Abstract
Chronic neuropathic pain (CNP) is a disabling medical condition that impairs the health-related quality-of-life of affected patients. A high prevalence of anxiety, depression, sleep disturbance and cognitive impairment has frequently been reported in association with CNP, making the management of this disease complex and often multidisciplinary. Dual-acting agents such as selective serotonin and noradrenalin reuptake inhibitors (SNRIs) are considered particularly useful in the modulation of pain and in treatment of the mood disorders frequently associated with CNP. Recent evidence suggests that the top-down inhibitory control of pain involves the engagement and enhancement of descending endogenous opioidergic, cannabinoid and serotonergic systems, with the effect of serotonin being particularly related to the receptor subtypes that are preferentially activated; indeed serotonin induces analgesia via activation of 5-HT7 receptors and hyperalgesia via activation of 5-HT3 receptors. Vortioxetine (VO) is a novel multimodal serotonergic antidepressant with a unique mechanism of action. It has been demonstrated recently in experimental and clinical studies to have efficacy on pain hypersensitivity and on mood disorders. This drug inhibits the serotonin transporter with a high affinity, antagonises the 5-HT3, 5-HT1D and 5HT7 serotonin receptors, and activates the 5-HT1A and 5-HT1B receptors. In clinical studies, VO has proved effective at a dose of 10–20 mg/daily in short- and long-term treatment of patients with chronic orofacial pain, demonstrating a higher rate of clinical response and remission, a better acceptability, safety rate and tolerability, and a lower latency of action compared with other antidepressants. In the light of these recent findings, VO may be considered as a new pharmacological treatment also in relation to various types of CNP, particularly in elderly patients with concomitant mood disorders and cognitive impairment. The purpose of this review is to provide an up-to-date overview of the pharmacology and clinical applications of VO and to highlight its potential therapeutic properties and advantages in the management of CNP.
Introduction
Chronic pain is one of the most widespread disorders throughout the world, with almost 18% of the general population in developed countries affected. 1 Despite its high estimated prevalence, chronic pain management is still not completely satisfactory, probably due to the variety of chronic pain conditions with different etiologies (neuropathic, visceral, musculoskeletal and cancer-related) and due to the pathophysiological mechanisms, which are only partially known.2,3
Neuropathic pain (NP) has been defined by the International Association for the Study of Pain (IASP) as ‘pain caused by a lesion or disease of the somatosensory nervous system’. 4 Patients typically experience a distinct set of symptoms, such as burning and electrical-like sensations, and pain resulting from non-painful stimulations (such as light touching) 5 ; the symptoms persist and have a tendency to become chronic and respond with increasingly less effectiveness to pain medications. 5
Despite the development of knowledge in relation to this topic, chronic neuropathic pain (CNP) continues to be a challenge for healthcare providers. 5 Indeed, CNP is an extremely complex phenomenon, which is composed of both a perceptive and an emotional component and is therefore often associated with anxiety, depression and sleep disturbance.6 –8 The coexistence of psychiatric comorbidities may aggravate the CNP symptomatology, which, if left untreated, contributes to worsen the quality-of-life of affected patients.9,10 Therefore, in recent years, CNP management has been focused not only on the modulation of pain but also on the treatment of potentially associated mood disorders and sleep disturbance. 5
In this context, the use of antidepressants (ADs) has been considered a valid therapeutic option in the treatment of CNP and healthcare providers have begun to use these drugs outside their regulatory approval, adopting an off-label regimen.11,12 Indeed, psychotropic drugs, particularly the selective serotonin and noradrenaline reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs) and selective serotonin reuptake inhibitors (SSRIs), which act as central neuromodulators, have been demonstrated to be effective in relieving pain and in improving anxiety, depression and quality of sleep.12 –17 However, the use of SSRIs and SNRIs and TCAs may be complex due to the difficulty in managing several potential side effects (for instance sleep disturbance, weight gain, QTc prolongation and sexual dysfunction), especially in elderly patients. 18 The incidence of such side effects may, in turn, impair the patient’s adherence to the treatment and undermine the achievement of the treatment goals. 19
Vortioxetine (VO) is a novel multimodal serotonergic antidepressant, approved in the last decade in many countries worldwide for the treatment of major depression.20,21
VO inhibits serotonin (5-HT) transporter (SERT), similarly to commonly used ADs, but, differently, it directly modulates the activity of 5-HT receptors. 21 Due to its peculiar pharmacological profile, VO was recently classified among ‘the other antidepressants’ in the World Health Organisation (WHO) ATC/DDD Index 2018. 22
Recent experimental and clinical studies have shown that VO exerts antidepressant and pro-cognitive activities and that it is also effective in modulating pain hypersensitivity.23,24 Indeed, the efficacy of VO in the treatment of CNP is produced through the enhancement of the serotonergic transmission, the simultaneous inhibition of the 5-HT3 receptors and the modulation of the 5-HT7 receptors.25,26 In this context, VO may cause analgesia mediated by the 5-HT7 receptors and hyperalgesia mediated by the 5-HT3 receptors.23,27 Moreover, VO has exhibited a good tolerability profile, particularly in the elderly, since it does not affect cardiovascular, endocrine parameters and body weight. 24
In light of the recent evidence about this promising new psychotropic drug, the aim of this review is to provide an up-to-date overview on the pharmacology and clinical applications of VO and to highlight its potential therapeutic properties and advantages in relation to CNP management.
Mechanism of action
Pharmacological and receptor binding profile
VO shows new and unique multimodal pharmacological properties. Indeed, it acts as an inhibitor of SERT, sharing this mechanism with SSRI, SNRI and TCA, as well as direct modulator of the activity of multiple 5-HT receptor subtypes.26,28
VO works as a modulator and stimulator of 5-HT, it also exerts its activity on the neurotransmission of the noradrenergic, dopaminergic, cholinergic, histaminergic, glutamatergic and gamma-aminobutyric acid (GABA)ergic systems in the relevant brain areas.29,30
The binding affinities and functional activities of VO in rats are generally similar to those in humans; however, its affinities for the 5-HT7 and 5-HT1A receptors in rats are 10- to 15-fold weaker at comparable doses, suggesting that the action of VO towards these receptors could be stronger in human subjects. 26
In humans, VO inhibits SERT with a high affinity (Ki = 1.6 nM), increasing the levels of 5-HT in the post-synaptic space, similarly to other antidepressants. However, it is the only drug that can modulate 5-HT receptor activity directly, being a full agonist of 5-HT1A (Ki = 15 nM), a partial agonist of 5-HT1B (Ki = 33 nM), and an antagonist of the 5-HT3 (Ki = 3.7 nM), 5-HT7 (Ki = 19 nM) and 5-HT1D receptors (Ki = 54 nM). 25 However, in rodents the binding affinity of VO is as follows: SERT (Ki = 8.6 nM); 5-HT1A (Ki = 230 nM); 5-HT1B (Ki = 16nM); 5-HT3 (Ki = 1.1nM); 5-HT7 (Ki = 200 nM); 5-HT1D (Ki = 3.7 nM). 26 Previous experimental studies have highlighted that the antagonism towards the three 5-HT receptors leads to an increase in 5-HT levels and, as a consequence, enhances the serotonergic neurotransmission in the different forebrain regions.31–33
Besides, the affinity of VO for the β1 adrenergic receptor is relevant only in the context of its side effects. 29 When tested against a panel of 70 other G-protein-coupled receptors (GPCRs), transporters, enzymes, ion channels and kinases, VO displayed no pharmacologically relevant activity. 26
VO’s binding affinity is dose proportional. Experimental clinical studies have shown that VO engages, preferentially, SERT and 5-HT3 at a lower dosage, between 5 and 10 mg, and engages all targets at a higher dosage of 20 mg. 29 In positron emission tomography (PET) studies in healthy volunteers, VO at doses of 5, 10 and 20 mg/day demonstrated dose-dependent SERT occupancy rates in the brain, namely 50%, 53–65% and 80%, respectively, for each dose with EC50 values ranging from 4.2 to 6.5 ng/ml.25,34
The rank order potency of VO is as follows: 5-HT3 > SERT > 5-HT1B > 5-HT1A = 5-HT7. 26
VO in the treatment of mood disorders, cognitive impairment and sleep disturbance
Table 1 shows the multimodal mechanism of action of VO.
Multimodal mechanism of action of VO.
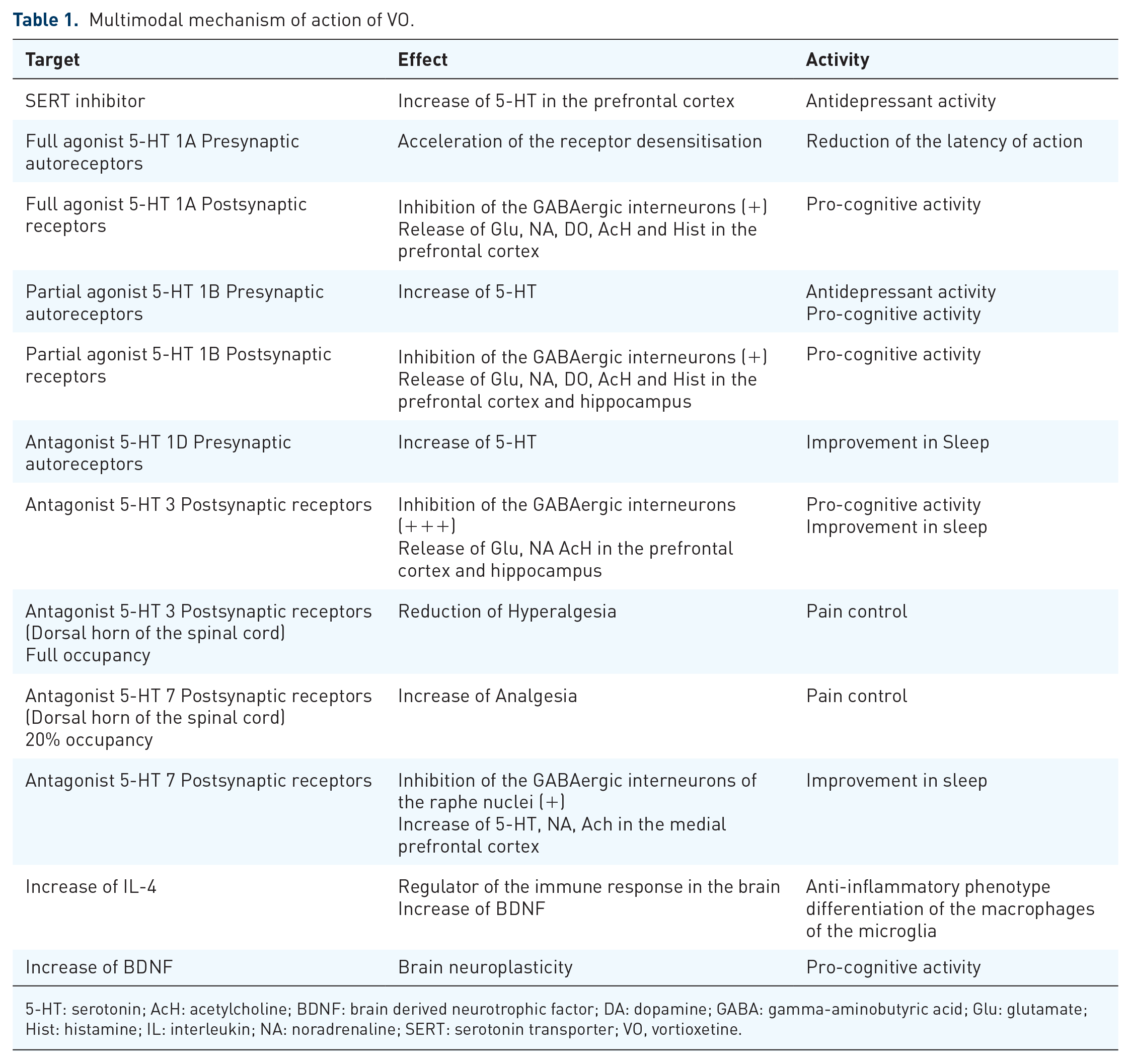
5-HT: serotonin; AcH: acetylcholine; BDNF: brain derived neurotrophic factor; DA: dopamine; GABA: gamma-aminobutyric acid; Glu: glutamate; Hist: histamine; IL: interleukin; NA: noradrenaline; SERT: serotonin transporter; VO, vortioxetine.
Experimental and clinical studies suggest that VO might exhibit simultaneously antidepressant, anxiolytic and pro-cognitive activities. 24 Indeed, the blocking of SERT causes a wide release of 5-HT that stimulates the presynaptic somatodendritic inhibitory 5-HT1A receptors of the raphe nuclei, creating a negative feedback loop that reduces, at the beginning, the antidepressant effect of the SSRIs and SNRIs. 35 Differently, VO beyond its action on SERT, works as an agonist of these receptors and accelerates the desensitisation and disinhibition caused by the 5-HT release, contributing to a reduction in the latency of action of this drug compared with other ADs.36,37
In addition, the antagonism of the 5-HT7 further enhances serotonergic transmission with a synergistic antidepressant effects. 38
Moreover, VO is an antidepressant with a pro-cognitive effect, independently from any improvement in mood. 24
In animal models, acute, sub-chronic and chronic administration of VO exerts positive effects across a range of cognitive tasks. VO achieved enhancements in acquisition and retention of contextual fear memory, object recognition memory and visuospatial memory in rats and mice.39–42
In humans, VO has demonstrated cognitive enhancing properties in seven short-term randomised controlled trials (RCTs) (6–12 weeks) ,43–52 three meta-analyses,45,47,53 and one open-label trial, 54 with significant improvements in processing speed, executive control, verbal learning and recall domain in young and elderly patients with moderate to severe depression. 55 These improvements, independent from the alleviation of depressive symptoms, represent a direct effect of VO and have been reported at doses across the clinically relevant dose range (5–20 mg/day) without a dose–response effect, suggesting that VO exerts its procognitive and antidepressant actions via separate mechanisms. 55
The pro-cognitive activity of VO is related mainly to the blocking of the 5-HT3 receptors located in a subset of the GABAergic inhibitory interneurons, with a subsequent reduction in the GABAergic transmission, which can in turn disinhibit pyramidal neuronal activity and increase glutamatergic neurotransmission, long-term potentiation (LTP) and neuroplasticity in the prefrontal cortex and in the hippocampus.27,56 Moreover, the partial agonism toward the 5-HT1B receptors increases the release of 5-HT, glutamate, acetylcholine and histamine, indirectly of dopamine and noradrenaline, particularly in the prefrontal cortex and hippocampus, while the antagonism of 5-HT7 increases the acetylcholine and noradrenalin levels in the medial prefrontal cortex.25,27 The stimulation of cholinergic and histaminergic neurotransmission may further contribute to the pro-cognitive effect of VO.55,57
The enhancing effect on noradrenergic neurotransmission is related not only to a partial agonism of 5-HT1B but also to the stimulation of the 5-HT1A and the blocking of the 5-HT3 receptors. 58 Similarly, the indirect selective increase in the levels of dopamine in the frontal cortex and hippocampus is related not only to the effect on the 5-HT1B receptors but also to the agonism of the 5-HT1A receptors, which are known to increase extracellular dopamine upon stimulation. 59 It has been demonstrated that VO considerably increases only the extracellular 5-HT levels in the nucleus accumbens in all the doses tested, without significantly affecting the noradrenalin and dopamine levels. 26 Furthermore, recent studies have suggested that VO, like monoaminergic ADs, is able to promote the synaptic neuroplasticity of the brain.56,60,61 Indeed, in experimental studies, VO can increase the levels of the brain-derived neurotrophic factor (BDNF), promoting neurogenesis, dendritic branching and dendrite spine maturation and forming functional synapses by mitochondrial support in the hippocampus dentate gyrus.62,63 Interestingly, this action was detected only after 1 day of treatment with VO, in contrast to fluoxetine, which induced similar effects after 7 days. 64 In addition, VO has proved to induce maturation of immature neurons by increasing dendritic length and the number of dendritic intersections in the dentate gyrus. 65 All these actions work in a synergistic way, leading to an overall enhancement of brain performance, particularly important in elderly patients where cognitive impairment could be an aggravating factor for depression and CNP. 55
VO has also been demonstrated to improve the quality of sleep by enhancing non-REM sleep and increasing slow-wave sleep (through its agonism on the 5-HT1A and antagonism on the 5HT-3 receptors) and by suppressing REM sleep (through its antagonism on the 5HT-7 and 5HT-1D receptors). 36 This effect should be considered carefully in the management of CNP where sleep disturbance is frequently reported. 66
VO in pain modulation
The mechanisms explaining how VO modulates pain are still not fully understood but it seems that the analgesic effect could be independent of its effect on depression and anxiety although the improvement in mood may potentially ameliorate the experience of pain and the ability to cope with pain.67,68
Recently, in two experimental studies VO has demonstrated its efficacy in modulating pain in two models of CNP.23,69
In the study of Zuena et al., VO venlafaxine and fluoxetine (all at 10 mg/kg, p.o.) were compared in modulation of pain in an established mouse model of NP.
VO has demonstrated a strong analgesic action in the chronic constriction injury model of NP, with an effect identical to that produced by venlafaxine, but different to that exhibited by fluoxetine. In detail, fluoxetine treatment in CCI mice had no effect on induced tactile allodynia on mice; instead, venlafaxine or VO caused a robust analgesia that was initially observed at day 7 and became substantial at day 12 of the treatment. 69
Subsequently, in the study of Micov et al., 23 VO induced analgesia in an oxaliplatin-induced neuropathy model in mice. The drug (at dosage of 1–10 mg/kg, p.o.) reduced significantly, in a dose-dependent manner, not only the mechanical allodynia in the von Frey test and the cold allodynia in the acetone test but also depressive-like behaviour in the forced swimming test in the tested mice. In this study, the analgesia produced by VO was found to be similar to that produced by duloxetine (at dosage of 1–15 mg/kg, p.o.), despite the difference in the mechanism of action between these two drugs. In detail, the maximum effect of VO was achieved from the 7th to 10th day after oxaliplatin injection and the effects were 60%, 71% and 88% for doses of 1, 5 and 10 mg/kg, respectively. The maximum effect of duloxetine was achieved from day 7 to day 14 after oxaliplatin injection. The effects were 40%, 62% and 81% for doses of 1, 5 and 15 mg/kg, respectively. 23
Despite the limited number of studies in this field, these results have suggested that VO could potentially be used in the prevention and treatment of chemotherapy-induced neuropathy, and also of other types of CNP, considering that, until now, duloxetine is currently the only drug of choice for the treatment of this condition.23,69
VO exhibits several actions that might explain its role in pain modulation. First, the blocking of SERT results in the up-regulation of biogenic amine neurotransmitters such as 5-HT and noradrenalin in the synaptic cleft of the central and peripheral nervous system, which can modulate pain transmission. Secondly, the direct modulation of receptor activity contributes to the allodynic action of the drug.70,71
Therefore, the increase in the 5-HT level seems to be a consequence of the SERT inhibition empowered by the 5-HT3 receptor antagonism and 5-HT1 A receptor stimulation, as each of these actions produces a greater 5-HT elevation than SERT inhibition alone. 72 In addition, in vivo studies on rats have demonstrated that the antagonism of the 5-HT3 receptors increases the noradrenalin levels in the hippocampus and that the agonism of the 5-HT1A receptors increases the noradrenalin levels in the hypothalamus and hippocampus.73,74
The increase of these neurotransmitters, especially 5-HT, and the downregulation and desensitisation of the pre- and post-synaptic receptors of the spinal dorsal horns over time contribute to a reduction in the ascending pain signals to the central nervous system (CNS). 75 Less time is needed for the desensitisation for VO compared with the SSRIs and SNRIs on account of its unique selective agonism toward the serotonergic receptor 5-HT1A. 36
In addition, the synergic increase of the levels of 5-HT, noradrenaline and dopamine in the CNS contribute to strengthening the function of the descending inhibitory system, which can regulate the ascending nociceptive pathway.76,77
Indeed, the descending inhibitory fibres that originate in the brainstem and terminate in the spinal dorsal horn can be either inhibitory or facilitatory of pain; these fibres suppress pain neurotransmission working through the release of 5-HT and noradrenaline. 78 Therefore a dysfunction of these systems can cause a dysfunction of the descending 5-HT and noradrenaline anti-nociceptive pathways and can exacerbate the chronic pain condition. 79
In condition of CNP, the serotonergic pathway descending from the lower brainstem to the dorsal horn of the spinal cord are hyperalgesic and this effect is mediated by the activation of 5HT3 receptors. 80 In detail, the top-down inhibitory control of pain is mediated by the 5-HT effect on the receptor subtypes that are preferentially activated, particularly the 5-HT induced analgesia mediated by the 5-HT7 receptors and the hyperalgesia mediated by 5-HT3. 69 Indeed, in rodents with peripheral neuropathy, the administration of the 5-HT3 antagonist resulted in a pain-suppressing effect, alleviating mechanical hyperalgesia and tactile allodynia.69,81 Therefore, in the experimental study of Zuena et al., 69 VO can reduce hyperalgesia, through blocking of the 5-HT3 receptors and the improvement in analgesia through enhancement of 5-HT transmission (despite blocking of 5-HT7 receptors). In fact, at a therapeutic dosage of 20 mg, VO saturates all the 5-HT3 receptors and blocks only 20% of the 5-HT7 receptors in rodents, 82 leaving the majority of the 5HT-7 receptors free and subsequently preserving analgesia mediated by these receptors.27,69
This further allows an increase in the 5-HT level, which, in turn, improves the descending modulation pain pathways. 80 On the basis of these findings, we hypothesise that 5 HT3 and 5-HT7 receptors could be fully occupied and partially occupied, respectively, at the dosage of 20 mg also in the human subjects, explaining why VO may potentially be effective as analgesic drugs in neuropathic pain.
VO may potentially be effective as analgesic drugs in neuropathic pain also in humans. However, this interesting hypothesis needs to be investigated further.
In addition, the modulation of 5-HT1A and 5-HT1B receptors could contribute to the anti-hyperalgesic action of 5-HT at the central and/or peripheral sites in CNP. 83 Indeed, the selective stimulation of the spinal 5-HT1A and 5-HT1B receptors decreases mechanical hypersensitivity and evokes the potential of the dorsal horn neurons in rats with peripheral nerve injury.23,84,85 However, the antagonism of the 5-HT1D receptors by VO is still much debated, considering that the role of these receptors in CNP modulation is rather inhibitory. 83
CNP is associated with structural and functional changes in the brain with an impaired connectivity of the hippocampus, amygdala and cortex. 86 Specifically, the hippocampus is a part of the brain limbic region critically implicated in modulating the emotional component of pain and is also particularly vulnerable to stress – its atrophy being a common finding in chronic pain and mood disorders. 87
In this context, the increase of the synaptic neuroplasticity of the brain induced by VO could play a pivotal role by reverting these brain alterations through neurogenesis. 88 Moreover, VO increases the level of the BDNF, restoring lost neurons, and potentially contributes to recovery from chronic pain.62,89–92
A growing body of evidence supports the hypothesis that an increased level of pro-inflammatory cytokines is crucial in the development of pathological pain; in experimental studies VO has been demonstrated to exhibit immunomodulatory properties, antioxidant activity and intrinsic anti-inflammatory effects. 93
This anti-inflammatory profile of VO is superior when compared with amitriptyline and S-citalopram. 94 Therefore, the analgesia induced by VO may also be explained in terms of a decrease of the neuroinflammation; this effect is mediated by modulation of microglia cells of the brain. 95
Indeed, the involvement of the microglia and astrocytes in some brain regions including the hippocampus, prefrontal cortex, amygdala, and nucleus accumbens has been considered in the pathogenesis of CNP.95–98
In normal circumstances, the microglia cells are resident inactive macrophages of the CNS (inactive or resting microglia) but after stress exposure and in CNP conditions the signalling between the microglia cells, astrocytes and neural cells is modified. Indeed, these nociceptive neurons can activate some subtype cells of the microglia (the M1 inflammatory phenotype) through the release of ATP chemokine and fractalkine.95,97
In turn, the activated microglial cells (M1) release proinflammatory cytokines such as tumour necrosis factor α (TNF α) and interleukin 1 β (IL-1β), which can contribute to the neuroinflammatory process, neuronal apoptosis and central sensitisation in chronic pain.99–101
Similarly, the activation of astrocytes mediates the neuroinflammation, induces neuronal hyperexcitability and plays a role in the maintenance of a status of chronic pain. 97
In contrast, the activation of another subtype of the microglia cells (the M2 anti-inflammatory phenotype) and astrocytes can promote an anti-inflammatory response, causing tissue repair and angiogenesis. 102
In this context, the modulation of communication among the microglia cells, astrocytes and neural cells with a concomitant shift of microglia from the M1 to the M2 phenotype may hold a key role in the improvement in pain perception, particularly in the hippocampus region.103,104
Recent experimental studies, have shown that VO could have a selective role in the maintenance of an anti-inflammatory status through the inhibition of the subtype of activated microglia (M1 phenotype) driving the shift versus an M2 anti-inflammatory phenotype, via 5-HT2b and 5-HT7 receptors, blocking neuroinflammation, enhancing neurogenesis and neuroplasticity.93,105
In addition, VO acts as an inhibitor of some subtypes of the activated hippocampal astrocytes, through the blocking of the 5-HT3 receptors expressed on the surface of these cells and increases the level of the vascular endothelial growth factor (VEGF), promoting a microvasculature of the hippocampus and in turn improving neuroplasticity. 106
Moreover, the blocks of the SERT, of the 5-HT3 and 5-HT7 receptors by VO contribute to further decrease the levels of the pro-inflammatory cytokines and subsequently neuroinflammation.107–109 In addition, the antagonism of the 5-HT3 receptors by VO has demonstrated a neuroprotective effect against oxidative stress.93,110
Dosage
VO is available as 5 mg, 10 mg and 20 mg tablets and drops. Doses of 5–20 mg were found to be effective, with doses of 20 mg exhibiting a greater clinical response. However, a lack of effectiveness was found at lower doses of 2.5–5 mg. 29
The recommended starting dose of VO is 10 mg/daily. However, dose adjustment should be considered on a patient-by patient basis until a clinical remission of the pain and an improvement of the mood disorders is achieved. Indeed, the progressive up-titration of the dosage until 20 mg/daily was proven to be efficacious and safe, as also suggested by several studies. 111 Nevertheless, the EU health authorities recommend initiating treatment in the elderly with a daily dose of 5 mg (and advise caution when prescribing VO at a dose of 10 mg/day), due to the fact that exposure to VO may increase by up to 27% in healthy volunteers aged 65 years as compared with those aged 45 years, who had received multiple doses of 10 mg/day. 112
Pharmacokinetics
The pharmacokinetic properties of VO have been studied in mice, rats, dogs, and in human subjects.30,112–116
In humans, the absolute bioavailability of VO, determined after intravenous and oral administrations to the same volunteers, was high (up to 75%) and independent of food intake. 117
VO shows a linear, dose-proportional and time-independent pharmacokinetics at doses from 2.5 mg to 75 mg. 118 It has an extensive volume of distribution as the plasma protein binding is 80–90%. It takes 3–16 h to attain maximum plasma concentration, with a terminal half-life of approximately 60–70 h, and a steady-state concentration in the plasma maintained for up to 2 weeks after drug administration. 112
VO is metabolised extensively in the liver, primarily by several cytochrome CYP450 enzymes (including CYP2D6, CYP3A4/5, CYP2C9, CYP2C19, CYP2A6, CYP2C8 and CYP2B6) with a subsequent glucuronic acid conjugation in pharmacologically inactive metabolites. In detail, CYP2D6 is the primary enzyme catalysing the metabolism of VO to its major metabolite (Lu AA34443), which is inactive. A second metabolite (Lu AA39835) is equipotent to VO as an inhibitor of human 5-HT transporter, but its concentration is much lower than that of the parent compound in plasma (i.e. metabolic ratio ⩽ 0.04), and it is not expected to have effects on the CNS based on a nonclinical pharmacology study. 119 Therefore, the clinical activity of the drug can be attributed solely to the parent compound. On account of its inability to inhibit or induce P450 enzymes, VO appears to have a relatively safe drug–drug interaction profile. 120
Intrinsic factors, such as age, sex, race, body size, and hepatic and renal impairment, had no significant effects on VO exposure, suggesting that no dosing adjustment is required for this drug.
However, as VO is a serotonergic agent, it should be used carefully in combination with other serotonergic drugs. 120 In particular, VO being a substrate for the cytochrome P450 enzyme CYP2D6 requires an adjustment of the dose when given in combination with CYP2D6 inhibitors (bupropion, fluoxetine or paroxetine). 119 In turn, co-administration with robust CYP inducers (carbamazepine, phenytoin or rifampicin) requires an increased dose of VO. 119
In addition, the co-administration of ketoconazole 400 mg/daily(CYP3A4/5 inhibitor) or fluconazole 200 mg/daily (CYP2C9, CYP2C19, and CYP3A4/5 inhibitor) with VO (10 mg/daily) increased the AUC(0–t) of the drug by 30 and 46%, respectively. However, given the modest increases, these findings were not considered clinically meaningful.119,121
Instead, the co-administration of ethanol, aspirin or omeprazole (CYP2C19 inhibitors) had no effect on exposure to VO.121,122
Safety, tolerability and drop-out
VO showed a favourable profile, both in short- and long-term clinical placebo-controlled studies, in terms of safety and tolerability at dosages of 5–20 mg. The most common adverse drug reactions (ADRs), reported in 5% of subjects, were, in order of incidence, nausea, headache, diarrhoea and dry mouth. It is well established that the most common ADR is nausea (affecting more than 1 in 10 people; 20.9/31.2%), generally with a mild or moderate severity.21,123,124 It has not been clarified how VO can induce this side effect, although nausea might be related to the serotonergic hyperactivation as consequence of the increase availability of 5-HT in the gastrointestinal tract and in the CNS or possibly to the genetic polymorphism of the monoamine oxidase A (MAOA-VNTR) which may influence the individual’s susceptibility toward this side effect.25,29 Generally, the nausea is usually transient, dose-related and more common in women and during the first week of treatment. 125
The incidence of haemorrhage in a short-term clinical study with a dose range of 10–20 mg/day was low, and similar between VO (1.7%) and placebo (1.2%) participants. 122
The good tolerability of VO has been demonstrated on account of the fact that no effects have been reported in the biochemical parameters, vital signs, body weight, heart rate, blood pressure or electrocardiogram parameters, including the QTcF interval. This aspect represents an important advantage of this drug, for both young and elderly patients, as it signifies an avoidance of any negative metabolic effects and related reasons for treatment discontinuation. 24
The incidence of sexual dysfunction was lower in relation to treatment with VO as compared with treatment with SSRIs. Interestingly, in a recent study this ADR was reported to be higher with a low dose of VO, namely 2.5 mg, compared with a 5 mg dose, with a higher incidence in men.126,127 In addition, switching patients already experiencing sexual dysfunction from previous antidepressants to VO led to a reduction in the incidence of sexual impairment while maintaining an adequate management of depressive symptoms. 128
On the other hand, VO has not been tested sufficiently in pregnant human subjects, but in animal studies decreased foetal weight and delayed ossification have been reported. 29
The sudden discontinuation of VO after 2 weeks of treatment showed no clinically significant withdrawal effects as compared with placebo (4.5–7.8% in the VO group versus 3.6% in the placebo-treated group). 129
Overall, VO has been demonstrated to be one of the most tolerable antidepressants, associated with the lowest rate of dropout, as suggested by a recent systematic review performed by Cipriani et al. 130
Clinical use
VO in the treatment of major depressive disorder
The efficacy of VO in the treatment of major depressive disorder (MDD) has been established in several short-term (6, 8 and 12 weeks) RCTs in adults at a dose range of 5–20 mg, where the higher doses were associated with increasing clinical effects.131–140
In RCTs in which active comparators were used, VO showed a significant superiority when compared with agomelatine and non-inferiority when compared with venlafaxina,132,141 escitalopram or duloxetine.43,131,142,143 The efficacy of VO has been confirmed also in a long-term clinical trial (52 weeks).144,145
Moreover, in patients who had exhibited an inadequate response to SSRIs or SNRIs before switching to VO, 146 this drug proved to be significantly superior to agomelatine in terms of reducing depressive symptoms in SSRI non-responders, 147 although not in SNRI patients. 141
A recent systematic review and network meta-analysis, which compared the efficacy and acceptability of 21 antidepressant drugs in the treatment of MDD, has shown that VO, together with agomelatine, amitriptyline, escitalopram, mirtazapine, paroxetine and venlafaxine, was more effective than other ADs. 130 These results are in line with previous meta-regression analyses performed by Llorca et al. and confirmed by Chen et al.121,148 Instead, in the study of Wagner et al., VO showed a similar efficacy to that of levomilnacipran and vilazodone. 149
In addition, VO showed the best tolerability profile,29,150 especially in patients of 65 years or older previously treated with SNRIs, and SSRIs, with a statistically lower number of overall ADRs compared with VO. 151 However, in a systematic review performed by Li et al., 152 VO was found to be less effective than duloxetine, even though patients treated with VO did not develop ADRs.
Despite the uncertainty about the choice of specific drug for the treatment of MDD, it is important to consider that patients treated with VO displayed an improvement in terms of a reduction of depressive symptoms, namely a full functional recovery and restoration of function in several domains of daily life activities after only 8 weeks of treatment, unlike treatment with placebo or duloxetine. 153 This result was confirmed by a meta-analysis of an elderly population where treatment with VO (15–20 mg) resulted in a significant improvement in the self-perception of physical health mental health and energy, and subsequently of quality-of-life, in MDD patients, compared with treatment with a placebo. 154
VO in the treatment of anxiety
The efficacy of VO in the treatment of generalised anxiety disorders (GAD) is still debated.
In the systematic review of Pae et al., 155 the authors suggest that VO may have a potential benefit in the treatment of GAD since VO was found to be more effective than placebo, particularly in patients with a severe form of GAD and with higher scores in the Hamilton anxiety rating scale (HAM-A) test. 156
However, in a recent systematic review and network meta-analysis of double-blind RCTs performed by Kong et al., 157 despite the tolerability of VO, its efficacy was comparable with that of placebo while the remission rate of agomelatine, duloxetine, escitalopram, paroxetine, quetiapine and venlafaxine was superior to that of placebo.
In relation to panic disorders and social disorders, preliminary results have shown a potential advantage in terms of efficacy in a long-term treatment with VO, despite no evidence about the optimal length of treatment and dosage having been reported.158–160
VO in the treatment of cognitive impairment
Several RCTs have demonstrated the efficacy of VO in terms of enhancing cognitive performance in patients with MDD – an effect considered independently of any alleviation of depressive symptoms. Indeed, the cognitive benefits of VO rely on its action on receptor activity, in particular 5-HT3 receptor antagonism.143,161–164
At a dosage of 10–20 mg, VO was superior to placebo in improving attention, memory, learning, processing speed and executive functions, especially in elderly populations. 143
These results were confirmed by a recent systematic review by Blumberg et al., 165 where only VO and bupropion have demonstrated pro-cognitive effects in patients with MDD whereas no effects were reported for tricyclic ADs, SSRIs and SNRIs.
Moreover, in a recent short-term (8 weeks) placebo controlled RCT, VO seemed to be efficacious both in monotherapy or as an adjunctive treatment to SSRI, also in the treatment of residual cognitive symptoms in patients affected by MDD. 51
VO in the treatment of sleep disturbances
VO showed efficacy in the treatment of sleep disturbance in preliminary results and in open label studies at a dosage of 10–20 mg. 166 Indeed, VO improved sleep quality, leading to a reduction in the scores of the Pittsburgh Sleep Quality Index, Epworth Sleepiness Scale and Insomnia Severity Index already after 8 weeks of treatment.166–168
Moreover, Wilson et al. analysed the sleep architecture in 24 healthy young men treated with VO (20 mg), VO (40 mg), paroxetine (20 mg) or a placebo. 169 All three active treatments significantly increased rapid eye movement (REM) onset latency and decreased the time spent in REM sleep.
VO in the treatment of CNP
Recently, in two pilot clinical studies, Adamo et al. tested VO in a sample of patients affected by chronic neuropathic orofacial pain (CNOP) with encouraging results, suggesting a new possibility in the management of these patients.111,168 Indeed, VO demonstrated efficacy in the relief of pain, anxiety, depression and sleep disturbance, improving the quality-of-life of the patients.111,168
No drug–drug interactions or side effects, such as QTc prolongation, sexual dysfunction or weight gain, were reported in these studies, in line with the findings from 11 double-blind RCTs on MDD. 170
These results were further confirmed in a long-term (12 months) RCT on 150 patients, where the same authors compared VO with four of the most frequently prescribed ADs in the treatment of CNOP: paroxetine, sertraline, escitalopram and duloxetine. 111 In this study, VO showed a faster-acting antidepressant activity and pain control compared with SSRIs and SNRIs. 111
In detail, VO and paroxetine showed the best effectiveness in reducing pain intensity, evaluated by the visual analogic scale (VAS), after 1 year of treatment (median scores at T0 were 10.0 and decreased to 0 after 12 months) compared with other antidepressants (sertraline, escitalopram, duloxetine) whose median scores after 12 months ranged from 1.0 to 3.0. Moreover, VO was the most efficacious in controlling pain quality evaluated by the total pain rating index (T-PRI) as the median scores dropped from 22.0 to 2.0 after 1 year of treatment, while all the others were able to reduce pain quality from 18.0–22.00 to 4.5–9.0.
In addition, VO presented the highest rate of clinical response and remission, and the best acceptability by the patients, safety profile and tolerability, compared with the other four ADs. 111 However, the authors revealed several differences between VO and the other ADs. Indeed, despite the fact that the acceptability of the treatment was good for all the drugs because no drop-out was reported before the sixth month, the patients who received VO were more enthusiastic about starting the treatment on account of its known pro-cognitive effect. 111 In addition, in long-term analyses, although all the ADs showed a reduction in scores for pain, anxiety and depression, only VO showed any achievement in terms of clinical response and clinical remission, with a functional recovery in 96.6% and 83.3% of patients after 6 and 12 months of treatment, respectively. 111 In addition, only 10% of patients treated with VO reported any side effects, in fact only nausea, and in most cases this resolved spontaneously after 2 weeks. However, abdominal pain, QTc prolongation, somnolence and sexual dysfunction were reported in treatment with other ADs. 111
Implications for the clinical care of CNP
Currently, ADs such as duloxetine, venlafaxine and amitriptyline are considered the gold standard drugs in the treatment of various pain syndromes, including CNOP, fibromyalgia, migraine, functional abdominal pain and chemotherapy-induced neuropathy, on account of their efficacy in pain management, especially in patients with a psychiatric comorbidity.130,171 Despite the utility of these drugs, patients frequently discontinue treatment due to the occurrence of ADRs – a factor that may influence the choice of a drug for a specific patient. 18 Therefore, VO could represent an important innovative option for the treatment of CNP and could be considered as the first line of treatment on account of its safety and tolerability profile, especially in elderly patients with cognitive impairment.
Conclusions
The management of CNP has become of the utmost importance in primary care due to the high prevalence of this disease in the general population and to the consequent increased demand for treatment. The frequent association of CNP with anxiety, depression and sleep disturbance has led clinicians to choose therapeutic strategies aimed not only at relieving the pain but also at treating the coexisting psychiatric comorbidities in order to provide a comprehensive management and therefore enhanced outcomes. 5
In this regard, VO is increasingly being considered as a treatment option. It is a novel drug that has proved to be well tolerated and effective in patients with CNOP in comparison with other ADs. In particular, VO has shown promising preliminary results in the treatment of CNOP and has therefore been suggested as a new frontier in the management not only of this disease but potentially of other chronic pain conditions. The use of VO presents several beneficial effects, especially in middle-aged or older patients, in whom VO improves several cognitive functions, along with providing a safe profile, good tolerability and lower latency of action.
Clinicians may choose to use VO (up to 20 mg/daily) either as a first line therapy or in the treatment of patients with an unsatisfactory response to other ADs or report the occurrence of ADRs. The treatment should be continued over the long term until the achievement of a clinical remission or response and a return to normal daily functioning, as suggested by a reduction in mood disorders, at least for 12 months because an early discontinuation of the drug may increase the risk of relapse and recurrence. 172 A careful tapering is recommended when remission has been achieved to avoid any withdrawal symptoms or a relapse of the disease. In patients who report nausea as a side effect and in non-responder cases, another AD may be considered.111,168
Footnotes
Author contributions
DA: conceptualisation; EC: methodology; NC: software; DA, MDM: validation; EC, NC: formal analysis; GP, EC: investigation; GP, DA: resources; GP, NC: data curation; DA, NC: writing – original draft preparation; DA, MDM: writing – review and editing; MDM: visualisation; DA, MDM: supervision. All authors contributed to the work and are familiar with the primary data; each has read the final version of the manuscript and approved its content. All the authors have agreed to have their name added to the paper.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Compliance with Ethical Standards
The content of the manuscript has not previously been published and is not currently submitted elsewhere. The manuscript is free of falsification, fabrication, plagiarism and self-plagiarism. All applicable principles of scientific integrity, including the ethical treatment of human subjects, have been followed.
