Abstract
The goal of treatment in patients with pulmonary arterial hypertension is to achieve a low risk status, indicating a favorable long-term outcome. The REPLACE study investigated the efficacy of switching to riociguat in patients with pulmonary arterial hypertension and an insufficient response to phosphodiesterase-5 inhibitors. In this post hoc analysis, we applied the REPLACE composite endpoint of clinical improvement to the placebo-controlled PATENT-1 study of riociguat in pulmonary arterial hypertension and its long-term extension, PATENT-2. Clinical improvement was defined as ≥2 of the following in patients who completed the study without clinical worsening: ≥10% or ≥30 m improvement in 6-minute walking distance; World Health Organization functional class I or II; ≥30% decrease in N-terminal prohormone of brain natriuretic peptide. At PATENT-1 Week 12, patients treated with riociguat were more likely to achieve the composite endpoint vs. placebo (P < 0.0001), with similar results in pretreated (P = 0.0189) and treatment-naïve (P < 0.0001) patients. Achievement of the composite endpoint at Week 12 was associated with a 45% reduction in relative risk of death and a 19% reduction in relative risk of clinical worsening in PATENT-2. Overall, these data suggest that use of the REPLACE composite endpoint in patients with pulmonary arterial hypertension is a valid assessment of response to treatment.
Keywords
Introduction
Pulmonary arterial hypertension (PAH) is a life-threatening disease characterized by increased pulmonary vascular resistance leading to progressive vascular remodeling, which can ultimately lead to right heart failure and death. 1 , 2 Current approved treatments for PAH (World Health Organization classification Group 1) include the soluble guanylate cyclase stimulator riociguat, phosphodiesterase type 5 inhibitors, prostanoids, prostacyclin receptor agonists, and endothelin receptor antagonists. 1 , 3 While cure is unlikely, the goal of treatment for patients with PAH is improvement to or maintenance of a low risk status as defined by clinically relevant parameters. 1 , 3
The 12-week phase 3 PATENT-1 study evaluated the efficacy and safety of riociguat in patients with PAH who were treatment-naïve or pretreated with endothelin receptor antagonists or non-parenteral prostanoids. Results showed that riociguat treatment significantly improved exercise capacity, functional status, time to clinical worsening, and hemodynamic parameters compared with placebo. 4 The benefits of treatment, as well as safety and tolerability, were maintained at two years in the long-term extension, PATENT-2. 5 , 6 The ongoing phase 4 REPLACE study will evaluate the efficacy of switching to riociguat in patients with PAH and an insufficient response to phosphodiesterase type 5 inhibitors treatment compared with the remaining patients who were on phosphodiesterase type 5 inhibitors. 7 The primary outcome of REPLACE is a composite endpoint of clinical improvement, which is assessed in all patients at Week 24. Here, we describe the REPLACE endpoint and apply it to the PATENT study population for validation as an additional tool to assess efficacy in future PAH clinical trials. The objective of this analysis was to assess whether the REPLACE composite endpoint could differentiate between patients who received riociguat and those who received placebo, including subgroups of treatment-naïve or pretreated patients, in PATENT-1. Additionally, we evaluated whether achievement of the REPLACE endpoint in PATENT-1 was associated with long-term outcomes in PATENT-2.
Methods
Study design and patients
This was a post hoc analysis of data from the randomized, placebo-controlled, phase 3 PATENT-1 study (NCT00810693). The study design and results have been published previously. 4 In brief, patients were randomized to receive placebo, riociguat up to 1.5 mg (capped exploratory dose), or riociguat up to 2.5 mg three times daily for 12 weeks. The primary endpoint was change in 6-minute walking distance (6MWD) at Week 12 vs. placebo. PATENT was conducted in accordance with Good Clinical Practice Guidelines and the Declaration of Helsinki, and the institutional review board at each participating center approved the protocol. All patients provided written, informed consent. Patients who completed PATENT-1 without ongoing riociguat-related adverse events were eligible to enter the PATENT-2 open-label extension in which former placebo patients received riociguat up to 2.5 mg three times daily, while patients who received riociguat in PATENT-1 continued to receive their optimum dose as previously determined. 8
REPLACE composite endpoint
The primary outcome of REPLACE is a composite endpoint of clinical improvement in patients who completed the study without clinical worsening (death, hospitalization due to worsening PAH, disease progression). Clinical improvement was defined by the presence of at least two of the following three criteria at the end of the 24-week study period: ≥10% or ≥30 m improvement in 6MWD; World Health Organization functional class (WHO FC) I or II; ≥30% decrease in N-terminal prohormone of brain natriuretic peptide. 7
Statistical analysis
Application of the REPLACE endpoint to PATENT-1
The REPLACE endpoint was applied to patients in the 2.5 mg–maximum and placebo arms of PATENT-1; patients in the exploratory 1.5 mg–maximum arm were excluded. Additionally, as the REPLACE endpoint was designed for patients in WHO FC III, the endpoint was separately applied to the subgroup of patients in the 2.5 mg–maximum and placebo arms who were in WHO FC III at PATENT-1 baseline.
For this post hoc analysis, the criteria for clinical worsening from PATENT-1 were applied, defined as any of: death, heart or lung transplantation, atrial septostomy, admission to hospital due to worsening of PAH (or start of parenteral prostanoids), start of new specific PAH treatment or modification of existing prostanoid treatment (increase in dosage or frequency of existing prostanoid therapy, or initiation of an intravenous prostanoid), persistent decrease of > 15% from baseline in 6MWD, or persistent worsening of WHO FC. Any patient who experienced a clinical worsening event in PATENT-1 was considered not to have achieved the REPLACE endpoint, regardless of other factors.
The ability of the REPLACE endpoint to differentiate between the treatment arms in PATENT-1 was assessed using Fisher’s exact test. Data were based on observed cases and no imputations were made.
Association between achievement of the REPLACE endpoint and long-term outcomes in PATENT-2
In PATENT-2, associations between achieving the REPLACE composite endpoint and survival (both overall survival and clinical worsening-free survival) were assessed using Kaplan–Meier and Cox proportional hazards analyses. The analysis was conducted on patients in the 2.5 mg–maximum arm of PATENT-1 who participated in PATENT-2, and as the REPLACE composite endpoint was designed for patients in WHO FC III at baseline, those not in WHO FC III at PATENT-1 baseline were excluded.
As freedom from clinical worsening is a component of the REPLACE endpoint, clinical worsening in PATENT-1 was excluded from the analyses of survival and clinical worsening in PATENT-2 to avoid bias in favor of responders. To account for potential bias due to differences at baseline, a propensity score model was applied using age, gender, race, smoking status, weight, height, 6MWD, N-terminal prohormone of brain natriuretic peptide, pretreatment, and alcohol use as well as treatment assignment as covariates. The overlap weights method was used for propensity score adjustment. 9
Results
Patients
This analysis included 380 patients who participated in the PATENT-1 study, comprising 254 patients who received riociguat 2.5 mg–maximum and 126 patients who received placebo. Of the 380 patients, 366 (96%) were in WHO FC II or III. In total, 167 patients (44%) were pretreated with endothelin receptor antagonists (primarily bosentan), and 27 (7%) had received prostanoid therapy (primarily inhaled iloprost), including three patients who received both an endothelin receptor antagonist and a prostanoid. The remaining 189 patients (50%) had not received any prior treatment for PAH. 4
Long-term outcomes were assessed in 340 patients who completed PATENT-1 and entered PATENT-2, of whom 231 patients were from the riociguat 2.5 mg–maximum arm and 109 patients were from the placebo arm of PATENT-1 who were in WHO FC III at PATENT-1 baseline.
Achievement of the REPLACE composite endpoint in PATENT-1
At PATENT-1 Week 12, 126 patients treated with riociguat (50%) and 32 patients treated with placebo (25%) achieved the REPLACE composite endpoint (Fig. 1a). The percentages of patients achieving each individual component of the REPLACE composite endpoint are provided in Supplemental Tables 1 to 3. Ninety-nine riociguat-treated patients (39%) and 73 placebo-treated patients (58%) did not improve (i.e. remained stable), while three patients (1%) and eight patients (6%), respectively, experienced clinical worsening events (Fig. 1a). Two riociguat-treated patients (1%) and three placebo-treated patients (2%) died during PATENT-1.
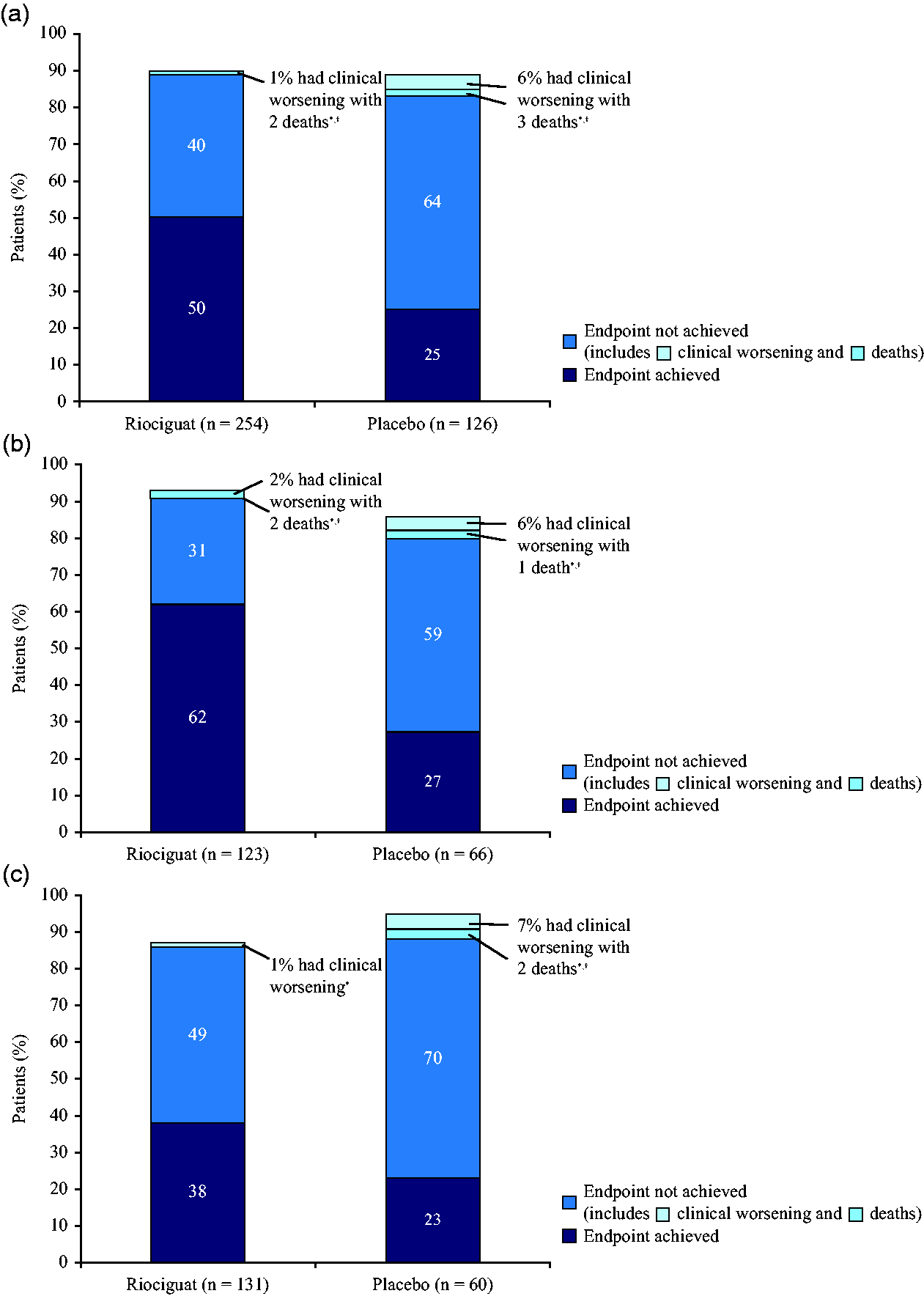
Proportion of patients achieving the REPLACE composite endpoint at Week 12 in PATENT-1 in (a) the 2.5 mg–maximum and placebo arms of the overall population, (b) treatment-naïve patients, and (c) pretreated patients. (a) *Patients who experienced clinical worsening are a subgroup of those who did not achieve the endpoint. †Deaths are a subgroup of clinical worsening. Bars do not total 100 as data for 26 patients (10%) are missing from the riociguat group; data for 13 patients (10%) are missing from the placebo group.
Similar results were observed in the subgroups of treatment-naïve and pretreated patients, although a higher proportion of treatment-naïve patients achieved the REPLACE composite endpoint during riociguat treatment. Of the treatment-naïve subgroup, 76 riociguat-treated patients (62%) and 18 placebo-treated patients (27%) achieved the REPLACE composite endpoint (Fig. 1b). Of the pretreated group, 50 patients (38%) and 14 patients (23%), respectively, achieved the composite endpoint (Fig. 1c).
In patients who were in WHO FC III at baseline, 51 patients receiving riociguat (36%) achieved the REPLACE composite endpoint at PATENT-1 Week 12 compared with five placebo-treated patients (9%) (Fig. 2). In treatment-naïve and pretreated patients who were in WHO FC III at baseline, similar results were observed, with a higher number of treatment-naïve patients achieving the REPLACE composite endpoint. Of the treatment-naïve subgroup, 27 riociguat-treated patients (49%) and four placebo-treated patients (16%) achieved the composite endpoint (Fig. S1a), while 24 riociguat-treated patients (28%) and one placebo-treated patient (3%) in the pretreated subgroup achieved the composite endpoint (Fig. S1b).
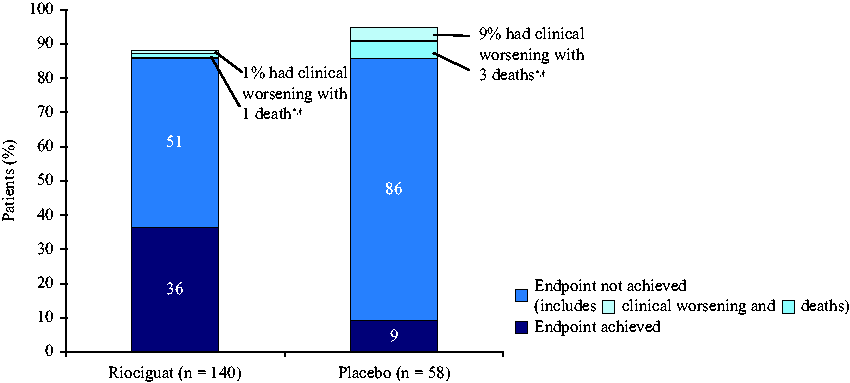
Proportion of patients in WHO FC III at baseline achieving the REPLACE composite endpoint at Week 12 in PATENT-1 in the 2.5 mg–maximum and placebo arms of the overall population.
Odds ratio analysis showed that patients treated with riociguat were more likely to achieve the REPLACE composite endpoint compared with those receiving placebo (P < 0.0001) (Fig. 3). This statement was also true for patients in WHO FC III at baseline (P < 0.0001), and in both the treatment-naïve (P < 0.0001) and pretreated (P = 0.0189) subgroups (Fig. 3).
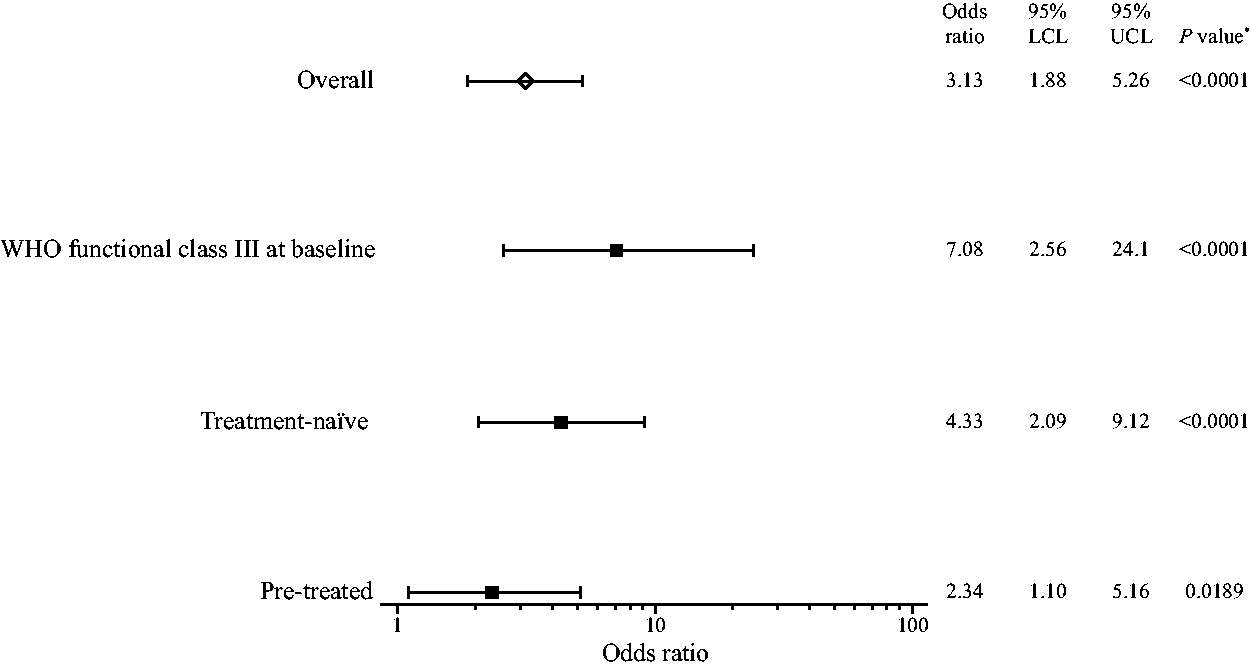
Odds ratios for achievement of the REPLACE composite endpoint at Week 12 in PATENT-1. Data were based on observed cases with no imputation. *P value is based on Fisher’s exact test.
Association of achievement of the REPLACE composite endpoint with long-term outcomes in PATENT-2 in patients receiving riociguat and in WHO FC III at baseline
Cox proportional hazards analyses with propensity scoring of riociguat-treated patients in WHO FC III at baseline showed that achievement of the REPLACE composite endpoint at Week 12 in PATENT-1 was associated with improved survival and clinical worsening-free survival (Fig. 4). Patients achieving the endpoint had a 45% reduction in their relative risk of death and a 19% reduction in their relative risk of experiencing a clinical worsening event compared with those who did not achieve the endpoint (Table 1). Given the small patient numbers and the low event rates in PATENT-2, it was not expected that any differences would be statistically significant.
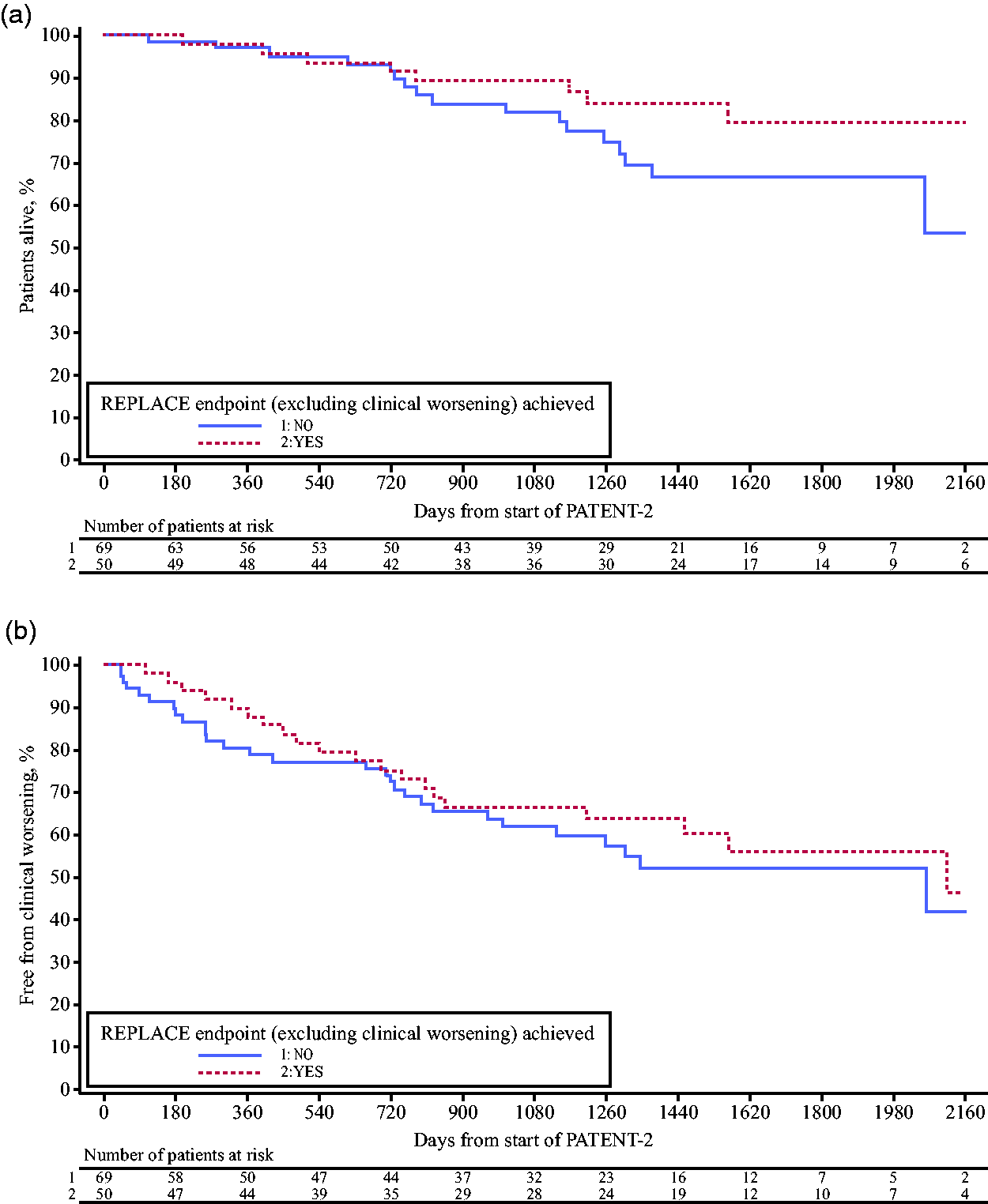
Association between achievement of the REPLACE composite endpoint (excluding clinical worsening) at PATENT-1 Week 12 and (a) survival and (b) clinical worsening-free survival in PATENT-2 in patients treated with riociguat in WHO FC III at baseline. (a) Survival. P value of log rank test: 0.1207. Data from patients in the 2.5 mg–maximum arm of PATENT-1 who participated in PATENT-2.
Cox proportional hazard ratios for survival and clinical worsening-free survival in PATENT-2 by achievement of the REPLACE composite endpoint (excluding clinical worsening) in patients in WHO FC III at baseline after 12 weeks in PATENT-1.
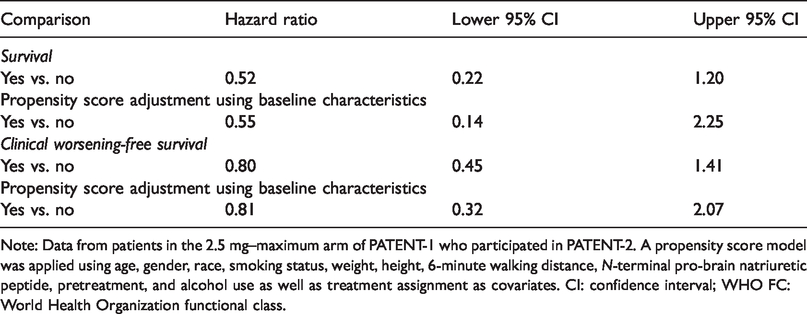
Note: Data from patients in the 2.5 mg–maximum arm of PATENT-1 who participated in PATENT-2. A propensity score model was applied using age, gender, race, smoking status, weight, height, 6-minute walking distance, N-terminal pro-brain natriuretic peptide, pretreatment, and alcohol use as well as treatment assignment as covariates. CI: confidence interval; WHO FC: World Health Organization functional class.
Discussion
In this post hoc analysis, we applied the REPLACE composite endpoint of clinical improvement to the PATENT-1 database to investigate whether the endpoint would differentiate between the treatment arms of this pivotal study. The results indicated that the REPLACE endpoint showed good discrimination between the treatment arms of PATENT-1, including in the treatment-naïve and pretreated subgroups, and in those patients in WHO FC III at baseline. Riociguat improved the likelihood of achieving the REPLACE composite endpoint in PATENT-1 compared with placebo, while a greater percentage of patients receiving placebo experienced no improvement. In addition to the differentiation between treatment groups in the short term, it is important to identify if achieving the composite endpoint is also meaningful for long-term outcomes. In this analysis, achieving the REPLACE composite endpoint was associated with survival and clinical worsening-free survival in PATENT-2.
The majority of clinical trials in PAH have used short-term improvement in parameters such as 6MWD 4 ,10–17 as surrogates for long-term outcomes; however, composite endpoints have also been used in several studies in PAH as a primary measure. In the AIR study of inhaled iloprost, a combined endpoint of improvement in WHO FC, 10% improvement in 6MWD, and absence of clinical deterioration at Week 12 was employed. 18 A greater proportion of patients achieved the composite endpoint when treated with iloprost compared with placebo. The phase 3/4 AMBITION event-driven study evaluated initial combination therapy with ambrisentan plus tadalafil (combination therapy group), ambrisentan plus placebo (ambrisentan monotherapy group), or tadalafil plus placebo (tadalafil monotherapy group) using a combined endpoint of clinical failure, defined as the first occurrence of death, hospitalization for worsening PAH, disease progression, or unsatisfactory long-term clinical response. A greater number of patients achieved the composite endpoint of clinical failure in the ambrisentan- and tadalafil-monotherapy groups compared with those receiving combination therapy. 19 A secondary endpoint of the study was the proportion of patients with a satisfactory clinical response (sCR) at Week 24. sCR was defined as a 10% increase from baseline in 6MWD, change to or maintenance of WHO FC I/II, and the absence of clinical worsening at Week 24. sCR was highest in the combination therapy group with 39% of patients achieving sCR, compared with 31% of patients in the ambrisentan monotherapy group and 27% of patients in the tadalafil monotherapy group. 19 In the phase 3 SERAPHIN study, the composite primary endpoint was the time from the initiation of treatment to the first event related to PAH (worsening of PAH, initiation of treatment with intravenous or subcutaneous prostanoids, lung transplantation, or atrial septostomy) or death from any cause up to the end of treatment. 20 A greater number of placebo-treated patients achieved the composite endpoint of death or hospitalization due to PAH compared with those treated with macitentan 3 mg and macitentan 10 mg.
In recent years, event-driven studies designed to address the potential limitations of single-parameter endpoints, such as lack of clinical relevance and inconsistent association with short- and long-term outcomes, have utilized combined endpoints of mortality and clinical worsening (itself a composite endpoint, now required by regulatory authorities to be included as an endpoint in PAH clinical trials).19–21 The composite endpoints applied in the SERAPHIN and AMBITION studies assessed death and clinical deterioration, while both the REPLACE and AIR study endpoints are fixed-term endpoints assessing clinical improvement rather than deterioration.
In addition to clinical worsening, the other components of the REPLACE composite endpoint – N-terminal prohormone of brain natriuretic peptide, WHO FC, and 6MWD – all have a proven relationship with long-term outcomes, supporting their use as indicators of clinical improvement in this endpoint. 6 , 22 Once validated, the REPLACE composite endpoint may be more widely used in clinical trials. Given that standard of care now involves risk assessment in clinical practice, with the aim of improving or maintaining (if low) a patient’s risk, it is clinically relevant and ethical to design endpoints of improvement that may be used in smaller studies with a shorter duration, rather than waiting for patients to deteriorate in large event-driven studies. 23 The discussion regarding the use of risk assessment as a potential clinical trial endpoint is ongoing, although trial endpoints are designed to assess an intervention, rather than the ongoing condition of a patient, and risk assessment may therefore be inappropriate as a trial endpoint.
The main strength of this analysis is the robust data set derived from a phase 3, randomized, placebo-controlled trial and long-term extension study. There are several limitations to this study; this was an exploratory post hoc analysis that assessed an endpoint that was not designed for the PATENT population. PATENT included a large proportion of patients in WHO FC II at baseline, whereas REPLACE included patients at intermediate risk in WHO FC III only. As the REPLACE endpoint was created for this patient population, the proportion of patients in the overall PATENT population meeting the endpoint was artificially high. In order to mitigate this, we also applied the REPLACE endpoint only to patients in WHO FC III at baseline. It is important to note that the definition of an endpoint should be appropriate for the intended study population; had the REPLACE endpoint specified any improvement in WHO FC rather than improvement to WHO FC I/II, preliminary analyses indicated that the response rate in PATENT would be similar to this analysis of WHO FC III patients only (data not shown). Moreover, the low rates of mortality and clinical worsening observed in PATENT-2 may have skewed the analysis of survival and clinical worsening-free survival, particularly as patients who were in WHO FC I, II, or IV at baseline were excluded from the analysis of long-term outcomes to mimic the REPLACE study population.
In conclusion, the REPLACE composite endpoint showed good discrimination between patients with PAH treated with riociguat and patients with PAH treated with placebo at Week 12 in PATENT-1, including subgroups of treatment-naïve or pretreated patients. Achieving the REPLACE composite endpoint while receiving riociguat was associated with improved survival and clinical worsening-free survival in PATENT-2 in patients who were WHO FC III at baseline. These data suggest that use of the REPLACE composite endpoint in patients with PAH is a valid assessment of response to treatment.
Supplemental Material
sj-pdf-1-pul-10.1177_2045894020973124 - Supplemental material for Assessment of the REPLACE study composite endpoint in riociguat-treated patients in the PATENT study
Supplemental material, sj-pdf-1-pul-10.1177_2045894020973124 for Assessment of the REPLACE study composite endpoint in riociguat-treated patients in the PATENT study by Gérald Simonneau, Hossein-Ardeschir Ghofrani, Paul A. Corris, Stephan Rosenkranz, Ekkehard Grünig, Jim White, Vallerie V. McLaughlin, David Langleben, Christian Meier, Dennis Busse, Frank Kleinjung and Raymond L. Benza in Pulmonary Circulation
Footnotes
Acknowledgments
The PATENT and REPLACE studies were sponsored by Bayer AG and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA. The authors thank Anthea Scothern, PhD of Adelphi Communications Ltd, Bollington, UK for providing medical writing support, which was funded by Bayer AG, Berlin, Germany in accordance with Good Publication Practice (GPP3) guidelines (![]() ). The authors would like to acknowledge Marc Lambelet and Britta Brockman of Chrestos Concept GmbH & Co. KG, Essen, Germany for their contribution to the statistical analysis.
). The authors would like to acknowledge Marc Lambelet and Britta Brockman of Chrestos Concept GmbH & Co. KG, Essen, Germany for their contribution to the statistical analysis.
Authors’ contributions
GS was responsible for the design of the post hoc analysis. DB was responsible for statistical analysis. All authors contributed to the data interpretation and drafting and revision of the manuscript. All authors approved the manuscript for submission.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: GS reports grants, personal fees, and non-financial support from Actelion, Bayer, GSK, and Merck. H-AG reports non-financial support, grants, and personal fees from Actelion; grants and personal fees from Bayer, Novartis Corporation, and Pfizer; and personal fees from Gilead Sciences, GSK, and Merck. PAC reports grants and personal fees from Actelion and Bayer, and personal fees from MSD. SR reports remunerations for lectures and/or consultancy from Abbott, Actelion, Arena, Bayer, Ferrer, GSK, MSD, Novartis, Pfizer, and United Therapeutics, and research support from Actelion, Bayer, Novartis, Pfizer, and United Therapeutics. EG reports grants and personal fees from Actelion and Bayer/MSD; grants from GSK, Novartis, and United Therapeutics, and personal fees from OrPha Swiss GmbH, SCOPE, and Zurich Heart House. JW reports no conflicts of interest. VVM reports grants, personal fees, and non-financial support from Actelion and Bayer; grants from Eiger and SoniVie; and personal fees from Arena, Caremark, Medtronic, MSD, and United Therapeutics. DL reports grants, personal fees, and non-financial support from Actelion and Bayer, personal fees from Merck and United Therapeutics; and grants from Northern Therapeutics. CM and FK are employees of Bayer AG. DB was an employee of Chrestos Concept GmbH & Co., KG, Essen, Germany at the time of the study and manuscript development. RLB reports grants from Actelion, Bayer AG, Eiger, Gilead, and United Therapeutics paid to his institution, and honoraria from Actelion, Bayer AG and Gilead.
Ethical approval
The PATENT study was carried out in accordance with Good Clinical Practice Guidelines and the Declaration of Helsinki. The study protocol was approved by the ethics committees of all participating centers, and all patients gave their written, informed consent.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The PATENT-1 and PATENT-2 studies were funded by Bayer AG, Berlin, Germany and Merck Sharp & Dohme Corp., a subsidiary of Merck & Co., Inc., Kenilworth, NJ, USA.
Guarantor
GS
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
