Abstract
Background:
More than 2% of the general population experience suicidal ideas each year and a large number of them will attempt suicide. Evidence-based therapeutic options to manage suicidal crisis are currently limited.
Objectives:
The aim of this study was to overview the findings on the use of ketamine and esketamine for the treatment of suicidal ideas and acts.
Design:
Systematic review.
Data Sources and methods:
PubMed, article references, and Clinicaltrials.gov up to June 30, 2022. Meta-analyses published within the last 2 years were also reviewed.
Results:
We identified 12 randomized controlled trials with reduction of suicidal ideation as the primary objective and 14 trials as secondary objectives. Intravenous racemic ketamine was superior to control drugs (placebo or midazolam) within the first 72 h, in spite of large placebo effects. Adverse events were minor and transient. In contrast, intranasal esketamine did not differ from placebo in large-scale studies. Limitations, clinical considerations, and opportunities for future research include the following points: large placebo effects when studying suicidal ideation reduction; small concerns about blinding quality due to dissociative effects; no studies on the risk/prevention of suicidal acts and mortality; lack of studies beyond affective disorders; no studies in adolescents and older people; lack of knowledge of long-term side effects, notably liability for abuse; no robust predictive markers; limited understanding of the mechanisms of ketamine on suicidal ideas; need for improved assessment of suicidal ideation in clinical trials; need for studies in outpatient settings, emergency room, and liaison consultation; need for research on ketamine administration; limited knowledge on the positive and negative effects of concomitant treatments.
Conclusion:
Overall, there is compelling evidence for a favorable short-term benefit-risk balance with intravenous racemic ketamine but not intranasal esketamine. The place of ketamine will have to be defined within a multimodal care strategy for suicidal patients. Caution remains necessary for clinical use, and pharmacovigilance will be essential.
Keywords
Introduction
In spite of a growing number of publications in suicidology over the last 30 years, 1 the prevalence of suicidal thoughts and behaviors remains high and therapeutic options limited. 2 The identification over the last two decades of the rapid antidepressive and anti-suicidal properties of ketamine3,4 has given rise to high hopes.
Ketamine was first synthesized in 1964 and originally used as a general anesthetic and analgesic veterinary drug since 1970.5,6 It is a racemic mixture containing equal parts of (R)-ketamine (or arketamine) and (S)-ketamine (or esketamine). It is a non-competitive antagonist of the N-methyl-D-aspartate receptor (NMDA-R), but pharmacological mechanisms of action extend beyond this receptor (e.g. AMPA receptors, opioid receptors, etc.) and possibly include a variety of intracellular alterations explaining their clinical effects. 7 It is available for various modes of administration including intravenous (IV), intranasal, intramuscular, intrarectal, and oral. This drug has several properties and indications, notably in acute and chronic pain treatment. 8 While initially described in anecdotal reports since the 1970s, its antidepressant properties have only been studied since the early 1990s. 9 In 2019, an intranasal spray of esketamine (Spravato®, Eskesia®) was approved for treatment-resistant depression and then the short-term treatment of moderate-to-severe depressive episodes by drug agencies, including the US Food and Drug Administration and the European Medicines Agency.
This article aims to review the scientific literature on the therapeutic and side effects of ketamine and esketamine on suicidal thoughts and behaviors. In our opinion, the recent publication of large studies justified an update. 10 To this aim, we systematically reviewed published randomized controlled trials (RCTs) and the most recent reviews and meta-analyses on this topic. Contrary to some previous reviews, we distinguished studies with a primary objective of reduced suicide risk from those in which suicidal improvement was only a secondary objective. These latter studies may lack statistical power and adequate measures of suicidal risk.
Our objective was to determine whether ketamine and esketamine can be recommended for the management of the suicidal crisis and highlight research direction based on the level of evidence and issues. Indeed, we want to underline here many problems about the investigation and treatment of suicidal thoughts and behaviors. These are notably difficult to define, assess, predict, understand, treat, and prevent, as discussed in Supplemental material. These issues should be integrated into the discussion about ketamine in suicidal thoughts and behaviors, and the interpretation of findings from clinical trials.
Methods
This was a systematic review. Writing of this article followed the Preferred Reporting Items for Systematic reviews and Meta-Analyses (PRISMA) guidelines (see checklist in Supplemental material). This review was not registered, and no protocol was prepared.
Search strategy
Studies were identified through searches in PubMed (suicid* AND ketamine OR esketamine) until June 30, 2022, ClinicalTrials.gov, and our own reference lists. We additionally considered several recent reviews within the last 2 years until June 30, 2022, including (1) one systematic review of systematic reviews, 11 two meta-analyses,12,13 and five reviews considered of good quality14–18 with (es)ketamine in suicidal ideation as the primary objective; (2) two reviews on interventions targeting suicidal risk and prevention;19,20 (3) and two Cochrane reviews,21,22 one meta-analysis 23 and one systematic review of meta-analyses 24 comprising secondary analyses of suicidal ideation.
Study selection
Eligibility criteria were as follows: double-blind randomized controlled trials (RCTs), using ketamine or esketamine (any administration modality and dose) versus a control drug, with suicidal thoughts or behaviors (but not non-suicidal self-injury) as primary or secondary objectives, in all types of mental disorders. We searched for articles published in English. Of the 1012 identified records (and 2 additional from reviews), 28 articles were finally selected (Figure 1). Two investigators (F.J. and R.C.) reviewed literature and extracted data into standardized spreadsheets: Countries of recruitment, year of publication, funding, sample size (total and per group), population including age, psychiatric condition, and setting, design (including parallel versus crossover), intervention including mode of administration, dose, duration of follow-up, and mono- versus multicentric feature, control drug, measure of suicidality, and main outcomes related to suicidality.
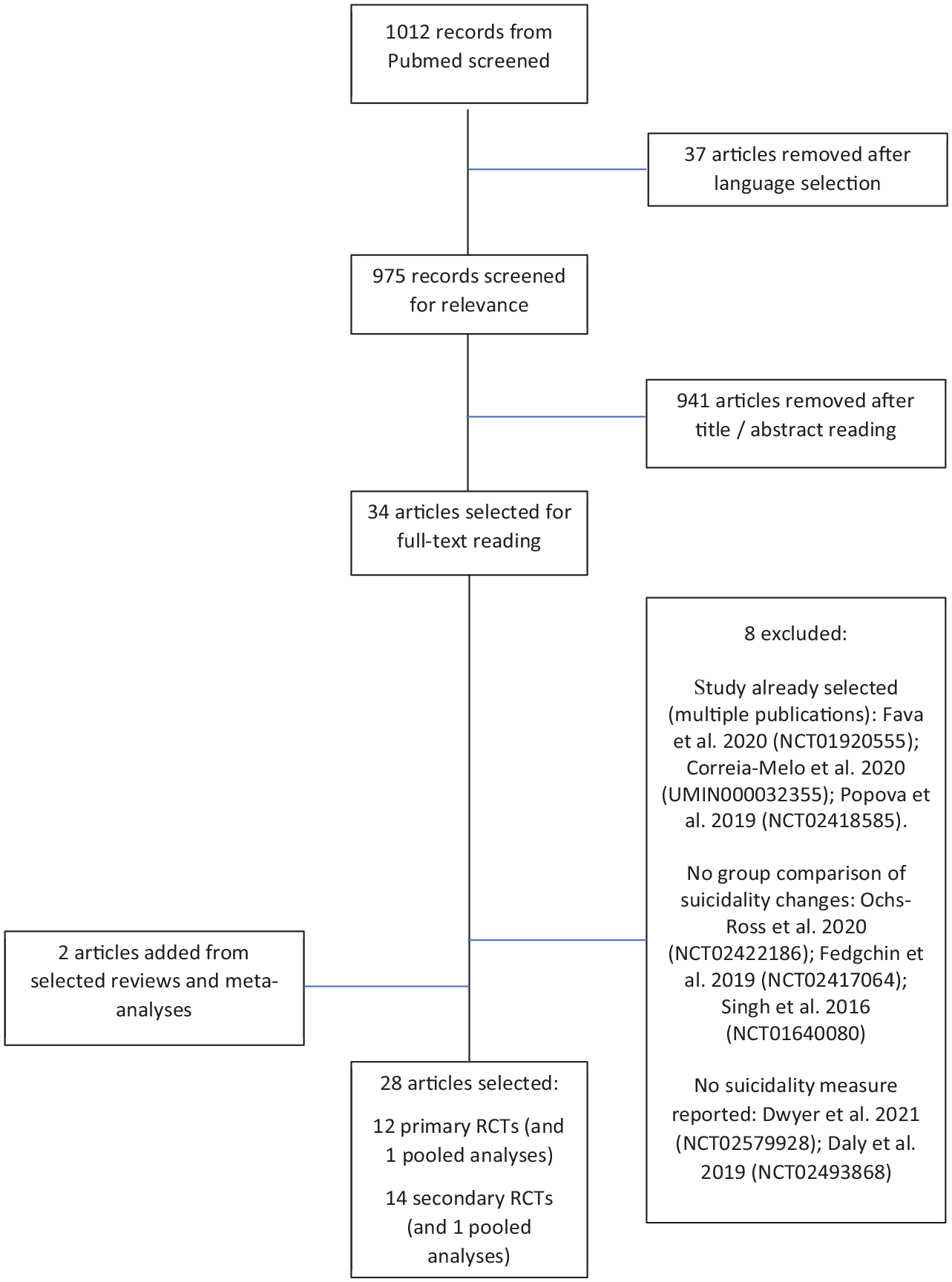
Flowchart.
Results
Global description of RCTs
No RCT investigated the effect of ketamine on the prevention of self-harming behaviors or suicide attempts or suicide mortality. Table 1 presents RCTs investigating the effect of ketamine on suicidal ideation as the main outcome (thereafter named ‘primary RCTs’) while Table 2 presents RCTs for which suicidal ideation was a secondary outcome (‘secondary RCTs’).
RCTs investigating the effect of ketamine on suicidal ideation as the primary outcome.
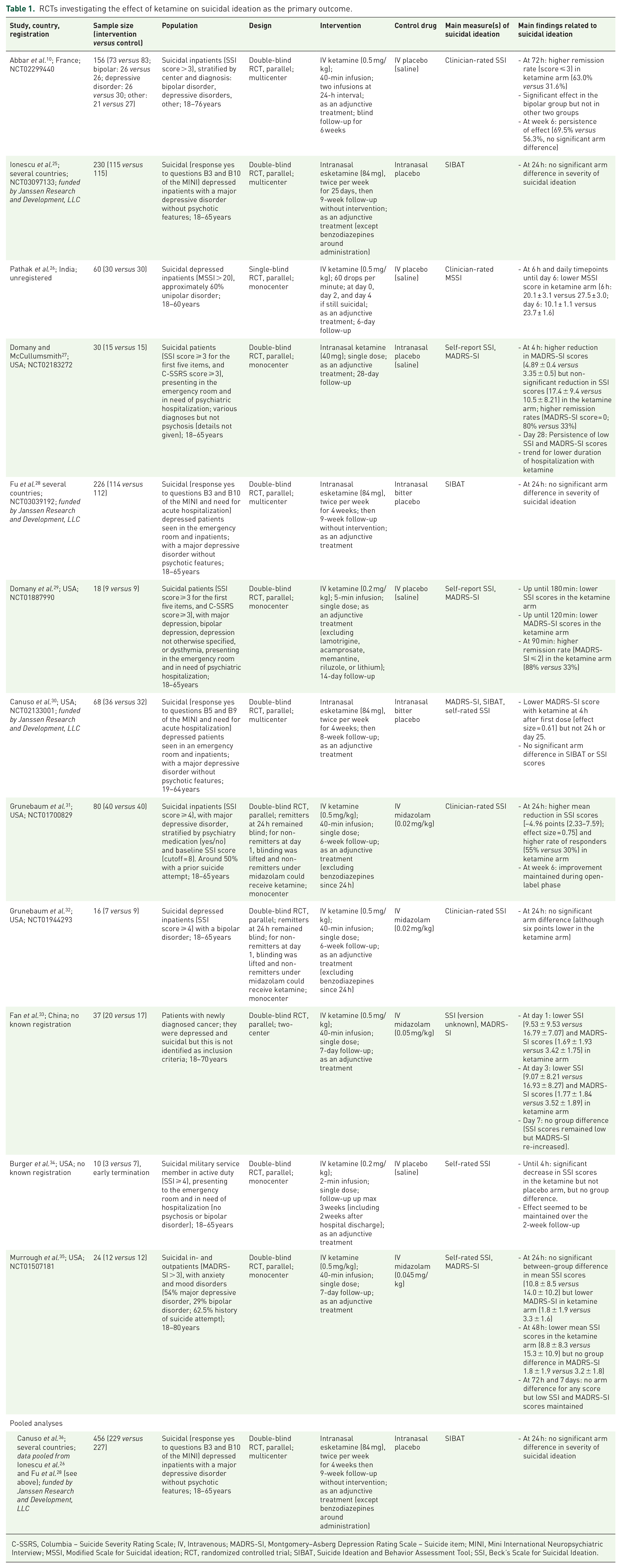
C-SSRS, Columbia – Suicide Severity Rating Scale; IV, Intravenous; MADRS-SI, Montgomery–Asberg Depression Rating Scale – Suicide item; MINI, Mini International Neuropsychiatric Interview; MSSI, Modified Scale for Suicidal ideation; RCT, randomized controlled trial; SIBAT, Suicide Ideation and Behavior Assessment Tool; SSI, Beck’s Scale for Suicidal Ideation.
RCTs investigating the effect of ketamine on suicidal ideation as a secondary outcome.
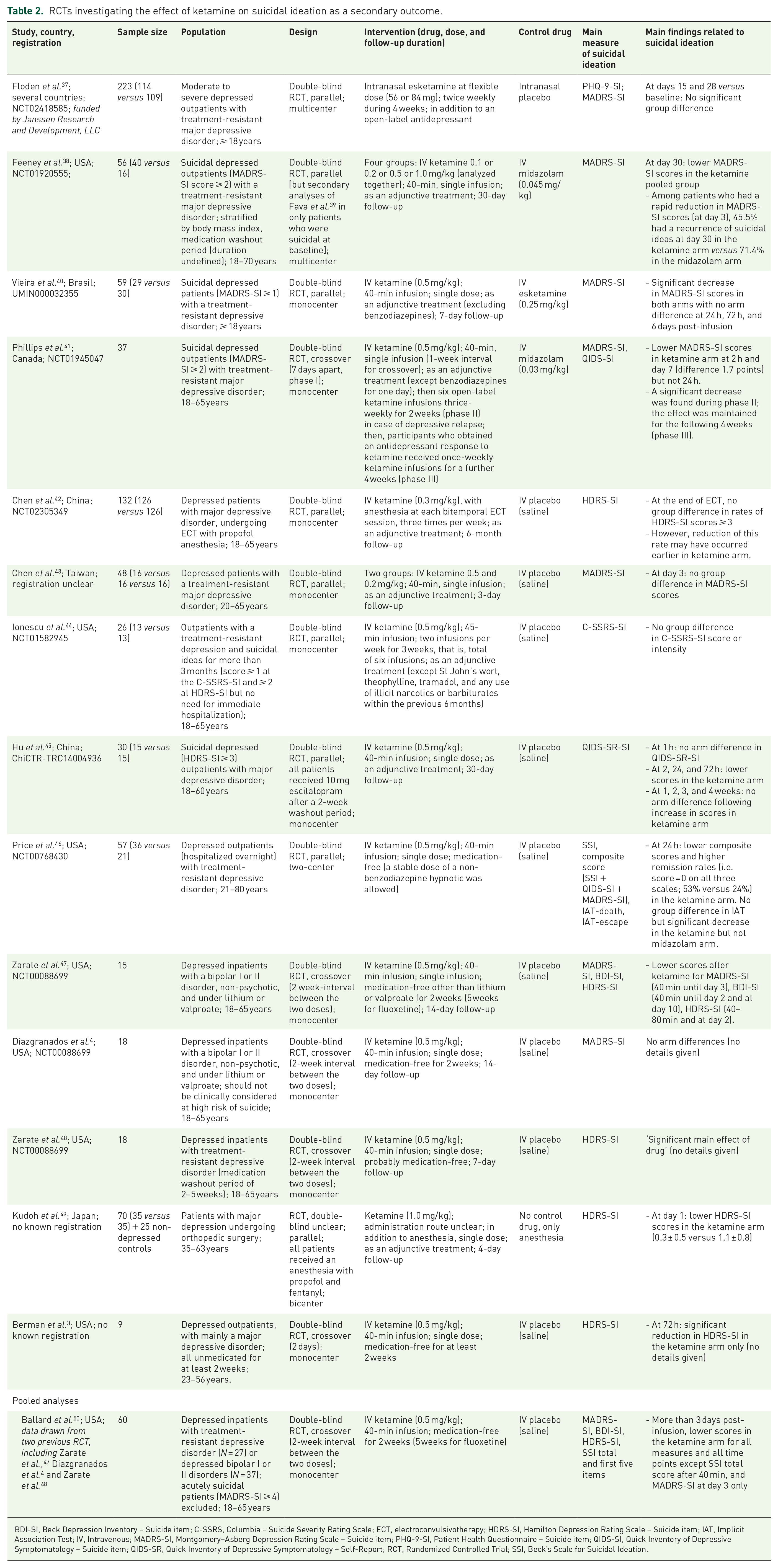
BDI-SI, Beck Depression Inventory – Suicide item; C-SSRS, Columbia – Suicide Severity Rating Scale; ECT, electroconvulsivotherapy; HDRS-SI, Hamilton Depression Rating Scale – Suicide item; IAT, Implicit Association Test; IV, Intravenous; MADRS-SI, Montgomery–Asberg Depression Rating Scale – Suicide item; PHQ-9-SI, Patient Health Questionnaire – Suicide item; QIDS-SI, Quick Inventory of Depressive Symptomatology – Suicide item; QIDS-SR, Quick Inventory of Depressive Symptomatology – Self-Report; RCT, Randomized Controlled Trial; SSI, Beck’s Scale for Suicidal Ideation.
We identified 12 primary RCTs published between 2015 and 2022 (total participants = 955) and 14 secondary RCTs between 2000 and 2022 (total participants = 798). Two additional articles reported analyses of pooled data from previously published RCTs (total participants = 516). Among the 26 studies, five RCTs used a crossover design (all secondary RCTs) and the rest a parallel design. Sample sizes ranged 10–230 in primary RCTs and 9–223 in secondary RCTs. Six primary RCTs (50%) and seven secondary RCTs (50%) included fewer than 40 participants. Four studies were multicentric among primary RCTs, and two among secondary RCTs. The United States accounted for 53.8% of all identified RCTs, and middle-income countries for 19.2%.
Population studied
Participants were mostly middle-aged adults (18–65 years), with a few studies allowing the recruitment of people above 65 years. None recruited adolescents, or specifically focused on young adults below 25 years or on people above 65 years. Gender representation was mixed in all studies.
Patients were suicidal at inclusion (with a specific suicidal inclusion criteria) in 11 primary RCTs (for 1 RCT, criteria about suicidal risk were not reported but patients were described as being suicidal) but in only 5 secondary RCTs (35.7%). The definition of suicidal ideation at inclusion varied greatly across these 17 studies: 5 used the Beck Scale for Suicidal Ideation (SSI, clinician or patient-rated), 2 studies combined it with the Columbia – Suicide Severity Rating Scale (C-SSRS), and 1 used the modified SSI (MSSI). Five studies used the suicide item of the Montgomery–Asberg Depression Rating Scale (MADRS). Three studies used specific items of the Mini International Neuropsychiatric Interview (MINI) combined with a need for hospitalization. Two studies used the suicidal item of the Hamilton Depression Rating scale (HDRS), combined with C-SSRS for one of them.
Patients were most often recruited during hospitalization or were hospitalized for the trial [92% of primary (one unknown) but only 28.6% of secondary RCTs]. Four studies recruited exclusively or partially from an emergency department.
‘Affective disorders’ were the main diagnoses for all studies. For the 12 primary RCTs, main diagnoses were various mood disorders (N = 4), bipolar disorders only (1), major depressive disorders only (4), depressed patients with various diagnoses at the emergency room (excluding psychosis; 2), and depressed patients with a newly diagnosed cancer (1). Among the 14 secondary RCTs, main diagnoses were treatment-resistant depressive disorders (8), major depressive disorder (2), bipolar disorder (2), various mood disorders (mainly major depressive disorder; 1), and depressed patients undergoing orthopedic surgery (1).
Intervention: mode of administration, drug and control condition
Among the 12 primary RCTs, one single dose of IV ketamine at 0.5 mg/kg over 40 min was used in four studies while one study used two infusions at 24-h interval and one study gave the possibility of two additional infusions after the first one according to the clinical response. Two studies used one single dose of IV ketamine at 0.2 mg over 2 or 5 min. One study used one single dose of intranasal ketamine at 40 mg. Intranasal esketamine at 84 mg twice a week for 4 weeks was used in the three studies funded by Janssen pharmaceutical company. The control condition always used the same protocol as for the intervention condition. IV or intranasal saline placebo was used in eight studies and IV midazolam at 0.02–0.05 mg/kg in four studies. In all these studies, drugs were given as adjunctive treatments. Four studies excluded benzodiazepines and one study excluded selected psychotropic drugs.
For the 14 secondary RCTs, one single dose of IV ketamine at 0.5 mg/kg over 40 min was used in seven studies. One study used a single dose of 1.0 mg/kg during the anesthesia for orthopedic surgery. One study had two groups of IV ketamine (single dose of 0.2 or 0.5 mg/kg) and another study had four groups (single dose between 0.1 and 1.0 mg/kg although data were pooled for the secondary analyses reported here). Three studies used repeated doses: one had a dose of 0.5 mg/kg twice a week for 3 weeks; one offered an initial single dose with possible repeated doses of ketamine over 6 weeks; and one used IV ketamine at 0.3 mg/kg during the anesthesia of each electroconvulsivotherapy (ECT) session. One study used repeated intakes of intranasal esketamine at 56 or 84 mg. The control conditions were an IV or intranasal saline placebo in 10 studies, midazolam (0.03 or 0.045 mg/kg) in 2 studies, IV esketamine at 0.25 mg in 1 study, and simple anesthesia in 1 study. Drugs were given as adjunctive treatments in nine studies, while a washout period was implemented in the five other studies. One study excluded benzodiazepines before intervention and one study various psychotropic drugs.
Scales used to measure outcomes
Different scales were used to measure suicidal ideation as a primary outcome. SSI (whether clinician- or patient-rated versions) was the main scale used. It was used alone in four primary RCTs and combined with the suicide item of MADRS in four studies. One study only used the MSSI. Finally, the three studies investigating intranasal esketamine used the Suicide Ideation and Behavior Assessment Tool (SIBAT).
In secondary RCTs, analyses over-relied on suicide items of depression scales. Four studies used only the suicide item of the HDRS, four studies used the suicide item of the MADRS [which was combined with the suicide items of Quick Inventory of Depressive Symptomatology – Self-Report (QIDS) in one additional study, the suicide item of Patient Health Questionnaire – Suicide item (PHQ-9) in one study, and those of Beck Depression Inventory (BDI) and HDRS in one further study], and one study used the suicide item of the QIDS. One study not only constructed a composite score from MADRS, SSI, and QIDS but also used a specific version of the neuropsychological test – Implicit Association Task. One study used the C-SSRS.
Duration of follow-up
The duration of follow-up was variable. In primary RCTs, durations were 7 days post-treatment (N = 3), 14 days (1), 28 days (1), 3 weeks (1), 6 weeks (3), and 13 weeks (3, intranasal esketamine studies). Most main outcomes were measured within the first 3 days. In secondary RCTs, durations of follow-up were also highly variable: 24 h (N = 1), below 30 days (9), 3 weeks (1), 4 weeks (1), 6 weeks (1), and 6 months (1).
Results of RCTs with suicidal ideation as primary outcome
Among the 12 RCTs for which suicidal ideation was a primary outcome, significant reduction in suicidal ideation was found within the first 72 h post-infusion in five studies.10,26,29,31,33 Among them, one large study reported higher overall remission rates (and not only response) versus placebo at 72 h: 63.0% versus 31.6%. 10 All used IV infusion of ketamine, at 0.5 mg/kg for 40 min in four studies and 0.2 mg/kg for 5 min in one study. Two studies used two infusions while the other studies used a single dose. The control drug was a saline placebo in three studies and midazolam in two studies. Diagnoses were various mood disorders in three studies, major depressive disorder in one study, and depressed patients with a cancer in one study. Although group differences tended to be non-significant with time, the maintenance of effect in the ketamine arm was observed over the observation period in all studies (6 days for two studies, 28 days for one study, and 6 weeks for two studies).
Two primary RCTs conducted in patients with various mood disorders27,35 yielded mixed results, depending on the clinical scale used and timepoints. Of note, both studies showed an initial decrease in suicidal scores that was maintained at 7 days for one study and 28 days for the other. Both used single doses of ketamine (one IV and one intranasally) in comparison to placebo or midazolam. Importantly, both studies had total sample sizes of only 24 and 30.
Five primary RCTs showed no significant effects on suicidal ideation as compared to the control arm.28,30,32,34 These include the three studies using repeated doses of intranasal esketamine versus placebo. Pooled analyses of two of these studies did not report positive effects. 36 The two other studies, one in bipolar disorder and one in ‘affective’ patients, used IV ketamine versus placebo or midazolam in only 10 and 16 participants, respectively. All demonstrated an initial decrease in suicidal ideation scores but insufficient for statistical group difference.
Results of RCTs with suicidal ideation as secondary outcome
Among the 14 RCTs for which suicidal ideation was a secondary outcome, seven studies reported a significant reduction in suicidal ideation at 24–72 h.3,38,45–47,49 One additional study 48 reported a positive effect on suicidal scores without giving details. One pooled analysis Ballard 50 of the two studies by Zarate et al.47,48 in addition to DiazGranados et al.4 confirmed a significant positive effect at day 3. All used a single dose of IV ketamine at 0.5 mg/kg (except one study for which doses ranged from 0.1 to 1.0; and one study at 1.0) in comparison to placebo in five studies, to midazolam in one study, and to control anesthesia in one study. Two studies reported a significant re-increase in suicidal ideation after a few days, while one study found a maintenance of effects at 4 weeks. Among these seven studies, two explicitly recruited suicidal patients. Three studies were conducted in patients with treatment-resistant depression, two with major depressive disorder, one with bipolar disorder, and one study in depressed patients undergoing orthopedic surgery.
One secondary RCT in treatment-resistant depression had mixed findings, 41 with non-significant group difference at 24 h post-infusion. However, a group difference was found at 2 h and 7 days in favor of ketamine. The effect was maintained at week 6. This study compared IV ketamine (0.5 mg/kg) to midazolam.
Finally, six secondary RCTs found no significant effect of ketamine4,40,42–44 {Floden et al., 2022, #143543}. Five used IV ketamine at 0.5, or 0.3 or 0.2 mg/kg (three single doses and two repeated doses) versus placebo for four studies versus IV esketamine for one study. One used intranasal esketamine at 56 or 84 mg. Four studies were in treatment-resistant depression, one in depressed patients necessitating ECT, and one in a small sample of patients with bipolar disorder.
Pooling data from five secondary RCTs, Ballard et al. 51 identified three groups: non-responders (29%), responders (44%), and remitters (27%). In responders and remitters, maximal improvements were achieved at 24 h.
Results of recent meta-analyses
Witt et al. 12 examined 15 trials/25 studies (including one with intranasal esketamine) published until December 2018 for a total of 572 patients. They reported a significant effect of ketamine versus control within 4 h post-infusion (10 studies), 12–24 h (10 studies), 24–72 h (8 studies), but not 72 h–2 weeks (11 studies), 2–4 weeks (5 studies), or more than 1 month (3 studies). For all analyses, a high heterogeneity was found.
Xiong et al. 13 reviewed publications until July 1, 2020. Nine studies testing the effect of a single dose of ketamine (including one with intranasal esketamine) were analyzed. Pooled analyses for all timepoints and formulation yielded a large effect size in favor of ketamine (Hedge’s g = 1.0). Effects of ketamine were also significant at 2, 4, and 24 h post-intake. Again, a high heterogeneity was observed.
Finally, Bahji et al. 23 compared the efficacy of racemic ketamine versus esketamine. ‘Suicidality’ was assessed in 11 trials published until December 2019. Overall, there was a significant effect of ketamine on ‘suicidality’ scores, even after accounting for publication bias [standardized mean differences = −0.50 (−0.82, −0.19)]. However, the effect was positive for racemic ketamine but not for intranasal esketamine.
Adverse events
We extracted adverse effects from the 12 primary RCTs identified here. Adverse events from six RCTs were available in articles (n = 288 patients) (Table 3). Four assessed only very short-term adverse events (minimum 2 h post-infusion and maximum 7 days post-infusion). Importantly, ketamine was mainly delivered combined with other psychotropic medication, and side effects in subgroups treated with ketamine in monotherapy were not provided. Altogether, adverse events were similar to those observed in studies designed for depression. The most frequent adverse events reported were sedation/drowsiness, nausea/vomiting, dizziness, headache, and elevated blood pressure. The three studies using repeated doses of intranasal esketamine25,28,36 reported similar adverse events as for racemic ketamine in the short term (Table 4).
Adverse events after racemic ketamine treatment for suicidal ideation in RCTs with suicidal ideation as the primary outcome.

BP, bipolar disorder; IV, intravenous, empty cell in case of non-reported effect; MDD, major depressive disorder; MDE, major depressive episode; ns, non-significant differences between groups.
In this study, mean scores (standard deviation) are given.
Adverse events after esketamine treatment for suicidal ideation in RCTs with suicidal ideation as the primary outcome.

MDD: major depressive disorder.
Discussion
In summary
Suicidal thoughts and behaviors are heterogenous, multifactorial, and complex phenotypes. Prediction of the transition from ideas to acts is weak suggesting that, until prediction is much improved, all suicidal patients should be considered at risk of acting out and adequate care should be implemented (and therefore available). Therapeutic options that are efficient to reduce the risk of suicidal acts with limited negative impact (side effects, financial cost, and stigma) for the patients and society may be more adapted to our low predictive capacities. Moreover, as most suicides occur in low- to middle-income countries, there is a special need for low-cost and easily available treatments. To date, there are limited options to manage the suicidal crisis and usual care often lacks strong evidence of efficacy. There are currently no pharmacological options available rapidly active on suicidal ideas. A multimodal approach to treatment of suicidal patients is recommended. Ketamine is a promising molecule to address some of these issues.
Our review confirmed that one single dose of IV racemic ketamine (but not intranasal esketamine) is an effective and rapid way to reduce suicidal ideation in many patients with a positive benefit-risk balance in the short term (less than 72 h) as discussed below.
Efficacy of ketamine on suicidal ideation
Indeed, we report that out of nine RCTs primarily investigating the effect of racemic ketamine on suicidal ideation, five found a significant reduction of suicidal ideation. Out of 13 RCTs using racemic ketamine and analyzing changes in suicidal ideation as secondary outcomes, 7 showed a significant reduction following ketamine. Negative findings in several studies could often be explained by low sample sizes or more complicated disorders including treatment-resistant depression and patients requiring ECT. Full suicidal remission was reached in 30–60% of cases.10,51 Most studies used a single IV dose, the mode of administration with the highest bioavailability, and effects were sustained over several days in most studies. Group differences were usually not statistically significant after 72 h, largely due to a reduction of suicidal ideas in the control group more than a secondary increase in the ketamine arm. These findings are in line with previously published meta-analyses.12,13
In contrast, the four large studies (including three as primary objectives) using intranasal esketamine were all negative, even when data were pooled. A recent meta-analysis confirmed the overall superiority of IV racemic ketamine over placebo or midazolam, and the lack of significant benefits in terms of suicidal ideation reduction of intranasal esketamine over the control group. 23
Adverse events
In the population examined in this review, ketamine was usually well-tolerated with only minor and transitory short-term effects. Few patients experienced an exacerbation of depressive symptoms or suicidal ideation during treatment.
Most previous reports about adverse events following ketamine intake in patients with psychiatric disorders come from the studies designed to treat depression and particularly treatment-resistant depression. Several reviews have been published.18,24,52–55 Side effects were more frequent with ketamine than placebo, usually of short duration (a few hours) following one single intake, and mostly minor. 52 These include headaches, dizziness, dissociation, and other psychomimetic symptoms, increased blood pressure, blurred vision, sedation or drowsiness, faintness or light-headedness, anxiety, elevated heart rate, nausea, urinary tract side effects, and cognitive side effects.
Studies assessing long-term side effects of ketamine and esketamine are lacking. Genito-urinary symptoms have been observed in recreational ketamine users with both dose and frequency effects. These adverse events ceased in most patients after cessation of the drug.56,57 Preclinical studies have reported brain lesions with repeated exposure to ketamine; however, there is no known report of cognitive disorders in patients treated with single or repeated infusion. 58 The potential for ketamine abuse in recreational users has been described. 59 However, whether single or repeated ketamine administrations in professionally controlled settings result in misuse or dependance remain to be investigated. Of note, preclinical studies suggest a lower abuse liability in (R)- versus (S)-ketamine. 59 Importantly, patients with a history of substance abuse are usually excluded from clinical trials.
In a recent systematic review of studies in suicidal patients, suicide attempts during follow-up occurred in 2.4% of the treatment group versus 1.5% of the control group. Exacerbation of depressive symptoms or suicidal ideation occurred in 2.4% and 2.2%, respectively. 18 Analysis of spontaneously reported post-marketing side effects with intranasal esketamine found a risk of potential serious events, including suicidal ideation [reported odds ratio (OR) = 24.03, 95% confidence interval (CI) = 18.72–30.84] and completed suicide (Reported OR = 5.75, 95% CI = 3.18–10.41). 60 More studies will be necessary to investigate the effect of ketamine in terms of suicidal behaviors.
Predictive clinical markers of response to ketamine
The identification of predictive markers of response to ketamine is limited by the small sample size of most studies. Globally, studies have been quite unconclusive.
In a meta-analysis, 23 subgroup analyses showed a significant effect on suicidality when treatment was adjunctive but not monotherapy (of note, most studies were not monotherapy). Moreover, there was no difference according to treatment-resistance, depression type (unipolar versus bipolar), or trial type (crossover versus parallel). 23 However, Abbar et al. 10 showed a highly significant effect of racemic ketamine on suicidal ideation at 72 h in bipolar disorder but not major depressive disorder or other affective disorders. Importantly, diagnostic groups were a priori defined in this study. Wang et al. 61 further suggested a faster reduction in suicidal ideas in depression with melancholic-anxious features.
Regarding baseline suicidal characteristics, Grunebaum et al. 31 found no interaction between response to ketamine and baseline levels of suicidal ideation. In contrast, Price et al. 46 found an increased response in patients with higher baseline suicidal ideation (but not non-suicide-related depression symptoms) and in those with past suicide attempts. Ballard et al. 51 reported that more suicidal ideation and a history of self-injury at baseline increased the likelihood for not being in remission of suicidal ideation after ketamine, while a history of sexual abuse had the opposite effect. Of note, a history of childhood maltreatment, a significant risk factor of suicidal thoughts and behaviors, was associated with an increased likelihood of response to repeated doses of ketamine in treatment-resistant depressive disorder. 62
Using Bayesian networks and data from a longitudinal open study in patients with chronic suicidal ideation, one group showed that younger age, female gender, normal body mass index, higher level of education, being single, being unemployed, previous suicide attempts, and suffering from a less severe level of depression all predicted a prolonged response to oral ketamine. 63 Finally, one study showed a good predictive capacity for the anti-suicidal response to ketamine by a combination of an early clinical response and early measures of kynurenic acid levels in serum. 64
Potential clinical mechanisms of action of ketamine on suicidal ideation
Mechanisms leading to the rapid reduction of suicidal ideas following ketamine intake remain largely unknown for several reasons: We have a limited understanding of the mechanisms of ketamine in general, a drug characterized by various pharmacological properties; a limited understanding of the processes underlying suicidal ideation; and a limited number of studies investigating the specific processes linking ketamine and suicidal ideation reduction. Therefore, only speculations can be made at this stage.
At the clinical level, the effect of ketamine on suicidal ideation may ‘simply’ be explained by its antidepressive effects. Indeed, suicidal ideation is highly associated with depression, and antidepressants other than ketamine reduce both suicidal ideation and the risk of suicide attempt and suicide in adults.65–67 Several studies have, however, suggested that the effect of ketamine on suicidal ideation may be partially independent of effects on other depressive symptoms,29,31,51,68,69 although this assumption is not supported by all studies.35,46 Moreover, it cannot be excluded that the lack of association is mainly due to error variance in different scales used.
Previous studies have suggested that ketamine in depressed patients may target specific symptoms, notably suicidal ideas, depressed mood, negative cognition, anhedonia, and lack of motivation. 70 In one qualitative study in suicidal patients, patients identified improvements in anxiety, ability to think and function as important mechanisms. 71 In a factor analysis, one recent study found that reduction in suicidal ideation following ketamine was strongly related to reduction in subjective/psychic depression, anxiety, and fatigue, but not somatic symptoms or complaints, sleep, or anger. 72 In patients with chronic suicidal ideation, oral ketamine over 6 weeks reduced the feeling of stress and increased the feeling of being able to cope with unexpected stressors. 73 In severe suicidal patients seen in the emergency room, Domany et al. reported reduced hopelessness after ketamine. 29 Murrough et al. 35 found a significantly higher reduction in panic and irritability in suicidal patients receiving ketamine versus midazolam.
Anhedonia may be another interesting target of ketamine explaining its anti-suicidal properties. One study found reduction of anhedonia partially explained the variance in the reduction of suicidal thoughts, 74 and anhedonia was found to mediate the effect of ketamine on suicidal ideation in depressed patients. 75
Mental pain is one of the core clinical factors associated with the emergence of suicidal ideations and suicidal behavior. 76 Abbar et al. 10 showed a mediating effect of mental pain on the effect of ketamine on suicidal ideation. This effect may be related to the opioid or serotonergic systems as both were previously associated with mental pain in depressed patients. 77 In contrast, a recent study pooling data from three RCTs found no association between psychological pain (or hopelessness) and suicidal ideation in the short- and long-term following ketamine administration. 78 Interestingly, the opioid agonist naltrexone reduces the anti-suicidal effect of ketamine. 79 Possibly, the effect of ketamine on peri- and subgenual parts of the anterior cingulate cortex, as a central region of both a putative mental pain network and suicidal ideation, could explain its ability to reduce the mental pain associated with suicidal ideation and acts. 77 Ketamine may modulate the activity of brain regions involved in top-down inhibition of pain centers. 81 In addition to their roles in cognitive control and reward-based learning, the anterior cingulate cortical regions are implicated in a circuit controlling the emotional aspect of pain. 82 While ketamine was found to improve cognitive control, a deficit associated with suicidal behavior83,84 and the suicidal transition, 85 these changes were not correlated with changes in suicidal ideation. 86
Clinical considerations and research direction
Findings from our review should, however, be interpreted in light of several unanswered questions. These have to be taken into account for clinical care if used in this indication (within local regulation rules). They also represent direction for future research. We refer readers to consensus statements published on the clinical use of ketamine in depression (see for instance).53,87 They notably recommend that ketamine is given by trained physicians in settings with cardiovascular monitoring and emergency material available even if the risk is low. We identified 12 major points of discussion.
A significant placebo effect on suicidal ideation reduction was found in RCTs. This was particularly the case in inpatients, with a considerable reduction in suicidal ideas over the first days of treatment explaining most of the lack of group difference after 3 days (and sometimes earlier e.g. in esketamine studies). This is also our experience. Hospitalization – and possibly empathetic social connection and support in general – has a potent impact on suicidal ideation in the short term, as regularly observed in clinical practice. If ketamine were indicated in the treatment of suicidal patients, its position in the clinical algorithm must be clearly defined to prevent inadequate or excessive use.
Blinding is not fully guaranteed in RCTs with ketamine. This is due to very recognizable side effects of ketamine, notably depersonalization. This could lead to a biased assessment of effects in studies. Use of midazolam instead of placebo is common in more recent studies to reduce this risk of bias and did not dramatically changed findings, suggesting that imperfect blinding is unlikely to explain group differences.
The definition and assessment of suicidal ideation raises important concerns. As discussed in the supplemental material, most questionnaires give a very global evaluation, lacking fine characterization, and none was built for repeated assessment of suicidal ideas, especially within very short periods of time (e.g. a few hours). Self-assessments and verbally based external assessments are also subjected to various limitations, notably in the case of suicidal ideation. Moreover, the fluctuating characteristics of suicidal ideas may necessitate more complex tools and analyses, such as ecological momentary assessment.88,89 Novel approaches have been proposed that should be tested in future studies with ketamine and other interventions. 90
All RCTs published with ketamine in suicidal ideation were conducted in affective disorders. Furthermore, most studies excluded depressive episodes with psychotic features. Thus, generalization of findings cannot yet be extended, notably to other mental disorders with a high suicidal risk, such as borderline personality disorders, schizophrenia, alcohol and substance abuse, or anorexia nervosa, among others. More research is necessary. Of note, one study 10 suggested that benefits of IV ketamine over placebo may be important in patients with a bipolar disorder (due to both a higher efficacy of ketamine and a low placebo effect) but less so in depressive disorders or in other affective disorders (due to both a lower efficacy of ketamine and a higher placebo effect). This will have to be confirmed.
RCTs with ketamine in suicidal ideation have mainly been conducted in middle-aged individuals. Future studies will have to be conducted in adolescents, the group for which suicide is a leading cause of death 91 and the suicide transition rate is the highest 92 , and in older patients, the age group with the highest rates of suicide. 93
No study specifically investigated the impact of ketamine on the prevention of suicidal acts and suicide and overall mortality. RCTs further suggest no clear advantage of ketamine over control drugs, with overall 2.2% of suicide attempts in the ketamine arm and 1.5% in the control arm in RCTs. 18 The low total number of participants, however, limits comparison of these rates, and large-scale studies and meta-analyses will be necessary to clarify this important point. Caution is, however, warranted. The rapid short-term positive effects of ketamine may lead to reduced emphasis on a multimodal and exhaustive (and therefore costly and time-consuming) treatment strategy (including psychiatric, somatic, and social assessment and care) and on good follow-up. This is especially true in a context of financial constraint on the health system and limited mental health service availability.
Long-term adverse events of ketamine remain largely unknown. This includes the risk of drug abuse. The risk of abuse has to be seriously investigated when considering the facilitated online access to ketamine. While it cannot be excluded that some patients may require repeated intake of ketamine for a sustained effect on depression or chronic suicidal ideation; for instance, it will have to be distinguished from intakes aiming at non-therapeutic benefits. 59 Preclinical and clinical studies suggest a low overall risk of abuse, and a higher risk of abuse with (R, S)- and (S)- than (R)-ketamine. 59
Most RCTs of ketamine in suicidal ideation were conducted in academic settings with most patients being hospitalized. This was, however, not the case for treatment-resistant depression with repeated doses of ketamine being administered in outpatient facilities. A few studies27,29 were conducted in the emergency room and suggest that ketamine may be given in these settings. Importantly, there is no strong evidence that hospitalization is an efficient means of suicide prevention, while one study suggested an association between outpatient service availability and lower suicide rates. 94 Studies in outpatient settings, day hospitals, or in somatic hospital departments as part of psychiatry liaison consultation are needed to test feasibility and effectiveness.
Robust markers predicting the efficacy of ketamine in suicidal ideation reduction are lacking. Preliminary findings reported in this review could not identify replicated markers of response or resistance. At this stage, it is therefore not possible to identify a subgroup of patients that should be prioritized.
Mechanisms explaining the action of ketamine on suicidal ideation are unknown. A few studies suggest that ketamine action may be mediated by an effect on mental pain, ‘psychic depression’, anxiety, and anhedonia.10,51,72 However, it is too early for definitive conclusions. There is also a need for clarification of the molecular and brain-network mechanisms in humans and for studies targeting endophenotypes of suicidal behavior in humans and animal models, 95 for instance, risky decision-making and cognitive control. 96 This may also help developing novel safer drugs.
The best therapeutic strategies when using ketamine for suicidal ideation will have to be investigated. While IV ketamine administration seems to outperform intranasal esketamine, and one single dose at 0.5 mg/kg may be sufficient in most cases in the short term, different administration designs will have to be directly compared. This will need to include variations in frequency, doses, and mode of administration. For instance, does a second consecutive dose of ketamine improve rates of remission? IV administration is currently not only cheaper than intranasal administration but also relatively difficult to implement. Another question pertaining to patients with chronic or recurrent suicidal ideation will be the benefit of repeated intakes of ketamine but also the risk of psychological and physical dependence.
The effects of concomitant medication on efficacy and safety necessitate more research. Most studies of ketamine in suicidal ideation have been conducted as an adjunctive treatment. However, how this influences efficacy and safety is still limited. A recent review of literature suggests reduced antidepressive efficacy of ketamine with benzodiazepines or lamotrigine, and possible interaction with antipsychotics but not lithium. 97
Limitations of the review
Although comprehensive, several studies may have escaped our attention, particularly those with the reduction of suicidal ideation as a secondary objective. Also, we chose to limit the search tools to PubMed, article references, and several published literature reviews. Moreover, it is always difficult to synthesize results measured in a variety of ways, on clinically heterogeneous populations, and recruited in different locations and countries. Different authors might therefore have a different conclusion from ours. Finally, we did not run a formal assessment of bias for each of the 28 selected studies.
Conclusion
Racemic ketamine (notably one single IV dose of 0.5 mg/kg) was superior to control drugs to reduce suicidal ideation within the first 72 h in several RCTs with this primary objective and in meta-analyses. Beyond this timepoint, no group difference with placebo was found in most studies, mainly due to a high placebo effect and more rarely to a re-increase in suicidal ideation. The short-term safety profile of ketamine was also very good, with minor and usually transitory side effects. In contrast, intranasal esketamine did not differ from placebo in terms of suicidal ideation reduction. Racemic ketamine, with its excellent benefit-risk ratio in the short term, therefore appears to be a valuable pharmacological intervention with a rapid effect. Many questions still need be addressed and many issues to be resolved, in terms of therapeutic strategy and long-term risks and management. Caution is warranted if ketamine should be integrated in clinical practice (within local and national regulation), and pharmacovigilance on a large scale will be essential. Finally, as ketamine is not efficient in all suicidal patients, alternative therapeutic options still have to be investigated.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253231151327 – Supplemental material for Ketamine and esketamine in suicidal thoughts and behaviors: a systematic review
Supplemental material, sj-docx-1-tpp-10.1177_20451253231151327 for Ketamine and esketamine in suicidal thoughts and behaviors: a systematic review by Fabrice Jollant, Romain Colle, Thi Mai Loan Nguyen, Emmanuelle Corruble, Alain M. Gardier, Martin Walter, Mocrane Abbar and Gerd Wagner in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
