Abstract
The Pulmonary Arterial Hypertension Symptoms and Impact Questionnaire (PAH-SYMPACT) is a PAH-specific patient-reported outcome scale assessing patients’ quality of life from four aspects: cardiopulmonary symptoms, cardiovascular symptoms, physical impacts and cognitive/emotional impacts. This study aimed to validate the Chinese version of PAH-SYMPACT and explore its relationship with risk stratification in patients with connective tissue disease-associated pulmonary arterial hypertension (CTD-PAH). In addition, 75 patients with CTD-PAH confirmed by right heart catheterization were invited to complete questionnaires including PAH-SYMPACT, the 36-item Medical Outcomes Study Short Form Survey (SF-36) and EuroQol five dimensions questionnaire (EQ-5D). The demographic, clinical, laboratory and treatment data were collected. The endpoint was treatment goal achievement status in 6–12 months after completing the questionnaires, defined as an integrated outcome. Participants’ mean age was 36.4 ± 11.9 years and the mean pulmonary arterial pressure was 38.9 ± 13.67 mmHg. The reliability of the PAH-SYMPACT domains ranged from 0.83 to 0.88. Results of factor analysis basically conformed the original PAH-SYMPACT. The treatment goal achievement (TGA) status in 6–12 months was significantly associated with physical impacts scores (odds ratio: 0.180, 95% confidence interval: 0.036–0.908, P=0.038). The Chinese version of PAH-SYMPACT is a reliable measurement to evaluate quality of life in CTD-PAH patients and is also a potential predictor of patient’s condition change in routine clinical practice.
Pulmonary arterial hypertension (PAH) is a severe complication of connective tissue disease (CTD) and can lead to significant mortality.1,2 It is increasingly acknowledged that PAH causes a range of symptoms (including fatigue, shortness of breath and chest pain) which affect many aspects of daily life. 3 Moreover, quality of life (QoL) assessment is recommended in routine clinical practice of CTD patients and should be considered to incorporate into treat-to-target strategy.4,5 Most QoL evaluation in PAH patients is generic or non-disease-specific. 3 However, a new PAH-specific questionnaire named Pulmonary Arterial Hypertension-Symptoms and Impact (PAH-SYMPACT) were currently validated. 6 But it has not been validated in Chinese and little is known about the QoL status in connective tissue disease-associated pulmonary arterial hypertension (CTD-PAH) patients.
The 2015 European Society of Cardiology (ESC) and the European Respiratory Society (ERS) pulmonary hypertension (PH) guidelines recommended a multidimensional stratification system to assess the mortality risk in PAH patients. 7 Moreover, subsequent validation studies of this multi-parameter risk assessment in idiopathic PAH and scleroderma-associated PAH proved it helps establish a prognosis at baseline and assess response to therapy.8–10 PAH-SYMPACT scales were proved to be sensitive to improvement in clinician-reported disease outcomes. 6 However, there is no previous research on the relationship between QoL and risk stratification in the follow-up evaluation of PAH patients. Treatment goal achievement (TGA) in our studies was defined using the same parameters as the risk stratification criteria and was proved to be associated with long-term survival in Chinese systemic lupus erythematosus-associated PAH patients. 11 The aim of this paper is to report the results of cross-cultural adaptation of the PAH-SYMPACT in Chinese and explore the association between risk assessment and QoL in CTD-PAH patients confirmed by right heart catheterization (RHC).
Methods
Study design and participants
Seventy-five patients with CTD-PAH visiting the outpatient clinic of Department of Rheumatology, Peking Union Medical College Hospital (PUMCH), during January 2019 and June 2020 were invited to complete the Chinese version of PAH-SYMPACT along with 36-item Medical Outcomes Study Short Form Survey (SF-36) and EuroQol five dimensions questionnaire (EQ-5D). Baseline demographic and clinical data including RHC and echo-cardiography (ECHO) results, WHO functional class (WHO-FC), N terminal-pro brain natriuretic peptide (NT-proBNP), medication and TGA status were obtained. RHC was performed to confirm diagnosis, determine severity and rule out left-sided heart disease. A follow-up evaluation of TGA status in 6–12 months was recorded as endpoint. This study received approval from the ethics committee of PUMCH and written consent was obtained from all participants.
The type of CTD was defined at enrollment as follows. Systemic lupus erythematosus (SLE) diagnosis was defined by the 1997 revised American College of Rheumatology (ACR) criteria. 12 Rheumatoid arthritis (RA) diagnosis was defined by the 2010 ACR/the European League Against Rheumatism (EULAR) classification criteria. 13 Systemic sclerosis (SSc) diagnosis was defined by the 1980 American Rheumatism Association criteria and confirmed by the 2013 ACR/EULAR classification criteria. 14 Primary Sjogren syndrome (SS) diagnosis was defined by the 2002 revised American-European Consensus Group criteria. 15 Undifferentiated CTD diagnosis was defined by the established consensus.16–18
PAH diagnosis based on RHC was defined by the 2015 European Society of Cardiology (ESC)/European Respiratory Society (ERS) guidelines as the mean pulmonary arterial pressure (mPAP) ≥25 mmHg at rest, pulmonary arterial wedge pressure (PAWP) ≤15 mmHg and pulmonary vascular resistance (PVR) > 3 Wood units. 19 The exclusion criteria included chronic pulmonary thromboembolism confirmed by CTPA or V/Q scan or significant interstitial lung disease and total lung capacity <60% based on the results from pulmonary function test and chest high-resolution computed tomography. The composite treatment goals were defined according to the ESC/ERS guidelines 19 as: (1) clinical symptoms: no signs of right heart failure, syncope or progression; (2) WHO FC I or II, or 6-min walking distance > 380–440 m; (3) serology: brain natriuretic peptide < 50 ng/L or NT-proBNP < 300 ng/L; and (4) cardiac imaging: normal right atrial area according to ECHO.
Quality of life assessments
The PAH-SYMPACT has two constructs: PAH symptoms and PAH impacts. The PAH symptoms domains are cardiopulmonary symptoms and cardiovascular symptoms; the PAH impacts domains are physical impacts and cognitive/emotional impacts. PAH-SYMPACT has 23 items (12 for PAH symptoms construct with a 24-h recall period and 11 for PAH impacts construct with a one-week recall period). Scores for each item has five options ranging from 0 to 4, with higher scores indicating greater symptom severity or worse impact.
The English PAH-SYMPACT was translated into Chinese using forward and backward methods by two translators who is a health professional and a certified independent translator. All physical activities involved in the items of PAH-SYMPACT were compatible with Chinese culture, and we translated them without substitution. The backward English version was compared with the original version by a native English speaker, which indicated no difference compared with the original version. All the researches constituted an expert panel to evaluated the cultural and conceptual equivalences of both English and Chinese versions. The finalized Chinese version was assessed throughout by Chinese clinical experts to ensure it is cultural appropriate and understandable.
Statistical analysis
The continuous variables tested by Shapiro–Wilk test were expressed as mean ± standard deviation or median (interquartile range: 25%–75%) as appropriate, while categorical variables were summarized as percentages. Confirmatory factor analysis (CFA) was used to test the conceptual framework of PAH-SYMPACT scale. The goodness-of-fit of the conceptual structure was evaluated with the comparative fit index (CFI) and the root mean square error of approximation (RMSEA). The psychometric validation included internal consistency and test–retest reliability analysis. Backward logistic regression analyses were performed to identify the predictive factors of TGA status in 6–12 months. The IBM SPSS version 25 and AMOS version 24 was used for data management and statistical analyses.
Results
Study population
Of 75 patients with CTD-PAH enrolled (Table 1), 98.7% were female, with the median PAH duration of 32.45 months. RHC revealed mPAP as 38.9 ± 13.7 mmHg, CI as 3.7 ± 0.9 L/min×m2 and PVR as 4.8 ± 3.1 WU. In addition, 75.8% of patients were WHO Fc I and II. The underlying CTD was SLE (56.7%), SSc (11.8%) and SS (15%). Moreover, 100% and 98.7% patients were treated with glucocorticoid and immunosuppressors, respectively. As for PAH therapy, 83.3% CTD-PAH patients were treated with PAH target therapy and 16.3% were prescribed with ≥2 targeted medications. The percentage for ERA (endothelin receptor antagonist), PDE-I (phosphodiesterase inhibitor) and PG (prostaglandin analogue) were 54.2%, 55.9% and 3.4%, respectively.
Patient baseline characteristic.

WHO-FC: World Health Organization-Functional Class; SLE: systemic lupus erythematosus; SS: Sjogren syndrome; SSc: systemic sclerosis; UCTD: undifferentiated connective tissue diseases; RA: rheumatoid arthritis; ILD: Interstitial lung disease; NT-proBNP: N-terminal pro-brain natriuretic peptide; PASP: pulmonary arterial systolic pressure; TAPSE: tricuspid annular plane systolic excusion; RV: right ventricular; LVEF: left ventricular ejection fraction; RHC: right heart catheterization; RAP: right atrial pressure; mPAP: mean pulmonary arterial pressure; PAWP: pulmonary arterial wedge pressure; PVR: pulmonary vascular resistance; CI: cardiac index; CO: cardiac output; CYC: cyclophosphamide; MMF: mycophenolate mofetil; TAC: tacrolimus; HCQ: hydroxychloroquine; ERA: endothelin receptor antagonist; PDE5-I: phosphodiesterase inhibitor; PG: prostacyclin analogue.
Internal consistency and CFA of PAH-SYMPACT
Summary of PAH-SYMPACT, SF-36 and EQ-5D domain scores are shown in Table 2. In the internal consistency assessment for each of the domain of PAH-SYMPACT (Table 3), four domains all had good results with a Cronbach’s α >0.80 (0.83–0.88). The interclass correlation coefficient (0.83–0.88) demonstrated good test–retest reliability. Results of the CFA supported the conceptual framework of PAH-SYMPACT (Table 4) with good model fit in PAH symptoms domain (CFI = 0.93, RMSEA = 0.11) and PAH impacts domain (CFI = 0.96, RMSEA = 0.09). Furthermore, the item-to-factor loadings exceeded the acceptable threshold of 0.3 except for “think clearly” (0.00), “walk slowly on a flat surface” (0.24) and “wash or dress self” (0.18).
Data of different quality of life questionnaires.
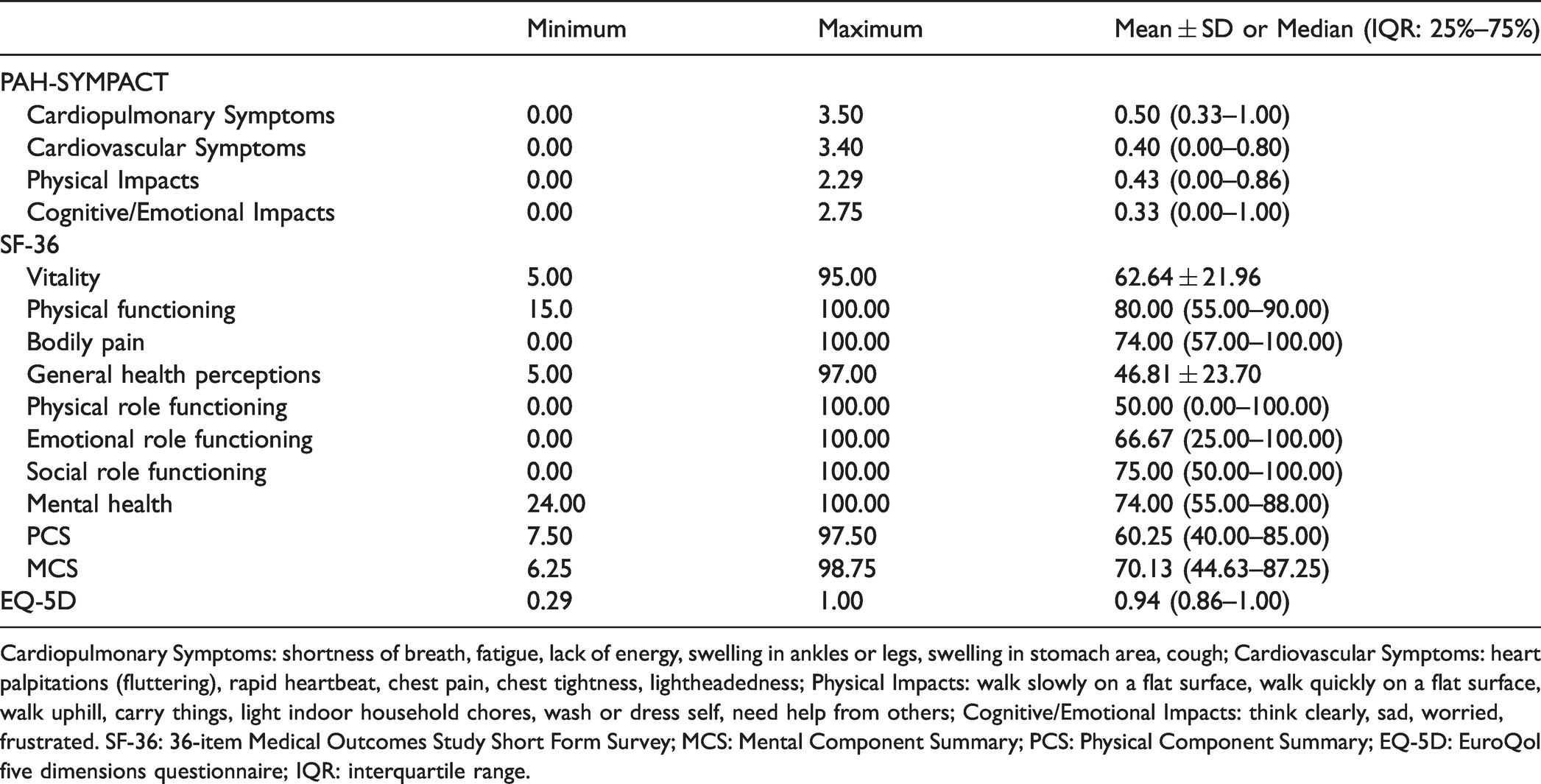
Cardiopulmonary Symptoms: shortness of breath, fatigue, lack of energy, swelling in ankles or legs, swelling in stomach area, cough; Cardiovascular Symptoms: heart palpitations (fluttering), rapid heartbeat, chest pain, chest tightness, lightheadedness; Physical Impacts: walk slowly on a flat surface, walk quickly on a flat surface, walk uphill, carry things, light indoor household chores, wash or dress self, need help from others; Cognitive/Emotional Impacts: think clearly, sad, worried, frustrated. SF-36: 36-item Medical Outcomes Study Short Form Survey; MCS: Mental Component Summary; PCS: Physical Component Summary; EQ-5D: EuroQol five dimensions questionnaire; IQR: interquartile range.
Internal consistency of domain scores.
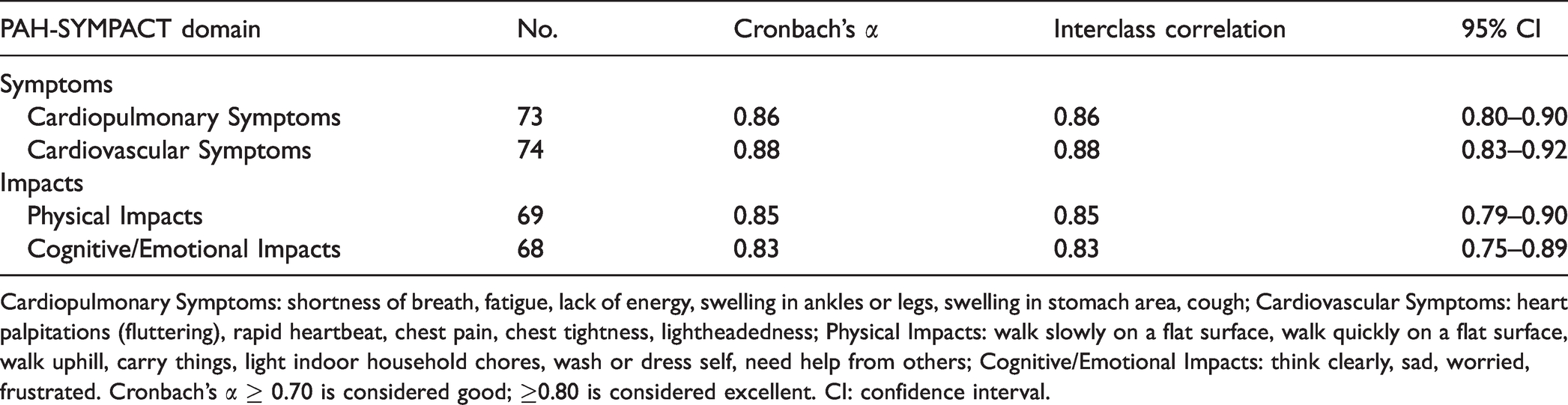
Cardiopulmonary Symptoms: shortness of breath, fatigue, lack of energy, swelling in ankles or legs, swelling in stomach area, cough; Cardiovascular Symptoms: heart palpitations (fluttering), rapid heartbeat, chest pain, chest tightness, lightheadedness; Physical Impacts: walk slowly on a flat surface, walk quickly on a flat surface, walk uphill, carry things, light indoor household chores, wash or dress self, need help from others; Cognitive/Emotional Impacts: think clearly, sad, worried, frustrated. Cronbach’s α ≥ 0.70 is considered good; ≥0.80 is considered excellent. CI: confidence interval.
Confirmatory fit analysis for PAH-SYMPACT.

CFI: confirmatory fit index; RMSEA: root mean square error of approximation.
TGA and quality of life
Patients who achieved TGA at baseline had significantly lower PAH-SYMPACT scores (Fig. 1) in cardiopulmonary symptoms domain (0.33 (0.13–0.67) vs. 0.67 (0.50–1.00), P<0.05), physical impacts domain (0.14 (0.00–0.43) vs. 0.57 (0.32–1.21), P<0.01) and cognitive/emotional impacts (0.00 (0.00–0.75) vs. 0.75 (0.44–1.25), P<0.01). Baseline characteristics were compared between patients who achieved TGA and patients who did not at baseline (Supplementary Table 1) or in 6–12 months (Supplementary Table 2). In multiple logistic regression analysis (Table 5), TGA status in 6–12 months was significantly associated with physical impacts scores (odds ratio: 0.180, 95% confidence interval: 0.036–0.908, P=0.038).
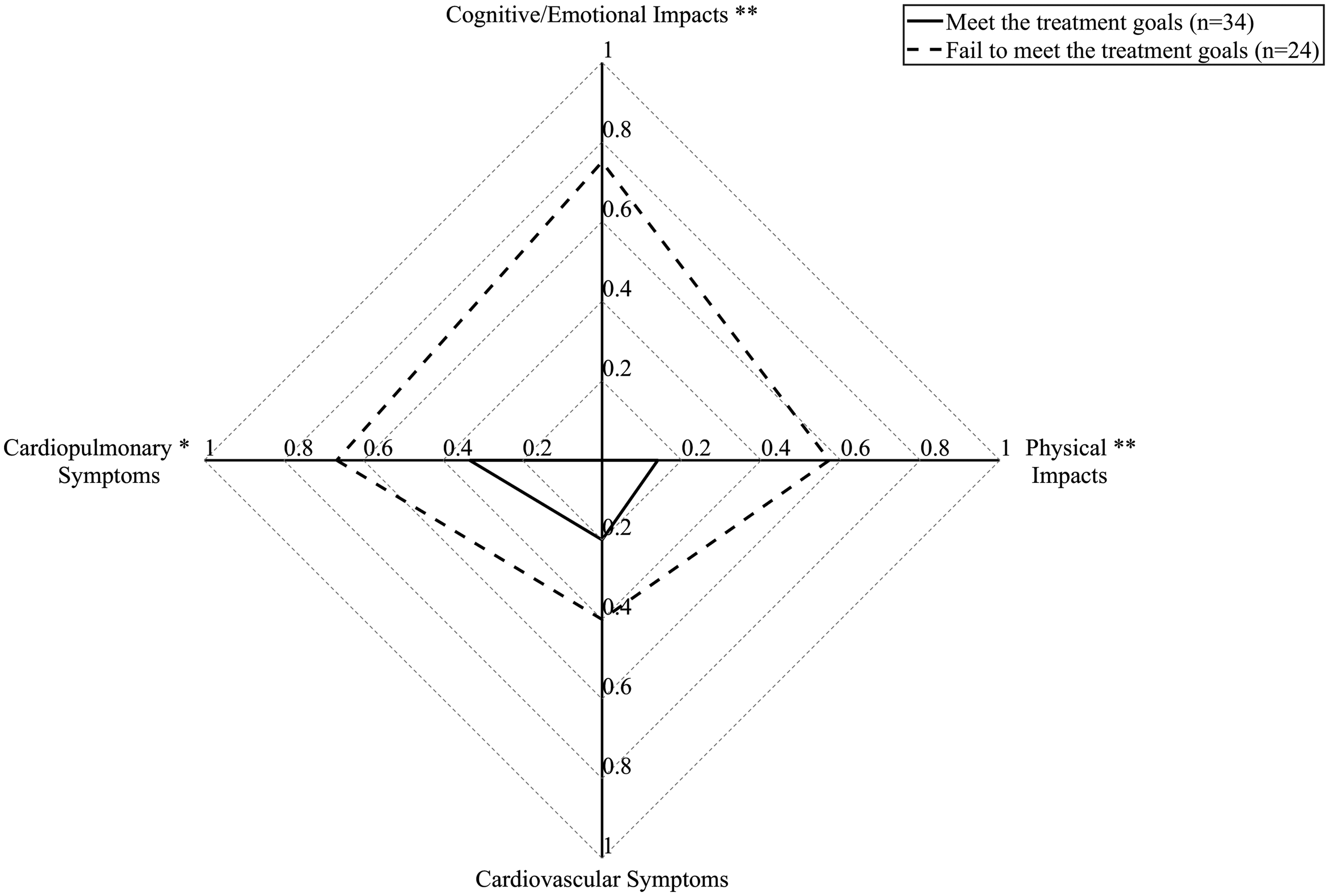
PAH-SYMAPCT scores in patients with CTD-PAH who met the treatment goals compared with those who did not meet the goals at baseline. *
Results of logistic regression analysis of relationship between quality of life and treatment goals achievements status in 6 to 12 months.
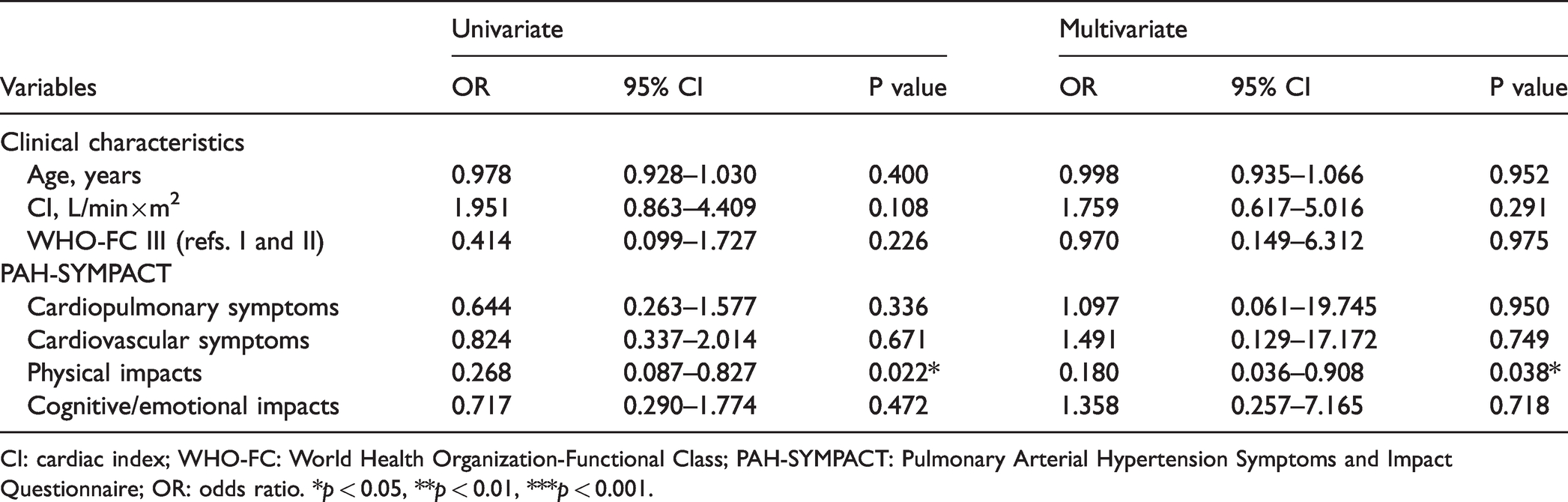
CI: cardiac index; WHO-FC: World Health Organization-Functional Class; PAH-SYMPACT: Pulmonary Arterial Hypertension Symptoms and Impact Questionnaire; OR: odds ratio. *
Discussion
This is the first study to indicate that PAH-SYMPACT have good validity and sensitivity in assessing QoL of CTD-PAH patients with RHC-based diagnosis. We also identify the relationship between baseline clinical characteristic, TGA outcomes and QoL scores. Furthermore, physical impacts domain in PAH-SYMPACT appears to be a potential predictive factor affecting TGA status in medium-term follow-up.
The Chinese translated version PAH-SYMPACT demonstrated fair psychometric properties in our study. But three items including “think clearly,” “walk slowly on a flat surface” and “wash or dress self” suggest poor factor loadings in CFA, especially the “think clearly” item. Except for the limited number of subjects, another reason for this situation might be the different underlying PAH etiology compared to the original validation trial. 6 The cognitive impairments in PAH patients may be affected by age 20 and CTD-PAH patients may have an earlier diagnosis and treatment than idiopathic PAH patients because of other inflammatory symptoms. 21 The relatively mild to moderate PAH symptoms (WHO-FC) of our patients is also a possible reason. Moreover, arthritis and musculoskeletal involvement in CTD-PAH patients (especially in SSc and RA) could also have a major impact on QoL, which could be confounding factors for PAH-specific QoL evaluation. Thus, the adaption of disease-specific QoL measurements into different types of PAH needs to be studied further.
In previous studies, the main clinical factors associated with QoL in PAH patients were WHO-FC and 6-minute walking distance (6MWT).22,23 Our study focused on the relationship among WHO-FC, CI, QoL and risk stratification during the follow-up. The results show that physical impacts scores are the predictive factors for TGA status in 6–12 months. Physical activity has been shown to significantly impact clinical outcomes in the management of autoimmune disease. 24 Our finding also suggest physical component of the PAH-SYMPACT questionnaire is a significant predictor of follow-up outcome. Hence, we propose that QoL evaluation deserves more attention in routine care visits, especially the physical functioning related domains. Our results underline that future studies should pay more attention to physical activity domain of QoL when trying to incorporate QoL in risk stratification criteria.
The goal-oriented treatment in PAH was originally proposed by Hoeper et al. in 2005 25 and “Treat-To-Target” approach was already recommended in the management of several rheumatological disease.26,27 Our data suggests that baseline life quality assessment is significantly related to TGA in patients with CTD-PAH, which is consistent with studies using other disease-specific QoL scales in PAH. 28 Thus, it is possible to incorporate QoL into the composite PAH treatment goals in the future. Our study reinforces the need for further research into appropriate PAH-specific QoL measurements and better strategy to optimize health-related QoL in long-term monitoring and therapy adjustment.
A major strength of our study lies in that it is the first research on QoL assessment in CTD-PAH patients and comprehensive evaluation of its relationship with RHC data and TGA outcomes. Our study has several limitations too. First, the lack of information about 6-minute walking distance, occupation, income status and repeated QoL assessment during follow-up need further investigation. Second, the sample size was relatively inadequate and potential confounding variables were not adjusted. Third, CTD-PAH treatment comprises immune-modulators and pulmonary vasodilators. The impact of medical therapies on TGA and quality of life needs to be further evaluated. Another limitation is that most of our CTD-PAH patients were WHO FC I/II/III, further researches are needed in severe PAH patients.
In conclusion, among patients with CTD-PAH, PAH-SYMPACT is a valid and reliable instrument for assessing the impact of the illness and its treatment on QoL. This study also provides a comprehensive analysis of relationship between QoL and clinical outcomes. Our finding also calls for continued monitoring of QoL status in CTD-PAH management which may be a potential way to remotely detect the deterioration of the disease.
Supplemental Material
sj-pdf-1-pul-10.1177_20458940211029899 - Supplemental material for Quality of life in ambulatory pulmonary arterial hypertension in connective tissue diseases and its relationship with risk stratification
Supplemental material, sj-pdf-1-pul-10.1177_20458940211029899 for Quality of life in ambulatory pulmonary arterial hypertension in connective tissue diseases and its relationship with risk stratification by Yue Shi, Yanling Meng, Xingbei Dong, Yang Liu, Yongtai Liu, Jinzhi Lai, Zhuang Tian, Jiuliang Zhao, Jinmin Peng, Qian Wang, Mengtao Li and Xiaofeng Zeng in Pulmonary Circulation
Footnotes
Authors’ contribution
J. P., Q. W., M. L. and X. Z. conceived and designed this study. Y. S., Y. M., X. D., Y. L., Y. L., J. L., Z. T. and J. Z. enrolled the patients, collected and analyzed the data, and wrote the manuscript. All authors have read and approved this final manuscript.
Ethical approval
This study received approval from the ethics committee of Peking Union Medical College Hospital.
Conflict of interest
The author(s) declare that there is no conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Chinese National Key Research R&D Program (grant numbers 2017YFC0907601, 2017YFC0907602 and 2017YFC0907603), “13th Five-Year” National Science and Technology Major Project for New Drugs of the Ministry of Science and Technology of China (grant number 2019ZX09734001-002-004), and Ministry of Science and Technology and CAMS Innovation Fund for Medical Sciences (CIFMS) (2019-I2M-2-008).
Guarantor
Not applicable.
Supplemental material
Supplemental material for this article is available online
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
