Abstract
It has been suggested pleural effusions may develop in right heart failure in the absence of left heart disease. The incidence and prognostic significance of pleural effusions in pulmonary arterial hypertension is uncertain. Patients with pulmonary arterial hypertension followed at our tertiary care center were reviewed. Survival was examined based on the subsequent development of a pleural effusion. A total of 191 patients with pulmonary arterial hypertension met the inclusion criteria. The prevalence of pleural effusions on initial assessment was 7.3%. Among patients without a pleural effusion on initial imaging and at least one follow-up computerized tomography (N = 142), pleural effusion developed in 27.5% (N = 39) of patients. No alternative etiology of the effusion was identified in 19 (48.7%) cases and effusions deemed related to pulmonary arterial hypertension occurred at an incident rate of 38.6 cases per 1000 person-years. Of these, 14 (73.7%) were bilateral, 3 (15.8%) were right-sided, and 2 (10.5%) were left-sided. Effusion size was trace or small in 18 patients (94.7%). Development of a new pleural effusion was associated with attenuated survival in unadjusted survival analysis (HR: 3.80; 95% CI: 1.55–9.31), multivariate analysis (HR: 5.13; 95% CI: 1.86–14.16), and after the multivariate model was adjusted for concomitant pericardial effusion (HR: 4.86; 95% CI: 1.51–15.71). Negative impact on survival remained unchanged when effusions more likely related to an alternative cause were removed from analysis. In conclusion, pleural effusions can complicate pulmonary arterial hypertension in the absence of left heart disease. These effusions are frequently small in size, bilateral in location, and their presence is associated with decreased survival. Attenuated survival appears independent of the risk associated with a new pericardial effusion.
Introduction
Pleural effusions are a common complication of a wide variety of disease processes. Simplistically, the development of a pleural effusion is related to either increases in pleural fluid entry, driven by Starling forces, or reduced pleural fluid clearance. 1 Given these physiologic principles, it has been historically held that pleural effusions in congestive heart failure are the result of severe left heart disease.2,3 However, an increasingly robust body of evidence supports the assertion that pleural effusions can occur with isolated elevations in right heart pressures in the absence of significant left heart disease.4–7
Chronic right heart failure in the absence of left heart disease can occur as a result of right ventricular infarction or progressive pulmonary hypertension. 8 In patients with pulmonary arterial hypertension (PAH), the occurrence of pleural effusions correlates with higher mean right atrial pressure compared to patients without pleural effusion. 7 While some effusions may be related to complications of connective tissue disease (CTD), literature suggests that these inflammatory processes cannot explain all effusions encountered in patients with PAH. 6 Although pleural effusions have been documented as complicating PAH, the precise incidence of this phenomenon has not been previously described. Moreover, despite the previously cited evidence, skepticism remains regarding the relationship of elevated right-sided cardiac pressures and subsequent effusion development.9,10
In addition to the development of pleural effusions, patients with PAH are known to have an increased proclivity to develop pericardial effusion, which likewise, can be multifactorial in etiology from elevated right-sided pressures or concomitant CTD. The development of either a pericardial effusion or a pleural effusion in PAH has been demonstrated to be associated with a worse overall prognosis.11–13 However, it is not clear if these risks are independent. In this study, we sought to define the incidence rate of pleural effusion in PAH, examine the prognostic significance of pleural effusions, and to further examine the risk factors for the development of a pleural effusion in this condition.
Methods
Records of patients ≥18 years of age referred to a comprehensive care pulmonary hypertension center between April 2002 and May 2018 were reviewed. Patients were included if they met the current diagnostic criteria for PAH based on a thorough clinical evaluation excluding other causes of pre-capillary pulmonary hypertension and right heart catheterization findings of mean pulmonary artery pressure >20 mmHg at rest, a pulmonary vascular resistance ≥3 Wood units, and a mean pulmonary capillary wedge pressure ≤15 mmHg. 14 Patients with significant obstructive or restrictive lung disease or chronic thromboembolic disease were excluded from the cohort. Included patients were required to have completed a six-minute walk test (6MWT), pulmonary function testing, thoracic computerized tomography (CT) scan, and right heart catheterization within three months of initial assessment.
All patients were followed within the comprehensive care pulmonary hypertension center at our Hospital. The study was approved by the Institutional Review Board (IRB# 17-2833) at our Hospital. Data were collected from the electronic medical record (Epic®), beginning at the time of the initial pulmonary hypertension clinic consultation and for up to five years following this initial assessment. Baseline demographic information and clinical data were collected. The presence of pleural or pericardial effusions and ascites was recorded based on the relevant radiographic reports. Data were collected for all included patients; however, those with a pleural effusion identified on baseline imaging (prevalent cases) or without follow-up thoracic CT were removed from the main analysis. All available clinical records, including patient history, cross-sectional imaging, diagnostic testing, and post-mortem examination (where applicable) of all patients with incident pleural effusion were examined to identify possible alternative etiologies for noted effusions. Cases (blinded for clinical outcome) were reviewed by an advanced lung disease pulmonologist (C.S.K.) and instances where an alternative etiology to PAH was thought as or more likely to have caused a patient’s pleural effusion were recorded. Where pleural effusions were present, thoracic CT images were reviewed separately by two advanced lung disease pulmonologists (C.S.K. and V.K.) to characterize effusion size (trace, small, moderate, or large). In cases of discordance, effusion size was collectively adjudicated by the readers.
The primary outcome of interest was overall survival in patients with a newly developed pleural effusion. Survival in patients with new pleural effusion not deemed related to an identifiable cause (other than PAH), and survival in patients with pleural effusions detected at the time of initial clinic evaluation were also examined.
Distribution of all continuous data was examined for normality using visual inspection and the Shapiro–Wilk test. Characteristics of the groups are presented using the mean and standard deviation for normally distributed data and compared between groups using the two-sample Student’s t-test. Data not normally distributed are presented as median and interquartile range (IQR) and compared using the Wilcoxon Rank Sum test. Categorical data are presented as counts with proportions and compared using Fisher’s exact test. Survival analysis was performed using the Kaplan–Meier method and the log-rank test was used to compare groups. The Cox proportional hazards model was utilized to calculate hazard ratios (HRs) with their 95% confidence interval (CI) to analyze individual parameter effect on survival. A multivariate model was constructed through consideration of clinically relevant variables and stepwise technique. To determine the effect on survival independent of risk possibly attributed by pericardial effusion, the final multivariate model was then stratified by the presence or absence of a coexisting pericardial effusion. All relevant statistical tests were two-tailed and a P value < 0.05 was considered statistically significant. All statistical analyses were performed using STATA version 14 (StataCorp LP; College station, TX, USA).
Results
There were 217 patients diagnosed with PAH during the study period; 26 patients were excluded for incomplete data and 191 patients with PAH qualified for the analysis and were included in the final cohort.
Analysis of prevalent cases
To explore the risk factors related to the development of a pleural effusion, patients in the cohort with a pleural effusion detected by thoracic CT performed at time of initial consultation (prevalent cases) were examined. A pleural effusion was identified as part of the initial consultation in 14 patients (7.3%). The demographic, clinical, and diagnostic data at the time of initial consultation of all 191 patients are presented in Table 1. Patients with a pleural effusion identified by thoracic CT at the time of initial consultation were statistically more likely to have a lower body mass index and higher mean right atrial pressure. Of the 14 patients with a pleural effusion at the time of initial consultation, 3 patients were felt to have another cause (malignancy, infection, and hypoalbuminemia). Prevalent pleural effusions were significantly associated with attenuated survival both before and after the removal of patients with a possible alternative etiology for the effusion (HR: 4.50; 95% CI: 1.68–12.06) and (HR: 4.06; 95% CI: 1.20–13.78), respectively.
Characteristics of all patients at time of initial consultation categorized by presence of a pleural effusion at initial evaluation.
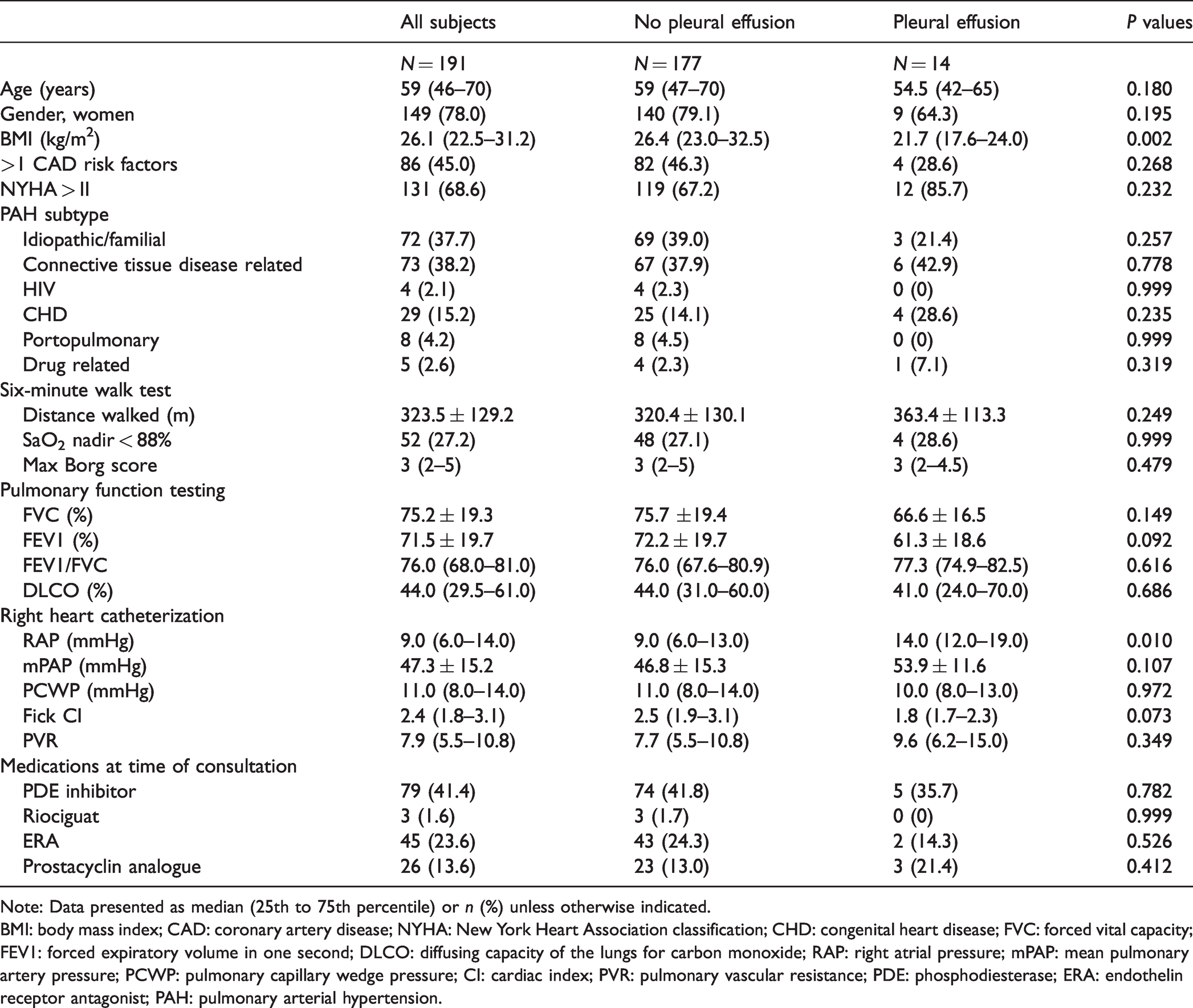
Note: Data presented as median (25th to 75th percentile) or n (%) unless otherwise indicated.
BMI: body mass index; CAD: coronary artery disease; NYHA: New York Heart Association classification; CHD: congenital heart disease; FVC: forced vital capacity; FEV1: forced expiratory volume in one second; DLCO: diffusing capacity of the lungs for carbon monoxide; RAP: right atrial pressure; mPAP: mean pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; CI: cardiac index; PVR: pulmonary vascular resistance; PDE: phosphodiesterase; ERA: endothelin receptor antagonist; PAH: pulmonary arterial hypertension.
Incident cases
Patients in the cohort without a subsequent thoracic CT (N = 35) and those with prevalent pleural effusions (N = 14) were removed from the survival analysis for incident cases. To determine the incidence of pleural effusion in PAH, we reviewed data of the remaining 142 patients spanning a median of 4.1 years (IQR: 1.9–5.0) resulting in 492.3 person-years of cumulative follow-up. The majority of these patients (80.3%) were female and the median age was 60.5 years (IQR: 48–71). The median number of total thoracic CTs performed during follow-up was 2.5 (IQR: 2–6) per patient. The baseline characteristics of these 142 patients stratified by subsequent development of a pleural effusion are presented in Table 2. Sixty-one (43.0%) patients had CTD-related PAH, whereas 81 (57.0%) patients had PAH from another cause (non-CTD-related).
Characteristics of patients at time of initial consultation categorized by development of an incident pleural effusion.
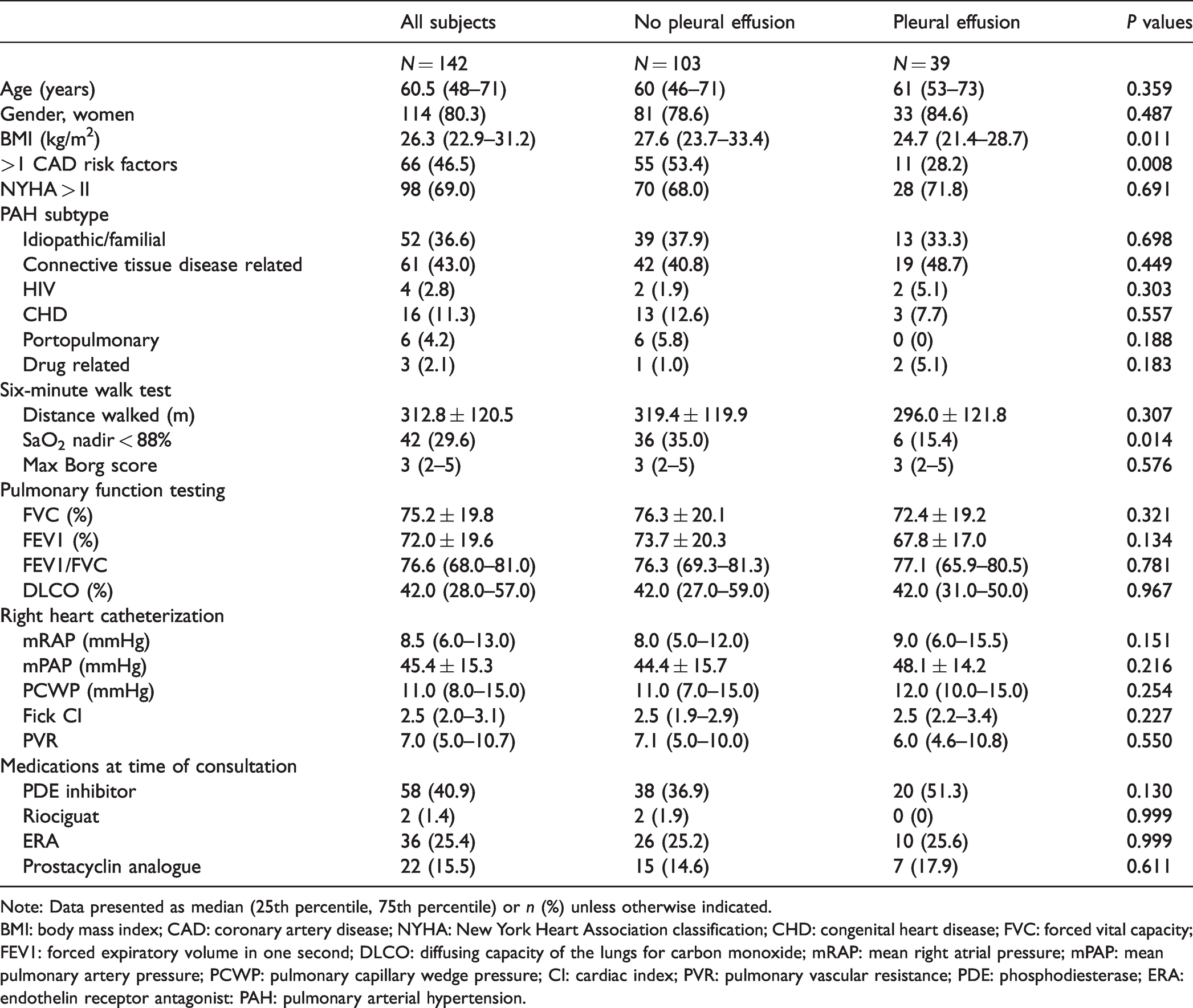
Note: Data presented as median (25th percentile, 75th percentile) or n (%) unless otherwise indicated.
BMI: body mass index; CAD: coronary artery disease; NYHA: New York Heart Association classification; CHD: congenital heart disease; FVC: forced vital capacity; FEV1: forced expiratory volume in one second; DLCO: diffusing capacity of the lungs for carbon monoxide; mRAP: mean right atrial pressure; mPAP: mean pulmonary artery pressure; PCWP: pulmonary capillary wedge pressure; CI: cardiac index; PVR: pulmonary vascular resistance; PDE: phosphodiesterase; ERA: endothelin receptor antagonist: PAH: pulmonary arterial hypertension.
Overall, 39 patients (27.5%) developed a pleural effusion during clinical follow-up which represents an incident rate of 79.2 cases per 1000 person-years. These patients were more likely to have a lower body mass index, have fewer baseline cardiac risk factors, and maintain their oxygen saturation above 88% without supplemental oxygen during the initial 6MWT. There was no significant difference in the incidence of pleural effusion between those with and without CTD (31.1% vs. 24.7%, P = 0.45). Change in distance ambulated during 6MWT six months after consultation compared to baseline was also compared between groups (Fig. 1). Patients who ultimately developed a pleural effusion and those who did not both demonstrated overall improvement in distance ambulated at six months after initial consultation. There was no difference in magnitude of improvement between the groups (18 m (IQR: –12 to 55) vs. 32 m (IQR: –8 to 70) P = 0.37).
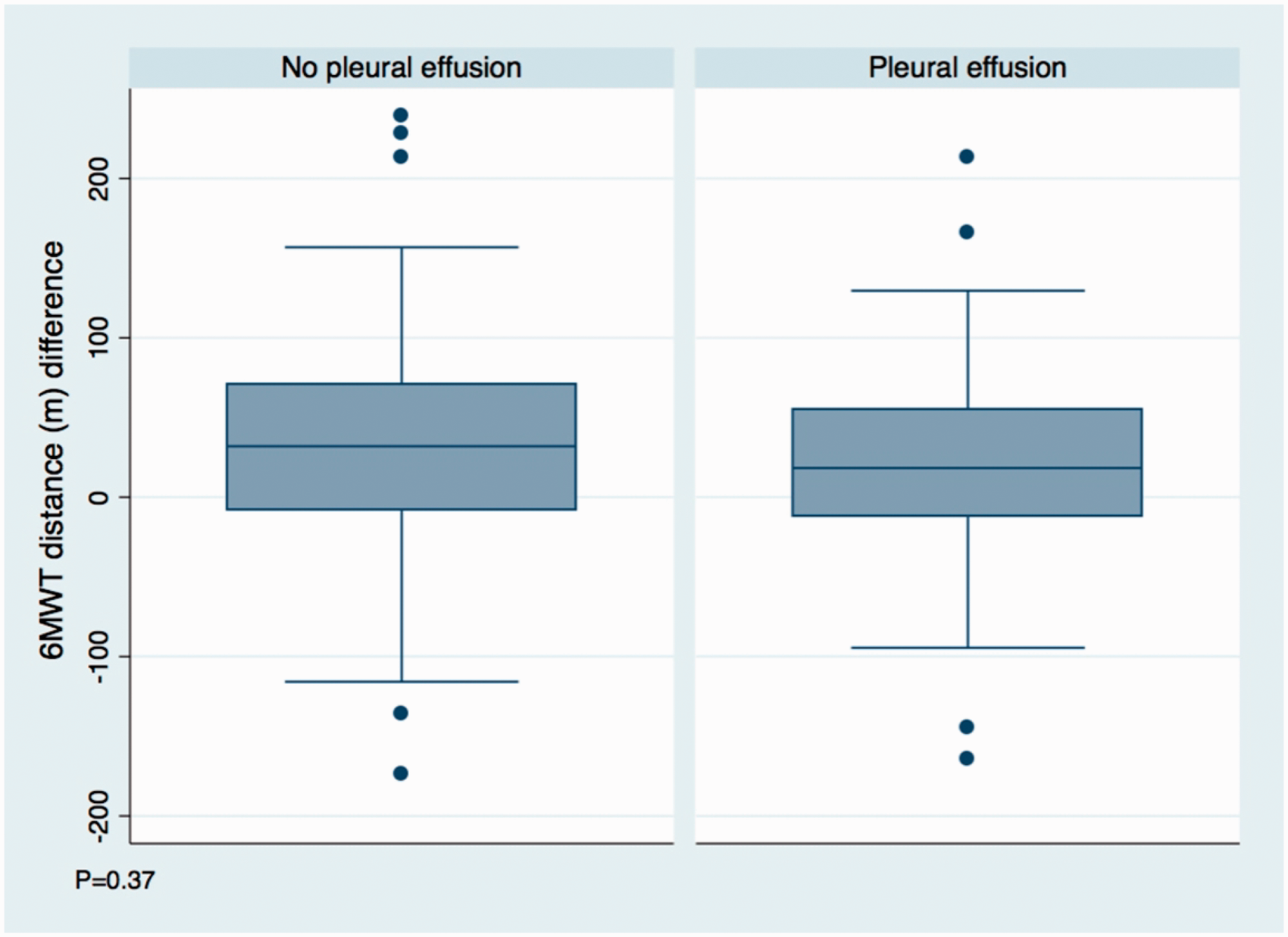
Box plot of difference in distance (m) ambulated during six-minute walk test between six months and baseline consultation grouped by development of pleural effusion during follow-up.
The characteristics of the pleural effusions noted during follow-up are included in Table 3. The majority (59%) of these pleural effusions were bilateral, while 9 (23.1%) were right-sided, and 7 (17.9%) were left-sided. Most effusions (89.7%) were either trace or small in size. Pericardial effusion and ascites were noted during the follow-up period in 25 (17.6%) and 14 patients (9.9%) respectively. At the time of pleural effusion diagnosis, 31 (79.5%) patients found to have a pleural effusion were being treated with a phosphodiesterase-5 inhibitor, 1 (5.3%) was taking riociguat, 20 (47.4%) were taking an endothelin receptor antagonist, and 6 (31.6%) were prescribed a prostacyclin analogue.
Characteristics of incident pleural effusions.

Twenty of the 142 (14.0%) patients in the cohort died. The development of any pleural effusion was associated with an attenuated overall survival (log-rank test, P < 0.01). Survival from time of initial consultation based on the presence or absence of incident pleural effusion is displayed in Fig. 2. The relationship between initial clinical factors and overall survival are included in Fig. 3. Gender, body mass index, distance walked during 6MWT (per 50 m), and diagnosis of idiopathic or familial pulmonary hypertension were included in the final model (Fig. 4). After multivariate adjustment (Table 4), the development of a pleural effusion remained associated with attenuated survival (HR: 4.65; 95% CI: 1.65–13.08).
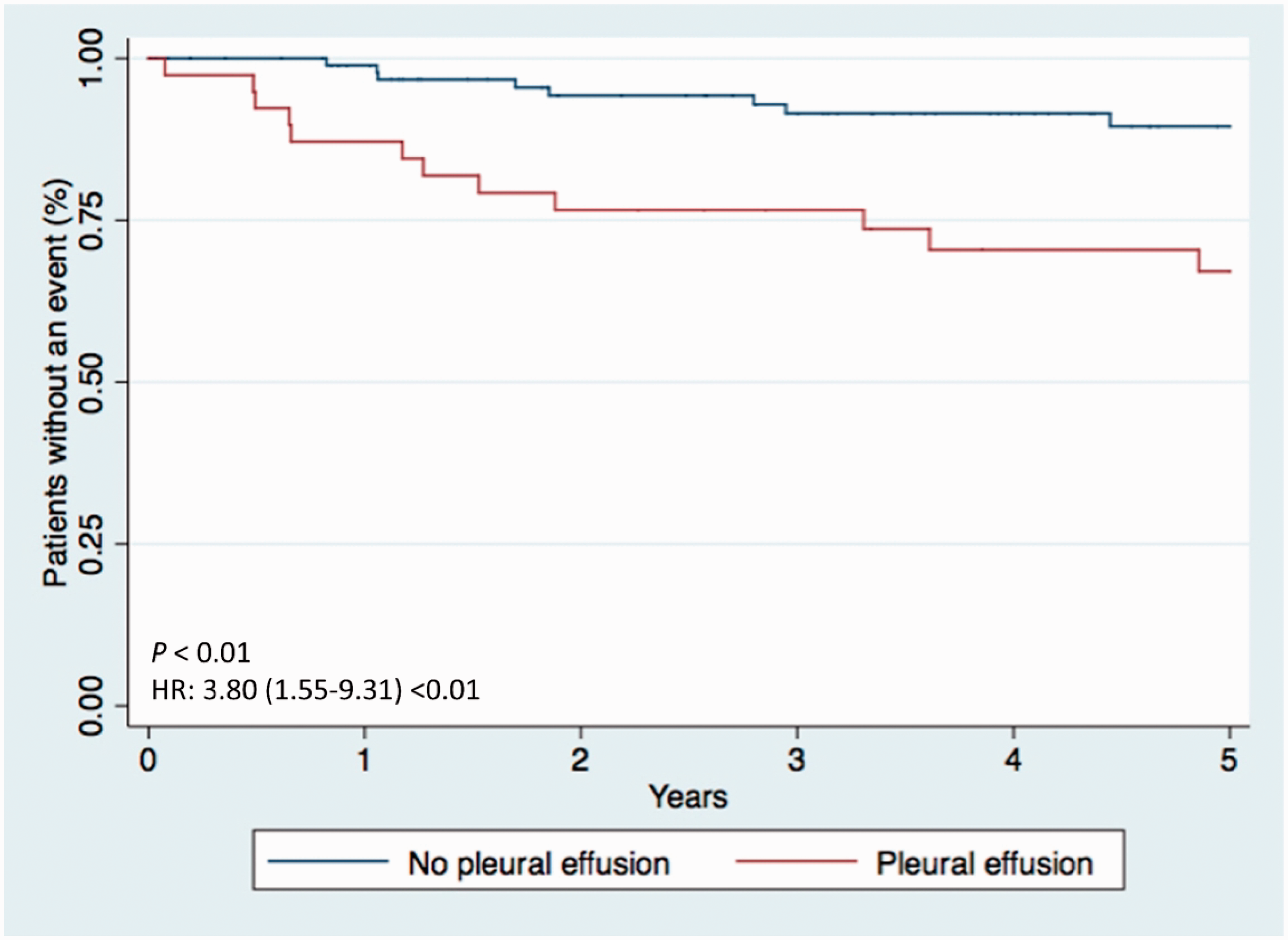
Kaplan–Meier survival curves from time of initial clinic visit based on the development of an incident pleural effusion.
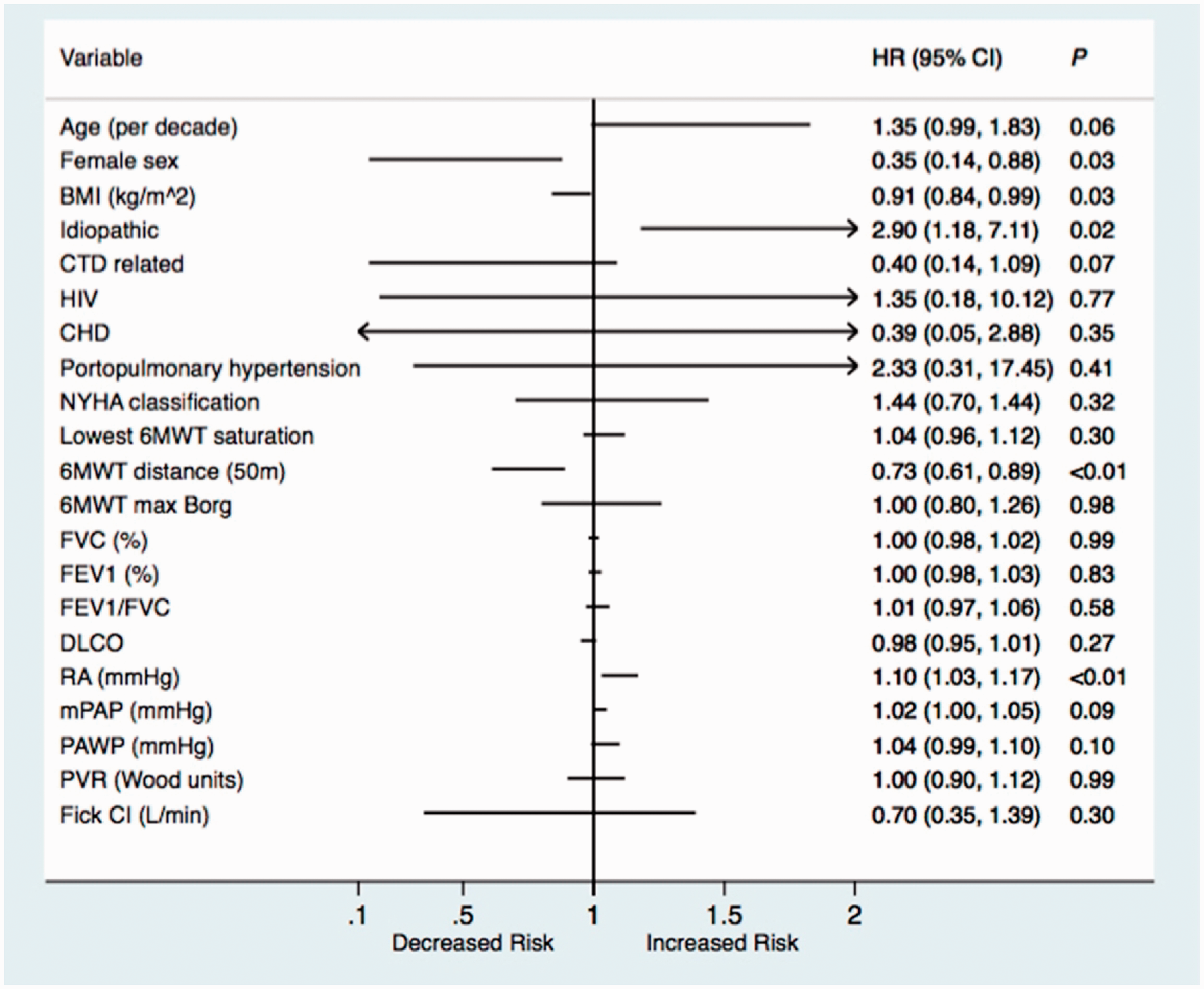
Univariate analysis of predictors of mortality over five years in patients with pulmonary arterial hypertension.
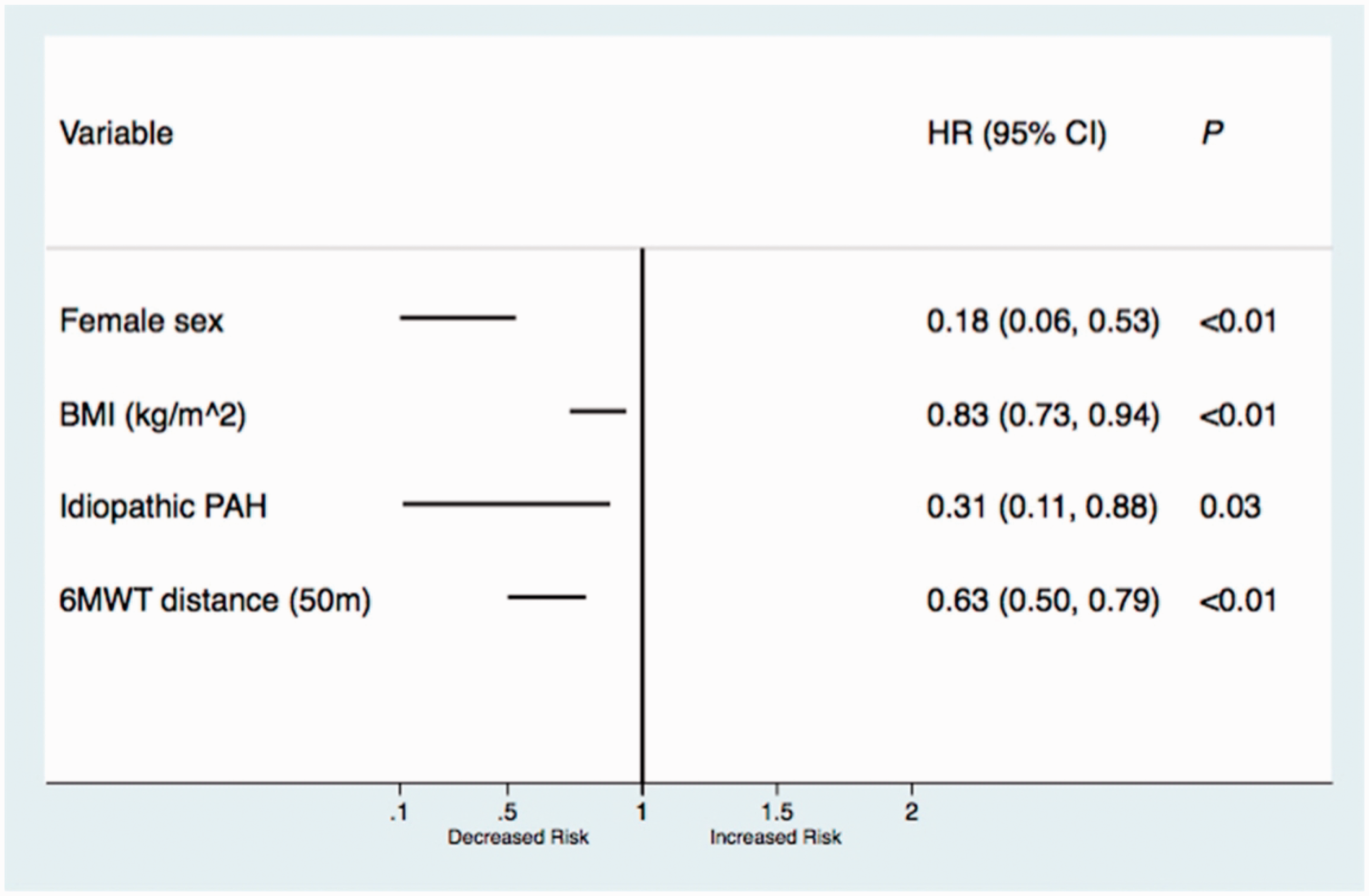
Multivariate predictors of mortality over five years in patients with pulmonary arterial hypertension.
Crude and adjusted hazard ratios for overall survival in the cohort.

Pleural effusions unrelated to PAH removed
Patients with incident pleural effusion were examined to determine a possible alternative etiology. Of the patients with an incident effusion, 20 pleural effusions were judged as likely or more likely related to another cause (Table 3). The most common alternative cause was infection (55%). After exclusion of pleural effusions with another etiology, pericardial effusions co-existed with pleural effusion in two instances (both pleural effusions were bilateral) and ascites co-existed with a bilateral pleural effusion in one instance. There were two additional cases where bilateral pleural effusions were concomitant with both pericardial effusion and ascites.
Survival from the time of initial consultation based on the presence or absence of the development of a pleural effusion, after the removal of those deemed unrelated to PAH, is displayed in Fig. 5. Incident pleural effusion remained associated with attenuated survival in univariate analysis (HR: 5.13; 95% CI: 1.86–14.16) and after multivariate adjustment (HR: 5.88; 95% CI: 1.91–18.06).
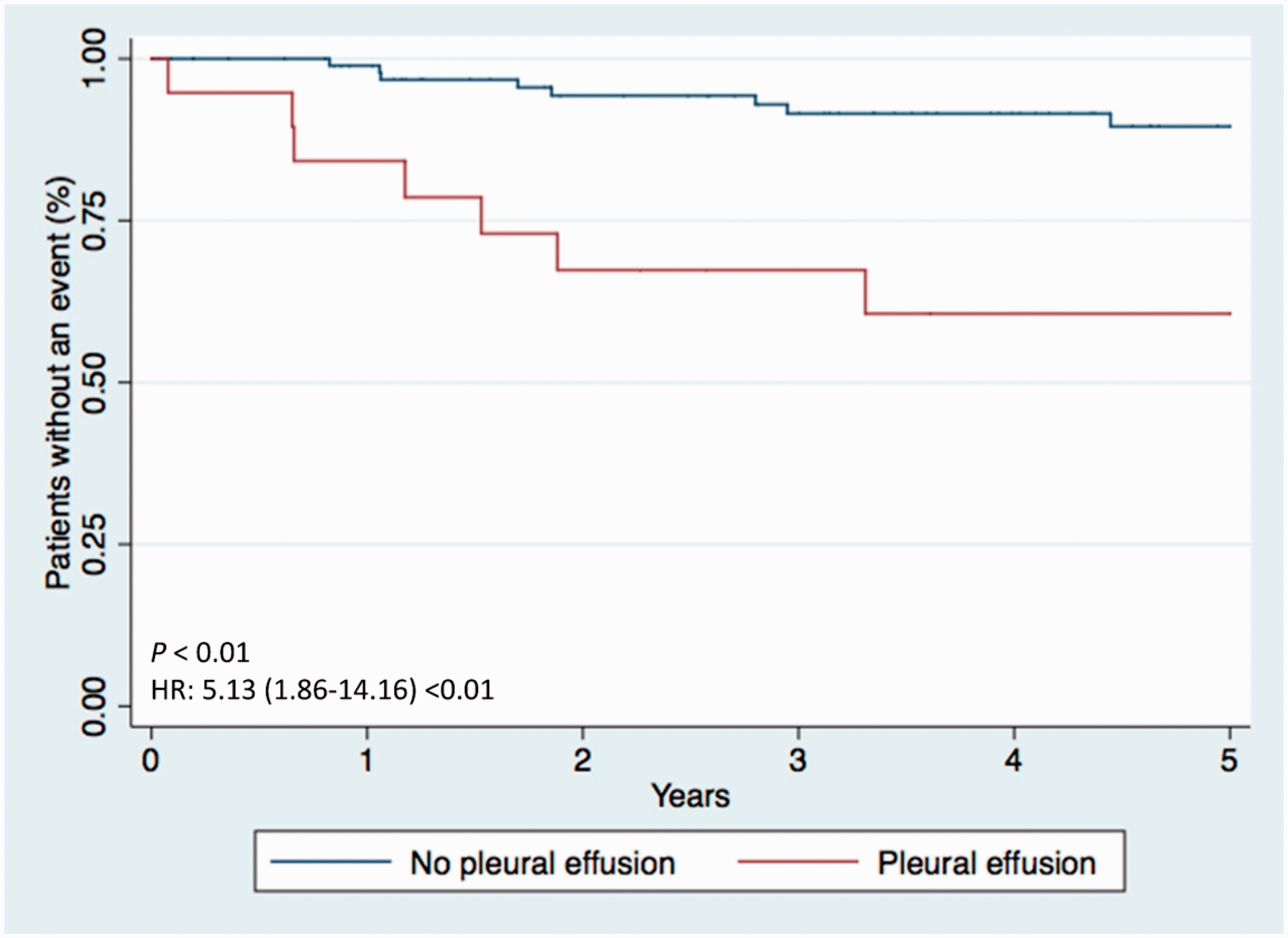
Kaplan–Meier survival curves from time of initial clinic visit based on the development of an incident pleural effusion. Patients with effusions thought unrelated to pulmonary arterial hypertension have been excluded.
Risk in excess of co-existing pericardial effusion
In order to determine if attenuated survival related to the development of a pleural effusion was in excess of the risk related to the development of a pericardial effusion, survival from time of initial consultation based on the presence or absence of incident pleural effusion was stratified by the presence or absence of a concomitant pericardial effusion. After adjustment of the multivariate model, an incident pleural effusion remained a significant predictor of attenuated survival in aggregate (HR: 4.58; 95% CI: 1.43–14.67) and after removal of patients with effusions deemed of alternate etiology (HR: 4.83; 95% CI: 1.45–16.07).
Discussion
Our study examined the clinical course of a cohort of 191 patients with PAH over time. We found the development of a radiographically diagnosed pleural effusion in the five years following initial consultation at our tertiary care center to be common; occurring in 27.5% of patients examined with an incidence rate of 79.2 cases per 1000 patient-years. Of the incident pleural effusions, 48.7% could not be explained by an alternative cause. Thus, effusions attributed solely to PAH occurred at an incidence rate of 38.6 cases per 1000 person-years. Pleural effusions were typically small in size and bilateral in location. The occurrence was similar in frequency between patients with CTD-related PAH and those with non-CTD-related PAH. Furthermore, pleural effusion could not be explained by simple volume overload or as a result of serositis as the majority of the pleural effusions occurred in the absence of concomitant pericardial effusion or ascites. In instances where pleural effusion occurred simultaneously with a pericardial effusion or ascites, their size and anatomic distribution did not suggest that they were secondary to these other fluid collections. 15
The development of a pleural effusion during treatment for PAH was associated with attenuated survival and this finding was independent of clinical factors at the time of diagnosis and the subsequent development of a pericardial effusion. Right atrial hypertension was significantly more common in patients with a pleural effusion at the time of initial evaluation compared to patients without effusions, and notably, mean pulmonary artery pressure and pulmonary capillary wedge pressure was not significantly different between these groups.
Both pleural effusions and pericardial effusions are well-documented complications of congestive heart failure. The development of a pericardial effusion related to congestive heart failure is thought mechanistically to be due to elevated pressure in the coronary sinus, Thebesian veins, or lymphatic circulation from right heart dysfunction. 9 Elevated right heart pressure in the absence of left heart disease has been demonstrated to result in the development of a pericardial effusion in both animal models and in patients with PAH.11,12,16 Alternatively, the development of a pleural effusion in congestive heart failure is theorized to result from the combined imbalance of arterial and venous connections to the visceral and parietal pleural from both systemic and pulmonary systems. Thus, it is conceivable that either left-sided congestive heart failure, right-sided congestive failure, or a combination thereof could result in the development of a pleural effusion. 9 Animal models have noted the development of pleural effusions in systems simulating both left and right heart failure.17,18 Other research suggests that pleural effusions can complicate right heart failure in the absence of left heart disease and this finding is likely to be related to worsening right heart dysfunction.4–7 However, this theory is not universally accepted and the correlation between isolated right heart failure and subsequent pleural effusion remains controversial.9,10 PAH represents an excellent model for the study of the effects of elevated right-sided pressures on the development of both pericardial and pleural effusions. However, coexisting comorbidities including CTD and use of diuretics in patients with PAH contribute to difficulty in examining the relationship between right heart failure and effusions. This study is novel in its goal of precisely quantifying the incidence of pleural effusion in patients with PAH. As demonstrated in prior studies, we found a notable difference between patients with and without a pleural effusion at the time of initial evaluation of an increased severity of right atrial hypertension with no significant difference in pulmonary capillary wedge pressure. Given the small sample size and multiple statistical comparisons, these finding should be interpreted cautiously. However, in the context of these prior studies, it provides additional evidence that right heart failure in the absence of left heart disease is likely a mechanistic cause of pleural effusion in this setting.
A growing body of literature, mostly from multiple large registries and meta-analysis, describes prognostic factors that portend increased risk of death and overall worse outcomes in PAH.19–23 Variables consistently associated with prognosis include New York Heart Association functional class, distance ambulated during 6MWT, brain natriuretic peptide concentration, cardiac index, and right atrial pressure. 24 Other factors, including World Health Organization subgroup and gender are included in commonly utilized risk assessment tools. 25 In our cohort, factors identified in univariate analysis to be associated with mortality included gender, body mass index, idiopathic or familial PAH, distance ambulated during 6MWT, and higher right atrial pressure at the time of initial consultation. Notable differences in our analysis compared to previous results were a lack of association between functional status and mortality in our cohort and the association of increased body mass index with improved prognosis. We suspect the former to potentially reflect referral bias to our tertiary care pulmonary hypertension clinic (i.e. the majority of patients referred to our clinic were New York Heart Association class > 2). The latter association of increased body mass index with improved prognosis is more complex and has been examined by a number of investigators with inconsistent findings.26–28 At this time, in our opinion, a possible link between improved clinical outcomes in obese patients with PAH remains unresolved and may reflect a true association with improved prognosis or potential confounding factors.
The presence of a pleural effusion in PAH has been associated with worse outcomes; however, this association has not been previously examined in detail. 13 Our study provides evidence that the development of a pleural effusion in PAH is related to worse outcomes and this effect is independent of other important clinical risk factors. This may be useful in clinical care for several reasons. First, given that most pleural effusions noted in this cohort were small and can be missed by traditional physical exam maneuvers, pleural effusions are readily detected via other non-invasive diagnostic testing. In this study, pleural effusions were identified via thoracic CT and our findings lend support to the utilization of serial thoracic imaging in the ongoing longitudinal care of patients with PAH. However, concern regarding long-term radiation exposure is a limitation to the application of serial thoracic CTs in clinical practice for many patients. Alternatively, point-of-care ultrasound is a validated, rapid, low-cost, non-invasive means for identifying pleural effusions and could be easily utilized in the longitudinal risk assessment of patients with PAH. 29 Furthermore, given the ease of clinical detection and the correlation to decreased overall survival noted in this population, the development of a pleural effusion may signify patients with PAH at higher risk of clinical deterioration and provide actionable therapeutic information. It is noteworthy that the incidence of pericardial effusion was less frequent than that of pleural effusion in this cohort. We speculate that in some instances, the development of a pericardial effusion may be a late manifestation of worsening right ventricular function and the development of a pleural effusion may represent an earlier sign of clinical deterioration. Further study is therefore important to determine if therapeutic changes at the time of effusion detection may translate to improved clinical outcomes.
This analysis has a few limitations. The study was retrospective and occurred within a single comprehensive care pulmonary hypertension center. Patients were followed after initial consultation at this center and many had already begun treatment for PAH at the time of referral. Given this, our results may have been affected by referral and survivorship biases. Patients included in the analysis were subject to real-world conditions and the effect of co-existing acute and chronic illness. Likewise, heterogeneous practice patterns related to clinical monitoring and treatment, and changes in therapy over time due to evolving clinical practice in the management of PAH may have influenced the results. Patient total body fluid volume and nutritional status at the time of serial thoracic imaging could not be standardized given the retrospective nature of the study and was inconsistently evaluated via laboratory means during the study period. Furthermore, the performance of thoracic CT was also not standardized. Arguably, many of the patients had diagnostic imaging ordered for cause; therefore, our estimate of the incidence of pleural effusions might be artificially inflated due to an “enriched” denominator. Additionally, though effusions were noted to be common, pleural fluid sampling was rare. Only one patient underwent a diagnostic thoracentesis, thus, these data were not available to aid in determining the etiology of pleural fluid or for mechanistic analysis of the relation of pleural effusion to PAH. We suspect that a lack of sampling in clinical practice was related to the overall small size of the majority of encountered effusions. Despite these limitations, we note that this is the largest systematic investigation of the incidence of pleural effusions in PAH to date and thus, believe the results are of clinical importance.
In conclusion, incident pleural effusions are common in patients with PAH. They are most frequently small in size, bilateral in location, and their presence is associated with a decreased overall survival. Likely, the development of a pleural effusion is related to progressive PAH and subsequent elevated right-sided cardiac pressures. The associated attenuated survival appears independent of the risk associated with a new pericardial effusion. Further prospective study is necessary to determine if therapeutic changes at the time of effusion onset could translate into improved survival.
Footnotes
Contributorship
A.C., V.K., and C.S.K. designed and implemented the study. A.V. and H.R. collected the clinical data. S.D.N., K.A., S.A., A.B., A.S., A.W.B., and O.A.S. contributed substantially to project design, data collection, data analysis, interpretation, and manuscript preparation. All authors contributed to analysis and review of the final manuscript. The views expressed in this article are those of the author and do not reflect the official policy of the Department of Army/Navy/Air Force, Department of Defense, or U.S. Government.
Ethical approval
The study was approved by the Institutional Review Board (IRB# 17-2833) at Inova Fairfax Hospital, Falls Church, VA, USA.
Guarantor
Abhimanyu Chandel.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
