg/kg i.p.) and instrumented for measurement of RV and systemic blood pressure. Briefly, a PE-240 polyethylene tube was inserted into the trachea to facilitate breathing. A customized pressure transducer catheter (SPR-513, Millar Instruments, Houston, TX), connected to a Millar Transducer Control Unit TC-510 and PL3504 PowerLab 4/35 data acquisition system (ADInstruments, Colorado Springs, CO), was inserted into the RV via the right jugular vein and right atrium. For systemic blood pressure measurement, the right carotid artery was cannulated with a fluid-filled PE-50 polyethylene catheter connected to a blood pressure transducer and, through a bridge amplifier, to the PowerLab system. A 30-minute stabilization period was permitted before a 30-minute recording of RV pressure and of RV functional parameters was conducted. At the end of pressure recording, the trachea catheter was connected to a Harvard Rodent Ventilator (Model 683; Harvard Apparatus, South Natick, MA), the thorax was opened, and lungs were flushed with saline (0.9% sodium chloride) via a needle inserted into the RV to remove the blood from pulmonary vessels. Animals were euthanized by anesthetic overdose, and heart and lungs were dissected and weighed.
Assessment of RV hypertrophy
The RV free wall was separated from the left ventricle (LV) and ventricular septum (S). RV hypertrophy was expressed as a wet weight of RV to LV + S (RV/LV + S). At the end of analysis, RV and LV + S were immersed in 10% buffered formalin for at least 72 hours before being embedded for histological analysis.
Histological and morphometric analysis of lung vessels and RV
The left lung from all animals was fixed in formalin and embedded in paraffin. A series of 4-μM tissue sections were dewaxed and stained with hematoxylin and eosin or Masson trichrome staining for histological and morphometric assessment. For morphometric analysis of pulmonary vessels (medial wall thickness), the pulmonary arteries were categorized depending on the external diameter, as described elsewhere. 31 Category I included arteries with an external diameter between 30 and 100 μm, and category II included arteries with an external diameter between 101 and 200 μm. Ten transversely sectioned pulmonary arteries within each category per each animal were digitally captured at 40× using an Olympus IX70 microscope and analyzed using CellSens Standard software. The total cross-sectional vessel area (TA) and the lumen area (LA) were measured, and vascular wall thickness was determined as the percentage of vascular cross-section area (TA – LA) from total vessel cross-sectional area. To assess the level of fibrotic changes (collagen deposition) in pulmonary arteries with external diameter 50–200 μm, the 4-μm pulmonary tissue sections stained with Masson trichrome were randomly scored by a blinded observer. Ten random images from each animal were scaled from 0 to 3, with 0 = no blue staining in pulmonary artery media (no fibrosis), 1 = mild blue staining in pulmonary artery media (mild fibrosis), 2 = moderate blue staining in pulmonary artery media (developed pulmonary artery media fibrosis), and 3 = strong pulmonary artery media blue staining (severe pulmonary artery fibrosis with loss of smooth muscle cells).
To detect fibrosis in RV tissue, RV sections were stained with Masson trichrome staining and scored as described elsewhere 32 in a blinded manner. The cardiac fibrosis score was as follows: 0 = no fibrosis; 1 = localized small amount of fibrosis; 2 = mild patch fibrosis; 3 = moderate diffuse fibrosis; 4 = severe diffuse fibrosis.
Immunofluorescence
Lung paraffin sections (4 μm in width) were exposed to 60°C for 30 minutes, deparaffinized in xylene, and run through graded alcohol to distilled water. Endogenous peroxidase was quenched with 0.3% H2O2 for 5 minutes, followed by two rinses with distilled water. Slides were pretreated with Target pH6 (Dako), rinsed in distilled water, incubated in Power Block (Biogenex Laboratories), rinsed in distilled water, placed in phosphate-buffered saline (PBS) for 5 minutes, incubated with primary antibody (anti-myeloperoxidase; Cappel ICN Pharmaceuticals) 1:2,000 for 30 minutes at room temperature, followed by two rinses in PBS, incubated with secondary peroxidase-labeled polymer conjugated to goat anti-rabbit immunoglobulin G (Envision +, Dako) for 30 minutes, and rinsed in PBS. Bound antibody was detected with the DAB+ substrate kit, (Dako). Hematoxylin was used for counter stain.
Myeloperoxidase activity in peripheral lung tissue
Myeloperoxidase (MPO) activity was measured by using fluorometric assay kit (BioVision) according to the manufacturer's protocol. Briefly, whole-lung tissues were pulverized in liquid nitrogen using a mortar and pestle, then homogenized in 4 volumes of MPO assay buffer (supplied in the assay kit) and centrifuged at 13,000 g for 10 minutes to remove the insoluble material. For the MPO reaction, 46 μL of MPO assay buffer was mixed with 2 μL of MPO substrate solution and 2 μL of MPO probe. The reaction mixture was then mixed with 50 μL of sample (or the positive control) and read using an excitation wavelength of 485 nm and an emission wavelength of 528 nm at T0 and again after incubation at room temperature (protected from light) for 60 minutes (T60). The amount of relative fluorescence units (RFUs) generated in 60 minutes (T60–T0) was then determined (ΔRFU). Protein concentrations in lung lysates were determined using the Bradford technique. MPO activity was then calculated as picomoles fluorescein per 60 minutes per milligram of protein, equivalent to picomoles per minute per milligram of protein, equivalent to microunits per milligram of protein.
Western blot analysis
Heart tissue was lysed and centrifuged at 6,000 g, and the supernatant was collected as previously described.33,34 Cell extracts were separated on 4%–20% polyacrylamide gradient gels and electrophoretically transferred to polyvinylidene fluoride membrane (Bio-Rad), then blocked with 1% bovine serum albumin in Tris-buffered saline. The membranes were probed using an antibody specific to Caveolin-1 (BD Biosciences). Reactive bands were visualized using chemiluminescence (ThermoFisher) using a Kodak 440CF image station. Band intensity was quantified using AlphaView SA image processing software. Protein loading was normalized by reprobing gels with a monoclonal antibody specific to β-actin (Sigma).
Measurement of superoxide levels in tissue
To detect superoxide generation in heart tissue, we performed electron paramagnetic resonance (EPR) measurements. Fresh-frozen tissues were pulverized in liquid nitrogen to form a fine powder. Then 4–6 mg of tissue powder was dissolved in 300 μL of EPR buffer (PBS supplemented with 5 μM diethyldithiocarbamate and 25 μM desferrioxamine; Sigma-Aldrich) at 0°C in a temperature-controlled cooling shaker (UltraCruz, Santa Cruz, CA) to generate a suspension of tissue particles containing intact, not lysed, cells. We found that, at 0°C, this tissue suspension does not generate a measurable amount of superoxide (data not shown). The dissolved powder was then split equally (75 μL) between four 1.5-mL eppendorf tubes containing only the spin probe, 1-Hydroxy-3-methoxycarbonyl-2,2,5,5-tetramethylpyrrolidine-HCl (CMH; 5 mg/mL), or CMH and the nitric oxide synthase inhibitor, ethylisothiourea (ETU; 1 mM). Each tube was then incubated for 2 minutes at 0°C. Thirty microliters of tissue suspension from each tube was then transferred to a glass capillary for 60 minutes at room temperature. Superoxide radical generation was then determined at room temperature using a Magnettech M100 instrument (Magnettech, Germany). Amplitudes of the EPR spectra were analyzed using ANALYSIS 2.0 software (Magnettech). To quantitate the amount of superoxide per milligram of protein, we performed a standard reaction of the superoxide-generating enzyme xanthine oxidase in the presence of xanthine and CMH. A reaction curve was generated with 1 mU of xanthine oxidase in the presence of xanthine (1 mM in PBS, pH 7.4, Sigma) in EPR reaction buffer containing CMH. This generated 1 nM/min superoxide generation rate over a 1-hour period at room temperature. Final superoxide generation in the tissue was then expressed as picomoles per minute per milligram of tissue.
Statistical analysis
Statistical calculations were performed using the GraphPad Prism software, version 4.01. The mean value (± standard error) was calculated for all samples, and significance was determined by either the unpaired or paired t test or analysis of variance (ANOVA). For ANOVA, Newman-Keuls post hoc testing or Bonferroni testing to compare the selected pairs of columns was used. A value of P < 0.05 was considered significant. For survival curve, the log-rank test for trend was used.
RESULTS
Our experimental protocol produced a significant difference in the survival rate between the male and female groups (Fig. 1). This observation is in accordance with clinical data, showing that, although females develop PH more often than males, they survive longer than males after PH has been diagnosed.20–23
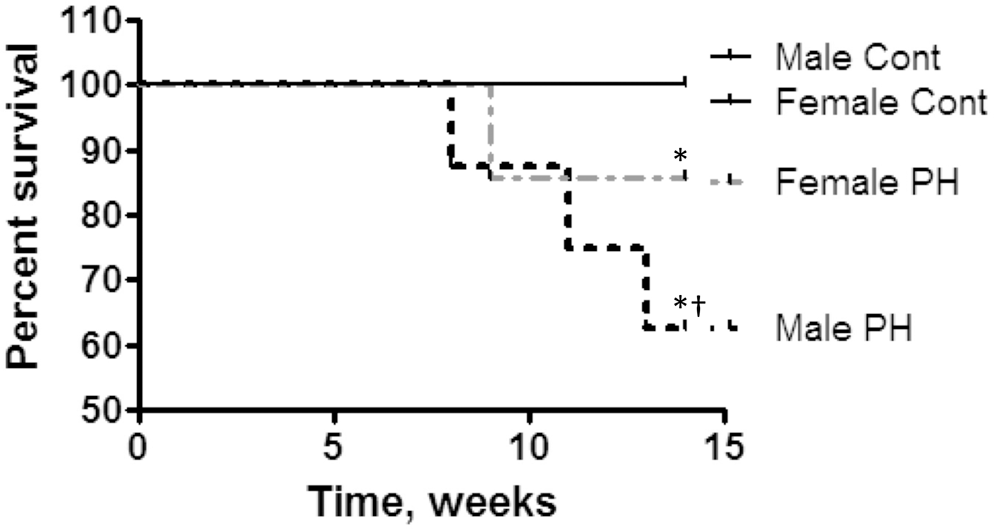
Sex-dependent differences in the survival rate of rats with pulmonary hypertension (PH). The survival curve of male and female rats during the 14-week study is shown. No deaths occurred in control groups (Cont) during this period of time. The survival rate in male but not female rats with PH was significantly different from that in controls. The survival rate in males with PH was also significantly different from that in female rats with PH. N = 6–10 rats in each group. Asterisk indicates P < 0.05 versus controls; dagger indicates P < 0.05 versus female rats with PH (log-rank test for trends).
By measuring the mean arterial pressure at the end of the study, we found the systemic blood pressure to be similar in all four groups (female, male, control, and SU5416-treated groups; Fig. 2A). However, there was a significant decrease in PAAT, assessed by echocardiography, in both females and males after 4 weeks of hypoxia (Fig. 2B), with only modest progression during return to normoxia. Because PAAT is thought to provide the most accurate estimation of pulmonary systolic blood pressure, 35 we conclude that there are no significant differences between the sexes in the development and progression of PH. This result was further supported using invasive hemodynamic measurements of RV peak systolic pressure (RVPSP) 14 weeks after SU injection (Fig. 2C). It should be noted that our data are in conflict with a previous report that indicated that the SU5416/hypoxia model induces a greater increase in RVPSP in female rats. 6 However, the conflicting data may be attributed to the use of different doses of SU5416. The high dose of SU5416 (200 mg/kg) used previously may promote greater apoptotic damage to the pulmonary endothelial cells. Because females are known to be more sensitive to endothelial damage, 19 the more severe conditions induced by a high dose of SU6416 could result in female rats developing worse PH.
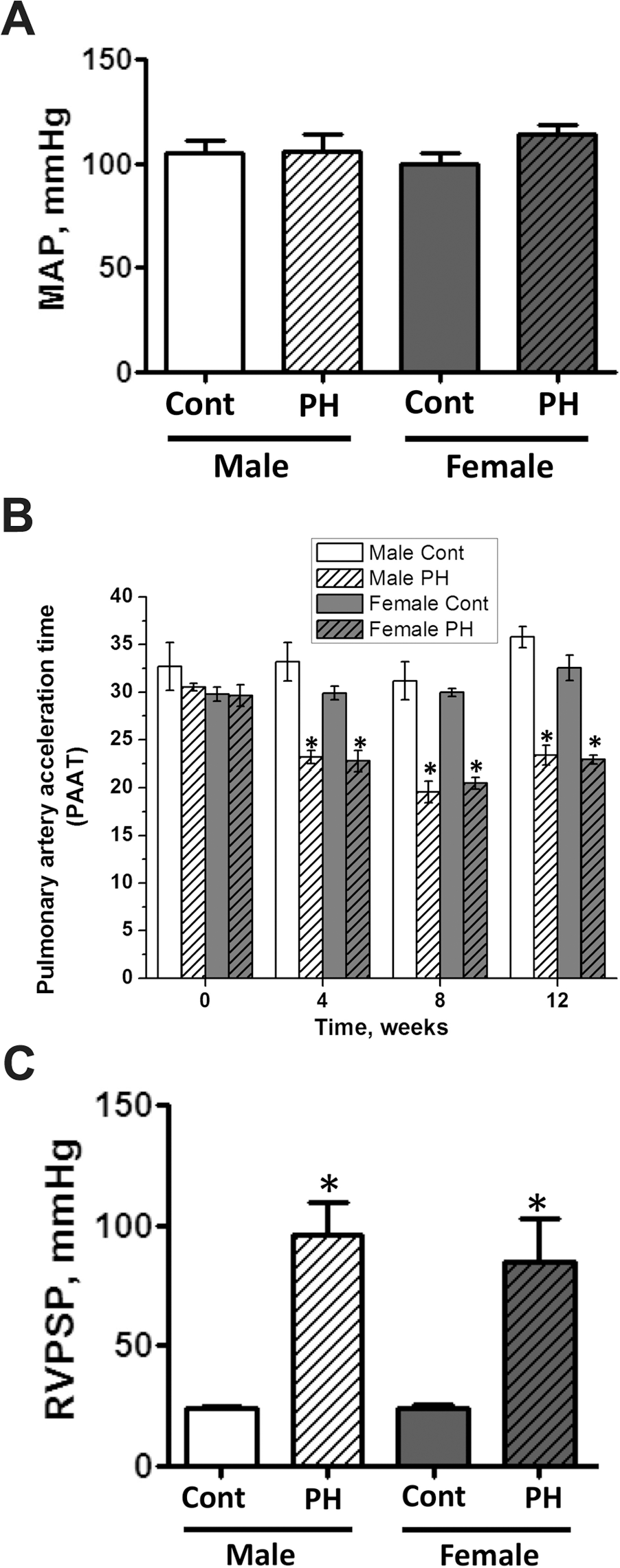
Systemic and pulmonary pressure in male and female rats with pulmonary hypertension (PH). The systemic pressure was evaluated by measuring the mean arterial pressure (MAP) in the right carotid artery. No significant differences were found between groups (A). Echocardiography identified a significant decrease in pulmonary artery acceleration time (PAAT) in both male and female rats with PH starting at week 4 of the study (B). The RV peak systolic pressure (RVPSP) was also significantly increased in both male and female rats at week 14 of the study (C). Results are expressed as mean ± SEM; N = 5–8 rats in each group. Asterisk indicates P < 0.05 versus control group (Cont).
The development of PH was associated with the remodeling of pulmonary arteries in both male and female rats (Fig. 3). However, females presented with more severe proliferation of the pulmonary parenchyma and small pulmonary arteries (Fig. 3D). To confirm that females have more pronounced pulmonary vascular cell proliferation, the vascular wall area was measured and calculated as a percentage of the total vessel cross-sectional area. The ratios of areas were used, because the thickness of the vessel wall proved to be unreliable due to the irregularity in shape of remodeled vessels. 36 Although we found a significant increase in vascular wall thickness in the pulmonary arteries between 30–100 μm and 101–200 μm in males, there was significantly greater thickening in female rats with PH (Fig. 3E).
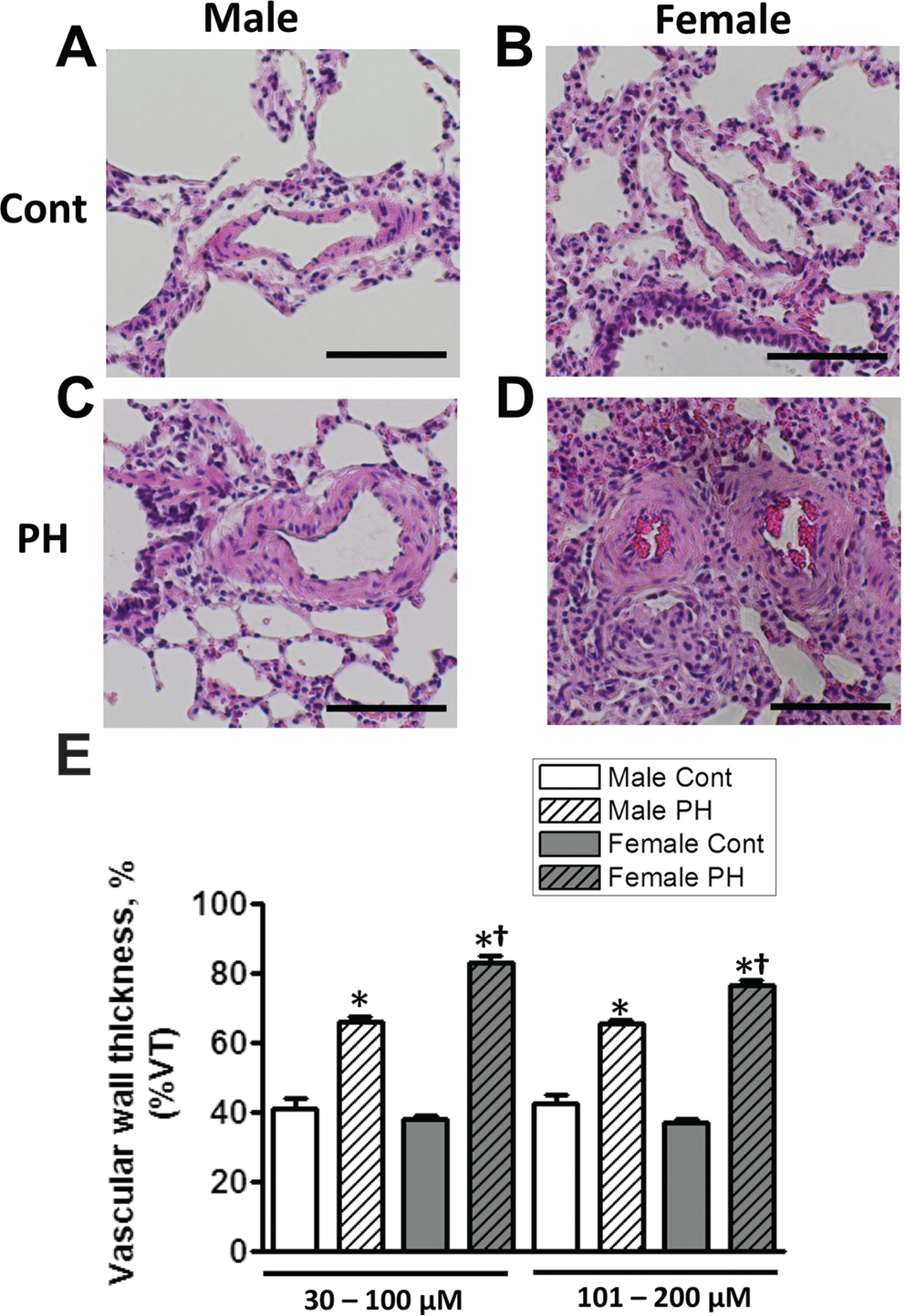
Female rats with pulmonary hypertension (PH) present with more severe angioproliferative changes in the lung. Representative images from hematoxylin-and-eosin-stained pulmonary arteries of control (Cont) male (A), control female (B), PH male (C), and PH female rats (D) are shown. Quantitative analysis of the vascular wall thickness (%VT) was significantly higher in both categories of pulmonary arteries examined (30–100 μm and 101–200 μm) in female rats with PH (E). Ten pulmonary arteries per each animal were analyzed. Results are expressed as mean ± SEM; N = 4 rats in each group. Asterisk indicates P < 0.05 versus Cont group. Dagger indicates P < 0.05 versus male PH group. The black bar corresponds to 100 μm.
The development of PH was associated with perivascular infiltration of the small pulmonary arteries by inflammatory cells in male (Fig. 4A) but not female (Fig. 4B) rats with PH. To confirm the more pronounced inflammatory component in males, lung sections were stained with an anti-MPO antibody, and total MPO activity in peripheral lung tissue was also measured. We identified high levels of MPO staining in the perivascular area of small pulmonary arteries only in male rats with PH (Fig. 4E). The pulmonary arteries of female rats (Fig. 4F) exhibited MPO staining findings comparable to those in control rats (Fig. 4C, 4D). Total MPO activity was also significantly increased in lung tissue prepared from male rats with PH (Fig. 4G). MPO activity did not significantly increase in female rats with PH (Fig. 4G).
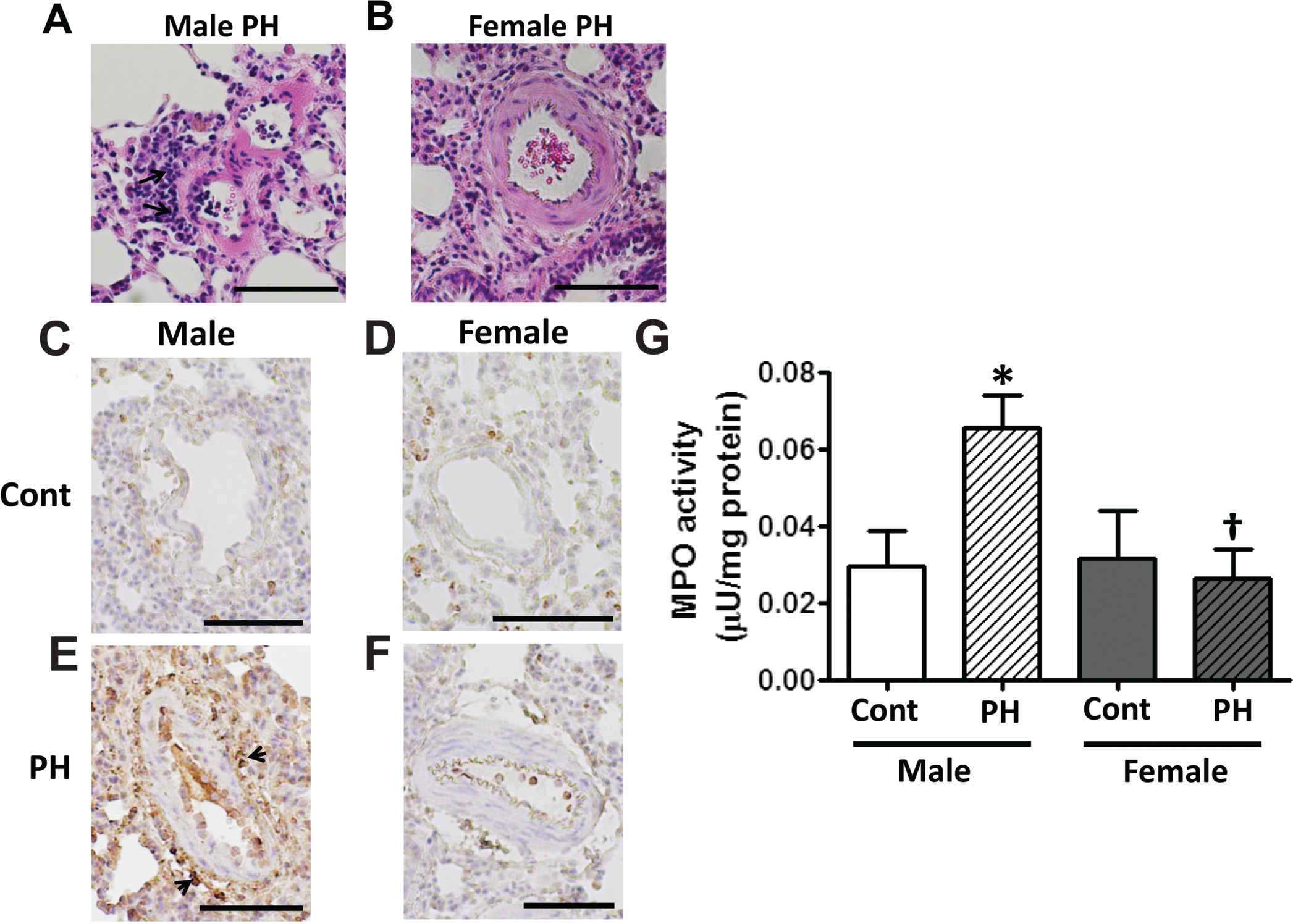
The inflammatory response is more pronounced in male rats with pulmonary hypertension (PH). Perivascular infiltration by inflammatory cells was primarily found in male (A; arrows) but not female (B) rats with PH. Immunohistochemical analysis of the lungs using a myeloperoxidase (MPO) antibody (brown) revealed significant accumulation of MPO staining in the perivascular area in male (E; arrows) but not female rats with PH (F). Neither male (C) nor female control (Cont) rats (D) exhibited significant MPO staining. A significant increase in MPO activity, measured in peripheral lung tissue, was also found in male rats with PH (G). Results are expressed as mean ± SEM; N = 5–8 rats in each group. Asterisk indicates P < 0.05 versus Cont group; dagger indicates P < 0.05 versus male rats that were injected with a single dose of the vascular endothelial growth factor receptor 2 antagonist, SU5416. The black bar corresponds to 100 μm.
PH also induced an abundant adventitial and medial fibrosis in male (Fig. 5A) but not female (Fig. 5B) rats.
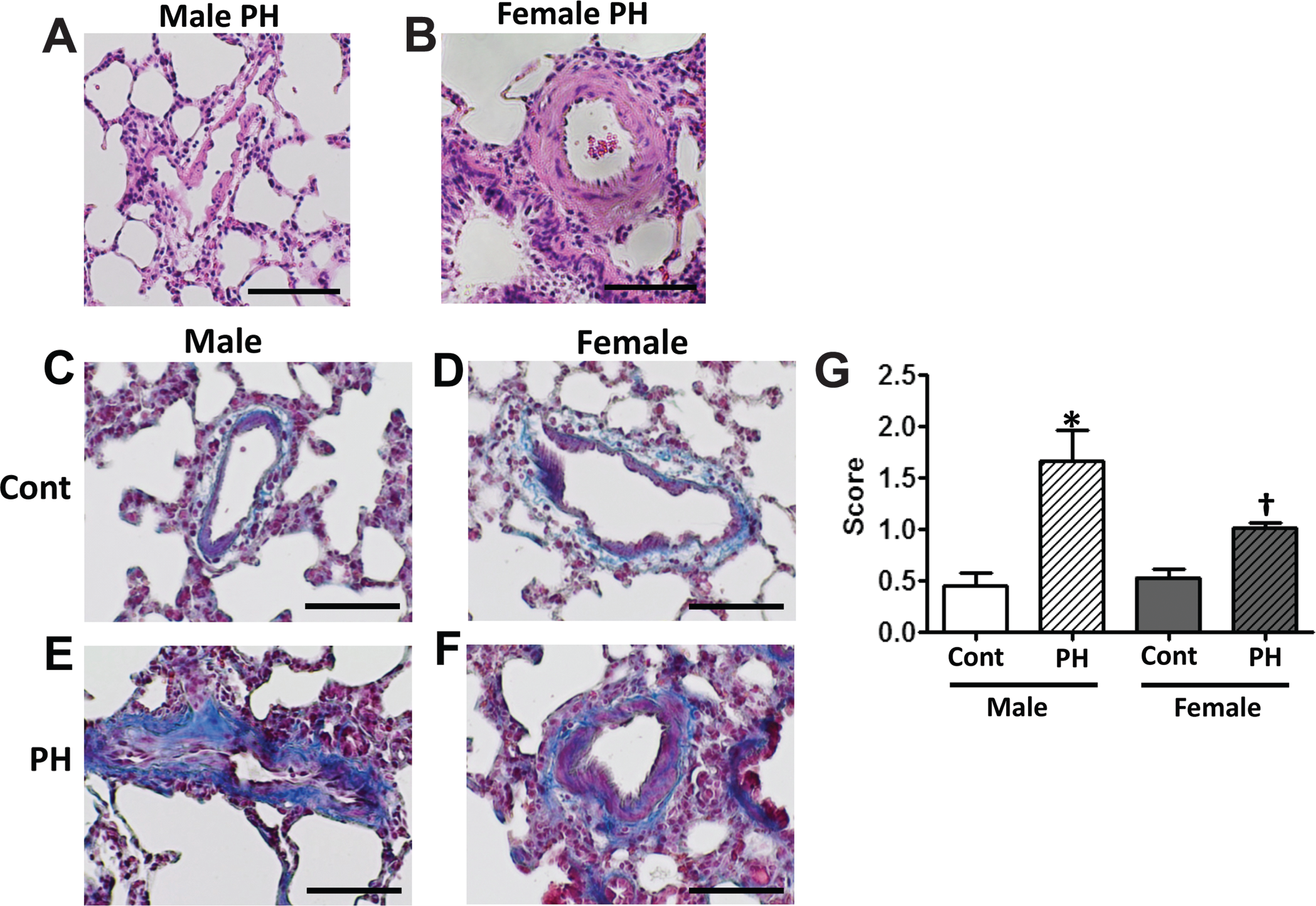
Male rats with pulmonary hypertension (PH) present with fibrotic changes in the adventitia and media of pulmonary arteries. Remodeling of the pulmonary arterial wall consisted of striking fibroproliferative changes in male rats with PH (A; arrows) and predominantly medial hypertrophy, without evidences of fibrosis, in female rats with PH (B). Masson trichrome staining identified low levels of extracellular matrix proteins in the adventitia of pulmonary arteries in control (Cont) male (C) and Cont female rats (D) as well as female rats with PH (F). However, abundant collagen accumulation was observed in the adventitia and media of pulmonary arteries from male rats with PH (E). Ten random fields (100×) for each animal were scored by a blinded observer. Scoring identified significant fibrotic tissue accumulation in male rats with PH and only mild fibrosis in female rats with PH (G). Results are expressed as mean ± SEM; N = 6 rats in each group. Asterisk indicates P < 0.05 versus Cont group. Dagger indicates P < 0.05 versus male rats that were injected with a single dose of the vascular endothelial growth factor receptor 2 antagonist, SU5416. The black bar corresponds to 100 μm.
The accumulation of collagen in the adventitia and media of small pulmonary arteries was confirmed by Masson trichrome staining (Fig. 5C–5F) and scored in a blinded manner (Fig. 5G). We identified significant deposition of matrix proteins in the pulmonary vascular wall of male rats with PH, with partial substitution of the smooth muscle layer with fibrotic tissue (Fig. 5E). In female rats with PH, medial hypertrophy was accompanied by only mild adventitial fibrosis with no signs of medial fibrotic changes (Fig. 5F). Scoring confirmed that the accumulation of connective tissue in the male rats with PH was significantly higher than that in female rats with PH. Although PH-induced fibrotic changes in pulmonary arteries are well described,36–38 this is the first evidence of the predominance of the development of lung fibrosis in the male sex, with females being resistant to fibrotic remodeling of the pulmonary arteries.
To evaluate the effect of sex on the development of RV hypertrophy, we monitored the thickness of RV free wall every 4 weeks by echocardiography. Our data indicate that female rats with PH develop mild RV hypertrophy only during their exposure to hypoxia (during the first 4 weeks of the study), whereas RV thickness was not further altered during the return to normoxia (Fig. 6A). In contrast, the male rats with PH showed a continuous increase in RV thickness under both hypoxia and normoxia conditions, and by week 12, male rats with PH presented with approximately twice the level of RV hypertrophy compared with females. These data were confirmed by measuring the RV/LV+S wet weight ratio (Fulton index) at the end of study (Fig. 6B). Together, our data suggest that female sex exerts a cardioprotective effect.
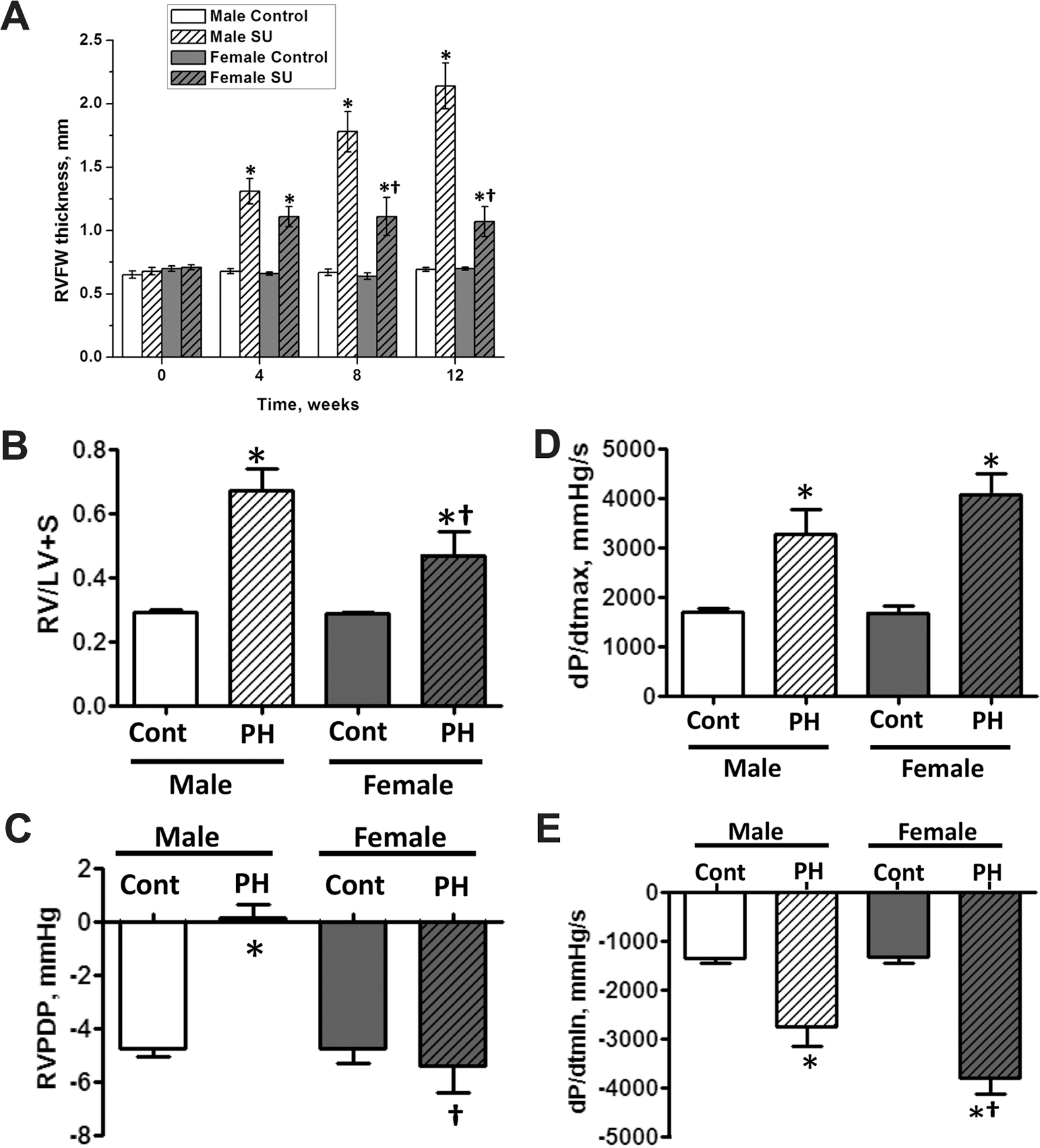
Right ventricle (RV) hypertrophy and function. The thickness of the RV free wall was measured by echocardiography. In male rats with pulmonary hypertension (PH), there was a time-dependent increase in RV thickness (A), whereas female rats had only a mild increase in RV hypertrophy during exposure to hypoxia (first 4 weeks) with no subsequent increase in RV thickness (A). The wet weight ratio of RV wall normalized on left ventricle plus septum (LV+S)—Fulton index—was measured at 14 weeks. The Fulton index was significantly higher in male rats with PH (B). Attenuation in the RV peak diastolic pressure (RVPDP) was also observed only in male rats with PH (C). RV contractility and relaxation were evaluated by measuring RV dP/dtmax and dP/dtmin, respectively. RV contractility was significantly increased in both male and female rats with PH (D). However, the increase in RV relaxation was significantly impaired only in male rats (E). Results are expressed as mean ± SEM; N = 5–8 rats in each group. Asterisk indicates P < 0.05 versus control (Cont) group. Dagger indicates P < 0.05 versus male rats that were injected with a single dose of the VEGF receptor 2 antagonist, SU5416.
To further investigate RV function, we studied RV diastolic pressure, RV contractility, and RV relaxation. In female rats with PH, the RV diastolic pressure was unchanged compared with controls (Fig. 6C). In contrast, male rats with PH presented with RV diastolic dysfunction. Indeed, the male group had a positive RV peak diastolic pressure (Fig. 6C). The dP/dtmin values in males were also significantly less negative than in females (Fig. 6E). Taking into account the similar increase in systolic RV pressure in males and females with PH, the difference in dP/dtmin suggests that the RV relaxation in males is significantly impaired compared with that in females. We also identified a nonsignificant trend of reduced RV contractility in males with PH (Fig. 6D), which may correlate with the development of RV systolic dysfunction.
To further investigate the mechanism underlying RV failure in males, we evaluated fibrotic changes. Masson trichrome staining revealed severe fibrosis in the RV of male rats with PH (Fig. 7C), whereas the deposition of collagen in the RV of female rats with PH was on a level with that observed in control rats (Fig. 7A, 7B, 7D). To assess the level of collagen deposition in each RV tissue, tissue sections were evaluated by a blinded observer. Scoring confirmed that fibrotic changes are apparent only in the RV of male rats with PH (Fig. 7E). The level of fibrotic tissue in female rats with PH was similar to that in controls (Fig. 7E). Taken together, our data suggest that PH in males is associated with the development of severe RV failure, whereas female rats with PH present with mild RV hypertrophy, a completely preserved RV morphology, and preserved RV function.
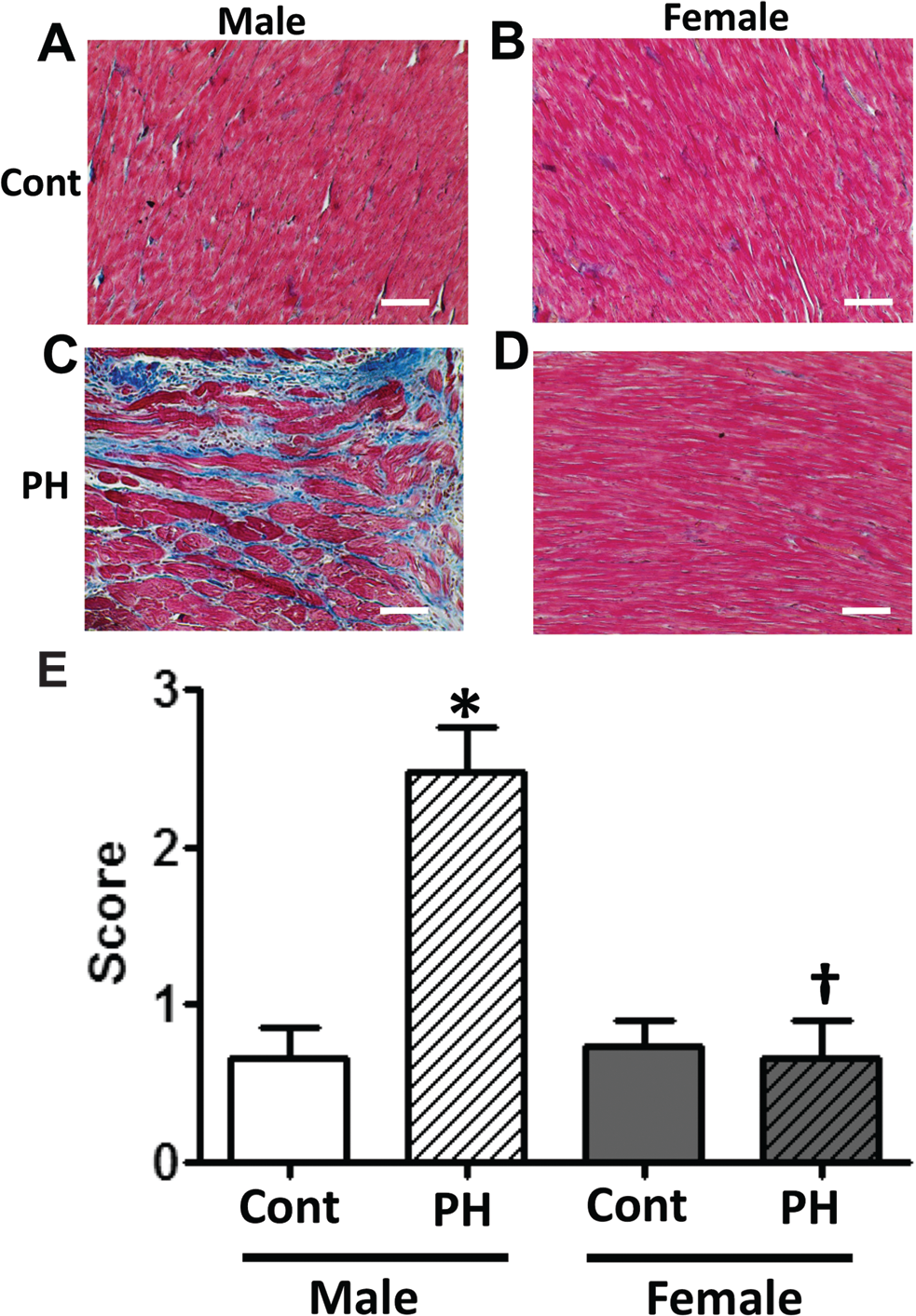
Severe myocardial fibrosis in the right ventricle (RV) of male rats with pulmonary hypertension (PH). Myocardial tissues, subjected to Masson trichrome staining, revealed significant accumulation of fibrotic tissue in the RV of male rats with PH (C), whereas the RV of female rats with PH (D) did not differ from controls (Cont; A, B). Five random fields (40×) for each animal were scored by a blinded observer. Scoring confirmed the development of severe RV fibrosis only in male rats with PH (E). Results are expressed as mean ± SEM; N = 6 rats in each group. Asterisk indicates P < 0.05 versus Cont group. Dagger indicates P < 0.05 versus male PH group. The white bar corresponds to 200 μm.
It has been recently shown that caveolin-1 expression is reduced in male rats in both the monocrotaline and sugen/hypoxia-induced rat models of PH. 39 To investigate whether differences in caveolin-1 expression could explain the differences in survival between males and females, we examine caveolin-1 protein levels in the heart. Our data indicate that control female rats had higher levels of caveolin-1 protein compared with control male rats (Fig. 8A). In addition, caveolin-1 protein levels were preserved in female rats with PH, whereas caveolin-1 protein levels were decreased significantly in male rats with PH (Fig. 8A). It is widely accepted that increased oxidative stress leads to activation of tissue fibrosis.40–42 Thus, we determined whether the sexual dimorphism associated with caveolin protein levels would modulate endothelial nitric oxide synthase (eNOS)-mediated superoxide generation (i.e, eNOS uncoupling). Our data indicate that there was a trend toward decreased eNOS-derived superoxide generation in the hearts of control female rats compared with control male rats (Fig. 8B), although this did not reach statistical significance. Furthermore, there was no significant increase in eNOS-dependent superoxide production in the hearts of female rats with PH (Fig. 8B).
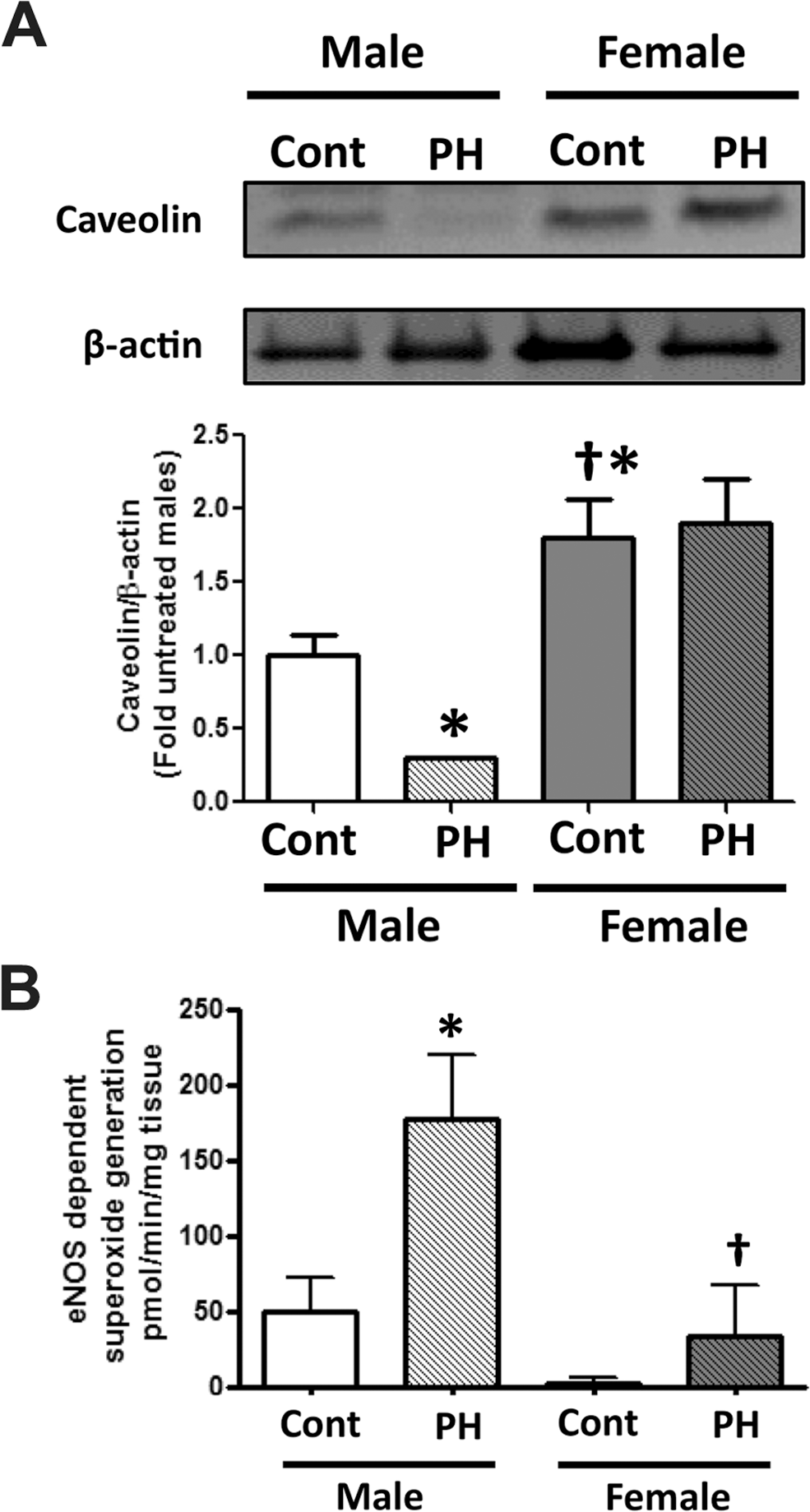
Caveolin-1 protein levels are decreased in male rats with pulmonary hypertension (PH). Western blot analysis demonstrated that caveolin-1 protein levels were higher in control (Cont) female rates compared with Cont male rats (A). Caveolin-1 protein levels were significantly reduced in male but not female rats with PH (A). Endothelial nitric oxide synthase (eNOS)-mediated superoxide production was also significantly increased in the hearts of male but not female rats with PH (B). Results are expressed as mean ± SEM; N = 3–4 rats in each group. Asterisk indicates P < 0.05 versus Cont group. Dagger indicates P < 0.05 versus male PH group.
However, there was a threefold increase in eNOS-derived superoxide generation in male rats with PH (Fig. 8B).
DISCUSSION
During the last decade, significant advances in our understanding of the pathology of PH have been achieved. The previously accepted “vasoconstriction” paradigm has transformed into our current concept of pulmonary vascular injury with subsequent proliferation of endothelial and smooth muscle cells that results in the obliteration of smooth pulmonary arteries and the formation of complex vascular structures known as plexiform lesions. However, our study highlights that these pathological changes are primarily associated with females with PH and that males with PH present with a significantly lower level of vascular proliferation. This may be due to the effects of female sex hormones, because estrogens have been shown to be damaging to the pulmonary vasculature, 5 especially when an imbalance between “good” and “bad” estrogen metabolites exists. 6 Therefore, estrogens may contribute to the initial pulmonary endothelial damage, which, in turn, is essential for the consequent vascular remodeling. In addition, the ability of estradiol (E2) to trigger angiogenesis through the up-regulation of pro-angiogenic VEGF gene transcription,13,14 and through stimulation of endothelial and smooth muscle proliferation,15,16 has been previously described. In contrast, testosterone appears to be protective for pulmonary vasculature and has been demonstrated to be a more potent pulmonary artery vasodilator than E2.43,44 Taken together, our data and previously published research indicate that females are more prone to develop angioproliferative pulmonary vascular disease than males. However, given the fact that males have a worse survival rate, it is possible that the extent of the vascular proliferative changes may play an important role in the development of PH but may not determine the progression of the disease or the survival time length.
Inflammation has been reported to be a prominent process in human PH and may contribute to progression of disease.45,46 Accumulation of inflammatory cells around remodeled pulmonary arteries as well as in adventitia, media, and plexiform lesions is well described.45,47,48 In our study, we found that the male sex is primarily associated with inflammatory cell infiltration, whereas females did not appear to develop a perivascular inflammatory response. There are several lines of evidence suggesting the importance of T and B cells in development of PH in animal models and in patients with PH.45,47,49 However, the contribution of monocytes and macrophages to PH is less understood. By measuring the expression and activity of MPO, which is an important product of activated neutrophils and macrophages, we were able to demonstrate that macrophages represent an important component of the inflammatory cell infiltrate. MPO has a number of biological effects that may contribute to pulmonary vascular injury and remodeling. These include the induction of severe oxidative stress and oxidative tissue damage, prolongation of inflammatory reactions, and propagation of vascular remodeling.50–52 Therefore, our finding that the male sex correlates with the accumulation of activated neutrophils and macrophages in the perivascular area of small pulmonary arteries coupled with increased MPO activity may be important and suggests that, in males, PH therapy may need to include an anti-inflammatory strategy. The recruitment of leukocytes in the vessel wall of lungs could also have a significant impact on the function and structure of pulmonary arteries, because activated macrophages and neutrophils can release reactive oxygen species that not only can induce damage to the surrounding cells but also can act as an NO “sink”. This would then compromise NO induced vasodilation and also potentially the vasoprotective properties of NO that include antithrombotic, anti-inflammatory, and antiproliferative actions. The oxidative and nitrative stress induced by active leukocytes could also stimulate the proliferative status in smooth muscle cells and fibroblasts through posttranslational activation of cell survival pathways 53 or through the release of various growth factors. 54 We also found that caveolin-1 protein levels were decreased only in the hearts of male rats with PH, and this correlated with increased oxidative stress due to eNOS uncoupling. Although caveolin-1 binding to eNOS is known to inhibit NO generation, these interactions have also been recently shown to attenuate eNOS uncoupling. 55 Indeed, the loss of caveolin-1 is sufficient to stimulate the development of PH in mice exposed to chronic hypoxia. 56 Thus, it is possible that maintaining adequate caveolin-1 protein levels protects female rats against developing fibrosis. However, the mechanism behind this ability to maintain caveolin-1 expression is unresolved and will need further study.
The proliferation of resident pulmonary artery adventitial fibroblasts and the attraction of circulating mononuclear cells with fibroblast-like properties to the sites of tissue injury are capable of inducing the production and accumulation of extracellular matrix proteins, especially collagen, in pulmonary arteries. 54 In this study, we found a correlation between leukocyte accumulation in the perivascular area of pulmonary vessels and the development of fibroproliferative changes in the adventitia and media of pulmonary arteries in male but not female rats with PH. From these data, we conclude that this inflammatory component may be an important factor in both vascular remodeling and the development of pulmonary fibrosis, although the known ability of male sex hormones to induce direct fibrotic changes in lungs should also be considered.57–59 The profound accumulation of these extracellular matrix proteins in the adventitia and media would severely increase pulmonary artery stiffness. Therefore, we speculate that the increase in pulmonary vascular resistance in males with PH would be additionally exacerbated by a decrease in compliance (increase in stiffness). Both the narrowing and stiffening of the pulmonary arteries contribute to RV afterload. Therefore, the ventricles of males with PH would have enhanced workloads, compared with the ventricles of females. Indeed, we found that the level of RV hypertrophy in male hearts significantly exceeded that observed in female hearts. In addition, male rats with PH developed high levels of RV fibrosis, which could also develop as a result of increased RV overload. It is well established that the cardiac fibrosis that develops after myocardial death in the hypertensive state is mainly attributable to increased afterload. Although there was an equal pressure load in both sexes during our study, the additional increase in pulmonary artery stiffness may be the factor that tips the balance in males toward RV failure. At the same time, the severity of the fibrotic changes in the RV of males with PH, coupled with previously published evidence that the pressure load itself is insufficient for development of RV failure, 60 suggests that other unknown mechanisms contribute to cardiac remodeling in males. Indeed, the sexual dimorphism associated with the development of cardiac fibrosis has been postulated clinically, 25 with more severe fibrosis observed in males. The possible mechanisms are traditionally attributed to sex hormones, although the data are limited and controversial. Thus, it is possible that the protective properties of estrogens or the deleterious role of androgens may be involved. In addition, a recent analysis of sex-dependent genes expressed in response to cardiac pressure overload revealed an important correlation, with genes activated in females being related to mitochondria and metabolism, whereas in males, the upregulated genes were related to matrix biosynthesis. 27 These data suggest that the female heart may have a better adaptation to maintain energy metabolism even in the presence of a decreased oxygen supply. This could potentially explain why we observed only mild RV hypertrophy and no fibrotic changes in female rats with PH. In contrast, the stimulation of matrix-regulating genes and genes with function related to cell growth, morphology, and differentiation may explain the development of severe RV hypertrophy and fibrosis in males with PH. The abundant accumulation of collagens in RV myocardial tissue would, in turn, lead to cardiac stiffness, and this could explain the development of the diastolic dysfunction we observed in males and has also been found in humans.
In conclusion, there is a strong sexual dimorphism in progression of PH. PH in female rats is primarily associated with angioproliferative changes in the pulmonary arteries in the absence of inflammatory and fibrinogenic processes in either the lung or heart. This correlates with preserved RV function and better survival rates. In contrast, in males with PH, inflammatory cell infiltration of the pulmonary arteries was associated with severe fibrosis in adventitia and media, profound myocardial fibrosis, impaired diastolic function, and lower survival rates. On the basis of these data, we conclude that, although the proliferative changes in pulmonary arteries play an important role in the development of PH, other pathological processes, such as inflammation and fibrosis, may also be a significant determinant of survival. This conclusion is in agreement with earlier clinical studies and may explain the phenomena of the poor survival rate of male patients with PH. Our data also provide a rationale for introducing sex-specific therapies for PH. Our data suggest that, in the female population, the primary focus should be on antiproliferative drugs, whereas in male patients, therapies may also need to include anti-inflammatory and antifibrinogenic strategies.
