Abstract
Background:
Postoperative delirium (POD) is associated with higher risks of postoperative complications and mortality (2- to 3-fold increase). Studies investigating the effect of intraoperative ketamine on POD risk have yielded conflicting results. This study aimed to assess the effects of intraoperative ketamine and its more potent version, esketamine, on POD.
Design:
Systematic review and meta-analysis.
Objective:
To evaluate the effect of intraoperative ketamine/esketamine on the incidence of POD.
Methods:
We adhered to PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines, and searched the PubMed, Embase, Medline (Ovid), Cochrane, Scopus, and Web of Science databases for the MeSH terms “ketamine” and “emergence delirium” from database inception to July 10, 2024. The primary outcome was POD incidence following general anesthesia. Data were analyzed using a common effects model, with between-study heterogeneity tested using the I2 statistic, and relative risk (RR) with 95% confidence intervals (CIs) for dichotomous data was used as the effect measure.
Results:
A total of 18 studies with a total of 1571 participants met eligibility criteria. A meta-analysis of all studies suggests that the intraoperative use of ketamine/esketamine may reduce the incidence of POD (RR = 0.71, 95% CI: 0.56, 0.90, p < 0.01). In the drug subgroup, esketamine demonstrated enhanced efficacy in preventing POD compared to ketamine (RR = 0.59, 95% CI: 0.38, 0.90, p = 0.02). In addition, subanesthetic doses of ketamine/esketamine (⩽0.5 mg/kg) contributed to POD prevention (RR = 0.52, 95% CI: 0.34, 0.79, p < 0.01), whereas higher doses (>0.5 mg/kg) showed no statistically significant effect (RR = 0.89, 95% CI: 0.66, 1.21, p = 0.46). Further analysis revealed additional benefits of ketamine/esketamine in reducing POD incidence in cardiac surgery (RR = 0.46, 95% CI: 0.31, 0.68, p < 0.01), in the elderly (RR = 0.68, 95% CI: 0.52, 0.91, p < 0.01), and in the first 24 h post-surgery (RR = 0.52, 95% CI: 0.29, 0.94, p = 0.03).
Conclusion:
Our findings suggest that perioperative administration of ketamine/esketamine had a protective effect against the incidence of POD, with esketamine demonstrating superior efficacy compared to ketamine. The treatment effect exhibited a dose-response relationship, with subanesthetic doses showing greater efficacy. Furthermore, ketamine/esketamine may offer additional benefits for patients with specific risk factors.
Introduction
Postoperative delirium (POD), a challenging-to-mitigate surgical complication, is defined as delirium occurring during hospitalization within the first postoperative week, characterized by acute impairments in cognitive function and attention. 1 It is classified into three subtypes: hyperactive (irritability, hallucinations), hypoactive (lethargy, reduced responsiveness), and mixed, with the hypoactive subtype often underdiagnosed due to its subtle symptoms. 2 While the general prevalence is relatively low in adult surgery (2%–3%), the incidence increases significantly in elderly patients (>60 years old) and those undergoing cardiac or major surgeries.3–5 Furthermore, POD is strongly associated with elevated mortality rates, accelerated cognitive and functional decline, prolonged hospital stays, and increased annual healthcare costs. 6 Consequently, POD has emerged as a critical public health concern, now formally incorporated into patient safety initiatives and recognized as a key metric for postoperative recovery quality assessment. 7
Despite the substantial health burdens imposed by POD, its underlying biological mechanisms remain unclear. 8 POD develops through the combined effects of risk factors including advanced age, major surgery, postoperative pain, neuroinflammation, and hemorrhage.9,10 The pathological mechanisms involve neuroinflammatory responses accompanied by glutamatergic system disruption, in which proinflammatory cytokine release and excessive N-methyl-D-aspartate receptor (NMDA) activation collectively contribute to neuronal injury. The synergistic interactions among these multifactorial etiologies complicate POD pathogenesis, making prevention and treatment clinically challenging. As a result, the prevention and management of POD remain difficult. Current therapeutic options remain limited in both prophylactic efficacy and interventional effectiveness, highlighting an urgent need for mechanistic studies to identify novel treatment targets and develop evidence-based interventions.
As a multimodal NMDA receptor antagonist, ketamine demonstrates unique therapeutic potential extending beyond its established applications in chronic pain management and treatment-resistant depression. Its primary mechanism involves a noncompetitive blockade of glutamatergic neurotransmission through NMDA receptor inhibition, thereby modulating excitatory synaptic plasticity and preventing glutamate-induced excitotoxicity. Mechanistically, its tripartite pharmacological profile encompasses anti-inflammatory, neuroprotective, and neuroplasticity-modulating properties, all of which are closely linked to glutamate homeostasis, making it a biologically plausible candidate for the prevention and treatment of POD. 11 Clinical evidence indicates that opioid consumption and inadequately controlled postoperative pain constitute independent predictors for POD.12,13 Importantly, some studies have shown that intravenous ketamine reduces opioid use and alleviates postoperative pain.14,15 The association between delirium and depression is complex and controversial. Some studies suggest that the two may share pathophysiologic pathways, such as glutamatergic dysregulation and neuroinflammation, which further supports ketamine’s therapeutic rationale. 16 Ketamine is known to have significant antidepressant effects,17–19 and S-ketamine (esketamine) is a racemic mixture of ketamine with stronger antidepressant effects and fewer side effects, and thus potentially anti-delirium effects.20,21 Moreover, the psychotropic properties of ketamine can complicate this potential relationship by causing adverse neuropsychiatric effects, ultimately leading to delirium. Not surprisingly, clinical studies on intraoperative ketamine use for POD prevention have yielded inconsistent results. 22
Therefore, we conducted a systematic review and meta-analysis to synthesize the existing evidence on the association between intraoperative ketamine/esketamine and the incidence of POD.
Methods
Protocol and registration
We followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. 23 Before conducting the study, we established the study protocol, including eligibility criteria, outcomes, and statistical methods, which has been registered in PROSPERO (CRD42023392286).
Eligibility criteria
Inclusion criteria were established before article reviews as follows: (1) adults (≥18 years) and undergoing general anesthesia; (2) intraoperative use of ketamine/esketamine vs. placebo/no intervention; (3) patients diagnosed with POD using standardized diagnostic criteria (Confusion Assessment Method (CAM), Confusion Assessment Method for the diagnosis of ICU delirium (CAM-ICU), Intensive Care Delirium Screening Checklist (ICDSC), Nursing Delirium Screening Scale (Nu-DESC), and Mini-Mental State Examination (MMSE)); (4) incidence of POD as an efficacy outcome using standardized measures.
Exclusion criteria were as follows: (1) previous cognitive impairment (dementia, psychosis, or psychiatric disorders) or cerebrovascular disease; (2) positive results of a delirium assessment one day before surgery; (3) sensitivity to ketamine/esketamine; (4) a history of substance abuse (potentially increasing the risk of serious complications, e.g., pheochromocytoma and aortic coarctation, from a significant increase in blood pressure during surgery); and (5) use of antipsychotic medications.
Information sources and search
A comprehensive literature search was conducted in PubMed, Embase, Medline (Ovid), Cochrane Library, Scopus, and Web of Science databases using MeSH subject terms “ketamine” and “emergence delirium” to retrieve articles published from inception to July 10, 2024. The search strategy can be found in Supplemental Material 1.
Study selection
Two reviewers (CCC and NZ) independently reviewed the titles and abstracts, retrieved and reviewed full eligible articles, and scored them for inclusion. Discrepancies between the reviewers were resolved with a third reviewer (JXH).
Data collection process
Data extraction was performed by two reviewers (CCC and NZ) using a standardized form, capturing demographic details, interventions, comparisons, preoperative and perioperative delirium scores, study methods, dose, and route of ketamine/esketamine administration.
Outcomes
We recorded changes in delirium severity from baseline to the study endpoint using a validated delirium rating scale (CAM/CAM-ICU/ICDSC/Nu-DESC/ MMSE) as the outcome measure. The primary endpoint was the occurrence of POD. The effectiveness of the intervention in preventing POD was evaluated by comparing the occurrence of POD in the intervention and control groups.
Risk of bias
The risk of bias was assessed using the Cochrane Collaboration bias assessment tool, 24 which examines potential selection, performance, detection, attrition, reporting, and other biases. Two reviewers (CCC and NZ) independently assessed the risk of bias in each included study, and a third reviewer (JXH) was consulted to resolve any disagreements.
Analytic methods
Both meta-analysis and forest plotting were conducted in R (version 4.2.1) using the meta package. POD was analyzed as a dichotomous variable (yes/no). The relative risk (RR) with 95% confidence intervals (CIs) was used as the effect measure for dichotomous data, with a p-value of <0.05 considered statistically significant.
In meta-analyses, I2 (calculated as I2 = (Q − df)/Q × 100%, where Q is Cochran’s heterogeneity statistic and df denotes degrees of freedom) quantifies the proportion of total variability in effect estimates attributable to true between-study heterogeneity, thereby guiding the selection of statistical models (fixed-effect vs random-effects). τ2 (tau-squared) estimates the absolute variance of true effect sizes across studies, complementing I2 by providing a clinically interpretable measure of heterogeneity magnitude. Cochran’s Q-test, which follows a chi-square distribution, evaluates the statistical significance of observed heterogeneity; a threshold of p < 0.10 is conventionally adopted to reject the null hypothesis of homogeneity. Together, these metrics synergistically address heterogeneity across statistical (Q-test), relative (I2), and absolute (τ2) dimensions, ensuring rigorous interpretation and methodological transparency.
We assessed the heterogeneity using the I2 statistic values according to Cochrane's guidelines as follows: <25% indicates low heterogeneity, 25%–50% indicates moderate heterogeneity, 50%–75% indicates high heterogeneity, and >75% indicates extreme heterogeneity. Given that the degree of heterogeneity among studies was initially unknown, a common effects model was used for the analysis. 25 Subsequent subgroup analyses were conducted based on factors such as ketamine versus esketamine, age, dose, type of surgery, and different time points. Publication bias was evaluated using funnel and Galbraith plots, which estimate the effect size against the standard error. 26 Sensitivity analysis was used to explore sources of heterogeneity when heterogeneity was large.
Results
Study selection
The The search strategy identified 1882 records. After duplicate removal, 864 unique records were searched by title and abstract. An additional 819 irrelevant records were excluded, leaving 45 articles for full-text review. Of these, 18 randomized controlled trials (RCTs) conducted between 2013 and 2024 met the final study inclusion criteria (Figure 1).27–44

Flow chart of selection of the included studies.
Characteristics of studies, participants, and interventions
Table 1 provides an overview of the study characteristics. The 18 RCTs involved 1571 patients. All were aged 18 years or older and had undergone surgery under general anesthesia. The studies can be categorized by the type of surgery as follows: six abdominal surgeries, four major cardiac surgeries, two orthopedic surgeries, one gynecological surgery, one thyroid surgery, one cranio-encephalic surgery, one breast surgery, and two studies with no restriction on the type of surgery.
Summary of study characteristics.
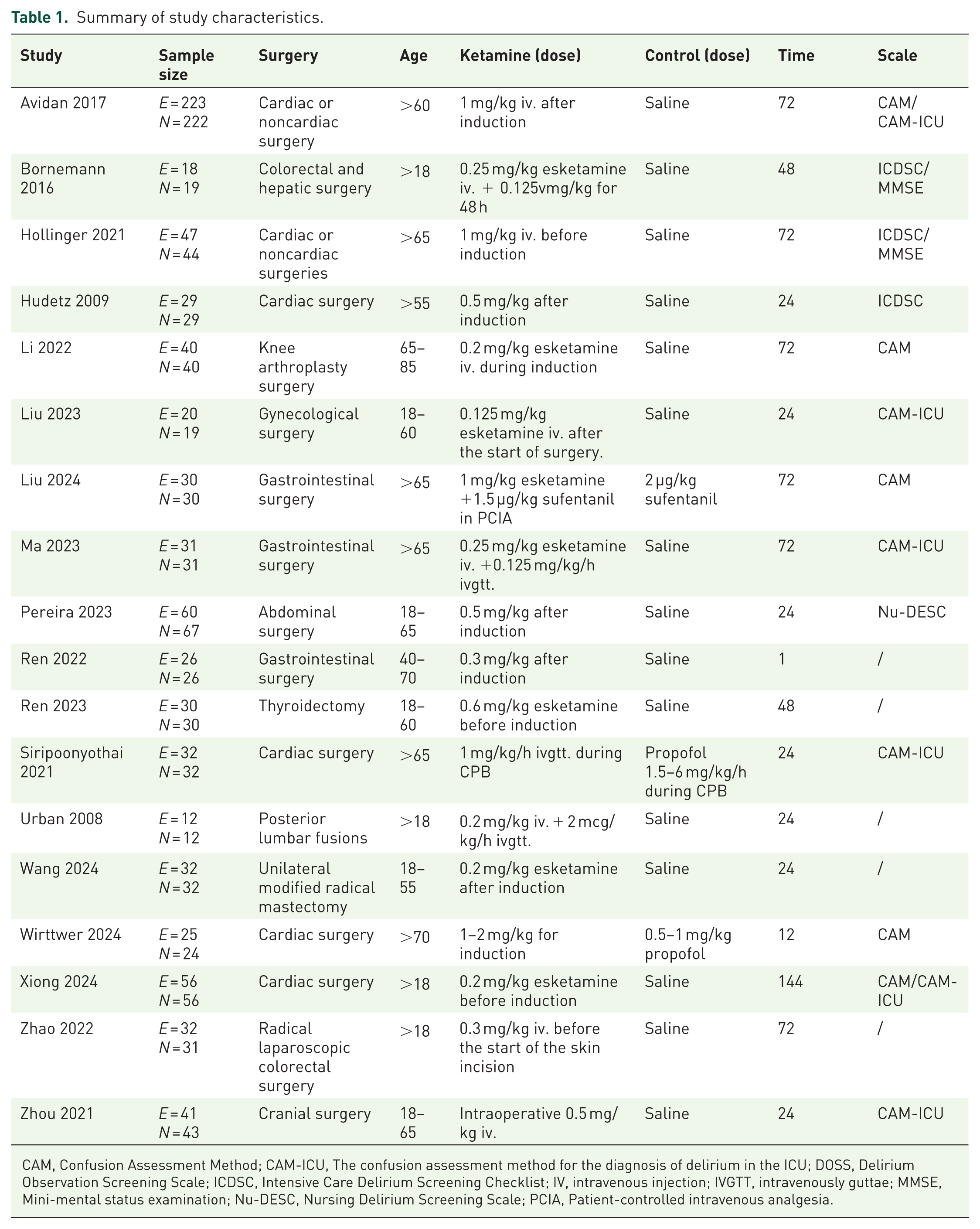
CAM, Confusion Assessment Method; CAM‑ICU, The confusion assessment method for the diagnosis of delirium in the ICU; DOSS, Delirium Observation Screening Scale; ICDSC, Intensive Care Delirium Screening Checklist; IV, intravenous injection; IVGTT, intravenously guttae; MMSE, Mini-mental status examination; Nu-DESC, Nursing Delirium Screening Scale; PCIA, Patient-controlled intravenous analgesia.
Study quality and risk of bias
We evaluated the quality of the RCTs included in the meta-analysis and determined that they were either of high or moderate quality, with none of the studies presenting “high-risk” factors (Figure 2).

Risk of bias. (a) Summary of methodological quality across all studies. (b) Detailed quality assessment for individual studies.
Publication bias
All studies fell within the 95% confidence intervals of the funnel plot and the Galbraith radial plot, and the funnel plot was relatively symmetrical, indicating that there was no significant publication bias (Figure 3).

Assessment of publication bias and heterogeneity. (a) Funnel plot of the effect of ketamine/esketamine in preventing POD. (b) Galbraith radial plot of the effect of ketamine/esketamine in preventing POD.
Effects of ketamine/esketamine on the incidence of POD
Meta-analysis of 18 RCTs (Figure 4) revealed that perioperative administration of ketamine/esketamine statistically significantly reduced POD incidence compared to controls (RR = 0.71, 95% CI: 0.56, 0.90, p < 0.01) under a common-effect model (I2 = 15%). Subgroup analyses demonstrated divergent efficacy patterns: ketamine alone showed no statistically significant benefit (RR = 0.77, 95% CI: 0.58, 1.02, p = 0.07; Figure 5), whereas esketamine exhibited clinically meaningful risk reduction (RR = 0.59, 95% CI: 0.38, 0.90, p = 0.02). Subanesthetic doses (single intravenous injection ⩽0.5 mg/kg) statistically significantly reduced POD incidence (RR = 0.52, 95% CI: 0.34, 0.79, p < 0.01; Figure 6), 36 while high-dose regimens (>0.5 mg/kg) showed no statistically significant effect (RR = 0.89, 95% CI: 0.66, 1.21, p = 0.46).

Forest plots of the effect of ketamine/esketamine in preventing POD.

Forest plots of differences in the incidence of POD between ketamine and esketamine.
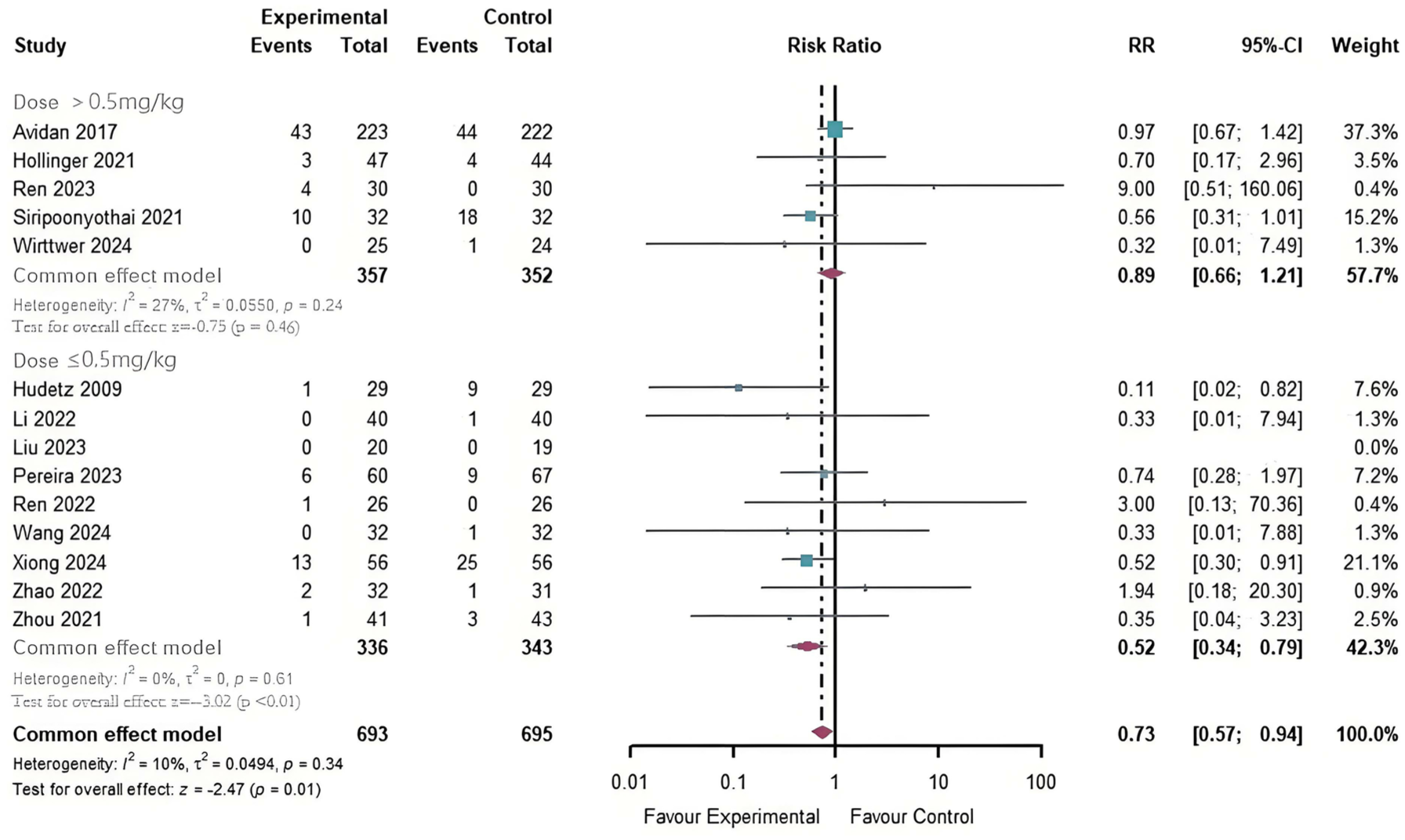
Forest plot of the effect of different doses of ketamine/esketamine in preventing POD.
Effect of ketamine/esketamine on the incidence of POD in patients of different ages and types of surgery
We created subgroups based on age (>60 years old), including nine studies, which are presented in the forest plot (Figure 7). Given that the inclusion criterion for the Hudetz study was age >55 years, and the mean ages of the experimental and control groups were 68 ± 8 and 60 ± 8, respectively, we determined that the study still met the criteria and included it in the analysis. The forest plot showed low heterogeneity (I2 = 25%) and indicated a beneficial effect of ketamine/esketamine on the incidence of POD in elderly patients (RR = 0.68, 95% CI: 0.52, 0.91, p < 0.01). In the subgroups (nine studies) with patients aged less than 60 years old, the results showed low heterogeneity (I2 = 13%) and no statistically significant effect of ketamine/esketamine on POD incidence in younger patients (RR = 0.78, 95% CI: 0.51, 1.18, p = 0.24).

Forest plots of the effect of ketamine/esketamine in preventing POD in the elderly.
Due to the large variety of surgery types in the included studies and to avoid having too few studies in each group, we pooled similar surgeries and focused on cardiac and general surgeries (Figure 8). In the cardiac surgery subgroup, heterogeneity was low (I2 = 0%), and ketamine/esketamine administration significantly reduced the incidence of POD (RR = 0.46, 95% CI: 0.31, 0.68, p < 0.01). The general surgery subgroup primarily included colorectal, gynecological, and thyroid surgeries. The results showed low heterogeneity (I2 = 25%), and ketamine/esketamine had no statistically significant effect on the incidence of POD (RR = 0.81, 95% CI: 0.48, 1.38, p = 0.44).

Forest plots of the effect of ketamine/esketamine in preventing POD in different surgery.
Effect of ketamine/esketamine on the incidence of POD at different time points
We divided the subgroups based on the time points of POD in each study into three groups: 24 h (within 24 h), 48 h, and 72 h (including beyond 72 h). Nine studies were included in the 24 h subgroup, which showed moderate heterogeneity (I2 = 27%). The results indicated that ketamine/esketamine could have beneficial effects (RR = 0.52, 95% CI: 0.29, 0.94, p = 0.03). In contrast, the 48 h subgroup (RR = 0.68, 95% CI: 0.38, 1.20, p = 0.18) and the 72 h subgroup (RR = 0.79, 95% CI: 0.59, 1.04, p = 0.09) did not show statistically significant effects (Figure 9).

Forest plots of the effect of ketamine/esketamine in preventing POD at different time points.
Discussion
The primary finding is that perioperative administration of ketamine/esketamine significantly reduces the incidence of POD compared to controls, underscoring the potential of NMDA receptor antagonists for POD prevention. Subgroup analyses revealed that esketamine may be more effective than ketamine in preventing POD. Moreover, the effects of ketamine/esketamine were dose-related, with subanesthetic doses (⩽0.5 mg/kg) significantly reducing the incidence of POD. In addition, the therapeutic benefit of ketamine/esketamine appeared to be more pronounced in elderly patients, cardiac surgery, and early-onset POD (<24 h postoperatively).
Compared to previous systematic reviews and meta-analyses, this review was updated to focus solely on POD as the primary outcome and includes a larger number of studies.31–38,40–43 Additionally, only published RCTs were considered, excluding observational studies and gray literature. Furthermore, subgroup analyses were conducted to explore various factors, including ketamine versus esketamine, dose, age, type of surgery, and different time points. A Bayesian network meta-analysis, encompassing 22 studies with 16,562 patients undergoing cardiac surgery, found that pre-anesthetic ketamine dosing significantly reduced the incidence of POD. 45 In a similar vein, another study demonstrated that ketamine is a safe and effective analgesic that does not increase the incidence of delirium when used perioperatively in total hip arthroplasty (THA) and total knee arthroplasty (TKA). 46 Despite the increasing number of studies on ketamine/esketamine and POD, its role remains controversial.47,48 One of the more compelling studies is an international multicenter RCT that recruited 672 elderly patients between 2014 and 2016, finding that administering a single anesthetic dose of ketamine during surgery did not reduce the incidence of POD. 49 However, the lack of surgical type restrictions in this study may limit the generalizability of conclusions. A recent retrospective study demonstrated that the administration of low-dose esketamine during thoracic surgery in elderly patients significantly reduced the incidence of POD and postoperative cognitive dysfunction, highlighting its neuroprotective potential. 50 In contrast, a meta-analysis of six studies concluded that the effect of ketamine on POD remains uncertain, but it may provide some protection against POCD (Postoperative cognitive dysfunction). 51 These contradictory findings may reflect methodological heterogeneity among the studies. Overall, differences in baseline characteristics such as patient age and disease status, as well as variations in POD evaluation criteria, ketamine/esketamine dosages, and types of surgery, may be influential factors contributing to the variability in the clinical efficacy of ketamine for POD.
While delirium can affect hospitalized patients across all age groups, current delirium prevention initiatives predominantly target older adults due to their elevated risk. 52 After conducting a subgroup analysis based on age stratification, we found that ketamine/esketamine had a protective effect on POD in older patients but not in younger. Age-related neurodegenerative changes increase the elderly patients’ vulnerability to intraoperative stress and anesthetic drugs.53–55 Ketamine, an NMDA receptor antagonist, protects by reducing neuronal hyperexcitability.56,57 Therefore, the neuroprotective effect of ketamine/esketamine may be more pronounced in elderly patients.
The type of surgery is also a significant risk factor for POD, as major surgeries tend to involve higher levels of perioperative inflammation, 58 and ketamine may help prevent POD by alleviating this inflammation. Our subgroup analysis indicated a protective effect of ketamine/esketamine on the incidence of POD in patients undergoing cardiac surgery. We hypothesize that ketamine/esketamine may provide better protection in patients with specific risk factors. Compared to ketamine, esketamine exhibits a superior affinity for NMDA receptors, which are crucial in mediating neuroprotective and anti-inflammatory effects, as NMDA receptor antagonists have been demonstrated to be neuroprotective.59,60 This implies that esketamine may be more effective in preventing POD. Our analysis further demonstrated that esketamine had a protective effect against POD, while ketamine did not. However, the current evidence remains inconclusive, and larger studies are needed to validate these findings in the future.
Inconsistent results may also reflect ketamine’s multifaceted and complex pharmacology. Ketamine is a commonly used anesthetic and analgesic agent, however, its use is associated with several side effects, including central nervous system disturbances (e.g., hallucinations and agitation), cardiovascular effects (e.g., hypertension, tachycardia), respiratory complications (e.g., laryngospasm and increased secretions), and a potential risk of dependence. 61 To reduce adverse effects, subanesthetic doses of ketamine are commonly administered for analgesia, combined with other agents such as benzodiazepines or anticholinergic drugs for anesthesia. Ketamine primarily inhibits NMDA receptors but also modulates multiple ion channels and interacts with the catecholaminergic, cholinergic, and opioid systems.62,63 Emerging evidence suggests that its beneficial effects on POD may involve anti-inflammatory, neuroprotective, and antidepressant mechanisms.64,65 Notably, inflammatory responses are implicated in POD pathogenesis and progression. 66 Surgically induced peripheral inflammation can affect inflammatory processes in the central nervous system. 67 Some studies have reported that ketamine administered during general anesthesia reduces postoperative concentrations of IL-6, TNF-α, and other inflammatory markers.68,69 In contrast, a recent study showed that postoperative levels of CRP, IL-6, and TNF-α in patients who received esketamine intraoperatively were not significantly different from those in a control group.70,71 Nevertheless, ketamine has well-documented psychoactive properties. Perioperative inflammation alone may not fully explain the complex mechanisms of POD. In conclusion, the complex mechanisms underlying the effects of ketamine/esketamine on POD require further exploration.
Limitation
The studies included in this analysis exhibited small-sample bias and low-quality designs. Although the heterogeneity among studies was relatively low, these issues may still affect the reliability of the results. Furthermore, due to the limited number of available studies, subgroup analyses could not be performed for certain specific types of surgery (such as intracranial surgery, orthopedic surgery, gynecological surgery, and thyroid surgery), the methods of administration, and the effects of ketamine and esketamine in different subgroups. Future research should be more targeted and in-depth to address these issues.
Conclusion
Our findings suggest that perioperative administration of ketamine/esketamine may be effective in reducing the incidence of POD. In particular, esketamine demonstrated greater efficacy than ketamine in mitigating the risk of POD. The efficacy of both drugs was dose-dependent, with subanesthetic doses (⩽0.5 mg/kg) proving more effective in preventing POD compared to higher doses. Ketamine/esketamine may offer enhanced benefits for patients with specific risk factors. However, given the limited number of studies, the variable quality of the evidence, and the heterogeneity in the types of surgeries included, further research is warranted to explore the physiological mechanisms of ketamine/esketamine in relation to POD, their effects on specific clinical outcomes, and the comparative efficacy of ketamine and esketamine.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251339378 – Supplemental material for Efficacy of intraoperative ketamine/esketamine in the prevention of postoperative delirium: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tpp-10.1177_20451253251339378 for Efficacy of intraoperative ketamine/esketamine in the prevention of postoperative delirium: a systematic review and meta-analysis by Chengchuan Chen, Na Zhou and Jixin Hou in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253251339378 – Supplemental material for Efficacy of intraoperative ketamine/esketamine in the prevention of postoperative delirium: a systematic review and meta-analysis
Supplemental material, sj-docx-2-tpp-10.1177_20451253251339378 for Efficacy of intraoperative ketamine/esketamine in the prevention of postoperative delirium: a systematic review and meta-analysis by Chengchuan Chen, Na Zhou and Jixin Hou in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
