Abstract
Background:
Previous studies reported higher incidences of venous thromboembolism and cardiovascular disease in schizophrenia patients and higher indicators of thrombosis, thrombocyte activation, and platelet dysfunction.
Objectives:
To check if first-episode schizophrenia (FES) patients have a hypercoagulable state and determine whether acute and chronic antipsychotics have the same effect on blood coagulation or fibrinolysis-related biomarkers.
Design:
Case-control study.
Methods:
A total of 81 participants were grouped in FES, chronic schizophrenia (CS), and healthy controls (HCs). In addition to demographic data and clinical characteristics, immunological analyses were performed to measure plasma levels of D-dimer, plasminogen activator inhibitor-1 (PAI-1), soluble P selectin (sP-sel), tissue plasminogen activator (tPA), thrombotic precursor protein (TpP), and von Willebrand’s disease factor (vWF).
Results:
Compared to HC group, FES patients showed higher PAI-1 (28.61 ng/ml versus 15.69 ng/ml), sP-sel (2.78 ng/ml versus 1.18 ng/ml), and TpP (15.61 µg/ml versus 5.59 µg/ml) along with a higher PAI-1/tPA (3.12 versus 2.00). Acute antipsychotic medication reduced higher PAI-1 (28.61 → 21.99), sP-sel (2.78 → 1.87), tPA (9.59 → 5.83), TpP (15.61 → 10.54), and vWF (383.18 → 291.08) in FES patients. However, plasma sP-sel and vWF in CS patients returned to the pre-treatment levels in FES patients, and PAI-1/tPA significantly decreased compared to FES patients.
Conclusion:
These results suggest a hypercoagulable state in FES patients and demonstrate contrast effects of acute and chronic antipsychotics on coagulation or fibrinolysis in schizophrenia patients.
Introduction
Schizophrenia is a chronic, severe, and disabling brain disorder with a life prevalence of approximately 1% of world population. 1 Patients with schizophrenia present positive and negative symptoms, as well as cognitive impairments that influence their thoughts, perceptions, speech, affect, and behaviors. It was estimated that people with schizophrenia die on average 20 years earlier than individuals without this illness.2,3 One possible risk factor for a shortened life span in people with schizophrenia is an increased risk of venous thromboembolism (VTE) 4 and cardiovascular disease. 5 These risk factors are directly or indirectly associated with the use of antipsychotics. For example, the risk of VTE in people using antipsychotics is higher than the general population.6–8 In a population-based cohort study, the schizophrenia cohort exhibited a 2.02-fold higher adjusted hazard ratio (HR) for developing deep venous thrombosis (DVT), and a 1.99-fold higher adjusted HR for developing pulmonary embolism (PE) compared to the non-schizophrenia cohort. Schizophrenia patients using first- or second-generation antipsychotics exhibited a higher adjusted HR for both DVT and PE development. 9 Patients with schizophrenia who receive long-term antipsychotic medication show evidence of an overall hypercoagulable and hypofibrinolytic state, which may contribute to their increased risk of VTE. 10 According to a recent review, 87.5% of patients taking clozapine had risk factors for VTE. 11
In addition to the antipsychotic drug-associated VTE risk factors mentioned above, schizophrenia itself has a tendency to increase the risk of VTE, as evidenced by elevated markers of thrombosis, thrombotic cell activation, and platelet dysfunction in antipsychotic-naïve patients with schizophrenia.12,13 This tendency is not surprising given immobility due to avolition, 14 and catatonia seen in a high proportion (9–17%) of acute schizophrenia patients. 15 Moreover, schizophrenia patients are often associated with hyperglycemia, hyperlipidemia, hyperhomocysteinemia, obesity, and nicotine dependence, which are risk factors for VTE.16,17 However, few studies have compared the patients with first-episode schizophrenia (FES) to those with chronic schizophrenia (CS) in terms of risk factors for VTE.
This study measured and compared several biomarkers relevant to coagulation or fibrinolysis in drug-naïve FES patients, CS patients, and healthy controls (HCs) matched with age and gender. The measured biomarkers include D-dimers, plasminogen activator inhibitor-1 (PAI-1), soluble P selectin (sP-sel), tissue plasminogen activator (tPA), thrombus precursor protein (TpP), and von Willebrand factor (vWF). In addition, effects of acute and chronic antipsychotic medications on these biomarkers in the FES and CS patients were assessed and compared. We aimed to know whether there were group differences in terms of clotting and fibrinolysis-related biomarkers, and to explore the effects of acute and chronic antipsychotics on these biomarkers in patients with schizophrenia.
Materials and methods
Participants
Participants in this study were 18–45 years old and categorized into three groups of drug-naïve FES patients, CS patients, and health controls, with 27 persons in each group. The sample size in each group was calculated by referring to a previous clinical study determining whether markers of thrombogenesis are increased in psychotic patients who have not yet been treated with antipsychotic medication. Each group of the referred study consisted of 25 participants. In the present study, we set a target power of 0.90, α value of 0.05 (two-sided). With these inputting data, the PASS 15 software (NCSS, Utah, American) figured out the sample size of 26 (persons) in each group.
The patients were hospitalized in the adult psychiatric unit of Shenzhen Kangning Hospital (SKH)/Shenzhen Mental Health Center during the period between May 2019 and May 2020, and healthy controls were adults matched for age and gender but had no history of mental disorders. All participants were informed of the purpose of the study before agreeing to participate in the study, which did not incur any additional costs to the participants.
All schizophrenia patients met the diagnostic criteria for schizophrenia as established by the DSM-5 (The Diagnostic and Statistical Manual of Mental Disorders 5th version) or ICD-10 (International Classification of Diseases 10th version). Their Positive and Negative Symptom Scale (PANSS) scored ≧60 points overall and were assessed by a certificated psychiatrist Tu Weifeng (TW). Patients with FES were those admitted to the hospital (SKH) for psychotic symptoms, but had never taken antipsychotic medications or been diagnosed with schizophrenia or any mental disorder. CS patients were hospitalized for a diagnosis of schizophrenia and had a disease course of ⩾5 years. Healthy controls had no mental disorder and no history of mental disorder in their immediate relatives. They matched with FES patients in age and sex. The exclusion criteria applied to all participants: (1) who had major physical conditions, such as gastrointestinal, liver, kidney, nervous system, blood disorders, and immune dysfunction; (2) who have been diagnosed with alcohol addiction or other psychotropic substance abuse; (3) who have undergone surgical operations within the last 3 months; (4) who are recently taking drugs that affect the hematopoietic system and regulate immune function; and (5) during pregnancy or lactation.
Study design
When enrolling in the study, all participants were asked to provide their demographic information, as well as a history of physical illnesses and mental disorders. Their body mass index (BMI) was calculated based on body weight (kg) and height (cm). PANSS was used to evaluate the severity of clinical manifestations of FES patients on admission day. Blood sample was collected from all participants after an overnight fasting for biochemical analyses to measure levels of serum fasting glucose (SFG), high-density lipoprotein (HDL), low-density lipoprotein (LDL), total cholesterol (TC), and triglycerides (TG). In addition, the levels of biomarkers related to coagulation and fibrinolysis in the blood of all participants were assessed. For FES patients, two batches of biochemical analyses were done with two blood samples collected on admission day (before antipsychotic medication) and after finishing acute antipsychotic (AAP) therapy for 4–8 weeks. For CS patients and healthy controls, only one batch of biochemical analyses were performed.
Antipsychotics or drug combination used for the schizophrenia patients
During a 4–8 weeks of hospitalization, the vast majority of patients with FES were treated with only one atypical antipsychotic, whereas two patients were treated with risperidone in combination with quetiapine or olanzapine. The medical records of CS patients show that more than half of them were treated with various combinations of drugs, including risperidone plus × (aripiprazole, clozapine, olanzapine, sulpiride, sodium valproate, or lithium). Dosage details for each drug and drug combination are shown in Table 1. In order to compare the antipsychotic dosages used in the FES and CS patients, we transferred dosages of all antipsychotics (except for risperidone) into risperidone dose equivalence, followed by statistical comparisons between the two groups of patients. The risperidone dose equivalence in the two groups were 6.94 (2.27) and 7.76 (3.34) mg/day, respectively. The difference is not statistically significant. Of note is, eight of CS patients were treated with an antipsychotic plus a mood stabilizer of either lithium or valproate in sustained release capsule. These patients did not respond well to antipsychotic(s), but did better in the combination of antipsychotic plus a mood stabilizer, which indeed reduced the dosage of the antipsychotic used for the patients as indicated by an averaged risperidone dose equivalence of 5.81 mg/day (1.70).
Drugs or drug combinations used for FES and CS patients.
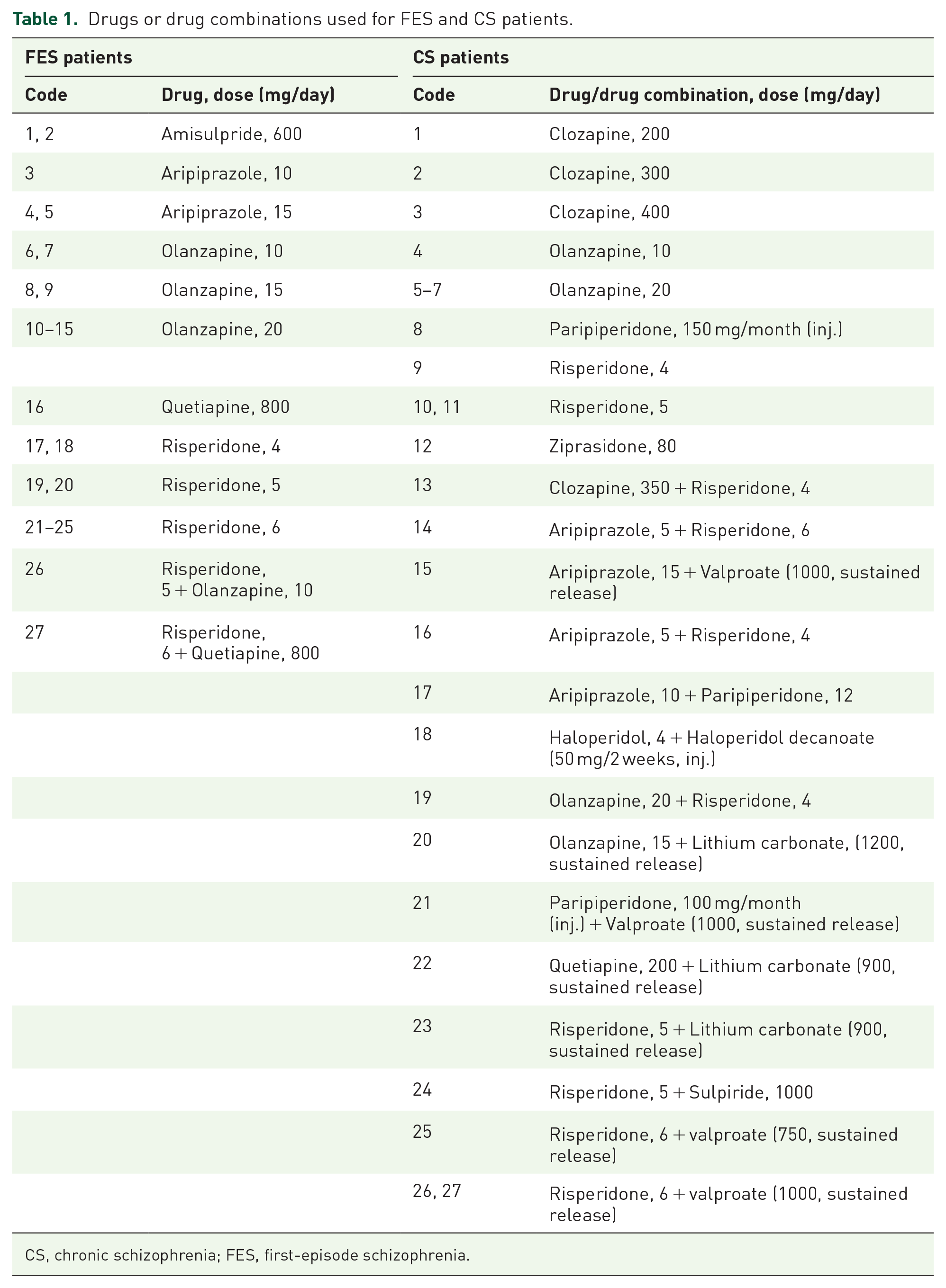
CS, chronic schizophrenia; FES, first-episode schizophrenia.
Biochemical and immunological analyses of blood samples
For biochemical analyses, peripheral venous blood samples (4 ml per person) were collected from cubital vein of all participants using a 21-gauge butterfly needle. SFG, LDL-C, HDL-C, TC, and TG levels were timely detected in the clinical laboratory of SKH. Plasma aliquots for other biomarkers were stored at −20°C until analyzed using commercial ELISA kits according to the protocol provided by the manufacturer (mlbio, Shanghai, China).
Statistical analysis
SPSS 25.0 (IBM, New York, American) was used for statistical analysis of all the data. The Shapiro–Wilk test was used to test the normality of a dataset. The data in normal distribution were expressed as mean ± SD, otherwise expressed as median (interquartile range). For inter-group (FES, CS, and HCs) comparisons, the Bonferroni method was employed following one-way ANOVA. For intra-group comparisons of FES patients (before-after comparisons), paired t-test was done. The comparisons of medians were done by performing the Mann–Whitney U-test. Enumeration data were expressed as percentages and compared by chi-square test. p ⩽ 0.05 was considered statistically significant.
Results
Baseline characteristics of the participants
A total of 81 persons participated in this study, including 27 drug-naïve FES patients, 27 CS patients, and 27 healthy controls matched for age and gender. As shown in Table 2, compared with healthy controls, patients with schizophrenia had fewer years of education and a family history of the disease. The CS group had higher BMI and TG levels than the other two groups. The level of SFG in CS group was higher than that in FES patients and healthy controls, but the difference was not statistically significant. HDL levels were lower in both FES and CS patients compared to healthy controls.
Demographic information and metabolism-related data of the participants.

Continuous values are presented as mean ± SD. TG data are presented as median (interquartile range). The values with superscript a resulted from one-way ANOVA (F); the values with superscript b resulted from chi square test (χ2); the values with superscript c resulted from the Kruskal–Wallis test (H).
BMI, body mass index; CS, chronic schizophrenia; FES, first-episode schizophrenia; HC, healthy control; HDL, high-density lipoprotein; LDL, low-density lipoprotein; SFG, serum fasting glucose; TC, total cholesterol; TG, triglycerides.
Hypercoagulable state in FES patients
The first objective of this study was to determine whether FES patients had hypercoagulability relative to healthy controls. We therefore measured plasma levels of biomarkers associated with coagulation or fibrinolysis, including D-dimer, PAI-1, sP-sel, t-PA, TpP, and vWF. As shown in Table 3, FES patients showed higher levels of PAI-1, sP-sel, TpP, and higher PAI-1/tPA value compared to healthy controls. Higher TpP reflects active thrombosis while elevated level of sP-sel is indicative of endothelium activation. Increased PAI-1 levels indicate reduced plasmin formation. These results indicate that the FES patients in this study were in a hypercoagulable state compared to healthy controls. The two groups were comparable in terms of tPA and vWF. D-dimer concentration in FES group was marginally higher (p = 0.085) compared to that of healthy controls.
Hypercoagulable state in first-episode schizophrenia patients.

Data are presented as median (interquartile range). Z values resulted from the Mann–Whitney U-test.
FES, first-episode schizophrenia; HC, healthy control; PAI-1, plasminogen activator inhibitor-1; sP-sel, soluble P selectin; tPA, tissue plasminogen activator; TpP, thrombus precursor protein; vWF, von Willebrand factor.
AAP medication improved the hypercoagulable state in FES patients
The next goal of this study was to examine the effects of AAP on the coagulable state in FES patients. Thus, all FES patients (n = 27) were treated with antipsychotics for 4–8 weeks, and biochemical analyses for the same biomarkers associated with coagulation and fibrinolysis were performed at the end of treatment. As shown in Table 4, higher levels of PAI-1, sP-sel, and TpP in FES patients were reversed after antipsychotic therapy for 4–8 weeks. Interestingly, plasma tPA and vWF levels were significantly reduced in FES patients after acute administration of antipsychotics. But the treatment did not change D-dimer concentration and PAI-1/tPA value of the FES patients.
Acute antipsychotic medications reverse the hypercoagulable state in FES patients.

Data are presented as median (interquartile range). Z values resulted from Mann–Whitney U-test.
AP, antipsychotic; FES, first-episode schizophrenia; PAI-1, plasminogen activator inhibitor-1; sP-sel, soluble P selectin; tPA, tissue plasminogen activator; TpP, thrombus precursor protein; vWF, von Willebrand factor.
In addition, we compared the FES patients following AAP to healthy controls in terms of the same biomarkers mentioned above. As shown in Table 5, significant differences existed between the two groups in values of PAI-1, tPA, TpP, and PAI-1/tPA, indicating that APP did not normalize all biomarkers relevant to coagulation and fibrinolysis in the FES patients.
The Comparisons between patients treated with acute antipsychotic medications and healthy controls.
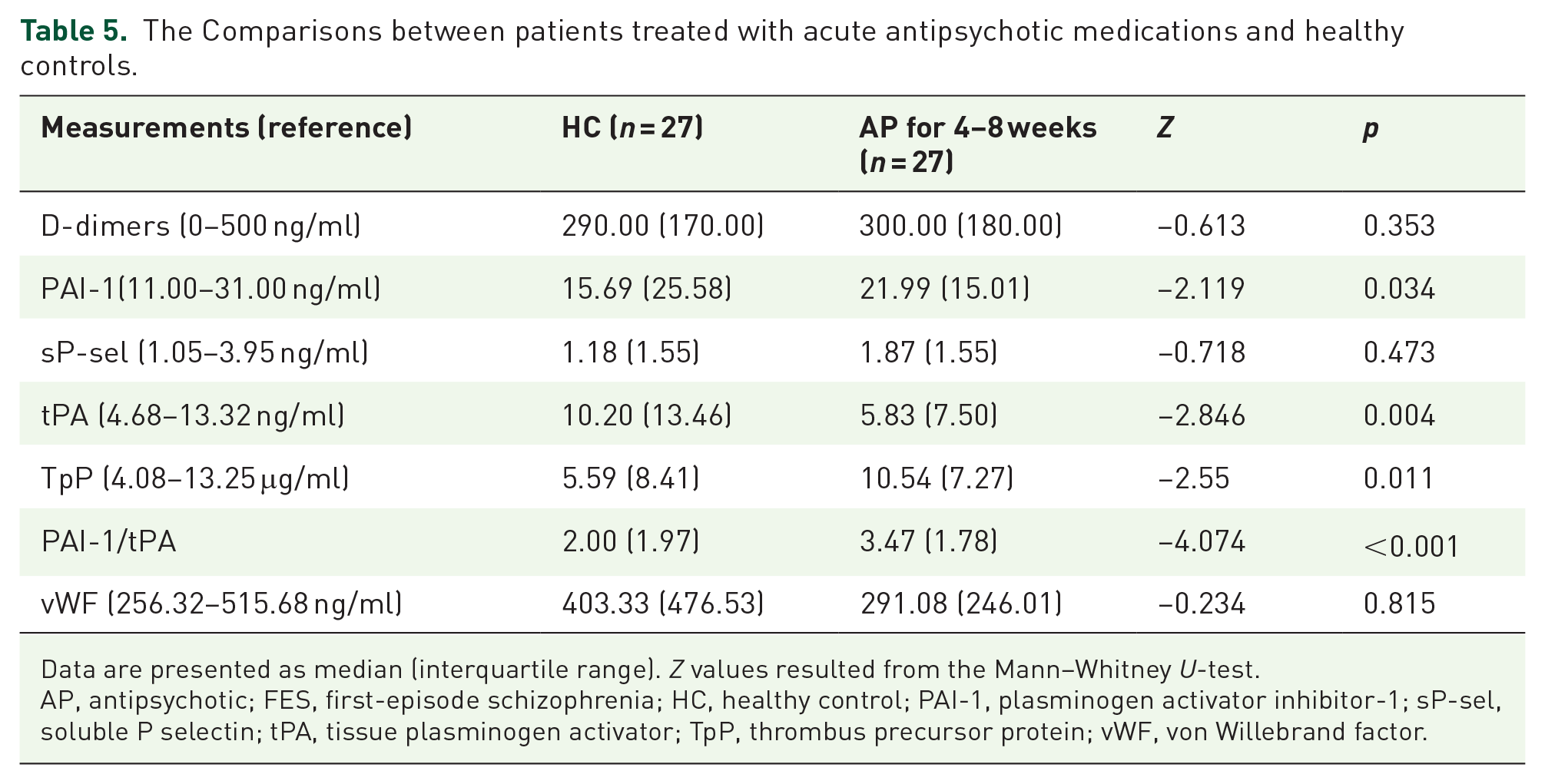
Data are presented as median (interquartile range). Z values resulted from the Mann–Whitney U-test.
AP, antipsychotic; FES, first-episode schizophrenia; HC, healthy control; PAI-1, plasminogen activator inhibitor-1; sP-sel, soluble P selectin; tPA, tissue plasminogen activator; TpP, thrombus precursor protein; vWF, von Willebrand factor.
Comparisons between CS and FES patients following AAP
The third goal of this study was to compare effects of acute and chronic antipsychotic medications on plasma biomarkers relevant to coagulation and fibrinolysis in schizophrenia patients. For this purpose, we compared the biomarkers of CS patients to those of FES patients before and after AAP. As shown in Figure 1, sP-sel level in drug-naïve FES patients decreased after AAP, but it returned to the pre-treatment level in CS patients [Figure 1(a)]. Similarly, levels of PAI-1, tPA, TpP, and vWF in drug-naïve FES patients decreased after AAP but returned to pre-treatment levels in CS patients [Figure 1(b), (c), (e), (f)]. However, AAP did not change tPAI-1/tPA value in patients with FES, whereas patients with CS had a reduced tPAI-1/tPA relative to patients with FES treated with AAP [Figure 1(d)].

Different effects of acute and chronic antipsychotic medications on coagulation and fibrinolysis biomarkers in the blood of patients with schizophrenia. (a) Effects on sP-sel levels. (b) Effects on PAI-1 levels. (c) Effects on tPA levels. (d) Effects on PAI-1/tPA. (e) Effects on TpP levels. (f) Effects on vWF levels. Data are presented as median (blue) + interquartile (red). Mann–Whitney U-test was done to make comparisons between groups. *p < 0.05, compared to FES. +p < 0.05, compared to AAP.
Discussion
The three main findings of this study are: (1) drug-naïve FES patients presented a hypercoagulable state and vascular endothelial cell injury as evidenced by higher levels of circulating PAI-1, sP-sel, and TpP, as well as higher PAI-1/tPA ratio compared to healthy controls; (2) the AAP therapy improved the hypercoagulable state and vascular endothelial cell injury in FES patients as the treatment reduced higher plasma levels of sP-sel, TpP, and PAI-1, as well as significantly decreased levels of tPA and vWF in the patients; and (3) chronic antipsychotic therapy increased the risk of thrombosis in the patients as CS patients showed higher plasma levels of sP-sel, tPA, and vWF than FES patients who received AAP medications.
As its name implies, PAI-1 functions as the principal inhibitor of tPA and urokinase (uPA), two activators of plasminogen. This protease inhibitor is mainly produced from the endothelial cells. Therefore, increased plasma level of PAI-1 in drug-naïve FES patients suggests the presence of endothelial cell damage, which may increase the risk for VTE. In support of this suggestion, FES patients showed a higher PAI-1/tPA ratio. 18 Indeed, elevated PAI-1 is considered a risk factor for thrombosis and atherosclerosis, whereas PAI-1/tPA ratio has been used as an index of abnormalities in thrombosis/fibrinolysis system. 19 Along with elevated PAI-1, circulating level of sP-sel in FES patients in this study was also higher than healthy controls, indicating platelet and endothelial cell activation.13,20
TpP has been identified as biomarker of active thrombosis in severe clinical conditions including myocardial infarction. 21 And TpP ELISA test had a 100% sensitivity for evaluating proximity deep thrombosis and PE. 22 In the present study, we found that FES patients showed higher levels of TpP, a marker of active thrombosis. Considering the data from the aforementioned two previous studies, the changes in PAI-1 and sP-sel, plus higher levels of TpP in FES patients in this study demonstrate a hypercoagulable state in these patients.
The fibrin DD is the primary enzymatic degradation product of fibrin by plasmin. As such, a negative D-dimer is now considered the biochemical gold standard for ruling out an acute episode of VTE in a patient with a low pretest probability for VTE. 23 In this study, the concentration of D-dimer in FES group was slightly higher than that in healthy control group (p = 0.085), but it was not significant, indicating that insignificant blood coagulation happened, at least in some patients of this group before blood collection. This explanation is consistent with previously reported elevated markers of thrombosis, thrombotic cell activation, and platelet dysfunction in patients with schizophrenia who have never taken antipsychotics.12,13 Of note, FES patients in this study were comparable to healthy controls in the demographic data and some clinical characteristics including age, BMI, SFG, TC, TG, and LDL, except for HDL which was lower in FES patients than controls. These data suggest that metabolic syndrome and obesity were not important contributors to the hypercoagulable state in young adult FES patients of the present study.
Although antipsychotic drugs increased VTE risk in previous studies, 24 few studies investigated effects of AAP agents. Of the available research, a time-matched case–control analysis with a cohort of 111,818 patients with at least one antipsychotic drug prescription is worth highlighting. This study revealed that current exposure to antipsychotic drugs was not associated with an increased risk of DVT, compared to nonusers. 25 Relevantly, an early study found no difference in coagulation factors between patients treated with clozapine (for 12 weeks) and the controls. 26 In the present study, 4–8 weeks of antipsychotic treatment effectively reduced higher levels of sP-sel, PAI-1, and TpP in patients with FES, but the PAI-1/tPA ratio did not change, suggesting that the treatment has an anticoagulant effect. In support of this suggestion, the treatment significantly decreased levels of vWF in the patients. This finding reminds us of the major role of vWF in blood coagulation via binding to other proteins, particularly factor VIII, and promoting platelet adhesion to wound sites demonstrated in a previous study. 27 Further supporting evidence for the beneficial effects of AAP is the decreased tPA in FES patients after the treatment. This serine protease (tPA) locates in endothelial cells and catalyzes the conversion of plasminogen to plasmin, the major enzyme responsible for clot breakdown. 28 Moreover, a recent study reported the reduction of plasma procoagulant activity in patients with schizophrenia during pharmacotherapy. 29 Collectively, the data presented in this study, plus those reported in the above-mentioned previous studies, demonstrate a protective effect of AAP on damaged endothelial cells in FES patients thus ameliorated the hypercoagulability in the patients.
Although the mechanism of protective action of AAP therapy on damaged endothelial cells in patients with FES is unknown, the anti-inflammatory and antioxidant effects of atypical antipsychotics are noteworthy. First, accumulating evidence demonstrates that schizophrenia is characterized by mild or chronic inflammation along with disturbances in metabolism.30,31 Second, metabolic syndrome/overload, which are associated with schizophrenia, evokes several stress reactions, such as oxidative, inflammatory, organelle, and cell hypertrophy. 32 Third, atypical antipsychotics were shown to suppress production of pro-inflammatory cytokines but upregulate interleukin-10 (an anti-inflammatory cytokine) in lipopolysaccharide-treated mice. 33 Both olanzapine and aripiprazole decreased mRNA and protein levels of IL-1β (interleukin-1β), IL-6, and TNF-α (tumor necrosis factor–α) in conditioned medium of stimulated primary human peripheral blood mononuclear cells from healthy donors. 34 Clozapine significantly increased the production of anti-inflammatory cytokines such as IL-10 and TGF-β (trnasforming growth factor-β) in blood leukocytes of healthy volunteers. 35 Fourth, atypical antipsychotics exert a strong antioxidant activity in experimental models of schizophrenia by rescuing the antioxidant system and increasing serum levels of the antioxidant enzymes superoxidase dismutase and glutathione as reviewed recently. 36 The anti-inflammatory actions of atypical antipsychotics, however, are not seen in patients after chronic antipsychotic treatment which induced weight gain and metabolic side effects. 37
Plasma sP-sel, tPA, TpP, and vWF in CS patients recovered to levels comparable to those of antipsychotic-naïve FES patients, but higher than those of FES patients who received AAP medication. Moreover, the PAI-1/tPA ratio significantly decreased because of the significant tPA increase. These data indicate the presence of a hypercoagulable state in the CS patients. They had a course of more than 5 years and was admitted to the psychiatric hospital due to recurrent psychotic episodes. These data added further evidence that chronic antipsychotics increase the risk of developing VTE in schizophrenia patients. This conclusion is in line with previous clinical reports linking the use of antipsychotics to VTE. 6
In explaining the beneficial effects of AAP on hypercoagulable state and vascular endothelial cell injury in FES patients and the relapse of blood hypercoagulable status in CS patients, the following issues merit special attention. First, CS patients have higher prevalence rates of co-morbidities including metabolic syndrome, diabetes, hypertension, obesity, and insulin resistance in Hagi et al. 38 On the one hand, these medical conditions are associated with the long-term use of antipsychotics, especially atypical drugs. 39 On the other hand, they are both risk factors for VTE and vascular injury.40,41 Second, smoking should not be ignored though this study did not collect the smoking-relevant information from the participants. Previous studies have reported that schizophrenia patients have extremely high prevalence of smoking compared to the general population and patients with other psychiatric diagnoses.42–44 And smoking has been demonstrated as a risk factor for VTE with a dose–response relationship.45,46 Therefore, it is plausible to suggest that smoking cessation during the hospitalization period of acute schizophrenia patients may help to ameliorate the hypercoagulable state and vascular endothelial cell injury in them, whereas re-smoking in some patients of CS group may contribute to the relapse of blood hypercoagulable status in these patients.
Limitations
The limitations of this study should not be ignored. First, the human cohort sample was relatively small. Therefore, caution should be taken in generalizing conclusions from this study. Relevant to the relatively small sample size, we could not analyze possible differences in effects of different antipsychotic treatment on the coagulation parameters in schizophrenia patients. Third, we were unable to analyze effects of confounding factors such as smoking as mentioned above. Fourth, the activities of daily living (ADL) of schizophrenia patients in the present study was not assessed although immobility due to avolition 14 is one of clinical manifestations of schizophrenia patients. This limitation disenabled us to rule out the contribution of ADL to the hypercoagulable state in FES patients and the abnormalities in coagulation parameters in CS patients. Nevertheless, the existent data confirmed the pre-existing abnormalities of coagulation parameters in FES patients and demonstrated the different effects of acute and chronic administration of antipsychotics on these parameters. These findings have potential implications for the prevention and intervention of VTE in schizophrenia patients.
Conclusion
In conclusions, drug-naïve FES patients have a hypercoagulable state and vascular endothelial cell injury; APP improves the hypercoagulable state and vascular endothelial cell injury in FES patients; chronic antipsychotic therapy increases the risk of thrombosis, which could be a secondary effect mediated by increased weight and body fat, as well as smoking.
