Abstract
Patients with pulmonary arterial hypertension associated with connective tissue disease (PAH-PAH-CTD) such as systemic sclerosis (SSc) have a poorer response to treatment and increased mortality compared with patients with idiopathic PAH. Current treatment options for PAH-CTD include prostanoids, phosphodiesterase type-5 inhibitors (PDE-5i), endothelin receptor antagonists, and the soluble guanylate cyclase stimulator riociguat. In this case series, we describe three patients with PAH-CTD related to limited scleroderma who were switched from a PDE-5i to riociguat due to insufficient clinical response. The switch to riociguat was associated with an improvement in respiratory and hemodynamic parameters and a favorable tolerability profile. These cases demonstrate that switching to riociguat is a therapeutic option in patients with PAH-CTD who have not achieved a satisfactory clinical response to a PDE-5i.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) remains a progressive disease with high associated morbidity and mortality. 1 Fortunately, a variety of options are now available to treat PAH, including prostanoids, phosphodiesterase type 5 inhibitors (PDE-5i), soluble guanylate cyclase stimulators (sGCS), and endothelin receptor antagonists (ERA). 2 Because of cost and ease of use, PDE-5is are often utilized as first-line therapy, with additional medications added sequentially or as part of upfront combination therapy.3,4 Unfortunately, significant heterogeneity exists in terms of treatment response, such that alternative therapeutics with different modes of action are often required. Riociguat, the first-in-class sGCS, was approved in 2013 by the U.S. Food and Drug Administration for the treatment of adults with PAH (World Health Organization [WHO] Group 1) and in those with inoperable or persistent/recurrent chronic thromboembolic pulmonary hypertension (CTEPH; WHO Group 4).5,6 Riociguat targets the nitric oxide (NO) pathway to enhance cyclic guanosine monophosphate (cGMP) production; however, unlike PDE-5i, which inhibits degradation of cGMP, riociguat sensitizes sGC to endogenous NO by stabilizing NO–sGC binding and also directly stimulates sGC independent of NO via a different binding site.7,8 As NO synthesis and signaling are reduced in patients with PAH, and continue to decline as disease progresses, riociguat may be an attractive substitute for patients not responding clinically to PDE-5i therapy.9–11 Use of riociguat with a PDE-5i is contraindicated.5,6
Heterogeneity to PAH-specific therapy and insufficient response to therapy can be particularly troublesome in PAH associated with connective tissue disease (PAH-CTD), especially if due to the scleroderma spectrum of diseases (PAH-SSc). 12 Compared with idiopathic PAH (IPAH) patients, individuals with PAH-SSc have increased mortality and a poorer response to treatment. 12
In the subset of PAH-CTD patients naïve to PDE-5i therapy in the Pulmonary Arterial Hypertension Soluble Guanylate Cyclase-Stimulator Trial-1 (PATENT-1; n = 111, 66 with SSc), the least-squares mean treatment difference in six-minute walk distance (6MWD) at 12 weeks with riociguat (maximum 2.5 mg TID) was 28 m compared with 36 m in the overall study population. 13 In patients with PAH-SSc receiving riociguat (n = 43), the mean increase in 6MWD at week 12 was 4 ± 43 m, but those patients receiving placebo (n = 16) showed a larger decrease of –37 ± 20 m. 13
In the PATENT-2 extension trial, at two years, mean 6MWD increased by 25 m from PATENT-1 baseline in patients with PAH-CTD (n = 70), while in the overall population, 6MWD increased by 47 m. 13 Improvements in 6MWD were largely maintained in the PAH-SSc subpopulation as well. 13
Survival rates at one and two years were comparable for patients with PAH-CTD relative to the overall population: PAH-CTD = 97% (95% confidence interval [CI] = 90–99) at one year and 93% (95% CI = 85–97) at two years; overall population = 97% (95% CI = 95–98) at one year and 93% (95% CI = 90–95) at two years. The safety profile of riociguat in PAH-CTD patients was similar to that in the overall population. 13
Whether riociguat can be used as a substitute for a PDE-5i, or even preferentially, in patients with PAH-SSc is currently unknown; furthermore, the recently completed RESPITE clinical trial (NCT02007629) 14 examining the effects of switching a PDE-5i to riociguat excluded patients with PAH-SSc as response to therapy is often mitigated in this subpopulation.15,16 In the current case series, we report the effect of switching to riociguat in three patients with PAH-SSc who had insufficient response to treatment with a PDE-5i.
Case descriptions
Case 1: Key measurements from serial RHC.
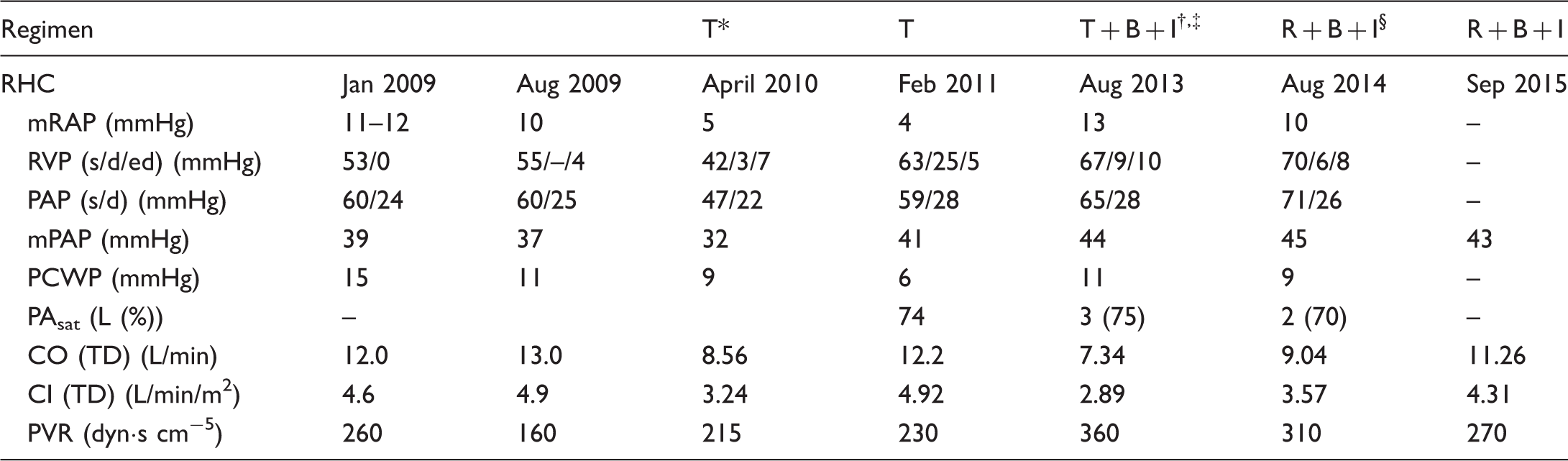
*Tadalafil was initiated in late 2009.
Bosentan was initiated after February 2011 RHC findings.
Imatinib was initiated after April 2013 diagnosis of CML.
Tadalafil was replaced with riociguat in November 2013 due to worsening hemodynamics.
B, bosentan; CI, cardiac index; CO, cardiac output; I, imatinib; mPAP, mean pulmonary artery pressure; mRAP, mean right atrial pressure; PAsat, pulmonary arterial oxygen saturation; PAP, pulmonary arterial pressure; PCWP, pulmonary capillary wedge pressure; PVR, pulmonary vascular resistance; R, riociguat; RHC, right-heart catheterization; RVP, right ventricular pressure; s/d/ed, systolic/diastolic/end diastolic; T, tadalafil; TD, thermodilution.
Case 1
A 33-year-old woman presented with Raynaud’s phenomenon in 2003. In 2005, she developed ischemia of the second digit on her right hand and was diagnosed with limited scleroderma (antinuclear antibody [ANA] positive, 1:1280; centromeric pattern; centromere Ab positive; topoisomerase 1 [SCL-70] negative). Three years later, at age 38 years, she developed progressive dyspnea on exertion (DOE)/shortness of breath (SOB) affecting exercise tolerance and activities of daily living; imaging revealed minimal interstitial lung disease and she required supplemental O2 (WHO functional class [FC] III). The results of an echocardiogram and computed tomography pulmonary angiogram in January 2009 showed mild right ventricular (RV) enlargement with depressed systolic function, dilated inferior vena cava, pleural effusion, diffuse mosaic attenuation pattern, interlobular septal thickening, and a pulmonary arterial systolic pressure (PAsp) of 58 mmHg; RHC revealed elevated right atrial pressure (RAP), right ventricular pressure, PAP, pulmonary capillary wedge pressure (PCWP), cardiac output (CO), and PVR (Table 1). PAH-CTD was diagnosed and the patient was treated aggressively with prednisone and mycophenolate mofetil due to SSc-associated skin tightening. An RHC in August 2009 showed improved cardiac output and PVR, but the patient remained WHO FC II–III with symptoms requiring supplemental oxygen at 2–3 L/min.
Tadalafil was initiated in late 2009 with stable-to-improved respiratory status (WHO FC II) and a RHC in April 2010 showed hemodynamic improvement with a cardiac index (CI) of 3.24 L/min/m2 (decreased from 4.9 L/min/m2); a slight increase in PVR was seen (215 mmHg versus 160 mmHg) and had improved compared with baseline (PVR = 260 mmHg). A follow-up echocardiogram in December 2010 showed an increase in RV systolic pressure (RVSP) (59 mmHg versus 45 mmHg in January 2010) and a hypokinetic right ventricle; an RHC in February 2011 showed slight increases in PAP and PVR relative to April 2010 (Table 1). Due to worsening symptoms (WHO FC III) and digital ulcerations, bosentan was added in 2011. Her respiratory status initially improved (WHO FC II–III), but pulmonary function tests (PFTs) in April 2012 showed a severe decrease in carbon monoxide diffusion capacity (DLCO; 8.6 mL/min/mmHg). A repeat echocardiogram in January 2013 showed an increase in left ventricular ejection fraction (LVEF) and flow reversal into the hepatic veins; the patient was diagnosed with chronic myeloid leukemia (CML) in April 2013 and treated with imatinib with an excellent response (remission). A follow-up RHC in August 2013 showed increases in PAP and PVR despite therapy with two approved PAH drugs (bosentan and tadalafil) and imatinib, which has been investigated for treatment of PAH (despite significant improvements in hemodynamics, it was not approved to treat PAH due to adverse events). 19 In November 2013, tadalafil was switched to riociguat (titrated to 2.5 mg TID without significant side effects) and the patient’s respiratory status improved (WHO FC II). Further evaluation in August 2014 showed decreased PVR with increased CO/CI); although the CO was elevated, the CI was actually low after accounting for the patient’s body surface area. She remained stable to slightly improved at the most recent evaluation in September 2015.
Case 2
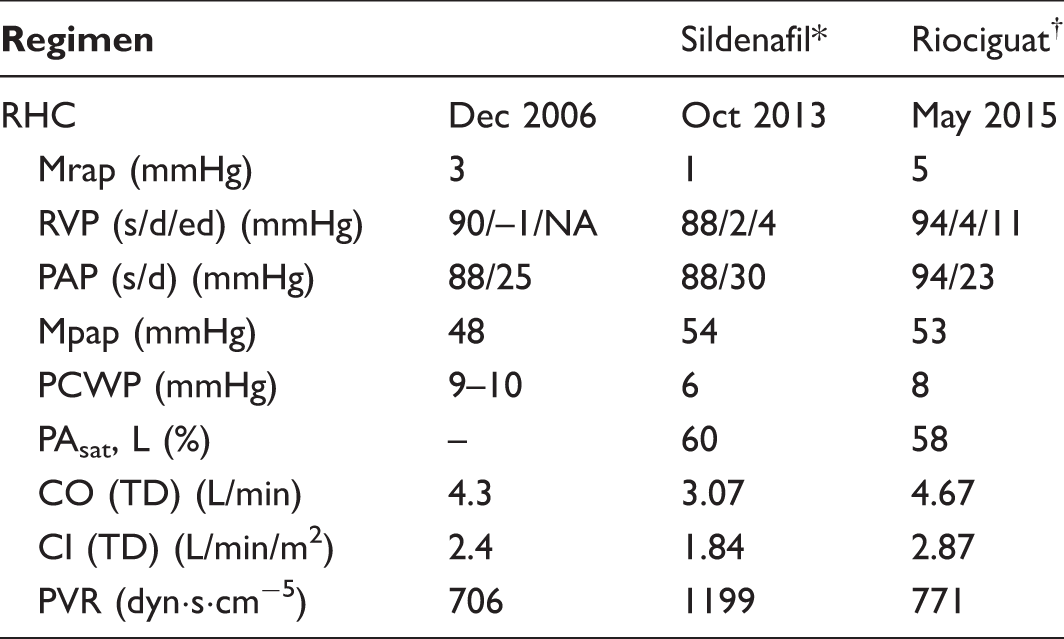
Sildenafil initiated in March 2007.
Sildenafil was replaced with riociguat in October 2013.
CI, cardiac index; CO, cardiac output; mPAP, mean pulmonary artery pressure; mRAP, mean right atrial pressure; NA, not available; PAsat, pulmonary arterial oxygen saturation; PAP, pulmonary arterial pressure; PCWP, pulmonary capillary wedge pressure; PFT, pulmonary function test; PVR, pulmonary vascular resistance; RHC, right-heart catheterization; RVP, right ventricular pressure; s/d/ed, systolic/diastolic/end diastolic; TD, thermodilution.
Case 3: Key measurements from serial RHC.
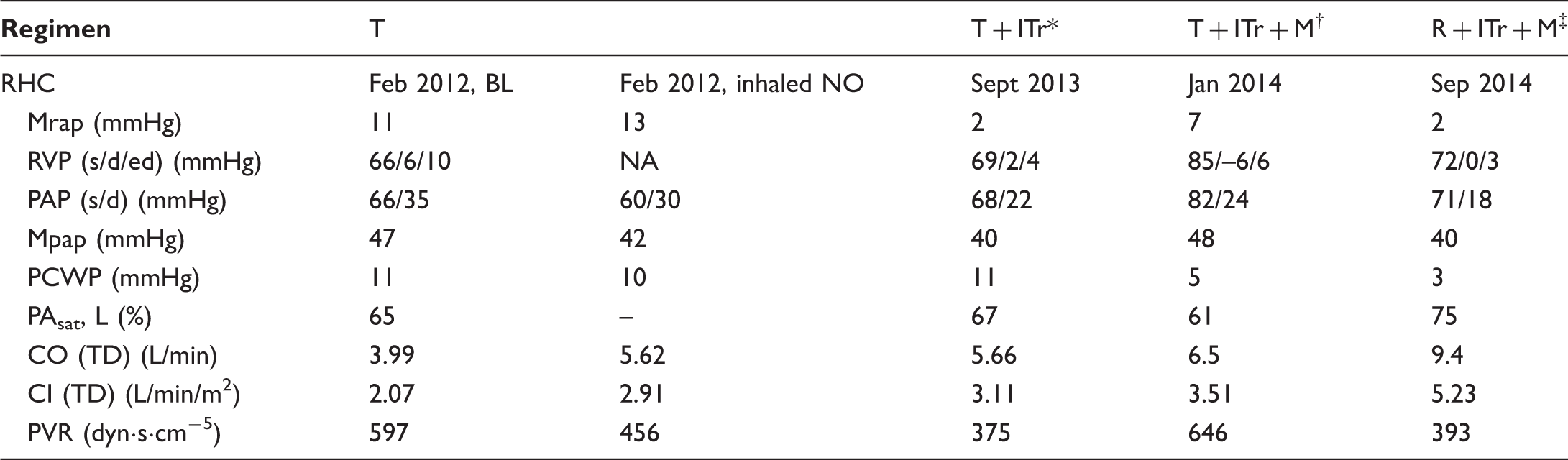
Inhaled treprostinil was added to tadalafil August 2012.
Macitentan was added to tadalafil and inhaled treprostinil in October 2013.
Tadalafil was replaced with riociguat in February 2014. Inhaled treprostinil and macitentan were maintained.
CI, cardiac index; CO, cardiac output; ITr, inhaled treprostinil; M, macitentan; mPAP, mean pulmonary artery pressure; mRAP, mean right atrial pressure; NA, not available; PAsat, pulmonary arterial oxygen saturation; PAP, pulmonary arterial pressure; PCWP, pulmonary capillary wedge pressure; PFT, pulmonary function test; PVR, pulmonary vascular resistance; RHC, right-heart catheterization; R, riociguat; RVP, right ventricular pressure; s/d/ed, systolic/diastolic/end diastolic; T, tadalafil; TD, thermodilution.
Case 3
A 67-year-old woman was diagnosed with PAH-CTD due to limited SSc (ANA+ 1:80, centromeric pattern, centromere Ab+, SCL-70 negative, RF+) in February 2012. Over the previous year, she had been experiencing DOE/SOB and required 2 L/min continuous supplemental O2. She had advanced FC III–IV symptoms at presentation as confirmed by PFTs (FVC = 73%, FEV1 = 70%, FEV1/FVC = 74%, TLC = 80%, DLco = 37%) and RHC, and severe RV dysfunction (RV ejection fraction 29% on cardiac magnetic resonance imaging [MRI]) (Table 3). She also had concomitant, transient, LV dysfunction (LVEF 35%) which was felt to be secondary to RV:LV interaction in the context of massive RV dilatation (RV end diastolic volume 283 mL). Coronary angiography did not reveal any significant coronary disease and MRI showed no edema or contrast enhancement of the left ventricle other than at the RV insertion points. Given the advanced symptoms and an elevated Registry to Evaluate Early and Long-term PAH Disease Management (REVEAL) score, she was initially treated with rapid titration of intravenous (IV) treprostinil, which resulted in elevated PCWP of 24 mmHg. IV treprostinil was discontinued, and she was subsequently started on tadalafil while the LV dysfunction was treated. Repeat echocardiogram in May 2012 revealed a normal LVEF of 65%. Inhaled treprostinil (9 breaths QID) was added to therapy six months later after LVEF had improved. Although she improved from February 2012 (WHO FC III, 6MWD = 253 m, N-terminal pro b-type natriuretic peptide [NT-proBNP] = 6112 pg/mL) to January 2013 (WHO FC II, 6MWD = 366 m, NT-proBNP = 823 pg/mL), her clinical status began to decline early in 2013 (FC III, 6MWD = 219 m with lowest O2 saturation 83%, NT-proBNP = 1361 pg/mL). RHC demonstrated persistent PAH (mPAP = 40 mmHg). Because of her previous inability to tolerate IV therapy, macitentan was added in October 2013. Although this led to some improvement (6MWD = 292 m without significant ambulatory desaturation), she remained WHO FC III with an increased NT-proBNP of 1501 pg/mL and an elevated REVEAL score of 12. RHC again demonstrated worsening PAP and PVR (Table 3). Because the patient was reluctant to add parenteral therapy, tadalafil was switched to riociguat (titrated to 2.5 mg TID) in February 2014. Following this change in therapy, WHO FC improved to FC II, 6MWD stabilized at 302 m, and, most notably, PVR decreased while CI increased. In terms of adverse effects, the patient experienced headache as well as reduction in blood pressure, requiring dose reduction in her antihypertensive agents. The patient remains on riociguat, macitentan, and inhaled treprostinil, which was increased to 12 breaths QID in mid-2015.
Discussion
Recently presented data from the RESPITE trial have shown that replacing PDE-5i therapy with riociguat may be of benefit in patients with PAH not responding adequately to their current therapy. 14 Treatment with riociguat was associated with a mean improvement in 6MWD of 31 m over 24 weeks, a mean change of –347 pg/mL in NT-proBNP and improved WHO FC in 54% of patients who switched to riociguat from a PDE-5i. Although patients with CTD were excluded from the study, results from phase 3 trials have already demonstrated efficacy in this patient population. 13
In these three cases, substitution with riociguat was associated with clinical and hemodynamic improvements similar to those seen in the PATENT-1 and PATENT-2 PAH-CTD and PAH-SSc subpopulations, although improvement in these subpopulations was less pronounced than in patients with IPAH. Previous studies in patients with PAH-CTD and PAH-SSc have also demonstrated decreased response to therapy in this patient population.15,16 In PATENT-1 and -2, patients with PAH-SSc randomized to placebo experienced significant deterioration in 6MWD—more so than the PAH-CTD population—whereas those receiving riociguat saw sustained improvement at the two-year cutoff. 13 Stabilization of 6MWD may be indicative of response to therapy in specific PAH subpopulations given the underlying pathophysiology of CTD and the effect of musculoskeletal involvement on exercise capacity and overall mobility.15,20 The REVEAL registry identified unique baseline risk factors associated with mortality in the SSc population, among them male sex, age > 60 years, systolic blood pressure < 110 mmHg, 6MWD < 165 m, mean right atrial pressure > 20 mmHg within one year, and PVR > 2560 dynċsċcm−5. 20
Also of potential interest to this patient population, sGC stimulation has been shown to inhibit transforming growth factor (TGF)-β–induced collagen release and fibroblast-to-myofibroblast differentiation in dermal fibroblasts from SSc patients and healthy volunteers.21,22 sGC stimulation also inhibited dermal fibrosis in mouse models of SSc (bleomycin-induced dermal fibrosis, an inflammation-dependent model; Tsk-1 mice, an inflammation-independent model; and mice expressing constitutively active TGF-β receptor I) as assessed by skin thickening, hydroxyproline content, and myofibroblast counts.21,22 Thus, the effect of riociguat in these patients, compared with a PDE-5i, might be due to its differential mechanism of augmenting the effect of NO and/or, to some degree, by the antifibrotic effect observed in preclinical models of SSc. This aspect of sGC stimulation is currently under further investigation in the ongoing RISE-SSc trial (NCT02283762).
In sum, these three cases demonstrate that in patients with PAH-CTD who have an inadequate response to a PDE-5i, switching to riociguat has the potential to improve clinical and hemodynamic parameters and showed a favorable tolerability profile in clinical trials and in our select patients.
Footnotes
Conflict of interest
AR reports personal fees from Bayer, United Therapeutics, and Actelion. RB reports speaker fees from Actelion and United Therapeutics. HF reports grants and personal fees from Actelion, Gilead, and United Therapeutics/Lung and personal fees from Bayer and Bellerophon.
Funding
Article processing charges for this article were funded by Bayer HealthCare Pharmaceuticals, Whippany, NJ, USA. Medical writing and editing assistance were provided by Adelphi Communications, LLC, and were funded by Bayer Healthcare.
