Abstract
The investigation of pediatric pulmonary hypertension (PH) drugs has been identified as a high priority by the United States National Institutes of Health (NIH). Studying pediatric PH is challenging due to the rare and heterogeneous nature of the disease. We sought to define the pediatric PH clinical trials landscape, to evaluate areas of trial success or failure, and to identify potential obstacles to the study of pediatric PH drugs. Interventional pediatric (ages 0–17 years) PH trials registered on ClinicalTrials.gov from June 2005 through December 2014 were analyzed. There were 45 pediatric PH trials registered during the study period. Median (IQR) projected trial enrollment was 40 (24–63), with seven trials (16%) targeting > 100 participants. Industry was the most common trial sponsor (n = 23, 50%), with only two (4.4%) NIH-sponsored trials. Phosphodiesterase inhibitors were the most frequently studied drug (n = 18, 39%). Single group study designs were used in 44% (n = 20) with an active comparator (parallel, factorial, or cross-over designs) in 25 trials, including 22 with randomization and ten that were double-blinded. Study outcomes varied markedly with inconsistent use of known surrogate and composite endpoints. One-third of trials (n = 15, 33%) were terminated, predominantly due to poor participant enrollment. Of the 17 completed trials, 11 had published results and only three efficacy trials met their primary endpoint. There are unique challenges to drug development in pediatric PH, including enrolling patients, identifying appropriate study endpoints, and conducting randomized, controlled, double-blind trials where the likelihood of meeting the study endpoint is optimized.
Introduction
Pulmonary hypertension (PH) in children is associated with significant morbidity and mortality, as well as a high societal healthcare burden.1,2 Yet very few PH drug therapies have been rigorously studied in children with PH. Although 11 different drugs are currently approved for the targeted treatment of pulmonary arterial hypertension (PAH) in adults, inhaled nitric oxide (iNO) remains the only FDA-labeled drug for the treatment of pediatric PH (and only in neonates).3,4 All other targeted PH therapies must be used off-label in children with safety, efficacy, and dosing extrapolated from adult studies.
Recognizing the necessity for pediatric PH therapies, in 2010 the U.S. National Institutes of Health (NIH) and U.S. Food and Drug Administration (FDA) prioritized pediatric PH drugs as an area of highest therapeutic need. 5 The prioritization process is intended to encourage clinical trials for drug labeling in children and adolescents. However, conducting these trials in children with PH can be challenging due to the heterogeneous nature of the disease and the choice of an optimal endpoint to gauge treatment response. This was clearly demonstrated by the first FDA-sponsored pediatric clinical trial of sildenafil, one of the most ubiquitous PAH pharmacotherapies. The Sildenafil in Treatment-Naïve Children, Aged 1–17 Years, with Pulmonary Arterial Hypertension (STARTS) trials showed an increase in patient mortality associated with the use of high dose sildenafil. 6 Interpretation of the STARTS trials, with respect to the safety and efficacy of lower dose sildenafil in children, has been controversial. After reviewing the trial results, the U.S. FDA placed a safety warning on the sildenafil drug label recommending against the use of sildenafil at any dose in children. 7 However, the European Medicines Agency (EMA) reviewed the same trial results and determined that sildenafil is safe and effective at lower doses. 8 Prominent members of the pediatric PH community in the U.S. and Europe agreed with the EMA decision; 9 subsequently the U.S. FDA clarified their position. They have since stated that there may be situations in which the benefit-risk profile of sildenafil may be acceptable in individual children. 10
Much of this confusion stems from the unique challenges associated with designing and conducting pediatric PH trials. Subsequent analysis of the STARTS trials has demonstrated factors that may have contributed to the difficulties in interpreting the trial results. Such factors include variability in the treatment response in patients of different ages and in different diagnostic cohorts, a lack of adequate study power for subgroup comparisons, no control group for comparison of long-term outcomes, and substantial variability in usage of additional PAH pharmacotherapies and patient follow-up among study centers.9,11
In light of the numerous challenges highlighted by the STARTS trials, we hypothesized that evaluating the pediatric PH clinical trials landscape would provide insight into appropriate trial design and identify the major obstacles to performing successful trials for the labeling of pediatric PH drugs. To this end, we evaluated ClinicalTrials.gov, a registry of clinical trials maintained by the National Library of Medicine. We sought to define the pediatric PH clinical trials landscape by evaluating: (1) enrollee demographics including age ranges and underlying disease entities; (2) trial demographics including size, duration of follow up and choice of study endpoints; and (3) trial success based on publication of results. To supplement these findings and further identify potential trial barriers, we also surveyed parents and providers.
Methods
Data source
ClinicalTrials.gov is a web-based registry of studies that is maintained by the National Library of Medicine (NLM) at the National Institutes of Health (NIH). Information on ClinicalTrials.gov is provided and updated by the sponsor or principal investigator of the clinical study. Studies are generally submitted to the website (that is, registered) when they begin and the information on the site is updated throughout the study. Each study record contains a set of data elements describing the study’s purpose, recruitment / enrollment, design, eligibility criteria, location, sponsor, and other protocol information; standard definitions are used. 12 The International Committee of Medical Journal Editors (ICMJE) began requiring registration of all interventional trials as a condition for publication on July 1, 2005, and the U.S. FDA has mandated registration of all interventional trials that include enrollment of study participants since September 27, 2007. As a result of these regulations, all clinical trials that enroll patients in the United States must be registered. Although registration is not similarly mandated for trials conducted outside of the USA, the ICMJE requirements influence many of these trials ensuring international representation.
Study selection
We searched ClinicalTrials.gov for all PH trials that included any pediatric age groups (age < 18 years) and were registered from July 1, 2005 (beginning of the ICJME requirements for publication) through December 31, 2014. PH trials were identified using the following search terms: pulmonary hypertension, ambrisentan, bosentan, epoprostenol, iloprost, sildenafil, sitaxsentan, tadalafil, treprostinil, and vardenafil. Only interventional trials were included. These trials are defined broadly by ClinicalTrials.gov to include drugs, devices, and non-invasive approaches such as surveys or education (www.clinicaltrials.gov/ct2/about-studies/glossary). The term “interventional” is intended to differentiate studies from those that are purely observational. Our search identified 296 potentially eligible trials. Of these, 83 were excluded as duplicate studies. After manual review of the remaining 213 studies, an additional 168 trials were excluded as they either represented adult trials but with an age range that did not exclude pediatric patients (most allowed for enrollment of adolescents ages > 16 years), or because we determined that they were not PH trials. The remaining 45 studies were included for analysis.
Data extraction
A dataset of all potentially eligible trials was downloaded into a study database. Variables extracted included: clinical trial registry number, study title, recruitment status, trial sponsors and collaborators, funding source, number of study sites, countries or study sites, conditions studied, interventions studied, genders included, age groups included, number of patients enrolled, trial design, phase of trial, dates of trial initiation and completion, dates trial was first received and last verified on Clinicaltrials.gov, primary and secondary outcome measures, and whether or not study results are reported on Clinicaltrials.gov. Publication of main trial results was assessed based on reporting in the publication field of ClinicalTrials.gov. When this data field was incomplete, a PubMed review using the ClinicalTrials.gov identifier was conducted to identify publications containing trial results.
Derived variables
Funding source was derived using information about the lead sponsor (the organization that oversees implementation of the study and data analysis) and collaborators (organizations that provide support including funding). Funding source was defined as NIH if the lead sponsor or any collaborators were from NIH and the lead sponsor was not from industry. Funding source was defined as industry if the lead sponsor was from industry or if any collaborators were from industry and there was no NIH involvement. The remaining studies were characterized as “other,” and had various sources of funding including institutional support and/or foundational support. Trials were classified as early phase (phase 0, 1, 1/2, or 2), late phase (phase 2/3, 3, or 4), or missing. For recruitment status, studies that were listed as either suspended, terminated, or withdrawn were analyzed as a single group. Classification of type of PH being studied was based on the World Health Organization (WHO) classification system 13 and derived from study inclusion criteria listed on the ClinicalTrials.gov website.
Outcome measures were classified into six categories, including: (1) hemodynamic data derived from cardiac catheterization or direct catheter measurement of pressures; (2) clinical endpoints, including laboratory markers of changes in clinical status, or clinical events (e.g. PH crises, death); (3) echocardiographic measures; (4) measures of exercise tolerance, such as six-minute walk test (6MWT); (5) NICU endpoints, which included measures of changes in oxygenation or changes in level of respiratory or circulatory mechanical support in studies specifically targeted to the neonatal population; and (6) measures of drug safety or pharmacokinetics, including serious adverse events or laboratory values that represented an adverse drug effect. In classification, the following assumptions were made: (1) hemodynamic changes were assumed to represent non-invasive hemodynamics unless catheter based study was reported; and (2) outcomes involving changes in pulmonary artery pressure or pulmonary vascular resistance were assumed to be catheter based unless otherwise reported. Multiple outcomes within the same category were counted as a single outcome.
Survey of trial investigators and patients with PH
To gain a more in-depth understanding of reasons for trial success or failure, the clinical trial investigators of included studies as well as a group of parents of children with PH were surveyed. For PH investigators, an online survey was sent out to either the contact person or principle investigator listed on ClinicalTrials.gov or the contact author of the published trial. Nine of 45 trials did not have a contact investigator listed on ClinicalTrials.gov. A separate online survey was sent to parents of patients with PH, made available to them via a social networking website. All survey responses were anonymous.
Analysis
Trial characteristics were described using standard summary statistics. Categorical variables were reported as proportions ± standard deviation and continuous variables as medians with interquartile ranges. Missing values were excluded from analysis. To describe trends in time, pediatric PH studies were evaluated over two time periods; January 2005 to December 2009 and January 2010 to December 2014. The period was derived using the date the trial was first registered with ClinicalTrials.gov. SAS Studio version 3.4 (SAS Institute) was used for all statistical analyses.
Results
Trial demographics
Demographics and diagnostic categories.
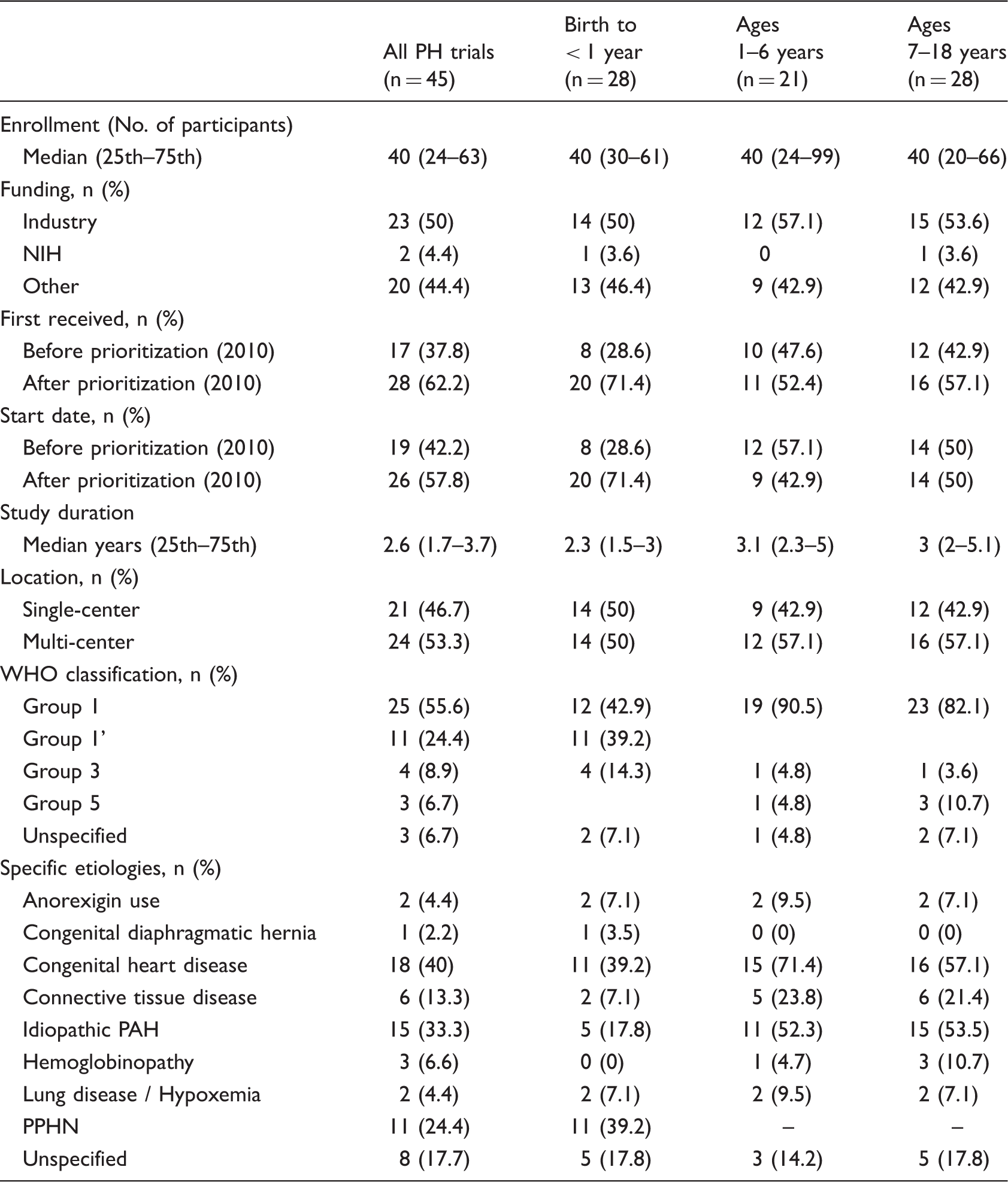
Neonates and infants were the most commonly studied age group and were represented in 28/45 (62%) of all trials, including 16 (36%) trials that enrolled only children age less than one year. Eight (18%) trials included only older children (age range, 7–18 years) and 11 (24%) trials enrolled across the entire pediatric age spectrum (age range, 0–18+ years). Half of the trials were industry-funded (n = 23), two trials (4.4%) were NIH-funded, and the remainder (44%) were funded by “other” sources.
Diagnostic categories and drugs studied
The vast majority of trials (36/45, 80%) evaluated children with WHO group I PH, including 18 (40%) in children with congenital heart disease (CHD)-associated PAH, 15 (33%) with idiopathic or familial PAH, and 11 (24%) with persistent pulmonary hypertension of the newborn (ppHN) (Table 1). Excluding trials without specified inclusion criteria and trials investigating only ppHN, 9/40 (23%) trials were limited to a single diagnostic cohort of WHO group I patients, with all other studies enrolling children across the spectrum of group I PH diagnoses. Those single cohort trials included three trials in patients with idiopathic or familial PAH and six trials of patients with CHD-associated PAH.
Drug therapies investigated are illustrated in Fig. 1. Most trials (n = 42, 93%) investigated a single agent. Phosphodiesterase-5 (PDE5) inhibitors were the most common investigational drug for all age groups, including 16 (35%) sildenafil trials and two (4%) tadalafil trials. Prostanoids were the second most studied class of PH therapy, with ten trials of iloprost (22%), and one each for epoprostenol and treprostinil. Endothelin receptor antagonists (ERAs) were investigated in eight (18%) trials, with six trials of bosentan and two of ambrisentan. iNO was the only drug showing any evidence of an age-group preference with two-thirds of iNO trials restricted to neonates.
Investigational drugs by age.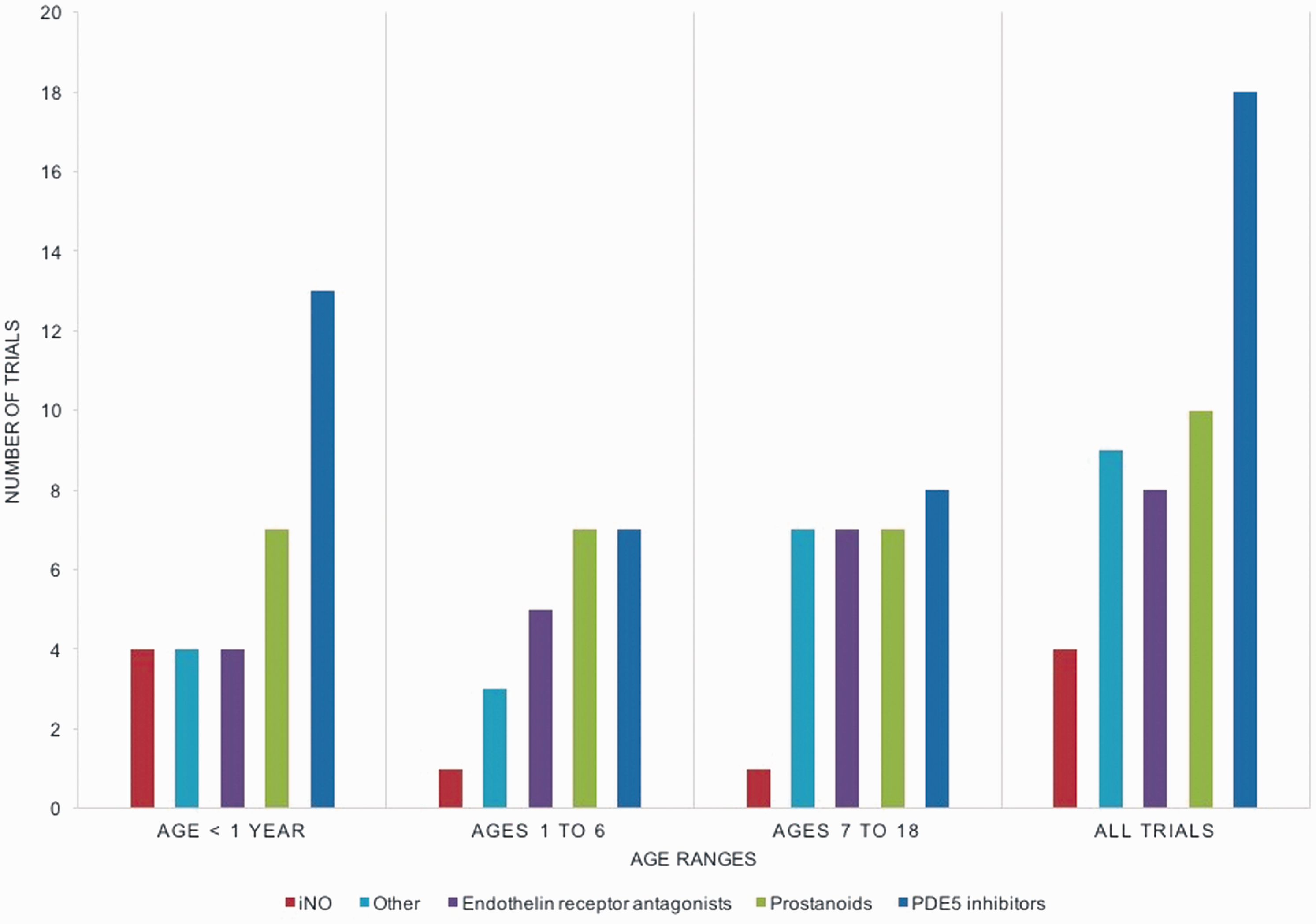
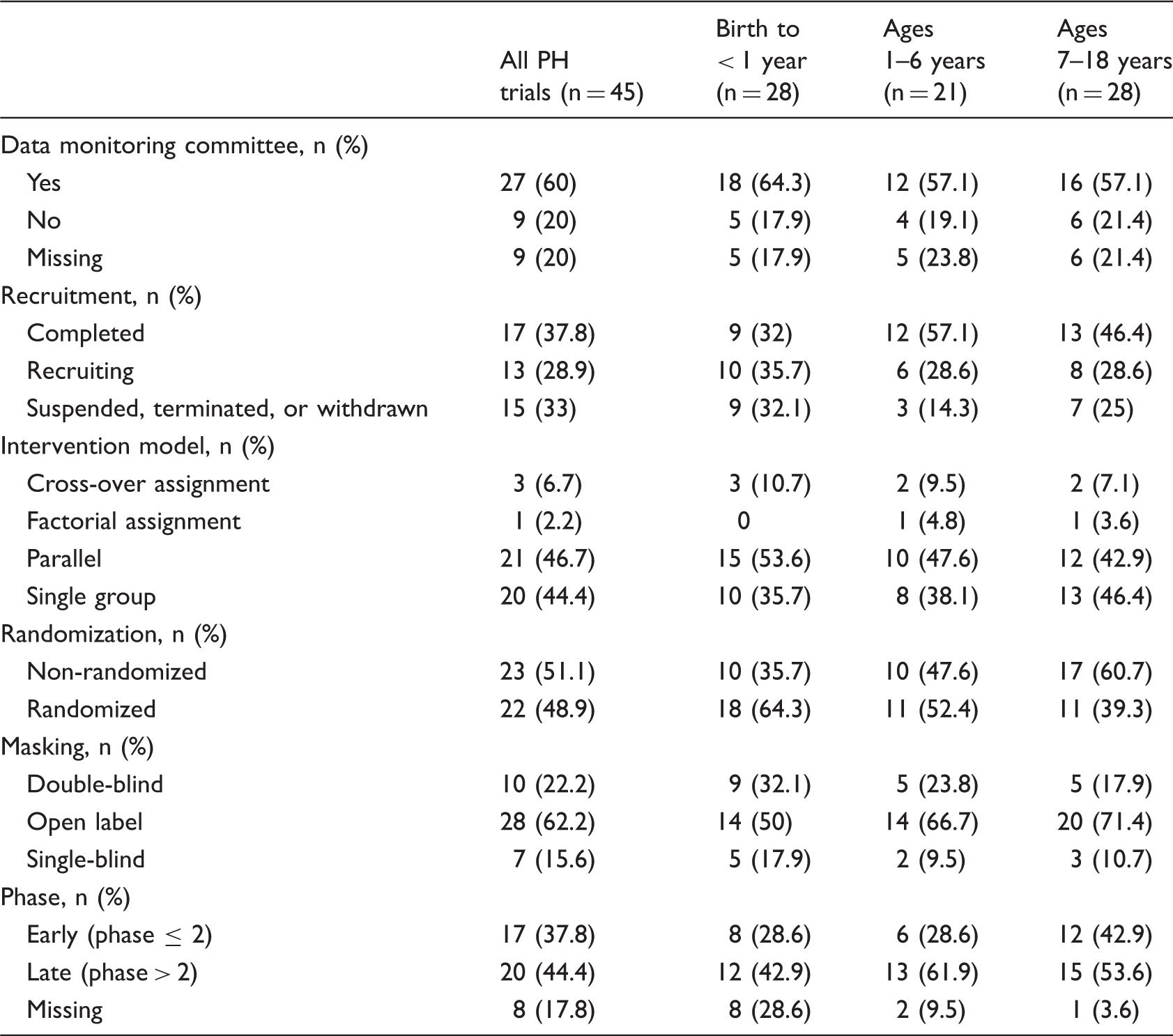
Trial designs
Trial designs, interventional models, and quality measures.
Outcome measures
The distribution of outcome measures for all trials, and for trials by age group, is shown in Fig. 2. Nearly all trials used two or more categories of outcome measures (43/45 trials, 96%). Clinical outcomes (30/45 trials, 67%) were the most frequently used endpoints. Individual clinical outcomes across trials were heterogeneous, including change in WHO functional class (n = 9), change in quality of life (n = 7), mortality (n = 9), other change in clinical status (n = 7), the occurrence of a PH crisis (n = 3), change in vital signs (n = 9), change in physical exam findings (n = 4), and change in B-type natriuretic peptide (BNP) (n = 5) or in some other laboratory value (n = 8). Pharmacologic and safety outcomes were used in 23/45 trials (51%) with fairly uniform measures among trials (area under the curve, maximum concentrations, adverse events).
Outcome measures by age.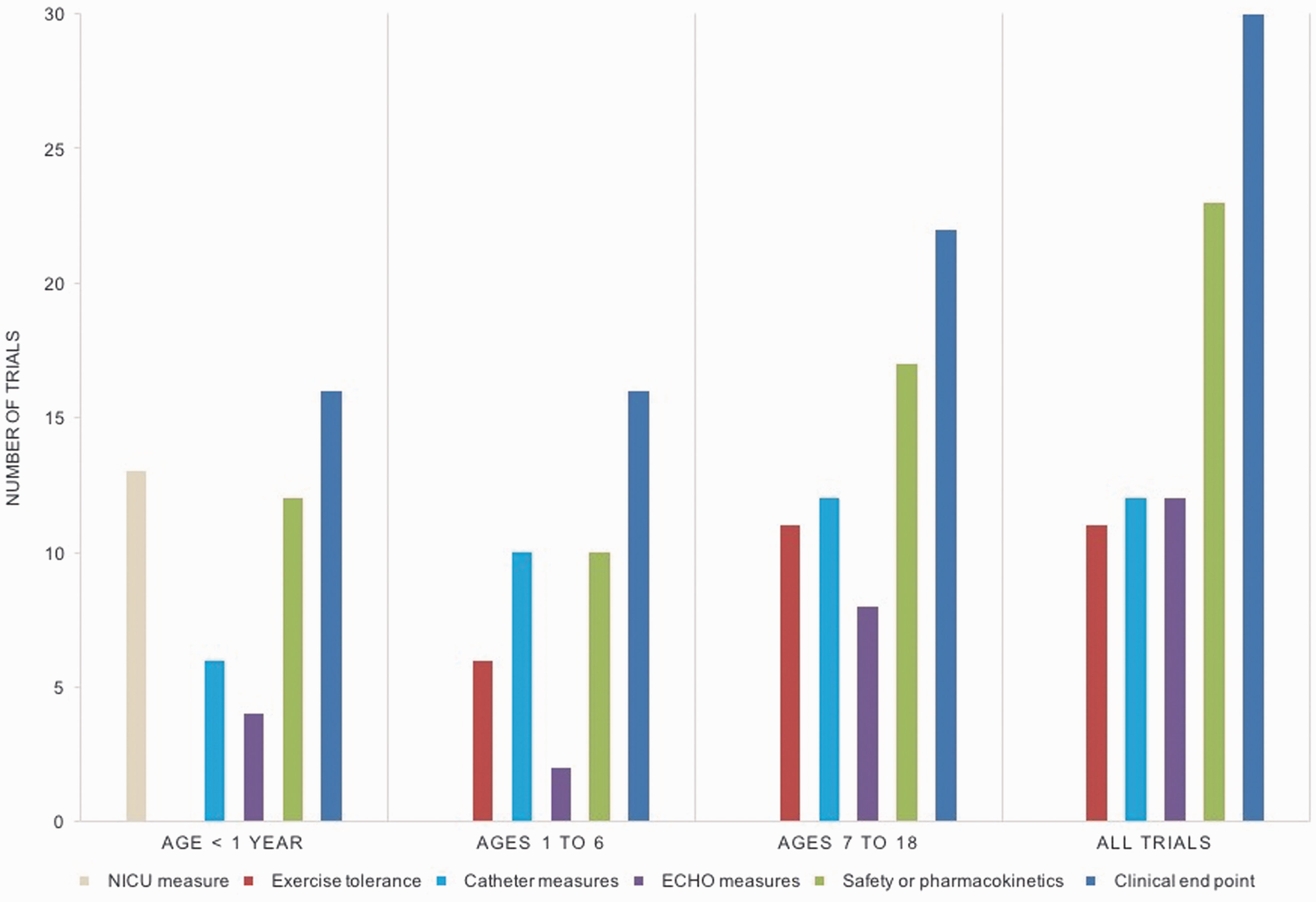
Exercise tolerance was evaluated by either 6MWT or treadmill in 11/45 trials (24%), with six (55%) of these trials including at least some patients aged less than seven years. Echocardiographic and cardiac catheterization-based endpoints were each used in 12/45 trials (27%). Lastly, trials that focused solely on neonates and infants used NICU specific endpoints, such as change in oxygenation indices or measures of ventilator support, in 13/16 trials (81%).
Trial completion and publication
One-third (n = 15, 33%) of trials were suspended, terminated, or withdrawn prior to completion. The most common reason for trial discontinuation was lack of adequate enrollment (n = 8, 53%). Additional reasons included a change in clinical practice preventing trial continuation (n = 1), a concern about inconsistent absorption of study drug (n = 1), and a primary study investigator leaving the sponsoring institution (n = 1). Four trials did not list a reason for discontinuation. Funding source was not an obvious driver of trial termination, as trials were terminated across the funding spectrum including 35% of industry-funded trials (n = 8/23), 50% of NIH-funded trials (n = 1/2), and 30% of trials with “other” funding sources (n = 6/20).
Characteristics and outcomes of published trials.

AEs, adverse events; CHD, congenital heart disease; iNO, inhaled nitric oxide; iPAH, idiopathic pulmonary arterial hypertension; PPHN, persistent pulmonary hypertension of the newborn; PVfO2, peak oxygen consumption; PVR, pulmonary vascular resistance; RIPC, remote ischemic preconditioning; TnI, troponin I.
PH survey responses
Thirty-seven parents of children/adolescents with PH participated in our patient survey and 8/36 investigators (all physicians) responded to the investigator survey. Nearly all PH investigator respondents were experienced trialists; seven had participated in three or more previous PH trials. Parent respondents represented children with significant disease pathology. Median age since diagnosis was four years (range, 0.9–16 years) and almost all had children currently taking either two (n = 9) or three (n = 26) PH drug therapies.
Parent (n = 37) and investigator (n = 8) survey responses.

Our parent survey perhaps provides some insight into mechanisms whereby recruitment can be improved. The pediatric PH community is very active as demonstrated by the rapid and excellent response of parents to our survey request.
Respondents were asked to rate on a scale of 1–10, categories were compiled as follows: minor/suboptimal 1–3, moderate/intermediate 4–6, major/optimal 7–10.
Although not a specific question, three investigators commented that they would consider composite clinical endpoint outcome measures as optimal.
iPAH, idiopathic pulmonary arterial hypertension; PVR, pulmonary vascular resistance.
Parent respondents almost universally agreed (94%) that clinical trials are important to understand the efficacy and safety of PH medications in children, even if already tested in adults. However, 72% of the sample felt comfortable with their doctors prescribing adult PH medications, regardless of whether they had been adequately tested in children. Overall, 86% of parents would consider letting their child participate in a clinical trial and none endorsed the statement that “it is unethical to do PH clinical trials in children.” In terms of barriers to trial participation, very few considered extra blood draws (n = 1), extra clinic visits (n = 0), extra non-invasive tests (n = 0), or the need to take a previously untested medication (n = 1) as prohibitive barriers to trial participation. Only eight respondents felt that extra cardiac catheterization procedures would be prohibitive, although the majority (n = 28) had some concerns about this requirement. The most common prohibitive barrier that parents identified was the possibility of randomization to placebo instead of study drug. Of the parents, 30% (11/37) considered this as a prohibitive barrier, with an additional 43% (16/37) noting that they would have concerns but would still consider having their child participate in a placebo-controlled trial.
Discussion
We hypothesized that a review of pediatric PH clinical trials would be helpful in the design of future pediatric PH trials. Our analysis of nine years of data from ClinicalTrials.gov highlights some of the challenges facing drug development for pediatric PH. We found that most pediatric PH trials are small and heterogeneous, enrolling a wide spectrum of patient cohorts and evaluating a variable collection of study endpoints. Moreover, recruitment of enough patients to achieve adequately powered results was a struggle for trials. This frequently resulted in premature trial termination. Less than one-quarter of trials analyzed have thus far made it to publication, and of those, only three met a primary efficacy endpoint. Our survey of investigators and parents suggest potential solutions to these issues, including the possibility of parent engagement to improve trial design and recruitment, and novel trial endpoints that could be considered for future pediatric PH trials.
Challenges for pediatric PH trials
Patient recruitment
Investigators in our survey highlighted patient enrollment as the biggest barrier to successful pediatric PH trials. This was supported by our ClinicalTrials.gov analysis. Despite most of the trials in our analysis being small (median projected enrollment of 40 participants), one-third were terminated. This was most commonly due to poor enrollment. An additional one-third of completed trials had lower than anticipated enrollment. In prior analyses of ClinicalTrials.gov, poor patient enrollment has consistently been identified as the major obstacle to study completion.14,15 An analysis of 114 randomized controlled trials in the United Kingdom found that less than one-third reached their target recruitment goal. 16 This contrasts with the reported termination rate for trials of common diseases, which is only 5–10%. In an analysis of ClinicalsTrials.gov, Bell et al. found that rare disease trials were more than twice as likely to be terminated early, yet the 13.7% termination rate that they reported for rare diseases is significantly lower than the 33% termination rate we found for pediatric PH trials. 17
Heterogeneous cohorts
Likely in an attempt to improve recruitment, two-thirds of pediatric PH trials in our analysis enrolled heterogeneous cohorts of PH patients, including different PAH diagnostic groups and/or wide age ranges. Broadening inclusion criteria is a reasonable approach to increase enrollment. However, overly heterogeneous trials can make the interpretation of trial results more challenging. This was highlighted by the STARTS trial, where older patients and patients with idiopathic PAH appeared to have worse outcomes than patients with CHD-associated PAH. Yet because the trial was not powered for subgroup comparisons, it has been difficult to interpret results.6,9 An additional heterogeneity-related concern identified in the STARTS trials relates to variability across sites. The majority of deaths in the STARTS trials occurred in patients living in poorer countries, potentially without access to second and third line PAH therapies. 18 Some have questioned whether it is appropriate to extrapolate these sildenafil-related safety concerns to patients expected to have access to more advanced therapies that can be initiated upon clinical worsening. This issue highlights the challenges of appropriate site selection. Multi-center trial-sponsors must consider multiple factors including costs, the diversity of the patient population, number of available participants, trial experience / infrastructure of each participating center, and eventual generalizability of study results.
Selection of outcome measures
The few successful trials in our analysis, at least in terms of being published and meeting intended endpoints, focused on short-term surrogate outcome measures such as invasive hemodynamic measurements or acute clinical changes in the ICU setting. Benefits of surrogate outcome measures include reduced cost, lower trial burden leading to improved patient recruitment, the potential to facilitate “higher power” study designs (e.g. cross-over or n of 1 designs), and easier correlation with pharmacokinetic measures (e.g. PK/PD trials). Data from these earlier phase trials can often provide insight into the appropriate design and conduct of larger-scale safety and efficacy trials (e.g. Phase III trials). Indeed virtually all of the currently approved adult PAH therapies underwent phase II studies prior to phase III, whereas none of the drugs that have gone straight to pivotal trials have met with regulatory approval. 19 However, for a surrogate outcome to be meaningful, it should ideally first be validated as an accurate measure of clinical status. 20 No rigorous validation studies have been performed in pediatric PH,19,21 and this is reflected in the wide array of outcomes used in the analyzed trials. Specific endpoints that pediatric PH experts have advocated for include WHO functional class, 6MWT, and BNP.8,21,22 We found these measures to be used infrequently, and are not without their limitations. 6MWT and WHO functional class are difficult to apply across all ages, 21 and BNP has not been well validated as an outcome measure in PH.
Potential solutions
Our survey data highlighted several potential approaches to improve pediatric PH trials. First, the responses from parents to our survey were overwhelmingly positive. The majority supported the conduct of PH clinical trials in children, even when participation in these trials would require additional clinic visits, studies, or even blood draws. The pediatric PH community is an active and engaged community; involving patient/parent advocates in the clinical trial process may help to improve trial design and encourage trial participation. This approach has been successful in other rare diseases that face similar challenges to pediatric PH. 23 Second, our investigator survey data, although limited, identified potential trial endpoints that could be explored. Investigator respondents supported use of quality-of-life surveys, changes in daily activity levels measured by activity monitors, and composite clinical event endpoints. These options are all promising, as they leverage increased study power either as continuous outcome measures (survey data or activity) or via increasing event rates (composite measures). The recently completed adult PH SERAPHIN trial used an event driven composite morbidity and mortality endpoint evaluating “time to clinical worsening,” with continued patient follow-up until a pre-specified number of outcome events were reached. This approach improves the likelihood of an interpretable trial outcome. Leaders in the pediatric PH community have advocated for a similar approach in future pediatric PH trials. 24 Another potential composite endpoint approach is the use of a global rank endpoint, where every participant in the trial is assigned a rank from best to worst based on a pre-specified list of outcomes. This approach improves study power and has been successfully applied to adult heart failure trials. 25 Similarly, activity monitors represent a potentially promising tool in clinical research. A query of ClinicalTrials.gov for physical activity monitors (i.e. devices such as a pedometer or “Fitbit”) yielded 2585 unique clinical trials currently registered on the site. Further research is perhaps warranted to validate this as an outcome measure in children and adolescents with PH. Finally, future PH trials could consider either stratified enrollment or covariate adjustment for baseline factors known to be associated with outcomes. These risk factors have been well demonstrated in previous analyses,26,27 and powering for stratified or covariate adjusted primary analyses may circumvent some of the challenges associated with enrolling a heterogeneous patient cohort.
Limitations
There are several limitations to this analysis. First, accuracy of the information on the ClinicalTrials.gov website is dependent on investigators submitted complete data. Some fields have missing or unsubmitted data, which can limit analyses. Second, not all studies are updated regularly, which may lead to inaccurately reported information. Third, trials not under US jurisdiction are not obligated to be reported and may have been missed in this analysis. Fourth, we excluded “adult” PH trials even though they included adolescent patients. Since the number of adolescents in these trials is typically small, we did not feel that they would be representative of the challenges facing pediatric PH trials. Finally, there are well-known limitations to surveys including skewed response rates and the inherent cross-sectional nature of the data.
Conclusions
We found that despite prioritization by major stakeholders, there have been relatively few successful pediatric PH trials over the past decade. Successful trial completion has been most significantly hindered by low patient recruitment. Other issues include the heterogeneous nature of pediatric PH, with many trials including multiple PH patient populations and variable study endpoints, and a relative lack of funding, particularly from the NIH. Prior reports have outlined strategies for improving PH trials. 19 Recommendations have included a need for consensus regarding optimal outcome measures, including validated surrogate outcomes and composite endpoints. There remains a need for creative trial designs to overcome the difficulties of studying a rare and heterogeneous patient population, and a need for long-term continuation of blinded dose-ranging for adverse event surveillance. More robust pediatric pharmacokinetic and safety data are also required. Our data support these recommendations and may be useful as a framework for designing future pediatric PH trials. Additionally, we found that parents of patients with PH recognize the importance of dedicated pediatric clinical trials and are interested in participating.
Footnotes
Conflict of interest
Richard Krasuski has consulted for Actelion and Bayer Pharmaceuticals and receives research funding from Actelion. Kevin Hill receives support from The National Center for Advancing Translational Sciences of the NIH (UL1TR001117).
Funding
Richard Krasuski receives research funding from Actelion. Kevin Hill receives support from The National Center for Advancing Translational Sciences of the NIH (UL1TR001117) and from the Gilead Cardiovascular Scholars Program.
