Abstract
Background:
Long-acting injectable (LAI) antipsychotics have been shown to improve adherence and clinical outcomes in schizophrenia treatment. However, issues with compliance and early discontinuation of LAIs remain a significant challenge in real-world settings. Understanding the factors influencing successful initiation and maintenance is essential to maximize their clinical benefits.
Objectives:
This study aimed to investigate factors associated with the maintenance of paliperidone LAI (PLAI) during the first year of treatment in a real-world clinical setting, focusing on initiation practices and baseline clinical factors.
Design:
This was a non-interventional, retrospective observational study conducted at three hospitals in South Korea. Data were collected from electronic medical records from January 2010 to January 2023.
Methods:
This study included 664 patients who initiated PLAI treatment. Kaplan-Meier survival analysis and multivariate Cox proportional hazards models were used to evaluate clinical and demographic factors influencing the 1-year discontinuation rate.
Results:
The 1-year discontinuation rate was 51.5% (342/664), with most discontinuations occurring in the early phase of treatment. Factors significantly associated with a lower risk of discontinuation included initiating PLAI with the standard starting dose of 150 mg (hazards ratio (HR) 0.766, p = 0.021), concurrent use of antipsychotics at baseline (HR 0.630, p = 0.019), a higher dose of concurrent antipsychotics (HR 0.985, p = 0.005), and outpatient initiation (HR 0.671, p < 0.001). Baseline clozapine use was associated with a lower risk of treatment discontinuation (HR 0.755, p = 0.096). A predictive model incorporating these factors demonstrated moderate ability to predict 1-year discontinuation (area under the curve (AUC) = 0.61)
Conclusion:
The findings highlight the importance of adhering to the standard dosing regimen for PLAI initiation and its potential as an augmentation agent in combination with other antipsychotics. Initiating PLAI in an outpatient setting and addressing adherence challenges early in treatment may enhance long-term treatment continuity in patients with schizophrenia.
Introduction
Schizophrenia exhibits a chronic deteriorating course, with frequent relapses.1,2 Relapse of schizophrenia, often resulting in re-admission, exacerbates the disease course and increases the disease burden.3,4 The long-term maintenance treatment of antipsychotic medications is the most crucial factor for preventing relapse in schizophrenia.5,6 Thus, many pharmacological studies on schizophrenia focus on relapse prevention, with non-adherence identified as the primary cause of relapse. 7
The introduction of long-acting injectable (LAI) formulations has provided the opportunity to improve adherence and thus long-term prognosis of schizophrenia. 8 LAI antipsychotics offer several advantages, such as ensuring consistent plasma levels, reducing the frequency of dosing, and enhancing patient compliance.9–12 Despite the expectations for superiority over oral antipsychotics, previous large randomized controlled trials (RCTs) failed to prove the benefit of LAI antipsychotics in preventing relapse of schizophrenia. 13 However, results from naturalistic mirror-image studies and real-world analysis found that LAI antipsychotics (compared to oral antipsychotics) better prevented readmission.14,15 Moreover, recent evidence suggests that early initiation of LAI treatment in first onset or early schizophrenia may be associated with good adherence, fewer relapses, and symptom improvements.10,16 Another important featured advantage of LAI is patient preference. When used as maintenance treatment in remitted schizophrenia patients, LAI antipsychotics have been reported to provide a better subjective treatment experience compared to oral antipsychotics. 17
Discontinuation of antipsychotics can occur due to various clinical factors, including adverse events and inefficacy.18,19 Therefore, the adequate management of adverse events, including subjective aspects, and the enhancement of treatment efficacy through the proper selection of antipsychotics are essential for reducing the risk of treatment discontinuation. Clinical treatment protocols have been modified by reference to accumulated post-marketing clinical experience, which can differ from the results of controlled trials for approval. The appropriate positioning of certain antipsychotics for each patient and the combined application of different antipsychotics depending on the patient’s individual clinical and demographic factors have been refined through clinical experiences. Despite the benefits of LAI antipsychotics compensating adherence problems, the compliance issue with LAI itself cannot be avoided either. LAI’s proper clinical utilization strategy is required to achieve the advantage of LAI use effectively.
Paliperidone palmitate is an LAI formulation of the second-generation antipsychotic (SGA) paliperidone. Paliperidone LAI (PLAI) is one of the representative LAIs being introduced since 2009. 20 Currently, formulations up to 6 months of duration are available: paliperidone palmitate once-monthly (PP1M), three-monthly (PP3M), and six-monthly (PP6M). The initiation schedule of paliperidone LAI typically involves a starting dose of 150 mg with PP1M (day 1), followed by 100 mg a week later (day 8), and then a maintenance dose every 4-week interval. An analysis conducted by Joo et al. using the South Korea Health Insurance Review Agency database found that among LAI haloperidol, paliperidone, and LAI risperidone, the 6-month continuation rate for LAI paliperidone was the highest at 57.5%. 21 Few observation studies have identified the risk factors of PLAI discontinuation. However, most of these studies have a small sample size and report results for either PP1M or PP3M. Studies including more detailed clinical data in the real-world clinical setting regarding the initiation and maintenance of PLAI are needed.
This study aims to investigate the clinical factors associated with 1-year maintenance of PLAI in patients with schizophrenia using data from three representative hospitals in South Korea. The findings may contribute to maximizing the clinical benefits of LAIs and developing optimal clinical applications. Detailed examinations of real-world clinical practices will provide insights into clinically optimized applications of LAIs, ultimately reducing the risk of PLAI discontinuation.
Methods
Study design
This was a non-interventional and observational retrospective study. We reviewed longitudinal data regarding PLAI prescription from three centers, including Seoul National University Hospital (SNUH), Seoul Asan Medical Center (AMC), and Nowon Eulji Medical Center (EMC). The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) guidelines for observational studies. 22
Study population
Patients with schizophrenia with a history of PLAI prescription were selected and examined. All patients with PLAI prescription history were selected, including either PP1M or PP3M. Subjects having the diagnosis of schizophrenia (International Classification of Diseases, 10th edition code F20) were included, and the patients aged under 18 years or having severe neurologic deficits or organic mental disorders were excluded.
Data collection
Clinical factors related to the continuation/discontinuation of the PLAI were retrospectively examined from electric medical records. Data were collected from January 2010 to January 2023. The prescription data of PLAI, along with other psychotropic agents, including other antipsychotics (APs), mood stabilizers (MSs), antidepressants (ADs), benzodiazepines (BZDs), anticholinergics (ACHs), and beta-blockers (BBs), were retrieved. In addition, information regarding the demographic characteristics and the care setting at PLAI initiation (inpatient or outpatient) was also collected. Within the data collection period, the initiation time points of PLAI varied among patients, resulting in inconsistent observation periods for each individual. Thus, subjects with over 1-year observational period were included for further analyses. The data of SNUH were retrieved from the clinical data warehouse (CDW) of SNUH, SUPREME (Seoul National University Hospital Patients Research Environment), and the data of AMC and EMC were derived from each hospital’s electronic medical record database.
Data processing and definition of variables
Based on the collected data, we organized each subject’s PLAI prescription schedules (doses and intervals) chronologically and summarized the usage patterns of concomitant medications. We included in our analysis the use and dosage of oral paliperidone or risperidone before switching to PLAI, baseline concurrent medications (APs, MSs, ADs, BZDs, ACHs, and BBs), and the starting dose at the initiation of PLAI. Specifically, for antipsychotics, we included the number of different antipsychotics and the total equivalent dose, calculated as olanzapine equivalent doses, 23 as variables. We also included baseline clozapine use. Additionally, we considered the clinical setting during PLAI initiation (inpatient versus outpatient) as a variable.
In our study, the primary outcome was the duration from the initiation of PLAI to its discontinuation. We considered a patient to have newly started PLAI if there were no previous PLAI prescription records and the interval between the first two injections was within 14 days. Switching between PP1M and PP3M was considered as a continuation, and a patient was considered as continuing treatment until the treatment was stopped or interrupted for a period longer than a predefined injection interval: 60 days for PP1M and 120 days for PP3M (i.e., until the 1-month additional period from expected injection date). Therefore, in this study, PLAI discontinuation was defined as follows: (i) if the period without a PLAI prescription exceeded the predefined interval (60 days for PP1M and 120 days for PP3M) or (ii) the end of the observation period. This analysis did not consider the re-introduction of PLAI after discontinuation, and only the first period of PLAI use was included for further studies.
Statistical analysis
The Statistical Analysis System SAS (SAS for XP PRO, Release 9.4 TS2 M3; SAS Institute Inc., Cary, NC, USA) and R software version 4.2.2 (The R Foundation for Statistical Computing, Vienna, Austria) were used for statistical analyses. For all statistical tests, p < 0.05 was considered significant.
Categorical variables were presented as numbers (%), and continuous variables were presented as mean ± standard deviation (SD). The independent samples t-test was used to compare continuous variables, and the chi-square test or Fisher’s exact test was applied to compare categorical variables.
Kaplan-Meier survival analyses compared patients’ 1-year continuation/discontinuation rate from PLAI initiation. The studies were performed using several significant clinical conditions. A log-rank test was used to assess the statistical significance between survival curves. The Cox proportional hazards model was conducted to identify the impact of relevant variables on the discontinuation of PLAI within 1 year. PLAI initiation setting, previous history of LAI use, whether or not the standard initiating doses were used, oral paliperidone/risperidone dose and whether or not they were reduced, baseline co-medications including APs, MSs, ADs, BZDs, ACHs, and BBs, were included as variables. Sex and age were adjusted for as the covariates in every step. Model evaluation was conducted using the area under the curve (AUC) metric, and stepwise regression was used to select variables.
Results
Study subject selection
A total of 1329 patients were identified to have prescription records of PLAI (SNUH, n = 830; AMC, n = 324; EMC, n = 175). Among them, patients whose first PLAI was prescribed at least a year before the data collection date were selected to ensure a minimum follow-up period of 1 year. Subjects were considered to have newly initiated PLAI if the first PLAI was PP1M, followed by the second PP1M within 14 days. Age < 18 years were excluded. Key inclusion criteria used for the screening and subsequent number of subjects are shown in Figure 1. As a result, a total of 664 subjects were included in the analysis.
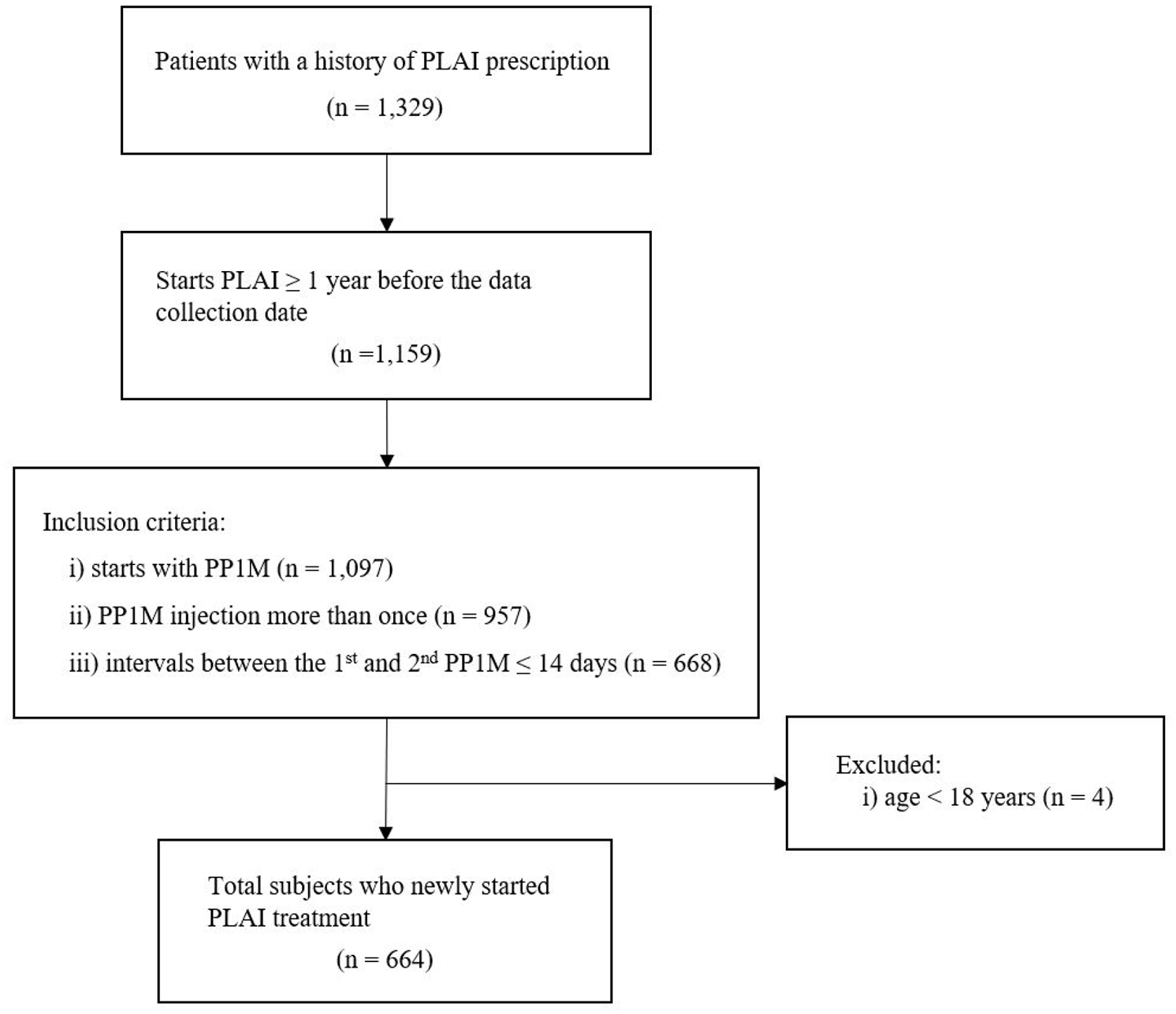
Subject screening and selection.
Information on paliperidone LAI use
Among the total subjects (n = 664), 44.7% (297/664) were male, and the mean age (±SD) was 37.14 ± 12.82 years. Firstly, the utilization pattern of PLAI without considering censoring (i.e., end of data collection) was analyzed. The average duration of PLAI maintenance was 687.80 ± 850.76 days (range, (65–4253)). The most common starting dose was 150 mg for the first PLAI (67.9%, 451/664), and 100 mg for the second PLAI (72.4%, 481/664). In total, 54.5% (362/664) of subjects followed the dosing schedule of 150–100 mg (first–second injection dose). Detailed information on the starting dose is shown in Supplemental Table 1. The average dose of PP1M, excluding the first and second doses, was 102.17 ± 32.49 mg. In most cases, the third injection dose of the subjects was equivalent or higher compared to their oral paliperidone/risperidone dose (80.2%, 467/582). Almost one-fifth of the subjects had a history of switching to PP3M (120/664) before the discontinuation, although around one-tenth of them (14/120) switched back to PP1M.
Figure 2(a) shows the time-to-discontinuation curve of PLAI within the total study period. Censoring was not considered. The discontinuation rate was higher in the early phase, with 12.3% (82/664) discontinuing right after the second injection.

Time to treatment discontinuation curves. (a) Total period and (b) 1-year period.
One-year discontinuation of PLAI
Figure 2(b) illustrates the time to discontinuation curve within 1 year from the initiation of PLAI. The 1-year discontinuation rate was 51.5% (342/664). Among them, 41.8% (143/342) were found to reuse PLAI at least once after the initial discontinuation.
We compared clinical variables between subjects who successfully maintained PLAI treatment over 1 year (Maintenance group) and those who stopped PLAI treatment within 1 year (Discontinuation group) from initiation (Table 1). Compared to the Discontinuation group, the Maintenance group showed a significantly higher proportion of starting the first dose than the standard dose (150 mg) (73.0% versus 63.2%, χ2 = 7.346, p = 0.008). However, there was no statistically significant difference in the proportion of subjects adhering to the standard dose for the first and second doses. The proportion of subjects switching from baseline oral paliperidone/risperidone to PLAI was higher in the Maintenance group (81.1% versus 71.6%, χ2 = 8.113, p = 0.005), with no significant differences observed in the equivalent ratio of baseline oral dose to the 1st injection dose. Among the total subjects, 91.6% (608/664) were concurrently taking at least one other antipsychotic in addition to oral paliperidone/risperidone at baseline. The Maintenance group had a higher number of concurrent antipsychotic medications (χ2 = 10.723, p = 0.013), and the total equivalent dose of concurrent antipsychotics was also higher in this group (23.32 ± 16.26 versus 20.05 ± 12.49, t = −2.780, p = 0.006).
Comparison between 1-year maintenance group versus discontinuation group.

Dose ratios and antipsychotic doses were calculated by the reference to the daily olanzapine-equivalent dose.
p < 0.05.
Oral PPD/RSP, oral paliperidone, and risperidone.
Risk factors for 1-year discontinuation of PLAI treatment
A Kaplan-Meier survival analysis was performed to compare the 1-year discontinuation of PLAI under various conditions. The log-rank test identified several significant predictors of PLAI discontinuation (Figure 3). Patients who initiated treatment with the standard starting dose of 150 mg had a significantly longer time to discontinuation compared to those who received other doses (p = 0.012). Outpatient initiation of treatment was associated with a longer duration of paliperidone use compared to inpatient initiation (p = 0.032). Prior use of oral paliperidone/risperidone at baseline was also significantly associated with lower discontinuation (p = 0.014). Patients with one or more co-prescribed antipsychotics at baseline had a significantly lower likelihood of discontinuation compared to those without co-prescribed antipsychotics (p = 0.001), and those receiving higher dosages of co-prescribed antipsychotics (⩾19.98 mg in olanzapine equivalents) showed better persistence (p = 0.003). In addition, co-prescription of clozapine at baseline was associated with a longer time to paliperidone discontinuation (p = 0.036).

Survival analysis for 1-year PLAI discontinuation based on clinical factors. A series of Kaplan-Meier survival curves showing 1-year discontinuation rates of PLAI treatment stratified by several clinical variables: (a) Standard starting dose of 150 mg versus other doses, (b) Treatment initiation setting (outpatient versus inpatient), (c) Use of oral paliperidone/risperidone at baseline, (d) Number of co-prescribed antipsychotics at baseline, (e) Dosage of co-prescribed antipsychotics at baseline and (f) Co-prescription of clozapine at baseline.
Using these variables, we conducted a multivariate Cox proportional hazards model analysis to investigate the risk factors associated with the 1-year discontinuation of PLAI (Table 2). As a result, initiating PLAI in an outpatient setting was significantly associated with a lower risk of 1-year discontinuation (HR 0.671, 95% CI (0.533–0.846), p = 0.001). Other variables with significant associations with a lower risk of 1-year discontinuation included initiating PLAI with standard starting dose (150 mg) (HR 0.766, 95% CI (0.611–0.961), p = 0.021), use of concurrent antipsychotics at baseline (HR 0.630, 95% CI (0.428–0.927), p = 0.019), and a higher dose of baseline concurrent antipsychotics (HR 0.985, 95% CI (0.975–0.996), p = 0.005). Baseline use of clozapine, although lacking statistical significance, was associated with a 25% reduction in hazard for 1-year discontinuation (HR = 0.755, 95% CI (0.542–1.051), p = 0.096).
Cox proportional hazards model for discontinuation of PLAI within 1 year from initiation.

The baseline antipsychotics equivalent dose was calculated as the olanzapine equivalent dose.
p < 0.05.
β, beta coefficient estimate; CI, confidence interval; HR, hazards ratio; PLAI, paliperidone long-acting injectable; SE, standard error.
Based on the results, we developed a model to predict the risk of PLAI discontinuation within 1 year using ROC curve analysis (Figure 4). The AUC for the model was 0.61 (95% CI (0.561–0.646)). The following is the mathematical formulation of the model:



ROC curve for the Cox proportional hazards model predicting PLAI discontinuation within 1 year.
Discussion
Through this study, we assessed real-world clinical practice of paliperidone palmitate (PLAI) among patients with schizophrenia and investigated the clinical factors associated with treatment discontinuation. Those who started with a dose of 150 mg PP1M had a 24% lower risk of discontinuation. Compared to patients using only oral paliperidone or risperidone at baseline, those using one other antipsychotic had a 43% lower risk, and those using two or more other antipsychotics had a 38% lower risk of discontinuation. The risk of treatment discontinuation decreased by 26% for each 1mg olanzapine equivalent dose increase in the baseline total dose of antipsychotics. Patients who used clozapine at baseline had a 29.3% lower risk of treatment discontinuation. In addition, patients who initiated PLAI during outpatient had a 33% lower risk of treatment discontinuation compared to those who initiated PLAI during admission. These findings provide valuable insights into the factors influencing PLAI discontinuation and will help to optimize treatment strategies for schizophrenia patients.
In this study, the 1-year discontinuation rate of PLAI treatment from the time of initiation was 51.5%. The discontinuation rates reported in previous non-interventional studies varied widely. A multicenter, observational prospective study in Italy reported a 33.9% discontinuation rate by 12 months from PLAI initiation. 24 A naturalistic cohort study performed in London, UK, reported a 65% 1-year continuation rate among 200 patients, excluding 10 follow-up loss. 25 A nationwide population-based study using the South Korea Health Insurance Review Agency (HIRA) database reported a 57.5% 6-month continuation rate after PLAI initiation, 21 similar to what we found, demonstrating about half of the patients initiating PLAI discontinued before 1 year period in South Korea. It is noteworthy that adherence to LAI cannot be warranted either. Therefore, promoting the clinical strategy of LAI utilization is also required to achieve the benefits of antipsychotic LAI.
The discontinuation rate of the present study should be considered in terms of several factors. In our study, follow-up loss was also considered treatment discontinuation; however, some patients may have continued PLAI treatment at other medical centers. Thus, the true discontinuation rate may be lower than reported. Most of the patients included in the present study were those who had maintained antipsychotic treatment before initiating PLAI. The three hospitals included in the study are specialized medical centers that primarily treat severe psychosis, with a high proportion of treatment-resistant schizophrenia patients referred from other institutions. Although this study did not assess the severity of symptoms or the presence of acute symptoms, the characteristics of this patient population may have influenced the discontinuation rate. Moreover, it was observed that approximately 20% of the patients who discontinued PLAI within 1 year after the first initiation subsequently resumed the treatment later, showing the re-introduction of PLAI despite the first failure of maintenance.
The standard dosing regimen for starting PP1M follows a two-injection initiation schedule designed to achieve therapeutic levels: 150 mg is administered on Day 1 and 100 mg on Day 8 via intramuscular injection, followed by a monthly maintenance dose (50–150 mg, with a suggested dose of 75 mg). In this study, one-third of the total subjects received a first injection dose other than 150 mg, and only 54.5% followed the standard regimen including the first and second injection doses. The observed off-label dosing patterns may reflect clinical decisions made by physicians based on their prior experience, considering patient condition or concerns about adverse effects such as akathisia. Notably, patients who initiated PLAI with the standard starting dose of 150 mg demonstrated a significantly lower discontinuation rate at 1 year, suggesting that adherence to the standard regimen is advantageous for treatment maintenance compared to off-label dosing. Relationship between correct initiation and successful continuation of PLAI is relatively limited. However, a naturalistic observational study by Attard et al. found that correct initiation was significantly associated with a lower likelihood of discontinuation, 25 further supporting the importance of following the recommended initiation protocol.
PLAI can be started in patients who have maintained various oral antipsychotic medications; however, it is recommended to take oral paliperidone or risperidone before starting PLAI to check tolerability and determine the proper dosage of PLAI.26,27 In the present study, the patients who switched to PLAI while maintaining oral paliperidone or risperidone had lower discontinuation rates. However, there was no significant association between the ratio of the oral paliperidone or risperidone dose and the PLAI initiation dose. The findings demonstrate that adherence to the proposed standard dosing schedule based on the pharmacokinetic study could reduce the treatment failure of PLAI.
When starting PLAI, patients who were concurrently having polypharmacy with other antipsychotics and those with a higher overall antipsychotic dose exhibited a lower discontinuation rate. As noted, most of the patients in the present study were those who have maintained antipsychotic treatment before initiating PLAI, and the study sites play a significant role in the maintenance treatment of the patients with moderate to high severity referred from various psychiatry clinics. Our findings suggest that starting PLAI in patients using high doses of multiple antipsychotics as a combination agent could be advantageous for maintaining the treatment of PLAI as well as pre-maintained oral antipsychotic medications. Notably, our study is the first to identify the association between baseline co-prescribed antipsychotics and treatment continuation with PLAI. Additionally, it was observed that patients using clozapine were more likely to continue PLAI treatment. According to Bioque et al.’s retrospective 6-month mirror-image study, combination treatment with clozapine and PLAI effectively reduced symptoms and improved functionality in patients with treatment-resistant schizophrenia. 28 We have published that a combination of risperidone LAI with clozapine enhanced the compliance of clozapine medication and reduced hospitalization. 29 The findings indicate that PLAI could be an essential treatment option to enhance the maintenance treatment of patients with schizophrenia as an effective augmentation agent in combination with multiple antipsychotic agents.
In the present study, we found that patients who initiated PLAI during admission (compared to those in the outpatient clinic) had a 1.5-fold higher risk of treatment discontinuation. In other words, starting PLAI in the outpatient clinic showed a more favorable outcome regarding PLAI maintenance. The current Korean healthcare system mandates hospital-based administration, and there is no system for community-based administration of LAIs. All injections are administered by nurses, and outpatients receive their injections in hospital injection rooms. Likely, inpatients were primarily in an active phase of the disease, exhibiting acute and perhaps more severe psychotic symptoms. 30 Patients who initiated PLAI in an outpatient clinic were likely more stabilized. 31 The results suggest that starting PLAI in a stable outpatient setting during maintenance treatment rather than an acute phase may lead to longer treatment retention. It appears that adequate patient preparation and ensuring the patient is well-informed about the LAI treatment are crucial for maintaining adherence.32,33 It could be assumed that more stabilized outpatients, who may also have better compliance after thorough explanation and consent, could lead to better treatment retention. Although data are limited, similar findings have been reported in previous studies. For example, in Taylor et al.’s study, initiating PLAI in outpatients reduced the risk of discontinuation by 49%, and switching due to previous poor tolerability increased the risk of discontinuation by 63%. 34
The STAR Network “Depot Study” results in 32 centers in Italy indicated that the most frequent reasons for discontinuation among all types of LAIs were the onset of adverse events (32.9%) followed by the participant refusal of the medication (20.6%). 24 In a naturalistic observation study conducted in the UK, the main reason for PLAI discontinuation was perceived ineffectiveness (52% of discontinuers), and ten subjects (5% of the total) discontinued due to adverse effects. 25 In the present study, when examining the longitudinal change of discontinuation rate, most discontinuations occurred within the initial 3 months, suggesting the need for further study on the reasons for early discontinuation. Although we could not identify the individual reasons for discontinuation, the fact that most discontinuations occurred shortly after initiation suggests that patient preferences and adverse events, such as akathisia or injection site pain, may play a significant role rather than a lack of efficacy. On the other hand, it can be inferred that if patients can overcome the early phase, it would be advantageous for treatment retention. Therefore, paying attention to adverse events is crucial, especially during the initiation phase. Future studies should examine whether taking proactive measures to reduce adverse events could be beneficial for maintaining medication adherence.
A predictive model based on the derived variables achieved an AUC of 0.61, highlighting its moderate ability to predict the 1-year discontinuation of PLAI. As the present study analysis used the data from electronic medical data, including demographic variables, medication data, and treatment setting, other clinical variables, such as adverse events, symptom characteristics, and other patients’ subjective aspects, were not included in the present study. Although the utilized data were limited, the present study has achieved a moderate predictive model. Further research combining the present model with the patients’ aspects mentioned above will provide a more clinically meaningful model for LAI utilization.
Limitations
The strengths of this study include a large sample size and a comprehensive examination of real-world clinical data, providing insights into the practical application of PLAI in schizophrenia treatment. The inclusion of multiple clinical centers adds to the generalizability of the findings. However, there are several limitations. The study is retrospective, which limits the ability to establish causality. Additionally, the study did not assess symptom severity or the reasons for discontinuation, which could provide further insights into factors influencing treatment adherence. Furthermore, follow-up loss was considered discontinuation, which may have overestimated the actual discontinuation rate, as some patients might have continued treatment elsewhere. Finally, while the study identifies critical clinical factors associated with PLAI discontinuation, as mentioned above, the predictive model’s AUC of 0.61 indicates moderate predictive ability, suggesting that other unmeasured factors may also contribute to treatment outcomes.
Conclusion
Initiation during the outpatient clinic, starting with a 150 mg dose, co-prescription of additional antipsychotics, a higher dose of baseline antipsychotics, and clozapine use, was associated with a reduced risk of discontinuation. The results provide valuable insights for optimizing treatment strategies. The findings suggest the importance of a sufficient starting dose of PLAI following the recommended starting method. Moreover, PLAI could be posited as an effective augmentation agent of choice in the maintenance treatment of patients with schizophrenia in real-world clinical settings.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253251333987 – Supplemental material for Investigation on the factors associated with maintenance of paliperidone long-acting injection in the real-world treatment of patients with schizophrenia
Supplemental material, sj-docx-1-tpp-10.1177_20451253251333987 for Investigation on the factors associated with maintenance of paliperidone long-acting injection in the real-world treatment of patients with schizophrenia by Nuree Kang, Ae Jeong Jo, Sung Woo Joo, Jung Sun Lee, Kyu Young Lee, Yong Sik Kim, Jae Hoon Jeong, Jeong Hoon Lee, Joongyub Lee and Se Hyun Kim in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-docx-2-tpp-10.1177_20451253251333987 – Supplemental material for Investigation on the factors associated with maintenance of paliperidone long-acting injection in the real-world treatment of patients with schizophrenia
Supplemental material, sj-docx-2-tpp-10.1177_20451253251333987 for Investigation on the factors associated with maintenance of paliperidone long-acting injection in the real-world treatment of patients with schizophrenia by Nuree Kang, Ae Jeong Jo, Sung Woo Joo, Jung Sun Lee, Kyu Young Lee, Yong Sik Kim, Jae Hoon Jeong, Jeong Hoon Lee, Joongyub Lee and Se Hyun Kim in Therapeutic Advances in Psychopharmacology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
