Abstract
Background:
Long-acting injectable (LAI) antipsychotic medications are used to optimise treatment outcomes in schizophrenia. Guaranteed medication delivery increases the responsibility of prescribers to monitor and manage adverse effects.
Methods:
In the context of a quality improvement programme conducted by the Prescribing Observatory for Mental Health, a clinical audit addressed documented side-effect monitoring in patients prescribed continuing LAI antipsychotic medication under the care of United Kingdom adult mental health services.
Results:
A total of 62 mental health services submitted data on 5169 patients prescribed LAI antipsychotic medication for more than a year. An assessment of side effects had been documented in the past year in 2304 (45%) cases. Post hoc analysis showed that extrapyramidal side effects were more likely to have been assessed and found to be present in those patients prescribed LAI haloperidol, flupentixol or zuclopenthixol. There was little other targeting of assessments to the known side effects profiles of individual LAI antipsychotic medications, but when dysphoria had been assessed it was most commonly found with LAI haloperidol treatment and when weight gain, sexual and prolactin-related side effects had been assessed, they were more often identified with LAI paliperidone.
Conclusion:
The data suggest a relatively low frequency of side-effect assessments, largely untargeted. This is likely to result in many adverse effects going unrecognised and unmanaged, thus failing to tackle their potential to confound mental state assessment and adversely affect physical health and adherence. Patients receiving LAI antipsychotic medication have regular contact with a healthcare professional who administers the medication, which provides an opportunity to potentially remedy this situation.
Keywords
Introduction
Maintenance treatment with antipsychotic medication more than halves the risk of relapse in people with schizophrenia, with a number-needed-to-treat of three, 1 one of the largest effect sizes associated with pharmacological interventions across all medical conditions, 2 although poor adherence to the medication can limit these beneficial effects. 3 Long-acting injectable (LAI) preparations of antipsychotic medication are commonly used in clinical practice to ensure medication delivery, and large database studies have confirmed that this strategy results in fewer relapses and re-hospitalisations compared with treatment with the same medication in oral form.4,5 In the United Kingdom (UK), between a quarter and a third of patients with schizophrenia under the care of the National Health Service (NHS) are prescribed LAI antipsychotic preparations, depending on the clinical setting, and the proportion is similar in most other developed countries. 6
With respect to the relative efficacy and tolerability of antipsychotic medications, network meta-analyses of randomised controlled trials (RCTs) of oral antipsychotic medications suggest that, clozapine aside, any differences in efficacy are relatively small and differences in side-effect profiles are more likely to be clinically relevant.7–9 However, it is difficult to be confident about the relative side-effect liability of individual antipsychotic medications as side effects are not always comprehensively and systematically assessed and reported in RCTs of antipsychotic medications. 10 Nevertheless, the absolute side-effect burden is likely to be high: when a large cohort of patients prescribed maintenance antipsychotic medication was systematically assessed for side effects, at least one treatment-emergent problem was noted to be present in more than three-quarters of cases. 11 This reinforces the need for careful clinical review of the tolerability of continuing antipsychotic treatment so that any side effects can be detected and remedial action taken.
We report here on the quality of side-effect assessment for patients prescribed continuing LAI antipsychotic medication in routine clinical practice in secondary mental health services in the UK.
Materials and methods
The Prescribing Observatory for Mental Health (POMH) invited all 64 member healthcare organisations in the UK to participate in a clinical audit as part of a quality improvement (QI) programme addressing the use of LAI antipsychotic medication for relapse prevention. 12 The practice standards for the audit were derived from the National Institute of Clinical Excellence (NICE) 13 and British Association for Psychopharmacology (BAP) 3 guidelines for the treatment of schizophrenia and related to the quality of care planning and medication review, including the nature and quality of the assessment of side effects in patients receiving LAI antipsychotic treatment for a year or more.
Each participating healthcare organisation was asked to identify a suitable QI sample of patients under the care of adult mental health services prescribed continuing LAI antipsychotic medication. 14 Audit of these patients’ clinical records, using a customised data collection tool, allowed each organisation to submit the following demographic and clinical data for each case: age, gender, ethnicity, documented psychiatric diagnosis, 15 the clinical setting in which care was provided and legal status. Information on the LAI antipsychotic preparation prescribed, including the dose and injection frequency, were also noted. With respect to the monitoring of side effects, the following data were collected for each case: whether there was a documented review of medication in the last year and, if so, whether the assessment of medication side effects had been recorded, specifically extrapyramidal side effects (EPS), weight gain, sedation, injection site reactions, and metabolic, prolactin-related, sexual and anticholinergic side effects, as well as dysphoric or discomfiting subjective experiences. For each of the documented side-effect assessments, it was as noted whether or not the particular side effect had been identified as present.
The audit data were collected by clinicians and clinical audit staff in participating services and were submitted pseudonymously on-line to POMH using Formic software. 16 Ethical approval is not required for audit-based QI initiatives. 17 The data were analysed using SPSS V26.0. 18
Results
A total of 62 mental health services submitted data relating to 5169 patients, all of whom had been prescribed an LAI antipsychotic medication for more than a year. Of these patients, 3393 (66%) were male, 3553 (69%) were White/White British and 4328 (84%) had a diagnosis of a schizophrenia spectrum disorder (ICD-10 F20-29). The mean age was 49 (SD 13) years; 197 (5%) patients were 25 years of age or younger and 565 (11%) were older than 65 years of age. At the time the data were collected, 4374 patients (85%) were under the care of a community mental health team (CMHT), 708 (14%) were detained in hospital under mental health legislation and the remaining 87 (2%) were informal inpatients. The depot/LAI antipsychotic medications prescribed were zuclopenthixol (n = 1289; 25%), flupentixol (1202; 23%), paliperidone (994; 19%), aripiprazole (n = 568; 11%), risperidone (503; 10%), haloperidol (375; 7%), fluphenazine (139; 3%), olanzapine (93; 2%) and pipotiazine (12; <1%). More than one of these preparations was prescribed in six cases.
Almost all (n = 560; 99%) of the LAI aripiprazole prescriptions were for monthly administration and, of these, 479 (86%) were for a dose of 400 mg (dosage range for monthly administration 200–400 mg). The other LAI second-generation antipsychotic (SGA) medications were also prescribed using relatively standard regimens in terms of both dosage interval and dose administered: 97% of LAI risperidone prescriptions were for 2-weekly administration (modal dose 50 mg; range 25–75 mg), and 85% of LAI paliperidone prescriptions were for 4-weekly administration (modal dose 150 mg; range 50–250 mg). With respect to the LAI first-generation antipsychotic (FGA) medications, 68% of LAI haloperidol prescriptions were for 4-weekly administration (modal dose 100 mg; range 25–400 mg) and 59% of LAI flupentixol prescriptions were for 2-weekly administration (modal dose 40 mg; range 20–600 mg) as were 55% of LAI zuclopenthixol prescriptions (modal dose 400 mg; range 50–1000 mg). The prescribed dose of LAI flupentixol was greater than the suggested optimal dose of 20–40 mg every 2 weeks in 224 (19%) cases. 19
An assessment of side effects had been documented in the past year in 2304 (45%) cases. Tables 1–4 provide information on the side-effect assessments associated with each of the LAI antipsychotic medications that was prescribed for more than 5% of the total national sample. Excluded from these tables are LAI fluphenazine and LAI pipotiazine (both have been discontinued in the UK) and LAI olanzapine (used infrequently due to the risk of post-injection delirium/sedation). 20 These tables include data for a total of 2866 patients prescribed an LAI FGA and 2065 patients prescribed an LAI SGA, of whom 2195 (45%) had an assessment of side effects in the past year documented in their clinical records.
Assessment of side effects documented in the clinical records in the past year for patients prescribed each LAI antipsychotic medication.

LAI, long-acting injectable.
Documented assessment of EPS in the past year and the co-prescription of anticholinergic medication for each LAI antipsychotic medication.

EPS, extrapyramidal side effects; LAI, long-acting injectable.
Documented assessment in the past year of weight gain and metabolic, prolactin-related and sexual side effects for each LAI antipsychotic medication.

LAI, long-acting injectable.
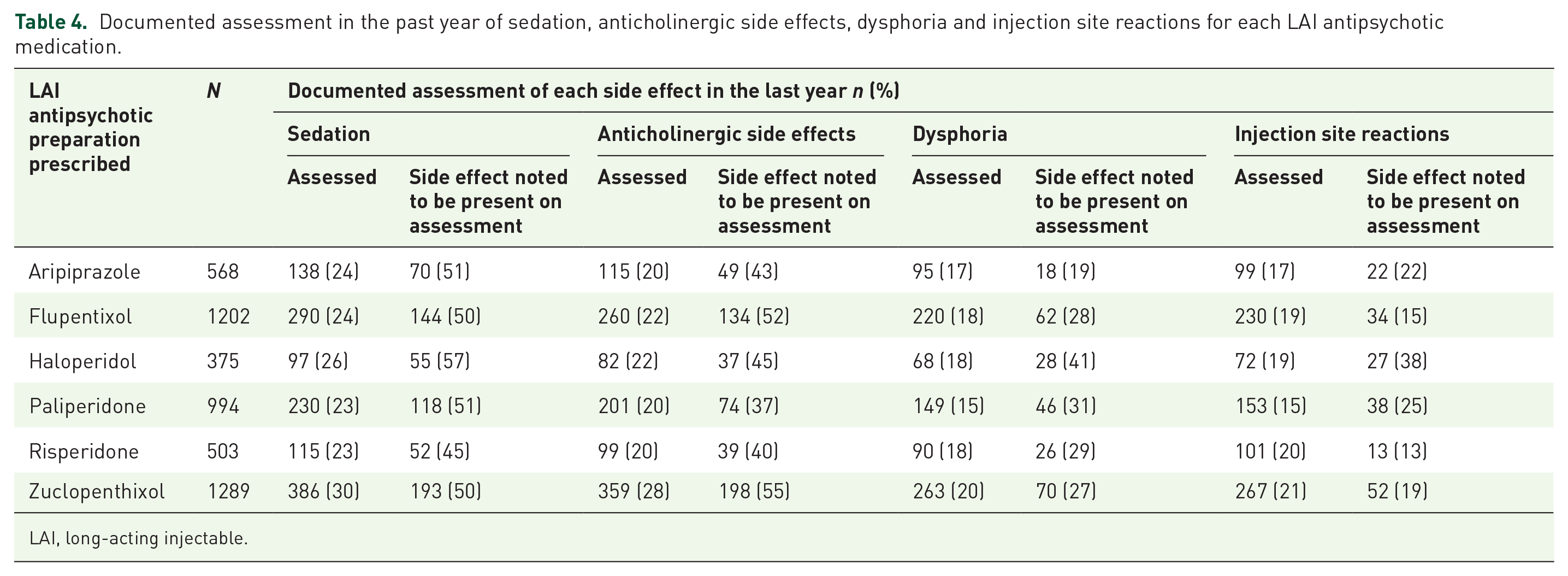
Documented assessment in the past year of sedation, anticholinergic side effects, dysphoria and injection site reactions for each LAI antipsychotic medication.
LAI, long-acting injectable.
Discussion
While our data were collected for the purpose of quality improvement in mental health services, both the high proportion of eligible mental health services that participated and the large number of cases for whom data were submitted allow the findings to be taken as a reflection of current prescribing practice for LAI antipsychotic medicines in the UK. 14 We discuss below the possible clinical implications of our findings regarding the LAI antipsychotic regimens prescribed and the side-effect assessments documented.
Dosage and dosage intervals for LAI antipsychotic medications
The LAI SGAs aripiprazole, risperidone and paliperidone have relatively narrow licensed dosage ranges and a fixed frequency of injection; for example, LAI aripiprazole has a recommended dosage range of 300–400 mg at a fixed interval of 1 month and paliperidone (as Xeplion®) a recommended dosage range of 25–150 mg, again at a fixed interval of 1 month. We found that the vast majority of prescriptions for these LAI antipsychotic medications were consistent with the manufacturer’s recommendations.
The licensed dosage ranges for the LAI FGAs are considerably wider and the injection intervals more variable, for example LAI flupentixol is licensed to be administered at doses that range from 20 mg every 4 weeks, to 400 mg weekly, an 80-fold difference. Consistent with this, we found that a wide range of doses and dosage intervals were prescribed for LAI FGAs. It has been suggested that these wide dosage ranges are not supported by the available evidence and may lead to some patients receiving unnecessarily high doses. For example, it has been proposed that the optimal dose of LAI flupentixol is probably between 20 mg and 40 mg every 2 weeks 19 ; we found that one in five prescriptions for this LAI antipsychotic medication were for doses above this upper threshold, thus potentially leading to an increased side-effect burden for little additional therapeutic benefit. The evidence suggests that most of the common side effects of antipsychotic medication, including EPS, are dose-related.11,21
Assessment of side effects
Network meta-analyses of RCTs of oral antipsychotic medicines suggest that differences in side-effect profiles are likely to be clinically relevant.7–9 For example, haloperidol and flupentixol have been shown to be more likely to cause EPS, while risperidone and paliperidone are more likely to cause weight gain and a raised plasma prolactin level. But there are few RCTs yielding data on the relative side-effect liability of LAI antipsychotic preparations. One example is an RCT of LAI haloperidol versus LAI paliperidone for the maintenance treatment of schizophrenia. 22 In this study, side effects were assessed systematically and around two-thirds of patients in each arm reported side effects that were rated as moderate to severe. But the nature of these side effects differed between the study arms, with akathisia detected more often in those participants assigned to LAI haloperidol, while weight gain (a mean difference between arms of 10 kg at 2 years) and raised plasma prolactin level were much more prominent in those receiving LAI paliperidone. These findings are consistent with the conclusions reached by the meta-analyses of oral antipsychotic medications described above and, further, the small number of RCTs comparing oral and LAI formulations of the same antipsychotic medication have reported similar side-effect profiles in each arm. 23 Thus, it seems reasonable to conclude that the side-effect profile of a particular antipsychotic medication will be much the same whether administered as an oral or LAI preparation.
Assessment of EPS and prescription of anticholinergic medication
When those patients in our audit sample who were prescribed an LAI SGA medication were assessed for EPS, these problems were noted to be present in more than half of cases. Those patients prescribed an LAI FGA were more likely to be assessed for EPS, and these assessments were more likely to find such side effects present. These data suggest that EPS remain a common clinical problem with all currently available LAI antipsychotic medications. However, only 1 in 10 patients receiving the LAI SGAs aripiprazole or paliperidone were also prescribed anticholinergic medication compared with between a quarter and a third who were receiving an LAI FGA. Possible explanations for this finding are that the nature of EPS found with LAI SGAs may differ (for example, akathisia is known to generally respond poorly to anticholinergic medication) or that the EPS symptoms do not reach a threshold considered by clinicians and/or patients to be sufficient to require remedial action using anticholinergic medication. Another possible explanation is that anticholinergic medication may be more often prescribed prophylactically for patients on an LAI FGA as such medications are perceived by clinicians to carry a greater risk of inducing EPS.
Overall, fewer than half of those prescribed regular anticholinergic medication had a documented assessment of EPS in the previous year. While the prevalence of anticholinergic prescribing in the sub-groups who had and had not been assessed for EPS was similar for LAI SGAs, anticholinergic medication was prescribed more often in the absence of an assessment in those patients who received the LAI FGAs flupentixol or zuclopenthixol. Taken together, these data suggest that the need for continuing treatment with anticholinergic medication is not reviewed routinely, and this is particularly so for those patients who are prescribed either LAI flupentixol or LAI zuclopenthixol. Our findings prompt further speculation that careful monitoring of EPS in patients prescribed these LAI FGAs might allow anticholinergic medication to be discontinued in a proportion, perhaps one patient in four, potentially reducing the burden of anticholinergic side effects to the level seen with other LAI antipsychotic preparations.
Assessment of weight gain and metabolic side effects
Of the LAI antipsychotic preparations available, network meta-analyses suggest that paliperidone and risperidone are associated with the greatest liability for weight gain and aripiprazole and haloperidol the least.8,9 We found that there was a documented assessment of body weight in the last year for only one patient in four overall, with little variation in this proportion between different antipsychotic LAI medications. Of those patients assessed, weight gain was noted to be present in more than a third and, as expected, this proportion was slightly higher with LAI paliperidone and LAI risperidone. Aripiprazole is considered to be relatively weight neutral,8,9 and our finding that weight gain was present in two-fifths of patients prescribed this LAI medication in whom this side effect was assessed led us to speculate that, for some patients, evident weight gain may have been the reason for prescribing this particular medication rather than a consequence of such treatment. In the absence of data relating to how much weight had been gained, we cannot know if the frequency of occurrence of this side effect differed across the LAI antipsychotic medications prescribed, but the findings of relevant network meta-analyses would suggest that this is likely to have been the case.8,9
An assessment of metabolic side effects in the last year was documented for only one patient in five overall, with no apparent targeting of particular LAI antipsychotic preparations. Where the outcome of these assessments had been recorded in the clinical records, metabolic side effects were present in two patients out of every five patients treated with LAI haloperidol, one patient in three treated with LAI paliperidone, and one patient in four who received any of the other LAI antipsychotic medications. This apparent hierarchy of risk is consistent with the findings of the network meta-analysis of oral antipsychotic medications conducted by Pillinger et al. 9
Assessment of prolactin-related side effects
Only around one in five patients had a documented assessment in the last year of prolactin-related side effects – a lower proportion than had been assessed for EPS or weight gain. Prolactin-related side effects are rarely obvious to the observing clinician; assessment requires focussed questioning, supplemented, if necessary, by a test for serum prolactin level. We found that those prescribed LAI risperidone or LAI paliperidone were no more likely to have a documented assessment than those prescribed LAI aripiprazole, despite the known differences in liability for prolactin elevation between these medications: aripiprazole has minimal, if any, effect on prolactin while risperidone and paliperidone are most likely to cause clinically relevant increases in serum prolactin, with LAI FGAs sitting between these two extremes. 8 These known relative liabilities are clearly reflected in our findings, with prolactin-related side effects identified with LAI aripiprazole in one in six of those assessed and with LAI paliperidone in almost one in two. The relatively low level of such side effects identified with risperidone is unexpected; the most likely explanation is that LAI risperidone is often used in relatively low doses and the effect on prolactin level is dose-related.
Sexual side effects are commonly associated with antipsychotic medication, and while the aetiology is complex, elevation of serum prolactin is a likely contributory factor. 24 As with prolactin-related side effects, focussed questioning is required to identify any problems. We found that fewer than one patient in five had a documented assessment for sexual side effects in the last year and this proportion was similar across all LAI antipsychotic medications. Where an assessment had been documented, sexual side effects were more often found in those patients treated with LAI paliperidone, being identified in almost two patients out of every five.
Assessment of sedation
Around one in four patients had a documented assessment in the past year addressing the sedative effects of LAI antipsychotic medication, with some modest targeting of these assessments towards those patients prescribed LAI haloperidol or LAI zuclopenthixol. Overall, sedation was noted to be present in around half of the patients in whom it was assessed; a slightly higher proportion of those prescribed LAI haloperidol and a slightly lower proportion of those receiving LAI risperidone, but these differences were very modest. The relative liability for sedation noted in the network analysis by Huhn et al. was lowest for paliperidone followed by aripiprazole then flupentixol, with zuclopenthixol associated with considerably more sedation than all other antipsychotic medications. 8 This hierarchy does not seem to be apparent in routine clinical practice, perhaps because in some cases where sedation was evident the dose of medication was titrated to achieve the best balance between efficacy and this objectively evident and potentially disabling side effect.
Assessment of dysphoria
Dysphoria, an unpleasant subjective state, is known to be associated with medications that are dopamine antagonists and such an experience may be a disincentive to accept continuing treatment with LAI antipsychotic medication. There are relatively few comparative data, but those that do exist suggest that dysphoria is up to three times more common with FGAs than with SGAs and is particularly associated with potent dopamine antagonists such as haloperidol. 25 The presence of dysphoria can be determined only through spontaneous patient reports and/or direct focussed questioning. We found that fewer than one in five patients had been asked about dysphoria in the last year, with no targeting of those patients prescribed LAI haloperidol. However, where assessments had been documented, dysphoria was noted to be present in between one-fifth and two-fifths of patients, depending on the LAI antipsychotic preparation, and, as expected, was identified most often in those prescribed LAI haloperidol and least often in those prescribed LAI aripiprazole. The relatively low levels of assessment for dysphoria mean that it is therefore likely to go undetected in the majority of cases.
Injection site reactions
Fewer than 20% of patients in our study had a documented assessment of adverse injection site reactions in the past year, but where such assessments were available the prevalence noted with LAI FGAs overall was consistent with the findings of Jones et al. 26 These authors systematically assessed all patients receiving an LAI FGA in local clinics and noted injection site reactions in 17%. Their finding suggests that, in current clinical practice, injection site reactions may not always be noted in the clinical records, perhaps because they may sometimes be considered as inevitable.
We found that injection site reactions were most often noted with LAI haloperidol, followed by LAI paliperidone and LAI aripiprazole. This finding with respect to LAI haloperidol is also consistent with the study by Jones et al., 26 who reported a higher prevalence of injection site reactions in those receiving LAI haloperidol compared with other LAI FGAs. These authors also noted an association between injection site reactions and both the number of injections administered and the volume of each injection. LAI haloperidol is not available in a concentrated formulation and so doses at the upper end of the licensed range require an injection volume of 3 ml. Our finding that injection site reactions were also more often recorded in those patients receiving LAI paliperidone or LAI aripiprazole may be partly related to the deltoid administration of these LAI SGAs medications in some patients, an injection site that may be associated with increased risk, but is not licensed and is rarely used for LAI FGAs.
Conclusion
Fewer than half of the patients in our sample had a documented assessment of side effects in the previous year and, except for EPS, there was no clear targeting of assessments in line with the perceived relative liability of different LAI antipsychotic medications to cause particular side effects. Our data also suggest that side effects may be more likely to be assessed in routine clinical practice when there are clear observable signs such as tremor, weight gain or sedation. Some other adverse effects, such as prolactin-related, sexual and metabolic side effects, are silent in that focussed questioning and/or blood tests are usually necessary to identify their presence. In current clinical practice, the vast majority of these side effects are liable to go undetected, particularly in those patients prescribed LAI SGAs, who appear to be less likely to have a documented side-effect assessment.
The potential consequences of side effects remaining undetected include failure to anticipate the implications for physical health and to take remedial action, and confounding of the clinical assessment of the mental illness, where unrecognised side effects may be mistaken for symptom of psychosis. Patients who receive continuing treatment with an LAI antipsychotic will be seen regularly by a member of the clinical staff to administer this medication, although the potential opportunity this presents for systematic monitoring of side effects does not seem to be taken for the majority of patients. 6 Further, simple screening tools, completed by patients, are available, which ensure more systematic and comprehensive assessment of side effects. 27 Their routine use has the potential to improve the detection of side effects and allow remedial action to be taken where necessary.
Key points
Fewer than half of those patients who were prescribed continuing LAI antipsychotic treatment had a documented assessment of side effects in the previous year.
The potential consequences are that a proportion of side effects remain undetected, with failure to anticipate the implications for physical health and confounding the assessment of mental state.
With the exception of EPS, there was no clear targeting of assessments in line with the perceived relative liability of different LAI antipsychotic medications to cause particular side effects.
Side effects may be more likely to be assessed in routine clinical practice when there are evident features such as tremor, weight gain or sedation.
There was limited use of pragmatic side effect screening tools despite their potential to enhance the detection of side effects.
Limitations of the study
Our data relate to patients under the care of adult mental health services who had been treated with an LAI antipsychotic medication for more than a year, as the focus of our QI efforts was the long-term monitoring of the side effects of such medication. However, our findings cannot be generalised to other patient groups or other healthcare settings.
Our data should not be interpreted as robust evidence for the relative liability of individual LAI antipsychotic medications for particular side effects or overall side effect burden. This is partly because of indication bias (the choice of LAI antipsychotic preparation for any given patient being based on the perceived efficacy and tolerability of that preparation in the context of each patient’s clinical circumstances) and partly because of factors biasing how and why clinicians conducted the documented assessments of side effects.
The data were collected from clinical records based on entries made by clinicians and provided only limited objective measures of the quality of side-effect assessment.
Footnotes
Conflict of interest statement
CP and BM have nothing to declare. TJC reported honoraria and travel support from Sanofi, Otsuka and Janssen. TREB reported participation in scientific advisory boards for Lundbeck, Newron Pharmaceuticals and Gedeon Richter/Recordati and receipt of an honorarium for a lecture from Janssen.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The work of POMH is funded wholly by subscriptions from mental health services in the UK.
