Abstract
A 52-year-old man with a past medical history of chronic myeloid leukemia (CML) in remission developed progressive shortness of breath over a two-month period. He was initially treated with dasatinib for four years, until developing pulmonary arterial hypertension (PAH) with pleural effusions. His symptoms improved after stopping dasatinib. He was then switched to bosutinib for approximately one year, which was then stopped before admission due to worsening shortness of breath. His initial workup showed bilateral pleural effusions with severe PAH and cor pulmonale. He had symptomatic improvement with PAH-specific therapy following discontinuation of the bosutinib.
The life expectancy of CML patients has increased in the era of the tyrosine kinase inhibitors (TKIs), and managing adverse events (AEs) of the TKIs and improving quality of life are becoming more important.
Pulmonary hypertension (PH) and pleural effusions are rarely reported AEs of bosutinib. More reports with PH and pleural effusions arising after bosutinib use in patients previously treated with dasatinib is furthermore concerning. In this era with novel chemotherapeutic agents, physicians ought to be weary of the significant morbidity implicated by these agents in the lives of patients.
Introduction
For most patients with chronic myeloid leukemia (CML), tyrosine kinase inhibitors (TKIs) have made this potentially devastating disease into a manageable chronic condition. 1 With better disease control and improved life expectancy, there is now more focus on improving the safety profile of these drugs. 2 Resistance, however, can be an important issue. 3 Novel agents like nilotinib, dasatinib, bosutinib, and ponatinib have been made to overcome this, 3 but this is not a completely benign change. These new agents can cause many cardiovascular adverse effects.3,4 After cessation of therapy with dasatinib, reversibility in pulmonary hypertension (PH) and pleural effusions may be possible, however persistence of pulmonary arterial hypertension (PAH) has been reported.5,6 Cognizance of these effects will lead to better decision-making in the selection of these novel chemotherapeutic agents and early detection of patients with these described adverse effects. The median age at CML diagnosis in the Western world is after the sixth decade; hence, the cardiovascular effects of BCR-ABL1 TKIs are important clinical considerations. 1 The purpose of this manuscript is to emphasize the adverse effects of bosutinib in patients with prior exposure to dasatinib.
Case report
A 52-year-old man with a medical history of CML developed progressive shortness of breath over a two-month period. He was unable to do his routine daily activities and experienced severe dyspnea with minimal exertion. His CML was diagnosed in 2011 and had been in remission. He was initially treated with dasatinib for four years, until developing PAH with pleural effusions. At that time, his hemodynamic measurements from right heart catheterization showed: pulmonary artery systolic pressure = 72 mmHg; pulmonary artery diastolic pressure (dPAP) = 49 mmHg; mean pulmonary artery pressure (mPAP) = 57 mmHg; pulmonary artery occlusion pressure (PAOP) = 13 mmHg; dPAP-PAOP = 36 mmHg; thermodilution cardiac output 4.4 L/min with an index 2.38 L/min/m2; and pulmonary vascular resistance (PVR) = 8.18 Woods units (WU; 654 dynes.s/cm5). The dasatinib was stopped and he was managed with ambrisentan. His symptoms as well as effusions improved. He was then switched to bosutinib, which was stopped after approximately one year of treatment due to worsening shortness of breath. He did not have repeat hemodynamic measurements before starting bosutinib. On examination, his blood pressure was 84/58 mmHg, pulse 75 bpm, respiratory rate 14 bpm, spO2 92%. He had jugular venous distensions, a loud S2, with normal S1. No murmurs were heard and air entry was reduced at both lung bases. Other aspects of the clinical examination were unremarkable. Autoimmune screen, ventilation perfusion scan, and HIV were unremarkable.
The patient’s white cell count was 4.96 thou/mm3; hemoglobin 11.6 g/dL; platelet 214 thou/mm3; sodium 138 mmol/L; potassium 3.4 mmol/L; creatinine 1.01 mg/dL; calcium 8.5 mg/dL. Autoimmune serologies including ANA, pANCA, cANCA, atypical pANCA, SCL-70, RNP antibodies, smith antibodies, RA latex turbid, rheumatoid factor, antichromatin antibodies, SS-A, SS-B, and DNA Ab were all negative. HIV was negative; NT pro BNP 5183 (0–125 pg/mL).
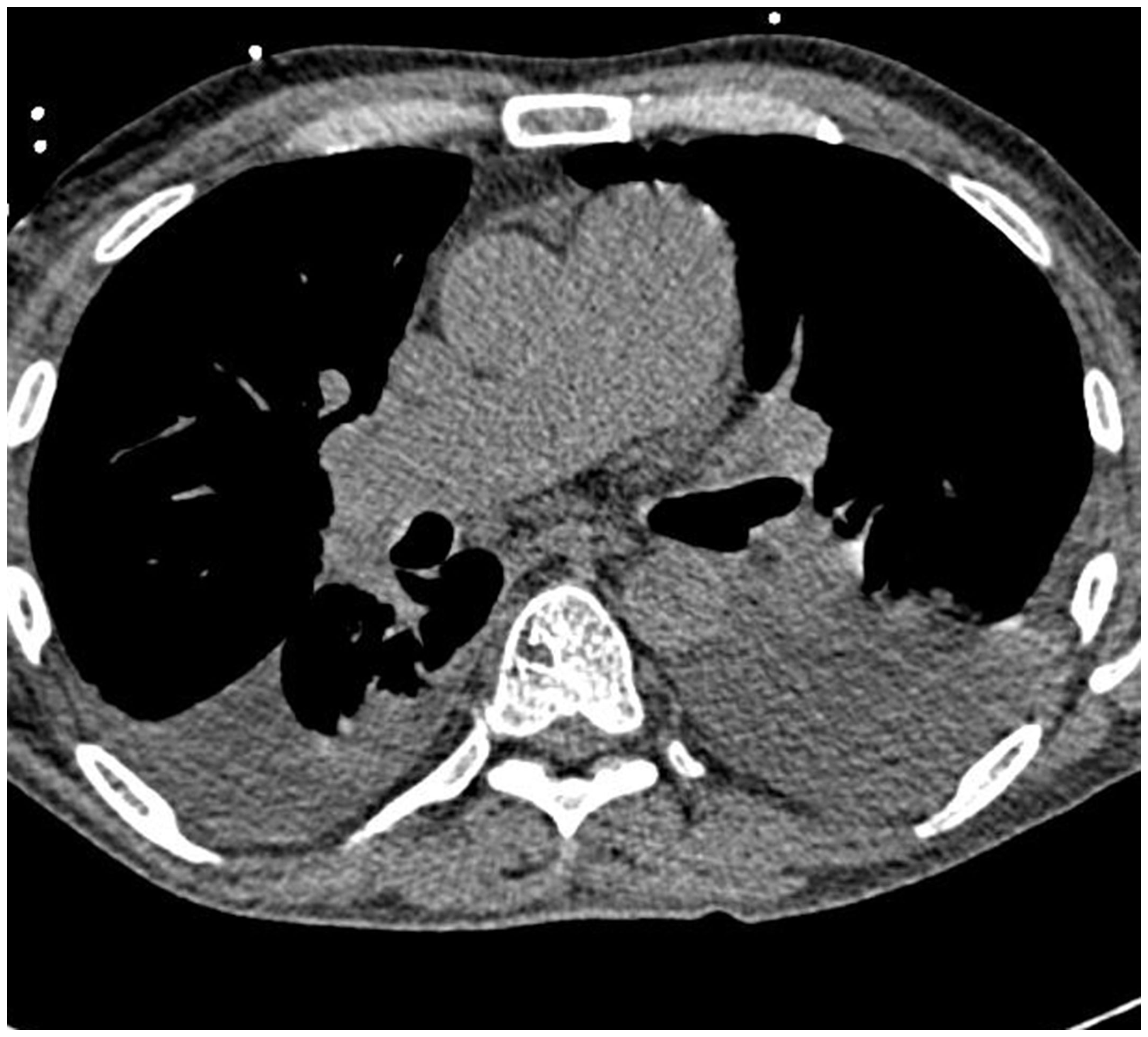
Echocardiogram demonstrated a severely dilated right ventricle (Fig. 1) with mildly reduced right ventricular ejection fraction, enlarged right atrium, flattening of the interventricular septum, mild tricuspid, and pulmonary regurgitation. There was normal left ventricular and left atrial size, with a left ventricular ejection fraction of 65% and negative bubble study. His ventilation perfusion scan showed low probability of pulmonary embolism. CT chest showed prominence of the pulmonary arterial trunk and main pulmonary artery (Fig. 2) with bilateral pleural effusions without evidence of interstitial lung disease.
Echocardiogram showing a severely dilated right ventricle. Axial CT chest showing bilateral pleural effusions with prominence of the pulmonary artery trunk, and main pulmonary artery.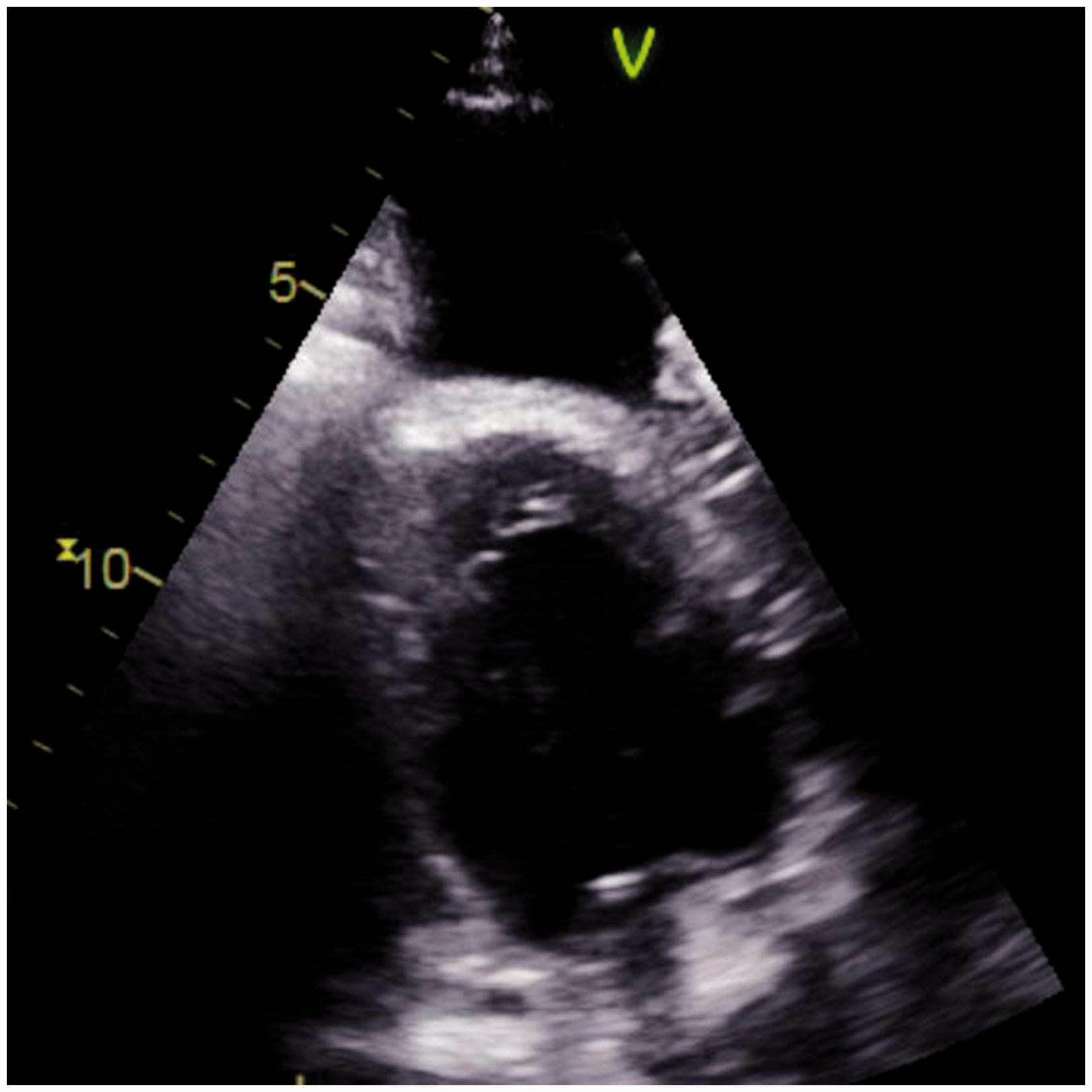
Hemodynamic measurements from a right heart catheterization showed: heart rate = 89 bpm; mPAP = 63 mmHg; right atrial pressure = 13 mmHg; pulmonary artery systolic pressure = 90 mmHg; dPAP = 48 mmHg; mPAP = 62 mmHg; PAOP = 18 mmHg; body surface area = 1.98; dPAP-PAOP = 30 mmHg; thermodilution cardiac output = 3.22 L/min with an index 1.64 L/min/m2; PVR = 13.66 WU (1093 dynes.s/cm5); systemic vascular resistance = 23.9 WU (1242 dynes.s/cm5). Therapeutic and diagnostic thoracentesis was done for the pleural effusions, which were consistent with exudative, lymphocyte-predominant pleural effusions. Flow cytometry of the pleural fluid did not reveal any evidence of malignancy.
He was managed with treprostinil titrated to a goal of 25 mg/kg/min, riociguat 1 mg t.i.d., ambrisentan 10 mg daily, and furosemide 40 mg b.i.d. Midrodrine 10 mg t.i.d. was added for blood pressure support.
His respiratory function improved allowing better WHO functional capacity. His NT pro BNP dropped to 1455 pg/mL after approximately four weeks of treatment.
Written informed consent was obtained from the patient for publication of this case report and the accompanying images. A copy of the written consent is available for review by the editorial board of this journal.
Discussion
TKIs have transformed the management of patients with CML, turning a potentially devastating disease into a manageable chronic condition.1,5 The “gold standard” of treatment for CML patients is imatinib, a BCR/ABL1 TKI. 3 The five-year survival rates with imatinib increased significantly from 40–50% to 90%.1,2 With this increase in life expectancy, both the CML patients and their treating physicians are now faced with improving quality of life and managing adverse events (AEs) of these TKIs. 7
The efficacy of second generation TKIs have surpassed imatinib, as they can induce more rapid and profound molecular responses, and overcome the resistance that can develop from imatinib use.1,3 Of these, nilotinib and dasatinib were approved for front-line therapy whereas bosutinib did not demonstrate complete cytogenic response when compared to imatinib and as such is presently a second-line agent. 8 Nilotinib has been reported to cause prolongation of QT interval and sudden cardiac death. As a result, there is a Black Box Warning against its use. Despite these advances in efficacy, the use of these agents has come with its own set of consequences. Major AEs include PH, pleural effusions, and peripheral arterial and venous events.3,9,10 Other cardiovascular adverse effects related to these agents include fluid retention, pleural-pericardial effusion, heart failure, QT prolongation, and coronary artery disease. 4
The mechanism by which PH and other vascular events come about is not well understood. 3 Some of these mechanisms described include: direct effect of these TKIs on vascular and/or perivascular cells causing vasospasm; 3 inhibition of SRC results in pulmonary vasoconstriction;6,11 direct pro-atherogenic and anti-angiogenic effects on endothelial cells; 3 mitochondrial reactive oxygen species production; 12 inhibition of fibroblast growth factor receptor and mitogen-activated protein kinases; 5 and apoptosis of endothelial cells. 5
In this report, we focus mainly on PH and pleural effusions with particular focus on their occurrence in patients treated with bosutinib after dasatanib.
PAH is a rare disease and drug-induced PAH contributes to an extremely small percentage of these patients. 8 Drug-induced PAH has been reported with dasatinib use and there is usually partial or complete reversibility after drug withdrawal.5,6 The first case of dasatinib-related PAH was reported in 2009. 1 There were nine patients with dasatinib-associated PAH reported in the 2012 French Pulmonary Hypertension Registry, mainly with moderate to severe PAH with symptoms and hemodynamic compromise.1,13 After withdrawal of the drug, improvements were observed; however two patients died in follow-up as a result of sudden death or cardiac failure. 1 The estimated frequency of dasatinib-associated PAH was 0.45%. 1
In the DASISION study, PAH was reported in 3% of patients on dasatinib and none of the patients on imatinib. 1 As a result of the above study, the US FDA issue a warning in 2011 regarding the cardiac and pulmonary risks of dasatinib. 14 In another recent study of 21 patients with dasatanib-induced PAH, there was improvement in PAH after withdrawal of the drug but persistence in over one-third of patients. 6
As previously reported, bosutinib is an excellent alternative in patients who are left without a suitable treatment option. 10 The non-hematologic toxicity is typically transient, with a low incidence of cardiovascular adverse effects reported. 1 However, of great concern is that bosutinib has been noted to cause marked worsening of pre-existing TKI-associated PAH. 11 With these new reports, physicians should be conscious of the morbidity imposed before initiating treatment with bosutinib in patients previously treated with dasatanib evidence by our case.
Dasatinib-induced exudative pleural effusion is another common AE with frequency in the range of 15–35%. 5 Bosutinib-induced pleural effusions are considered to be rare; however, an incidence of 8% in patients treated with bosutinib. 5 The majority of those patients experienced this toxicity when prior TKI therapies were used. 2
Due to these adverse effects, dasatinib and bosutinib should be avoided in those patients with a history of pleural effusion or PH. 10 It has been advised that patients be evaluated for signs and symptoms of cardiopulmonary disease before and during dasatinib treatment. 14 Furthermore, due to the persistence of PAH in patients after dasatinib use, ongoing surveillance of their hemodynamics is important. 6 Because the median age at CML diagnosis in the Western world is more than 60 years, when cardiovascular disease is common, the cardiovascular effects of BCR-ABL1 TKIs are critical factors in therapy decisions. 1 Two reports recommended that echocardiogram with Doppler flow studies would serve as adequate non-invasive assessment for high-risk patients before starting dasatinib and for patients with cardiopulmonary symptoms on treatment.1,2 Additionally, in patients treated with PAH medications for dasatinib-induced PAH, hemodynamic surveillance may be useful in detecting relapse after PAH therapy is stopped. 6 Unfortunately our patient did not have repeat hemodynamics before starting bosutinib; however, we advocate that this suggestion should be extended to other TKIs as well, in particular bosutinib.
Patients treated with dasatinib followed with bosutinib.

Conclusion
Worsening of pre-existing PAH and lymphocyte-predominant exudative pleural effusions is an uncommon adverse effect of bosutinib in patients previously treated with dasatinib. Bosutinib should be avoided in patients with pleural effusions and PH thought to be due to dasatinib. Screening for PH in at-risk patients should be advocated.
Footnotes
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
