Abstract
Introduction:
The Prescribing Observatory for Mental Health initiated a quality improvement (QI) programme on clozapine use in UK mental health services.
Methods:
Clinical audits conducted in 2019 and 2021.
Results:
Sixty-three participating NHS Trusts/healthcare organisations in 2019, and 61 in 2021, submitted treatment data for 6948 and 8155 patients, respectively. In both audits, high-dose and/or combined antipsychotic medications had been prescribed immediately before initiating clozapine in over a quarter of patients recently started on clozapine. In patients who were tobacco smokers and recently discharged from a smoke-free ward, the impact of the potential change in smoking status had been considered in the care plans of just under one-third in 2019 and just over a half in 2021. For community patients, their Summary Care Records (SCRs) included their clozapine prescriptions in 58% of cases in 2019 and 72% in 2021.
Conclusions:
Three QI issues were identified. (1) Antipsychotic regimens with limited evidence for efficacy in treatment-resistant schizophrenia were prescribed for over a quarter of cases before starting clozapine. Use of such strategies may delay clozapine treatment, potentially reducing the likelihood of a therapeutic response. (2) While anticipation of the consequences of a change in smoking status on plasma clozapine concentration following discharge from hospital showed improvement over time, even in 2021 it was not evident for nearly a half of relevant cases. (3) While inclusion of clozapine in the SCR also improved over time, even in 2021 it was missing for more than a quarter of community patients.
Introduction
The Prescribing Observatory for Mental Health (POMH) runs audit-based quality improvement (QI) programmes for UK mental health services. Audit data on aspects of pharmacological treatment are collected by participating healthcare organisations, principally NHS Trusts. The information collected allows for the assessment of performance against evidence-based practice standards, usually derived from relevant national guidelines, such as those generated by the National Institute for Health and Care Excellence (NICE) and the British Association for Psychopharmacology (BAP). However, in addition to the data directly related to the practice standards, selected contextual clinical data are collected. These data provide a broader picture of treatment and management and have the potential to improve the understanding of the variables that influence prescribing practice.
This article reports on the findings of two clinical audits, conducted in 2019 and 2021, as part of the POMH QI programme on the use of clozapine (a receptor antagonist: D2, 5-HT2, NE alpha-2) in UK mental health services (Prescribing Observatory for Mental Health, 2019, 2021). The methodology of the 2019 audit has been reported previously (Barnes et al., 2020a). Analysis of the data collected in both the audits identified possible areas for improvement in relation to the antipsychotic medication regimens prescribed before starting clozapine, the management of clozapine treatment during a change in smoking status and the inclusion of clozapine in a community patient’s Summary Care Record (SCR).
Method
As part of a POMH audit-based QI programme addressing the use of clozapine in UK adult mental health services, a baseline clinical audit was conducted in 2019 and a re-audit in 2021. All POMH member Trusts/healthcare organisations were invited to take part. For both audits, the same standardised, bespoke data collection tool was used by all the clinical services participating. The tool was designed to capture information from the clinical records related to clinical performance against seven evidence-based practice standards, which had been derived from the NICE Clinical Guideline CG178, ‘Psychosis and Schizophrenia in Adults: Prevention and Management’ (NICE, 2014) and ‘The Maudsley Prescribing Guidelines in Psychiatry’ (2018), 13th edition. Five of these practice standards referred to medication review and monitoring of physical health and adverse effects, and performance against these standards has been reported elsewhere (Barnes et al., 2020a). Another practice standard concerned the ‘off-label’ prescription of clozapine, and the remaining standard addressed the management of a change in tobacco smoking habit: ‘For patients started on clozapine as inpatients, there should be consideration of the implications for dosage of any change in smoking status on discharge’. In addition, contextual data relating to clinical practice were collected, including psychiatric diagnoses, age at diagnosis of schizophrenia, when clozapine was initiated, and the antipsychotic medication regimen prescribed immediately before clozapine treatment was started. Furthermore, when a patient was under the care of a community team, services were asked to check whether the SCR contained the information that clozapine was being prescribed.
In each participating Trust and healthcare service, data on clinical practice were collected by clinicians and clinical audit staff from the clinical records of a sample of patients who were prescribed clozapine and under the care of adult mental health services. These data were pseudonymous within the Trusts but submitted anonymously to POMH, using Formic Software (2016). The key fields in the data collection tool were mandatory, preventing incomplete submissions. Ethical approval was not required for such an audit-based QI initiative (Health Research Authority, 2017). The data were analysed using SPSS (IBM Corp., 2012).
Results
For the baseline audit in 2019, 63 participating NHS Trusts/healthcare organisations submitted data related to the use of clozapine in 6948 patients prescribed this medication under the care of adult mental health services. In the 2021 re-audit, 61 NHS Trusts/healthcare organisations submitted data on the treatment of 8155 such patients.
The clinical and demographic characteristics of the baseline and re-audit patient samples are shown in Table 1. The majority of patients in the baseline audit sample were male, between 26 and 55 years of age, and under the care of a community mental health team. The vast majority had a diagnosis of schizophrenia, with the most common comorbid psychiatric diagnoses being personality disorder and substance misuse. The clinical and demographic characteristics of the patients in the re-audit sample are very similar.
Demographic and clinical characteristics of the patients in the baseline audit (2018) and re-audit (2021) samples.
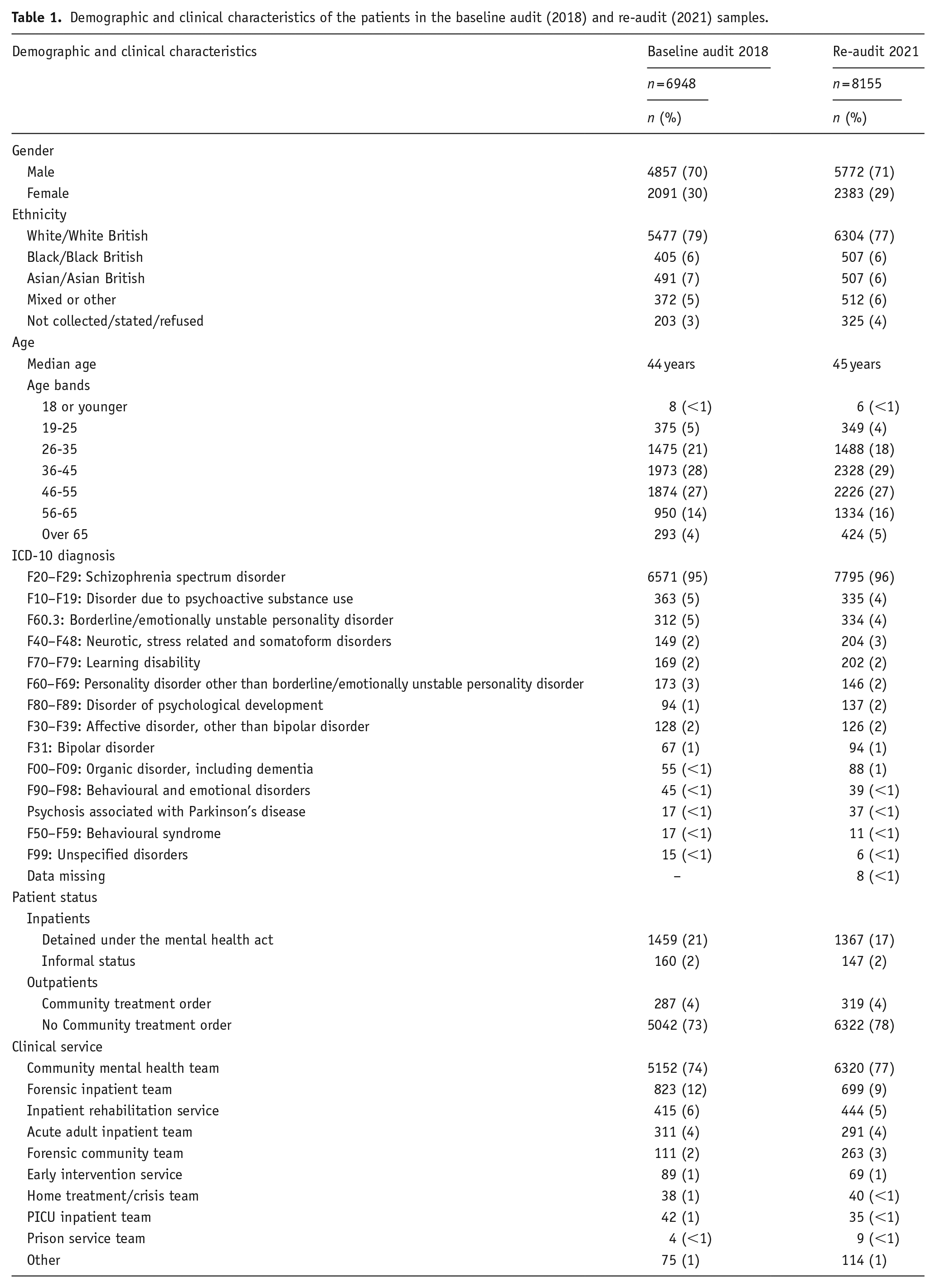
Of the 6571 patients in the baseline audit who had a diagnosis of schizophrenia, data relating to the age at which this diagnosis was made and the date that clozapine treatment was started were available for 3849 (59%). For the 7795 patients at re-audit, these data were available in 4393 (56%). In both these subsamples, the median age at diagnosis was 25 years, half had been diagnosed between 21 and 32 years of age, and the median time from the initial diagnosis of schizophrenia to the initiation of clozapine was 4 years (interquartile range 1–10 years at baseline and 1–9 years at re-audit).
Antipsychotic regimen before starting clozapine
In the subsample of patients treated with clozapine for less than 18 weeks (481 patients in the baseline audit and 396 in the re-audit), information was collected on the antipsychotic regimen prescribed immediately before clozapine was started. Regarding the route of administration, the majority (57%) of patients had been prescribed an oral antipsychotic medication only, whereas an LAI antipsychotic medication had been prescribed for 21% of patients, and a combination of oral and LAI antipsychotic medication for 13% of patients. The remaining 9% of patients had not been prescribed any antipsychotic medication. These percentages were the same in the two audits. Almost two-thirds of the patients in this subsample had been prescribed antipsychotic monotherapy within the licensed dosage range (Joint Formulary Committee, 2021), 65% in the baseline audit and 63% at re-audit, whereas more than one antipsychotic medication was prescribed for 21% and 23%, respectively, and a single antipsychotic in high dose for 6% and 5%, respectively.
Change in smoking status
In the subsample of patients treated with clozapine for between 18 weeks and 1 year (599 patients in the baseline audit and 458 at re-audit), 47% and 50% of patients, respectively, were documented as tobacco smokers. In the baseline audit, of the 77 smokers who had been discharged from a smoke-free ward, the implications of a potential change in smoking status on plasma clozapine concentration, and consequent adjustments to clozapine dose, had been considered in the care plans of 25 (32%). At re-audit, 53 smokers had been discharged from a smoke-free ward and the implications of a potential change in smoking status had been considered in 28 (53%). In the baseline audit subsample, clozapine was included in 58% of the SCRs, whereas at re-audit the respective proportion was 72%.
Accuracy of the Summary Care Record
In the subsample of patients treated with clozapine for more than a year who were under the care of a community mental health team (3,902 patients in the baseline audit and 4,738 at re-audit), information was collected about whether their SCRs (NHS Digital, 2020) identified clozapine as a currently prescribed medication.
Discussion
The similarity between the demographic and clinical characteristics of the baseline audit and re-audit patients may partly reflect the presence of some of the same patients in the subsamples treated with clozapine for more than a year, given that clozapine is typically a relatively long-term treatment. However, as the two audits were conducted 18 months apart, and the early treatment sample was defined by a clozapine treatment duration of less than 18 weeks, there would have been minimal, if any overlap in the early treatment subsamples. Overall, the comparability of these two large audit samples suggests that the information collected provides a representative picture of current clozapine prescribing practice in the UK.
Trusts participating in this POMH QI programme were provided with a customised report after each audit, showing their local performance data benchmarked against the performance of other participating mental health services and the total sample. They were asked to review these reports, identify areas where they fell short of best practice and consider appropriate QI initiatives. We report here on three clinical QI issues that arose from the data collected, which related to, first, the nature of the antipsychotic regimen immediately before clozapine treatment is started, secondly, anticipation of the impact on plasma clozapine concentration of a change in smoking status and, thirdly, the need to check that, for each community patient, the SCR contains the information that clozapine is currently prescribed.
Antipsychotic regimen before starting clozapine
Details of the antipsychotic regimen prescribed immediately before initiating clozapine were collected for those patients who had started clozapine relatively recently, to maximise the reliability of the data. At both the baseline audit and re-audit, just over half of the patients had been switched from other oral antipsychotic medication to clozapine, most often a single antipsychotic in a standard dose, but just over a quarter had been switched from high-dose antipsychotic medication or combined antipsychotic medications. Whether high-dose/combined antipsychotic regimens have any therapeutic superiority for treatment-resistant schizophrenia remains uncertain, but their routine use is not supported by current clinical guidelines (Barnes et al., 2020b; NICE, 2014). The relatively common use of these strategies in patients with refractory symptoms may partly explain the delay of several years commonly seen in clinical practice between the treatment-refractory nature of the illness becoming evident and the initiation of a trial of clozapine (Howes et al., 2012; Stokes et al., 2020; Taylor et al., 2003). The baseline and re-audit findings are consistent with this in that the median time from the initial diagnosis of schizophrenia to starting clozapine treatment was 4 years. There is evidence to suggest that the longer treatment with clozapine is delayed, following a diagnosis of treatment-resistant illness, the lower the likelihood of response (Üçok et al., 2015; Yoshimura et al., 2017).
Change in smoking status
The hepatic enzyme CYP1A2 is a major metabolic pathway for clozapine, and this enzyme is induced by the polycyclic aromatic hydrocarbons in tobacco smoke, rather than nicotine. Thus, people who smoke tobacco tend to have lower plasma concentrations of clozapine for any given dose administered than those who do not (Desai et al., 2001; Kroon, 2007). For example, the data analysed by Rostami-Hodjegan et al. (2004) suggest that female non-smokers will have a mean plasma concentration of 350 ng/ml with a daily clozapine dose of 265 mg, whereas female smokers will require 435 mg to achieve the same plasma concentration. Similarly, male non-smokers will have an average plasma clozapine concentration of 350 ng/ml with a daily dose of 325 mg, whereas male smokers will require 525 mg to achieve the same plasma concentration. But the confidence intervals around these mean values are wide, making it impossible to accurately predict for any individual patient the change in plasma clozapine concentration consequent upon an alteration in their smoking habit. A systematic review and meta-analysis by Wagner et al. (2020) concluded that plasma clozapine concentration was reduced by around one-third in smokers compared with non-smokers, although heterogeneity across the study outcomes was high. Overall, these findings strongly support the need for monitoring of plasma clozapine concentration and assessment of mental state following a change in smoking status (Qurashi et al., 2019; Wagner et al., 2020).
Specifically, close monitoring is indicated for any smoker prescribed clozapine who is discharged from a smoke-free ward. If such a patient were to re-start smoking but stay on the same dose of clozapine, it is likely that their plasma clozapine concentration would be significantly lower than the concentration achieved while in the smoke-free environment, and potentially down to a sub-therapeutic concentration. Therefore, the discharge care plan should outline the steps that should be taken to monitor any change in smoking status and the implications this may have for the dose of clozapine that is prescribed. In the baseline audit, such a plan was in place for less than one-third of smokers discharged from a smoke-free ward. At re-audit, the respective proportion was just over half, but that still meant that almost half of such patients might be at an increased risk of relapse should they resume smoking tobacco after leaving hospital. Wherever applicable, participating trusts were asked to review their clinical pathways for patients who are smokers and prescribed clozapine.
Summary Care Record
For each patient registered with a general practitioner, key clinical information is recorded in an SCR, which is populated from the clinical records generated in primary care as well as any correspondence received from hospital specialists. The SCR can be accessed by healthcare professionals, such as doctors in Accident and Emergency departments, to inform their clinical assessment and treatment plan. If a patient’s general practice records have inaccurate or missing information about their current medication regimen, specifically whether clozapine is prescribed, this could have serious implications for clinical management and monitoring (Parker and Somasunderam, 2010). For example, if clozapine were not documented in a patient’s SCR, the presentation in primary care or an acute medical setting of clinical symptoms suggestive of agranulocytosis, myocarditis or severe constipation might not be associated with clozapine treatment, or an interacting medicine might be prescribed, all of which could lead to a poorer outcome for the patient.
Across the two audits reported in this article, the proportion of patients under the care of a community mental health team for whom the SCR included clozapine increased markedly, from just over half to nearly three-quarters. It is uncertain to what extent this improvement might be attributable to a raised awareness of the issue in participating NHS Trusts/healthcare organisations prompted by receipt of their customised baseline audit report and the QI suggestion that any local gaps in the SCR should be discussed with primary care. But there is some evidence to suggest that the feedback regarding local SCR data may have encouraged some healthcare organisations to instigate processes to try and improve the accuracy of the SCR (Evans et al., 2019) or to boost established initiatives in this area (Butterworth and Shah, 2021; Pan Mersey Area Prescribing Committee, 2019). Participating Trusts were asked to reflect on the potential causes of any of their patients not having clozapine included in the SCR, such as a failure to notify primary care that clozapine was prescribed or a failure of primary care to upload this information onto the SCR because the medicine was supplied by mental health services.
Footnotes
Acknowledgements
Thanks are due to all the clinicians and clinical audit staff from the UK mental health services who collected and submitted data as part of this quality improvement initiative, and to Gavin Herrington, the POMH-UK programme manager.
Author contributions
TREB, CP, JMK and JHM developed the audit tools and interpreted the data. TREB, CP, MK and OD organised the audit, and MK and OD led the data analysis. All authors drafted and/or critically revised the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TREB has been a member of an advisory board for Gedeon Richter. JHM has received research funding and hospitality from H Lundbeck. JMK has received consulting fees from Alkermes, Allergan, Dainippon Sumitomo, H Lundbeck, Intra-Cellular Therapies, Janssen Pharmaceuticals, LB Pharmaceuticals, Merck, Minerva, Neurocrine Biosciences, Otsuka, Reviva, Roche, Saladex, Sunovion, Takeda and Teva, has ownership interest in LB Pharmaceuticals, Vanguard Research Group and North Shore Therapeutics, has received royalties from Up to Date, has received honoraria for lectures from Dainippon Sumitomo, H Lundbeck, Janssen Pharmaceuticals, Otsuka, Saladex and Teva and has received grant support from Janssen, H Lundbeck, Otsuka and Sunovion. CP, MK and OD have no interests to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The Prescribing Observatory for Mental Health is funded solely through subscriptions from member healthcare organisations (principally NHS Trusts).
