Abstract
Background:
The paliperidone palmitate 6-month (PP6M) long-acting injectable formulation is currently the longest dosing interval available for schizophrenia treatment.
Objective:
To compare treatment outcomes between a real-world external comparator arm (ECA; NeuroBlu database) and the PP6M open-label extension (OLE) clinical trial arm.
Methods:
The ECA comprised patients receiving PP 1-month (PP1M) or PP 3-month (PP3M) for ⩾12 months without a relapse. The PP6M OLE arm included patients with PP1M treatment prior to randomization who completed the 12-month double-blind PP6M study on either PP3M or PP6M relapse-free. Inverse probability treatment weighting (IPTW) was used to study time-to-relapse (primary outcome) and change in Clinical Global Impressions-Severity (CGI-S) score (secondary outcome).
Results:
At 24 months, 3.9% (7/178) of patients in the PP6M cohort experienced a relapse versus 15.6% (26/167) in the ECA. Time-to-relapse was longer in the PP6M cohort versus the ECA at 12-, 18-, and 24-months across the different weighting methods; median time-to-relapse was not reached in both cohorts. Hazard ratio (HR) for relapse was significantly lower for the PP6M cohort versus the ECA throughout the duration of the study [HR at 24 months: 0.18 (95% CI: 0.08–0.42), p < 0.001]. At 24 months, change in CGI-S score for the PP6M cohort was 0.76 points lower than the ECA (p < 0.001). Results were similar in a sensitivity analysis using propensity score matching (PSM); IPTW resulted in larger sample sizes in balanced dataset than PSM.
Conclusion:
Consistent findings across weighting and matching methods suggest PP6M efficacy in reducing and delaying relapses and long-term symptom control compared to PP1M/PP3M in usual-care settings. Additional confounds, such as greater illness severity and more frequent comorbidities and comedications in the ECA, were not fully controlled by the applied statistical methods. Future real-world studies directly comparing PP6M with PP3M/PP1M and adjusting for these confounders are warranted.
Keywords
Introduction
Patients with schizophrenia often experience recurrent relapses that negatively impact long-term cognitive and social functioning.1–4 A higher incidence of relapse has been observed in patients who are non-adherent to antipsychotic treatment, have lower household income or are unemployed.5–10 Non-adherence or gaps in medication use limits the translation of treatment benefits from randomized controlled trials (RCTs) into real-world (RW) settings. Moreover, RW settings often include more severely ill patients, including those with somatic comorbidities, such as metabolic syndrome and cardiovascular disease, concomitant use of mood stabilizers or antidepressants or history of substance use and suicide attempts. Patients with these characteristics have a higher risk of relapse and are often excluded from RCTs. 11 Poor adherence to antipsychotic use increases the risk of relapse, hospitalization, and suicidal attempts and has serious consequences for treatment outcomes as well as healthcare resources.12,13
The atypical antipsychotic paliperidone palmitate (PP) is a long-acting injectable (LAI) used for the acute and maintenance treatment of patients with schizophrenia. 14 The efficacy, safety and tolerability of paliperidone 1-month (PP1M) and 3-month (PP3M) formulations have been established in several randomized controlled trials, in the respective long-term extension studies and in the RW settings as well as in a non-inferiority study comparing PP3M with 6-month (PP6M) LAI formulations.5–7,15–21 In a controlled non-inferiority RCT setting, PP6M was similarly effective in preventing relapses over a 12-month period compared to PP3M. 21 Evidence indicates that long-term treatment adherence in schizophrenia improves when treatment injections are less frequent.5,6,22 Additionally, even after medications are discontinued, there is a reduced risk of relapse on long-acting versus oral antipsychotic treatment. 23 PP6M has the longest available dosing interval of 6 months and is intended for use in patients with schizophrenia previously stabilized on adequate treatment with PP1M or PP3M. Thus, the use of PP6M with less frequent dosing may allow patients who have limited access to healthcare to have more consistent medication coverage. 21
The aim of this study was to compare treatment outcomes in patients with schizophrenia receiving PP6M in an open-label extension (OLE) study (NCT04072575) 24 with patients from an ECA derived from patients receiving PP1M/PP3M in a RW setting. We hypothesized that in RW settings the longer injection interval of PP6M would translate into significant advantages in preventing or delaying relapse in patients with schizophrenia receiving PP1M or PP3M as part of usual care.
Methods
Patients and study design
The ECA (PP1M/PP3M cohort) was created using RW data and compared to data from an OLE of a double-blind, active-controlled randomized-controlled study for PP6M.
PP6M cohort
Patients in the PP6M cohort were derived from the OLE phase (NCT04072575) of the double-blind phase III noninferiority study of PP6M versus PP3M. 21 The OLE study was conducted in Argentina, Hong Kong, Italy, Poland, Russian Federation and Ukraine. Patients who were adequately treated with PP1M for at least 4 months or PP3M for one 3-month cycle, and who remained relapse-free during the double-blind phase entered this OLE and continued the same PP6M dose (700 mg eq. or 1000 mg eq.) for up to 24 months (Figure 1). Patients who received a ‘moderate’ dose in the double-blind phase (i.e. PP3M = 350 mg eq. or PP6M = 700 mg eq.) received PP6M 700 mg eq. as an initial dose during this OLE. Patients who received a ‘higher’ dose (i.e. PP3M = 525 mg eq. or PP6M = 1000 mg eq.) received PP6M 1000 mg eq. as an initial dose during this OLE. The PP6M dose level was allowed to be adjusted every 6 months based on the clinical judgment of the treating physician.

Study design.
External comparator arm
The ECA was created using RW data and was drawn from patients in the NeuroBlu Database Version 21R1 (Holmusk Technologies Inc., New York, NY, USA), an RW deidentified electronic health record (EHR) database 25 from >25 behavioral health centers across the United States, representing >560,000 patients treated by >11,000 clinicians who use the MindLinc EHR system. The NeuroBlu database contains demographic and clinical information including comorbid psychiatric diagnoses, prescribed medications, outcome measures such as hospitalizations, Clinical Global Impressions-Severity (CGI-S) scores, 26 and clinical labels derived from Mental Status Examination(MSE) records. Patients in the ECA had a schizophrenia diagnosis per the International Classification of Disease, 9th Edition (ICD-9) or ICD-10, had been prescribed either PP1M and/or PP3M for at least 12 months from treatment initiation, and had a CGI-S score available at baseline. Patients were excluded if they had a record of relapse within 12 months prior to the index date, or if they were diagnosed with bipolar disorder after receiving a schizophrenia diagnosis. Patients with other mental health comorbidities, such as substance use disorder, major depressive disorder, bipolar disorder, attention-deficit/hyperactivity disorder, obsessive-compulsive disorder, generalized anxiety disorder, borderline personality disorder, post-traumatic stress disorder, were not excluded.
To ensure comparability to the OLE PP6M cohort, patients in the ECA were selected with similar demographic and disease characteristics, and treatment-related factors as patients in the OLE study, and in accordance with inclusion and exclusion criteria aligned with those of the OLE (Supplemental Table 1). Covariates considered for ECA selection included sex, age at baseline, CGI-S score at baseline, PP dosage (low, moderate, high) and previous hospitalization to help adjust for potential confounding and account for differences in baseline characteristics between cohorts to minimize selection bias.
Endpoints and assessments
The primary endpoint was time-to-relapse following at least 12-month stabilization of treatment. Relapse was defined as one or more of the following: psychiatric hospitalization or Emergency Department visits for schizophrenia; the presence of MSE natural language processing (NLP) labels associated with suicidality and violent behavior (in ECA); patient inflicting deliberate self-injury or exhibiting violent behavior resulting in suicide attempt, clinically significant injury to themselves or another person, or significant property damage or suicidal or homicidal ideation and aggressive behavior (in PP6M cohort). The secondary endpoint was change in the CGI-S clinical scale by time point. The outcomes of the PP6M cohort were also evaluated for the ECA.
Statistical analysis
Cohort balancing
To achieve cohort balancing, the PP6M and ECA cohorts were combined into a single dataset of baseline characteristics and assigned a variable to account for treatment group origin. A propensity score (PS), which provided the predicted probability of being in the OLE cohort relative to the ECA as a function of these covariates, was calculated for each patient using multivariable logistic regression. To eliminate the association between confounders and treatment, PS scores were used to reweight patients in the PP6M and ECA cohorts. This inverse probability treatment weighting (IPTW) was defined as the inverse of the estimated PS for the PP6M cohort, and the inverse of one minus PS (1-PS) for the ECA. These weighted scores yielded an estimated average treatment effect. 27 To improve the efficiency of the binary IPTW estimation, a trimming factor of α = 0.1 was used in the Psweights package in R for the combined weighted dataset.28,29 Covariate balance was evaluated before and after weighting using standardized mean differences, where <10% was considered negligible.
Analysis of treatment effects
Descriptive statistics were reported for baseline characteristics. Continuous variables were summarized as means ± standard deviations (SD) or medians with interquartile ranges, as appropriate. Categorical and ordinal variables were described using frequencies and percentages. IPT-weighted Kaplan–Meier survival analysis was conducted to estimate the primary endpoint, time-to-relapse, for both cohorts. The median time-to-relapse, 95% confidence interval and p-value of a log-rank test are reported. Group differences were also examined using Cox proportional hazard model, and the corresponding hazard ratio (HR) and its 95% CI are reported. These analyses were repeated using stabilized weighting 27 to improve the precision of estimated effects. The secondary endpoint, change in CGI-S score from baseline to follow-up, was assessed using IPT-weighted mixed linear modeling (MLM) accounting for both fixed (e.g. gender, treatment modality, treatment dose, etc.) and random effects of the independent variables. Sensitivity analysis was performed using PS matching to create 1-to-1 comparable groups.
Results
Patient disposition
The study included 178 patients in the PP6M cohort. Of the 174 patients from the ECA, five patients were on PP3M, and 162 patients on PP1M. The combined dataset comprised 345 patients [PP6M: 178; ECA: 167 (95.9%)]; seven patients from the EHR dataset were excluded because they did not receive a PP1M or PP3M injection post-baseline (Figure 2).
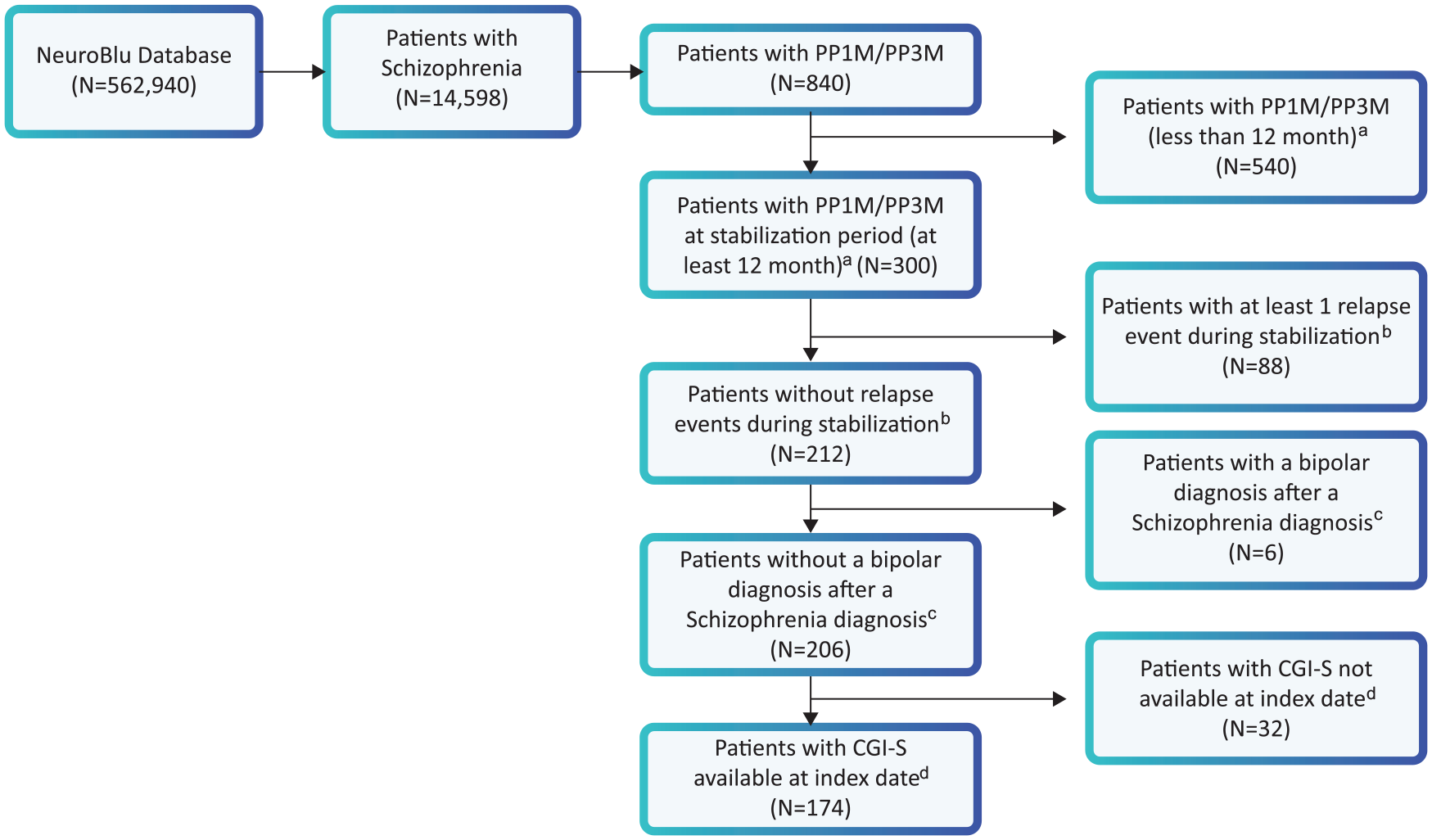
Attrition diagram for external comparator arm (PP1M/PP3M cohort).
Demographic and clinical baseline characteristics
The mean age at baseline was 40.4 years for the PP6M cohort and 42.7 years for the ECA, and most patients in both cohorts were male (74.2% and 70.8%). The PP6M cohort was predominantly White (98.9%). Of the patients in the ECA, 69.5% were White and 20.3% were Black (Table 1).
Demographics and baseline characteristics of PP6M and ECA cohorts.

CGI-S, clinical global impression-severity; PP1M, paliperidone palmitate 1-month; PP3M, paliperidone palmitate 3-month; PP6M, paliperidone palmitate 6-month; SD, standard deviation.
There were significant differences (p < 0.05) in baseline CGI-S scores and positive and negative symptoms between the two cohorts (Table 1). In the PP6M cohort, 18.5% of patients were moderately ill (baseline CGI-S score 4) and none had a CGI-S score that indicated severe/extreme severity of the disease. In the ECA, 75.5% had moderate-to-extreme severity of disease (baseline CGI-S score 4–7). The mean (SD) time from diagnosis of disease was 4509.2 (3239.3) days in PP6M and 790.6 (753.6) days in ECA. In the ECA, 86.6% of patients had up to 2 psychiatric comorbidities and 58.1% of patients had at least 1 psychiatric comorbidity. The most commonly occurring psychiatric comorbidities in the ECA were substance abuse disorder (33.9%) and major depressive disorder (8%). The PP6M cohort had no comorbidities recorded since patients with a non-schizophrenia primary active diagnosis were excluded in the preceding randomized controlled trial.
There were significant differences between the treatment-related baseline variables of the cohorts. These included average time from diagnosis to index date (PP6M: 12.4 years versus ECA: 2.2 years), previous hospitalization (56 days versus 10.4 days), and proportion of patients on high treatment dosage strength (53.9% versus 44.9%). Treatment duration after baseline was similar in both cohorts at 1.9 years. Significantly more patients in the ECA were on concomitant medications at baseline than in the PP6M cohort (Table 1).
Time-to-relapse
Proportion of patients experiencing relapses was less in the PP6M cohort as compared with the ECA (Table 2). Psychiatric hospitalization was the most common reason for relapse (Supplemental Table 2). The time-to-relapse was longer for the PP6M compared to the ECA across the different weighting methods [Figure 3(a)–(c)]. The median time-to-relapse was not reached in either treatment groups.
Proportion of patients with relapse.

CI, confidence interval; ECA, external comparator arm; IPTW, inverse probability treatment weighting; PP1M, paliperidone palmitate 1-month; PP3M, paliperidone palmitate 3-month; PP6M, paliperidone palmitate 6-month.
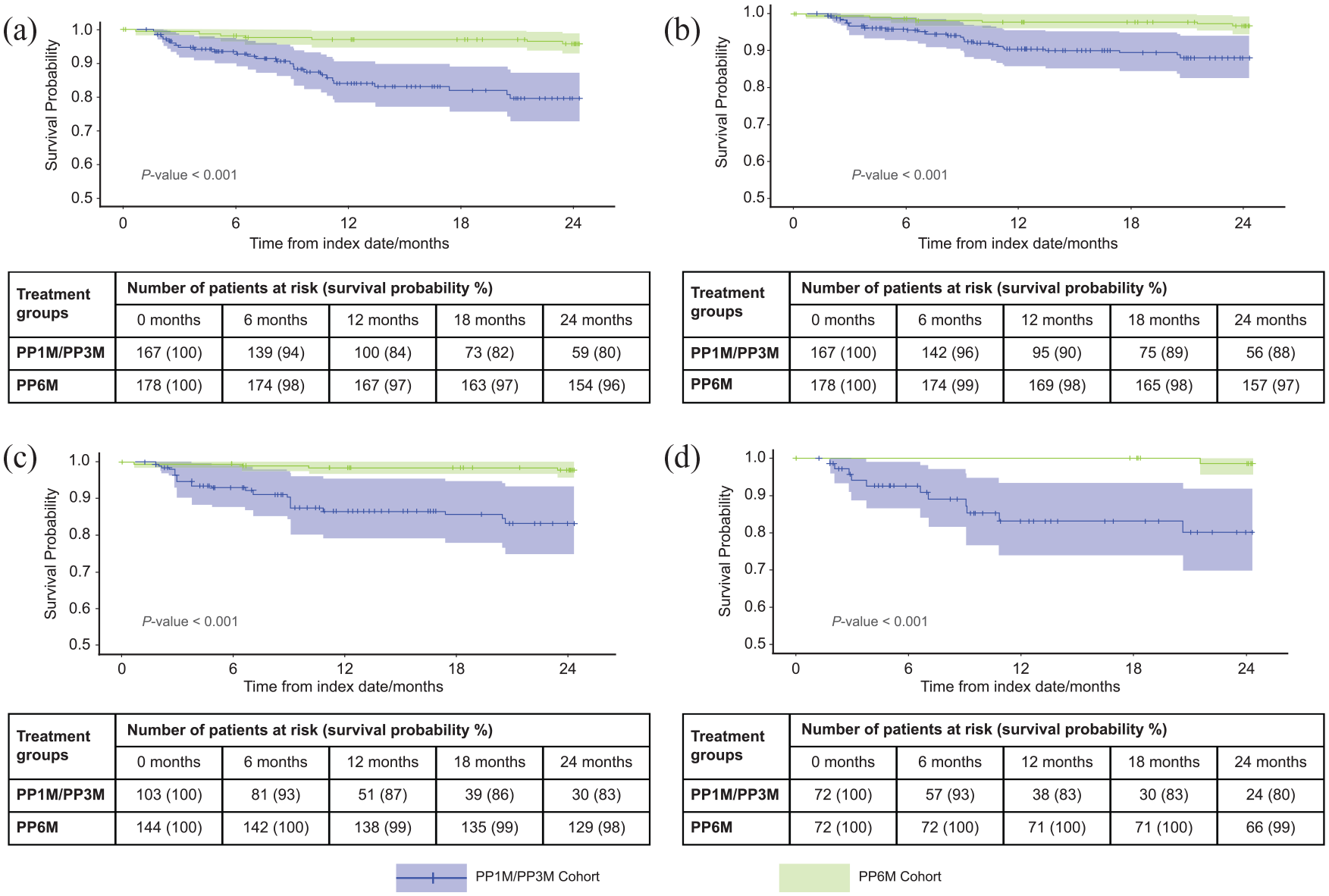
Time-to-relapse (Kaplan–Meier curves): (a) unweighted, (b) IPTW, (c) IPTW with optimal trimming, and (d) PS-matched.
Risk of relapse
Patients in the PP6M cohort were at significantly lower risk of relapse as compared with patients in the ECA at all time points assessed. The difference in risk of relapse was evident from month 12 and was maintained through 24 months of the follow-up, consistently across the weighting techniques (Table 3). In the unweighted dataset, at 24 months the risk of relapse for the PP6M cohort was 82% less than for the ECA (HR: 0.18, 95% CI: 0.08–0.42, p < 0.001). Similarly, in the IPT-weighted and IPT-weighted with optimal trimming datasets, the risk of relapse in the respective PP6M cohorts was 75% (HR: 0.25, 95% CI: 0.10–0.60, p = 0.003) and 88% (HR: 0.12, 95% CI: 0.04–0.36, p < 0.001) less than the ECA at 24 months.
Relapse-free survival using different balancing methods.

CI, confidence interval; IPTW, inverse probability treatment weighting.
Sensitivity analysis: Propensity score matching
As noted above, the two study cohorts – the EHR derived ECA and the PP6M cohort – were inherently different in terms of baseline characteristics. The PS-matched cohort was relatively small compared with the combined cohort, driven mainly by the OLE study inclusion criteria of patients that had no other psychiatric comorbidities. The distribution of PS before and after matching was well-balanced (Supplemental Figure 1), as observed by the improvements in the standardized mean differences (SMDs) post-matching for most variables (Table 4).
Summary of balance statistics.
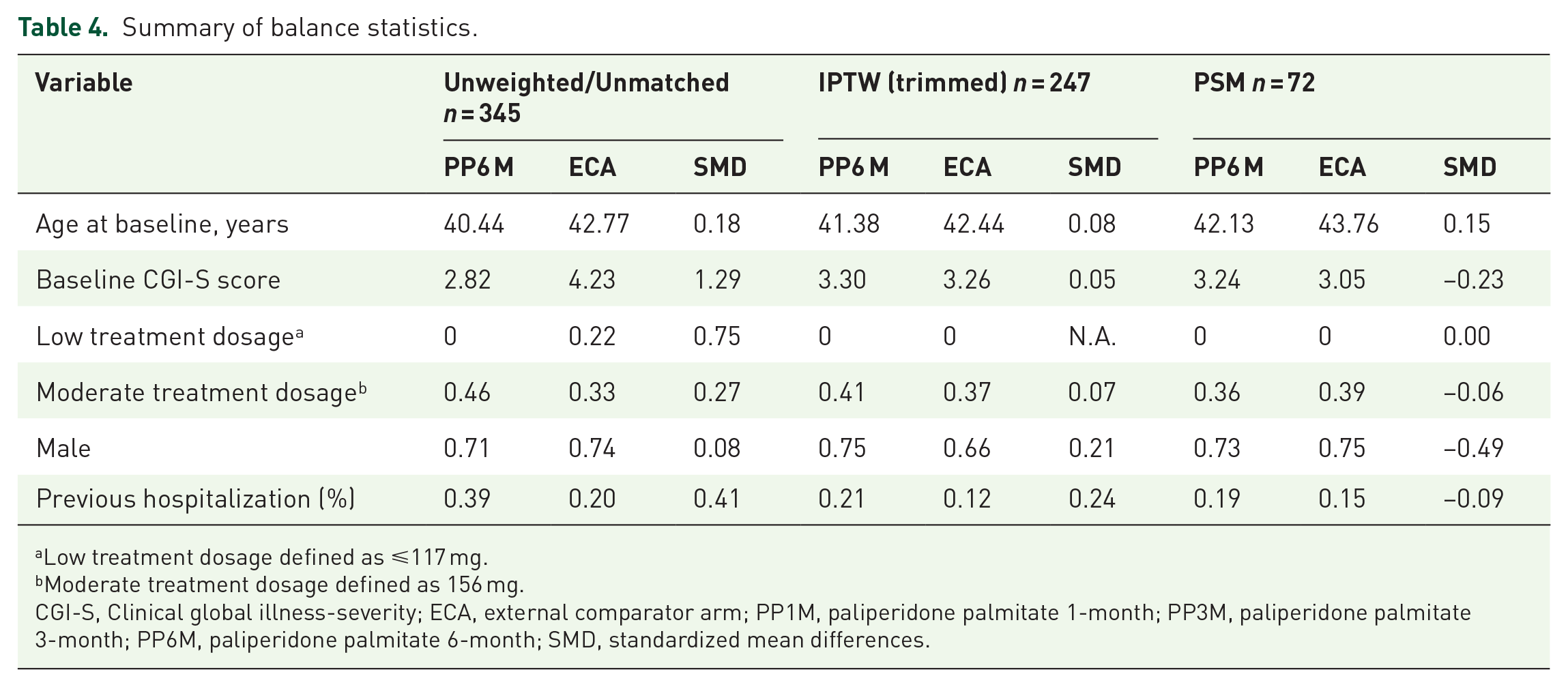
Low treatment dosage defined as ⩽117 mg.
Moderate treatment dosage defined as 156 mg.
CGI-S, Clinical global illness-severity; ECA, external comparator arm; PP1M, paliperidone palmitate 1-month; PP3M, paliperidone palmitate 3-month; PP6M, paliperidone palmitate 6-month; SMD, standardized mean differences.
Consistent with the findings from other weighting techniques, median time-to-relapse was longer for the PP6M cohort when compared to the ECA (log-rank p < 0.001, Figure 2) and fewer patients in the PP6M cohort experienced a relapse (% with relapses: 1.4% versus 15.2%). The hazard of relapse for the PP6M cohort was 94% less than the ECA (at 24 months, HR: 0.06, 95% CI: 0.01–0.48, p < 0.001; and incomputable at 12 and 18 months since there were no relapses).
Change in CGI-S
In the combined cohort (PP6M and ECA), 72.5% patients (79.8% PP6M and 47.2% PP1M or PP3M) had no change in CGI-S score at 24 months. The results from the MLM model showed that at 24 months, the CGI-S scores of PP6M cohort was on average 0.76 lower than the ECA [95% CI: (−1.01 to −0.52), p < 0.001]. The decrease in the mean CGI-S score was numerically larger in the ECA compared to PP6M cohort [at 24 months: −0.17 (1.34) versus −0.09 (0.44), p = 0.82; Supplemental Table 3]. An intra-class correlation of 0.99 suggested that this multi-level model was appropriate (p < .001). Baseline CGI-S as random effect had a significant effect (p < 0.001), with 99.2% variance explained by the model when CGI-S was included as a random effect.
Discussion
In this study, for the first time, data from patients receiving PP1M or PP3M for the treatment of schizophrenia in routine clinical practice were used as an ECA to evaluate the comparative effectiveness of PP6M in an OLE study. The ECA was derived from the NeuroBlu database, 25 which provided a patient pool closely mimicking that of the PP6M OLE study. Our findings indicated that patients in the PP6M cohort had significantly longer time-to-relapse and had lower risk of relapse as compared with the ECA (PP1M/PP3M cohort). Different weighing techniques like IPTW, IPTW with trimming, using stabilized weights and sensitivity analysis using PSM consistently demonstrated that PP6M was beneficial.
RW data can be leveraged to develop external control groups for single-arm studies. 30 Data from ECA studies complementing clinical trial data have been used in oncology and rare pediatric indications to support recent regulatory approvals.31–33 The inclusion criteria for the ECA and the OLE PP6M study were aligned, and confounding covariates were carefully selected to help adjust potential differences between the cohorts. Different statistical methods were applied, and further sensitivity analyses performed to ensure robustness of the findings.
There were inherent differences between the two cohorts in terms of demographics (race), disease duration/chronicity, disease severity (baseline CGI-S score), psychiatric comorbidities, comedications and PP treatment dosage. Patients from the ECA cohort had an observed disease evolution of about 2 years in the database, compared to a recorded period of >12 years for the PP6M OLE cohort; this difference may have impacted relapse rates. The PP6M cohort included patients outside the United States, had more patients with negative symptoms characteristics of longer episodes of schizophrenia and had no other psychiatric comorbidities or comedications. Presence of negative symptoms in patients with schizophrenia has been associated with poor functional outcome and long-term morbidity,34–36 as well as relapse risk. 37 In contrast, the ECA cohort consisted of patients only from the US, had a recent diagnosis in the database, severe illness, high comorbidity burden, and hence, may have had a higher risk of relapse. This finding of more severe illness characteristics in RW than RCT samples is consistent with data from Finland and Sweden. 11
IPTW was used for assessing the primary and secondary endpoints, and PSM was used for sensitivity analysis to assess the primary endpoint. While both methods resulted in an improved balance between the two cohorts, IPTW resulted in a larger sample size in the balanced cohort as compared to PSM (n = 167 versus n = 72). The cohort balancing strategies resulted in assigning large weights in the case of IPTW (due to a lack of common support) and a smaller number of matched pairs in the case of PSM (n = 72; less than half the ECA cohort had no other psychiatric comorbidities). The HR for relapse in patients in the PP6M cohort compared to the ECA cohort was 0.18 (unweighted data), representing an 82% reduction in the risk of relapse in the PP6M cohort. Across the weighting techniques, the point estimates generated by IPTW (0.25; 75% reduction), IPTW with trimming (0.12; 88% reduction), as well as using stabilized weights (0.25; 75% reduction), and the sensitivity analysis using PSM (0.06; 94% reduction) were similar and showed treatment benefits with PP6M. PP1M and PP3M LAIs are given earlier in the treatment trajectory, more likely treating the acute phase of schizophrenia when illness severity is high and predomiantly presents as positive symptoms. This is corroborated by the shorter disease duration and higher baseline CGI-S score in the ECA cohort than the PP6M cohort. Patients in the PP6M cohort had disease for >12 years while patients in the ECA cohort had recent diagnosis (~2 years). For the PP6M cohort, patients had a mean CGI-S score of 3.1 at baseline of the double-blind phase III study, received PP6M or PP3M through the study and had stable disease at the start of OLE PP6M administration.
During this study, the majority of the patients in the PP6M cohort demonstrated disease stability as assessed by no change from baseline in CGI-S score at 24 months. Altogether, improvement in CGI-S mean score in both cohorts was nominal and the difference was not significant. This finding is similar to the PP6M phase III study, which found on average no change in CGI-S score at follow-up (12 months) for patients in the PP1M/PP3M and PP6M cohorts. 21 This result contrasts with findings from other observational/open-label studies where significant improvements were observed in CGI-S score at follow-up (24 months) in patients on PP1M and PP3M therapy.3,38
Limitations
Results from this study need to be interpreted within the context of several limitations, especially considering that this analysis compared clinical trial data to a RW ECA. First, unlike RCTs, adjustment for confounding is restricted to the known confounders from the PP6M OLE data and the EHR database. It becomes difficult to control residual confounding from unmeasured or unknown characteristics/variables, such as treatment persistence, adherence, continued access to healthcare resources, socio-economic status, social/family support systems etc. 39 The potential effect of these additional factors on outcomes was not analyzed in this study. Therefore, residual confounding related to the less real-world nature of the PP6M clinical trial OLE data and overrepresentation of clinical features increasing the risk for relapse on the RW ECA dataset population cannot be excluded. Conversely, patient’s use of the NeuroBlu Database clinics may have reduced access to LAI formulations, with either sicker or less severely ill patients being initiated on PP1M or PP3M, possibly leading to a biased estimate of the relationship between treatments and incidence of relapses. Second, since lack of stability and comorbid substance use have been associated with symptom exacerbation during LAI treatment,40–42 these differences could have driven results in favor of PP6M. However, several different statistical approaches were used to mitigate against this effect that each yielded consistent results. Third, the inherent incompleteness of RW data derived from EHR posed challenges in matching the two cohorts. All inclusion/exclusion criteria from the open-label PP6M study could not be applied to the EHR-derived patients, notably the strict limitations on comorbidities and co-medications used in the double-blind study that preceded the OLE could not be consistently applied to EHR-derived patients. The criteria that both men and women should use active contraception during the study was not considered as the corresponding data were not recorded in the EHR. Fourth, for patients derived from EHR database who met the inclusion/exclusion criteria, a minimum follow-up duration criterion was not applied as that would have limited the number of patients eligible for the ECA having the primary outcome of interest, that is, time-to-relapse. Fifth, EHRs reflect RW patterns of care, and thus the data quality varies. Though efforts were made to collect patient information comprehensively, some relevant information recorded in the PP6M study was not collected in the EHR, such as medical history and comorbidities due to differences in clinician recording patterns across patient visits. However, this lack of documentation does not necessarily imply an absence of disease or comorbidities. Sixth, time-to-relapse was the primary endpoint of the study, but relapse and hospitalizations occurring in out-of-network hospitals or clinics were not captured in the EHR data used for this study. Seventh, the generalizability of findings from this study to other patient subgroups and therapeutic modalities are not known and will warrant further analysis. Our analysis assumed that patients in the PP1M/PP6M ECA were 100% adherent to medications prescribed in the EHR, which may not have been the case. Eighth, information on patients who constitute the ECA but were lost to follow-up during the study period were not available and data imputation was not applied. Ninth, we restricted our analysis only to the time-to-relapse and change in CGI-S as the primary and secondary outcomes, respectively. Other efficacy and safety endpoints, such as the Personal and Social Performance scale, treatment-emergent adverse events, extrapyramidal symptoms, suicidal ideation and behavior, and impact on medical resource utilization, could not be assessed due to lack of comparable EHR data. Tenth, RW ECA database included those patients who were treated in US, whereas the PP6M study was based on ex-US patients. Finally, the NLP-derived MSE labels used in this study warrant external validation. The MSE NLP labels were derived from clinician notes while the Positive and Negative Syndrome Scale (PANSS) is a systematic measure of the nature and severity of symptoms obtained through a structured questionnaire. This approach limits a direct comparison between the MSE NLP labels and PANSS and conducting the inferential analyses between the study arms. These limitations highlight the challenges of comparing clinical trial data with RW ECA and the need for direct comparison studies under real-world settings.
Conclusions
This ECA study, deriving comparator arm data from a RW setting, further confirmed the efficacy of PP6M in reducing and delaying relapses in patients with schizophrenia. The consistency of findings across the weighting and matching methods further suggests the effectiveness of PP6M in relapse prevention and symptom control. The increase in time-to-relapse for PP6M is potentially linked with improved adherence by providing continuation of therapy for a longer duration than shorter frequency LAIs such as PP1M or PP3M. The 6-monthly frequency (two injections per year) of the PP6M formulation may facilitate better adherence and less delay in receiving the injections, which will decrease the risk of relapse even as compared to monthly or every-3-months injections, as studies have shown that receiving <11 PP1M injections per year increases relapse rates.43,44 Additional confounds, such as greater illness severity and more frequent comorbidities and comedications may not have been fully controlled by the applied statistical methods. Hence, future RW studies should directly compare PP6M with PP3M and PP1M once sufficient PP6M RW data are available.
Supplemental Material
sj-pdf-1-tpp-10.1177_20451253231200258 – Supplemental material for Comparative effectiveness study of paliperidone palmitate 6-month with a real-world external comparator arm of paliperidone palmitate 1-month or 3-month in patients with schizophrenia
Supplemental material, sj-pdf-1-tpp-10.1177_20451253231200258 for Comparative effectiveness study of paliperidone palmitate 6-month with a real-world external comparator arm of paliperidone palmitate 1-month or 3-month in patients with schizophrenia by Ibrahim Turkoz, Joshua Wong, Benjamin Chee, Uzma Siddiqui, R. Karl Knight, Ute Richarz and Christoph U. Correll in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
Our esteemed colleague Dr. Dean Najarian passed away during development of this manuscript. He was a pivotal contributor to this work and will be greatly missed. We acknowledge Uma Kundu, MPharm, CMPPTM (SIRO Clinpharm Pvt. Ltd., India), for medical writing assistance, and Ellen Baum, Ph.D. (Janssen Global Services, LLC) for additional editorial support.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
