Abstract
Background:
Major depressive disorder (MDD) and generalized anxiety disorder (GAD) are frequently comorbid.
Aims:
To assess the effectiveness of vortioxetine in patients with MDD and comorbid GAD.
Methods:
Open-label, 8-week study (NCT04220996) in 100 adult outpatients with severe MDD and severe comorbid GAD receiving vortioxetine as first treatment for the current depressive episode or switching to vortioxetine due to inadequate response to another drug for depression. Vortioxetine starting dosage was 10 mg/day, with forced up-titration to 20 mg/day after 1 week. Response was defined as ⩾50% decrease in Montgomery–Åsberg Depression Rating Scale (MADRS) and/or Hamilton Anxiety Rating Scale (HAM-A) total score from baseline; remission defined as MADRS and/or HAM-A total score ⩽10.
Results:
Clinically meaningful and statistically significant improvements from baseline in symptoms of depression and anxiety, and overall functioning and health-related quality of life, were observed after 8 weeks’ vortioxetine treatment (all changes p < 0.0001 vs baseline). At week 8, rates of MADRS response and remission were 61% and 35%, respectively. Corresponding rates of HAM-A response and remission were 55% and 42%. Response on both the MADRS and HAM-A scales was achieved by 52% of patients; 31% achieved remission on both scales. Vortioxetine dose up-titration was well tolerated; no unexpected adverse events were reported.
Conclusion:
Vortioxetine demonstrates effectiveness in significantly reducing symptoms of both depression and anxiety in patients with severe MDD comorbid with severe GAD. Findings support increasing vortioxetine dosage to 20 mg/day early in the course of therapy, and show that this may be achieved without compromising tolerability.
Keywords
Introduction
Major depressive disorder (MDD) and generalized anxiety disorder (GAD) are frequently comorbid, and individuals experiencing comorbid GAD represent one of the largest patient groups within the overall population with MDD (Dold et al., 2017; Hasin et al., 2018; Kessler et al., 2005; Lamers et al., 2011; Moffitt et al., 2007; Saha et al., 2021; Zhou et al., 2017). When comorbid, MDD and GAD are associated with more severe symptoms, greater impairment in functioning and health-related quality of life (HRQoL) and greater risk of suicide ideation than seen in patients with either condition alone (Dold et al., 2017; Gili et al., 2013; Kessler et al., 2008; Lamers et al., 2011; Pfeiffer et al., 2009; Shepardson et al., 2019; Zhou et al., 2017). Patients with comorbid MDD and GAD are also considered more difficult to treat, requiring longer treatment duration, having higher rates of treatment withdrawal and achieving lower rates of remission than those with either condition alone (Dold et al., 2017; Kelly and Mezuk, 2017; Kessler et al., 2008).
Vortioxetine is a novel drug for depression with a multimodal mechanism of action, acting both as an inhibitor of the serotonin (5-HT) transporter as well as a modulator of several 5-HT receptor subtypes (5-HT3, 5-HT7 and 5-HT1D receptor antagonist, 5-HT1B receptor partial agonist and 5-HT1A receptor agonist) (Gonda et al., 2019; Sanchez et al., 2015). Vortioxetine has been shown to be effective and well tolerated for the treatment of depressive, cognitive and physical symptoms in patients with MDD across the approved dose range of 5–20 mg/day (Christensen et al., 2018; Gonda et al., 2019), and has also demonstrated anxiolytic effects in patients with MDD experiencing severe concurrent anxiety (Baldwin et al., 2016; Chokka et al., 2021). In patients with GAD, a short-term, randomized controlled trial demonstrated that vortioxetine 5 mg/day was significantly more efficacious than placebo for the relief of anxiety symptoms (Bidzan et al., 2012). Vortioxetine 5 or 10 mg/day has also been shown to be efficacious and well tolerated as maintenance treatment for the prevention of relapse in patients with GAD (Baldwin et al., 2012). However, other studies in patients with GAD conducted entirely in US populations have yielded conflicting results (Mahableshwarkar et al., 2014a, 2014b; Rothschild et al., 2012). Geographic differences in clinical trial outcomes have been reported for many effective drugs for depression, in particular for the US, which may arise from variation in patient characteristics, diagnostic and clinical practices, study design and trial setting (Chang et al., 2008; Dunlop et al., 2012; Khin et al., 2011; Niklson and Reimitz, 2001; Vieta et al., 2011; Welten et al., 2015).
Vortioxetine has been shown to have dose-dependent effects on symptoms of depression and concomitant anxiety in patients with MDD; greatest clinical benefits are achieved at a dosage of 20 mg/day (Baldwin et al., 2016; Christensen et al., 2021; Iovieno et al., 2021; Thase et al., 2016). However, the effectiveness and tolerability of vortioxetine have not been demonstrated in patients with a diagnosis of both MDD and GAD. This study was undertaken to assess the effectiveness and tolerability of vortioxetine 20 mg/day for the relief of symptoms of depression and anxiety in patients with MDD comorbid with GAD; patient functioning and HRQoL were also assessed.
Methods
Study design and participants
This was a multicentre, open-label effectiveness study in adult outpatients with MDD comorbid with GAD (NCT04220996). The study included a 2-week screening period, an 8-week open-label, flexible-dose treatment period and a 4-week safety follow-up period. Patients were aged 18–65 years with a primary diagnosis of MDD (Diagnostic and Statistical Manual of Mental Disorders (5th ed.; DSM-5) criteria (American Psychiatric Association, 2013), confirmed using the Mini International Neuropsychiatric Interview); duration of current major depressive episode (MDE) <12 months; comorbid GAD (diagnosed according to DSM-5 criteria before the current MDE); Montgomery–Åsberg Depression Rating Scale (MADRS) total score ⩾22 and Hamilton Anxiety Rating Scale (HAM-A) total score ⩾20. Patients with treatment-resistant depression (i.e. inadequate therapeutic response to ⩾2 previous drugs for depression administered at the recommended dose for ⩾6 weeks) and those with a primary diagnosis of any other current psychiatric or Axis I disorder were excluded.
Patients were receiving vortioxetine as their first treatment for the current MDE or switching to vortioxetine due to inadequate response to another drug for depression. Treatment with vortioxetine was initiated at the recommended starting dose of 10 mg/day, with forced up-titration to 20 mg/day in all patients after 1 week. Subsequent dose reductions were permitted based on individual tolerability/response. A patient-specific log was used to track study medication dispensed to and returned by patients at each study visit. Patients considered by the investigator to be non-compliant with study medication were to be withdrawn from the study.
In patients switching to vortioxetine due to inadequate response to another drug for depression, prior treatment was to be discontinued at least 2 weeks before administration of the first dose of study medication (baseline), except for selective serotonin reuptake inhibitors (SSRIs) or serotonin–norepinephrine reuptake inhibitors (SNRIs), use of which was permitted until baseline. If needed, gradual dose reduction was recommended during the screening period in order to reach the minimum therapeutic dose at baseline, in line with the local prescribing information. If switching from paroxetine, the last dose should have been taken no less than 1 week before the baseline visit. Fluoxetine was disallowed within 5 weeks before baseline.
Concomitant use of benzodiazepines was only permitted during the study in patients who were on a stable dose before baseline. Benzodiazepines were not to be taken the night before a study visit, and episodic use was not permitted. All other drugs for anxiety were to be discontinued before baseline. Use of monoamine oxidase inhibitors and other psychotropic agents (including herbal medications such as St. John’s Wort, kava kava, valerian and Ginkgo biloba) was not permitted within 2 weeks before baseline or during the study period. Concomitant use of antipsychotic medication was not permitted during the study; antipsychotic medications were to be discontinued at least 2 weeks before baseline (⩾6 months for depot formulations). Use of sedatives/hypnotics was not permitted within 1 day before baseline or during the study period, with the exception of zolpidem, zopiclone and zaleplon, use of which was permitted for a maximum of two nights per week, excluding the night before a study visit. Patients taking or likely to require any other medication that could potentially interfere with study drug assessment or the conduct or interpretation of the study were excluded from participation.
The study was conducted in accordance with the principles of Good Clinical Practice and the Declaration of Helsinki. Local ethics committee approval was obtained at all participating sites and all patients provided written informed consent.
Study assessments
Study assessments were conducted at baseline and at weeks 1, 4 and 8, with a follow-up safety assessment at 12 weeks.
Clinicians assessed symptom severity using the MADRS, HAM-A and Clinical Global Impression (CGI) scales, and functioning using the Functioning Assessment Short Test (FAST). The FAST is a clinician-rated scale designed to assess patients’ problems in daily functioning over the past 14 days across six domains: autonomy, occupational functioning, cognitive functioning, financial issues, interpersonal relationships and leisure time (Rosa et al., 2007). FAST total score ranges from 0 to 72 points, with higher scores indicating greater impairment in functioning. Proposed ranges for mild, moderate and severe functional impairment are 12–20, 21–40 and >40 points, respectively (Bonnín et al., 2018).
Patients assessed anxiety and depressive symptoms using the Hospital Anxiety and Depression Scale–Anxiety (HADS-A) and Hospital Anxiety and Depression Scale–Depression (HADS-D) subscales, and HRQoL using the Quality of Life Enjoyment and Satisfaction Questionnaire (Q-LES-Q) long-form. The Q-LES-Q long-form provides a comprehensive subjective assessment of HRQoL across eight domains: physical health, subjective feelings, work, household duties, school/course work, leisure time activities, social relationships and general activities (Endicott et al., 1993). The general activities domain of Q-LES-Q long-form may be used as a short-form version of the Q-LES-Q (Stevanovic, 2011). Two domains assessing satisfaction with medication and overall satisfaction and contentment are scored separately. Q-LES-Q scores are expressed as a percentage of the maximum score possible; higher scores signify better HRQoL.
All investigators received thorough training regarding administration and rating of the MADRS, HAM-A, CGI and FAST before being authorized to participate in the study.
Safety
Treatment-emergent adverse events (TEAEs) were assessed over the entire study period and summarized by Medical Dictionary for Regulatory Affairs preferred terms. The incidence of TEAEs occurring on or after the day of vortioxetine dosage increase to 20 mg/day was also assessed in patients who maintained this dose until study completion or withdrawal; this was compared with the incidence of TEAEs occurring at or after 1 week for patients who remained on vortioxetine 10 mg/day throughout the study.
Statistical analysis
Planned enrolment was 100 patients; with this sample size, and an assumed standard deviation of 10 points on the change in MADRS total score, precision with a 95% confidence interval of 4 points on the MADRS total score was expected. Effectiveness was analysed in all eligible patients who received at least one dose of vortioxetine and had a valid baseline and at least one post-baseline assessment for MADRS total score (full analysis set). Safety was analysed in all enrolled patients who received at least one dose of vortioxetine (all treated patients set). Analyses were performed for the overall population and by treatment history (i.e. receiving vortioxetine as first-line treatment or switching to vortioxetine due to inadequate response to a previous drug for depression).
The primary study endpoint was change from baseline to week 8 in MADRS total score; secondary endpoints were the corresponding changes in HAM-A, CGI-Severity (CGI-S), HADS-D, HADS-A, FAST and Q-LES-Q scores, and CGI-Improvement (CGI-I) score at each visit. Rates of response and remission for MADRS and HAM-A after 8 weeks of vortioxetine treatment were also evaluated. Response was defined as ⩾50% decrease in MADRS and/or HAM-A total score from baseline; remission was defined as MADRS and/or HAM-A total score ⩽10 at week 8.
For all effectiveness endpoints (except CGI-I), least-squares (LS) mean change from baseline was estimated using a mixed model for repeated measurements (MMRM), with week and site as fixed factors and baseline score as a covariate. The interaction between week and baseline total score was included in the model and an unstructured covariance matrix was applied. For CGI-I, LS mean CGI-I score was estimated by a similar MMRM, but with baseline CGI-S score as the baseline score covariate.
Descriptive and partial Pearson’s correlation coefficients were calculated to assess correlations between HAM-A and MADRS total scores and FAST total and Q-LES-Q general activities scores. Partial correlation coefficients were calculated using the baseline scores for the relevant scales as covariates. Analyses were conducted using SAS statistical software (version 9.4; SAS Institute Inc., Cary, NC, USA), with significance set at p < 0.05.
Results
Study population
This study was conducted at 17 sites in five countries (Poland, Estonia, France, Spain and South Korea) between 27 December 2019 and 9 March 2021. At baseline, patients had on average severe symptoms of depression (mean ± standard deviation (SD) MADRS total score, 29.5 ± 4.7) and anxiety (HAM-A total score, 28.6 ± 4.9), and severe impairment of functioning and HRQoL (Table 1). Of the 102 patients enrolled, 100 received at least one dose of vortioxetine and were included in this analysis. Twenty-three patients were receiving vortioxetine as first-line treatment and 77 were switching to vortioxetine due to inadequate response to prior therapy with another drug for depression (Supplementary Table 1). Most patients were switching from an SSRI (n = 56, most commonly escitalopram (n = 26)). Ninety-seven patients completed the study; two discontinued treatment primarily due to adverse events (dysgeusia, n = 1; hypersensitivity, n = 1) and one patient withdrew consent. No patients were withdrawn from the study due to non-compliance with study medication.
Patient demographics and disease characteristics at the start of vortioxetine treatment (all treated patients set).
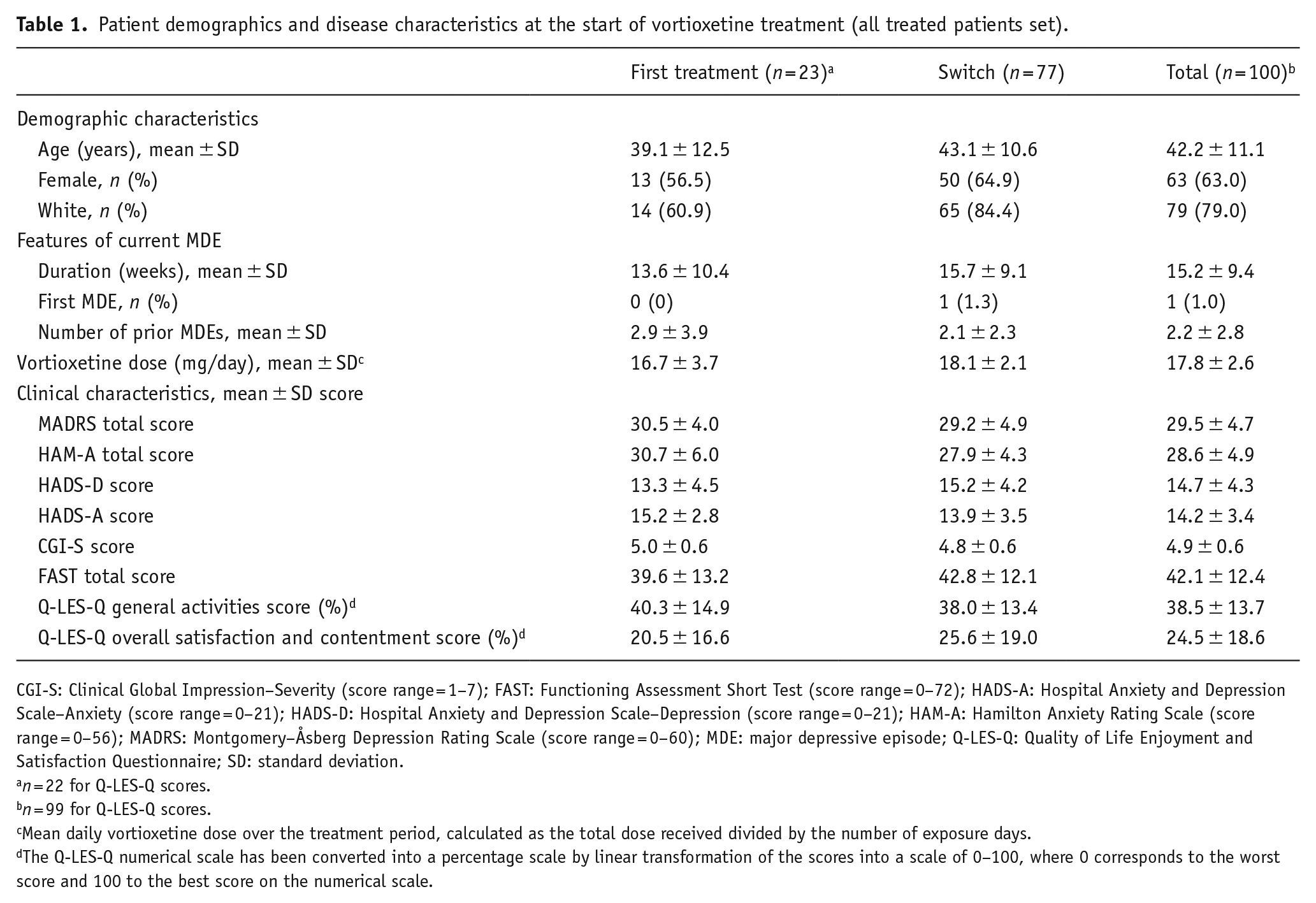
CGI-S: Clinical Global Impression–Severity (score range = 1–7); FAST: Functioning Assessment Short Test (score range = 0–72); HADS-A: Hospital Anxiety and Depression Scale–Anxiety (score range = 0–21); HADS-D: Hospital Anxiety and Depression Scale–Depression (score range = 0–21); HAM-A: Hamilton Anxiety Rating Scale (score range = 0–56); MADRS: Montgomery–Åsberg Depression Rating Scale (score range = 0–60); MDE: major depressive episode; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; SD: standard deviation.
n = 22 for Q-LES-Q scores.
n = 99 for Q-LES-Q scores.
Mean daily vortioxetine dose over the treatment period, calculated as the total dose received divided by the number of exposure days.
The Q-LES-Q numerical scale has been converted into a percentage scale by linear transformation of the scores into a scale of 0–100, where 0 corresponds to the worst score and 100 to the best score on the numerical scale.
Twenty-one patients discontinued treatment with a drug for anxiety prior to initiating treatment with vortioxetine (five in the first-treatment group and sixteen in the switch group). Five patients continued to take alprazolam after initiation of vortioxetine treatment (one in the first-treatment group and four in the switch group). One patient in the switch group was also receiving diazepam and continued to take this after initiation of vortioxetine treatment. No patients initiated concomitant treatment with a drug for anxiety during the study.
After 1 week of treatment, vortioxetine dosage was increased to 20 mg/day in 94 patients. In all, 87 patients were receiving vortioxetine 20 mg/day at week 4 and continued on this dose until study completion or withdrawal. Only four patients remained on vortioxetine 10 mg/day throughout the study. The mean ± SD dose of vortioxetine over the 8-week study period was 17.8 ± 2.6 mg/day.
Effectiveness
Clinically and statistically significant improvements in clinician- and patient-assessed symptoms of depression and anxiety were observed after 8 weeks of vortioxetine treatment (p < 0.0001 for all scales; Figure 1). For all scales, statistically significant improvements were seen from week 1 (all p ⩽ 0.0007). Results according to vortioxetine treatment line (first treatment or switch) are shown in Supplementary Figure 1 and Table 2.
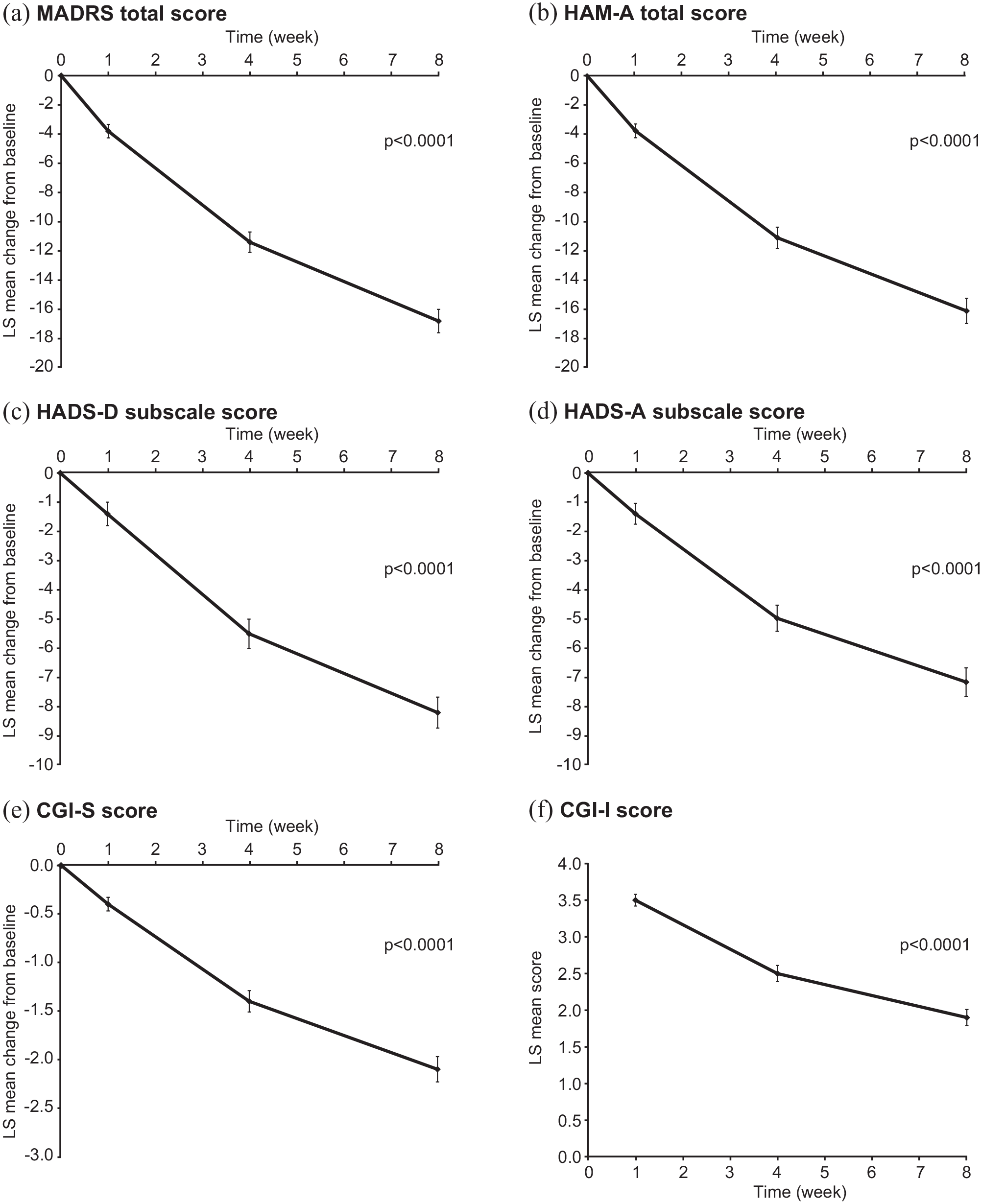
Panels (a) to (e): LS mean (SE) change from baseline for (a) MADRS total score, (b) HAM-A total score, (c) HADS-D subscale score, (d) HADS-A subscale score and (e) CGI-S score; panel (f): LS mean (SE) CGI-I score (full analysis set). For panels (a) to (e), p-values are at week 8 vs baseline; for panel (f), p-value is at week 8 based on a test of CGI-I score = 4 (no improvement).
Results of the mixed-model repeated measurements analyses for key effectiveness outcomes after 8 weeks of vortioxetine treatment (full analysis set). For all values except CGI-I, LS mean (SE) change from baseline is shown; for CGI-I, LS mean (SE) score is shown.
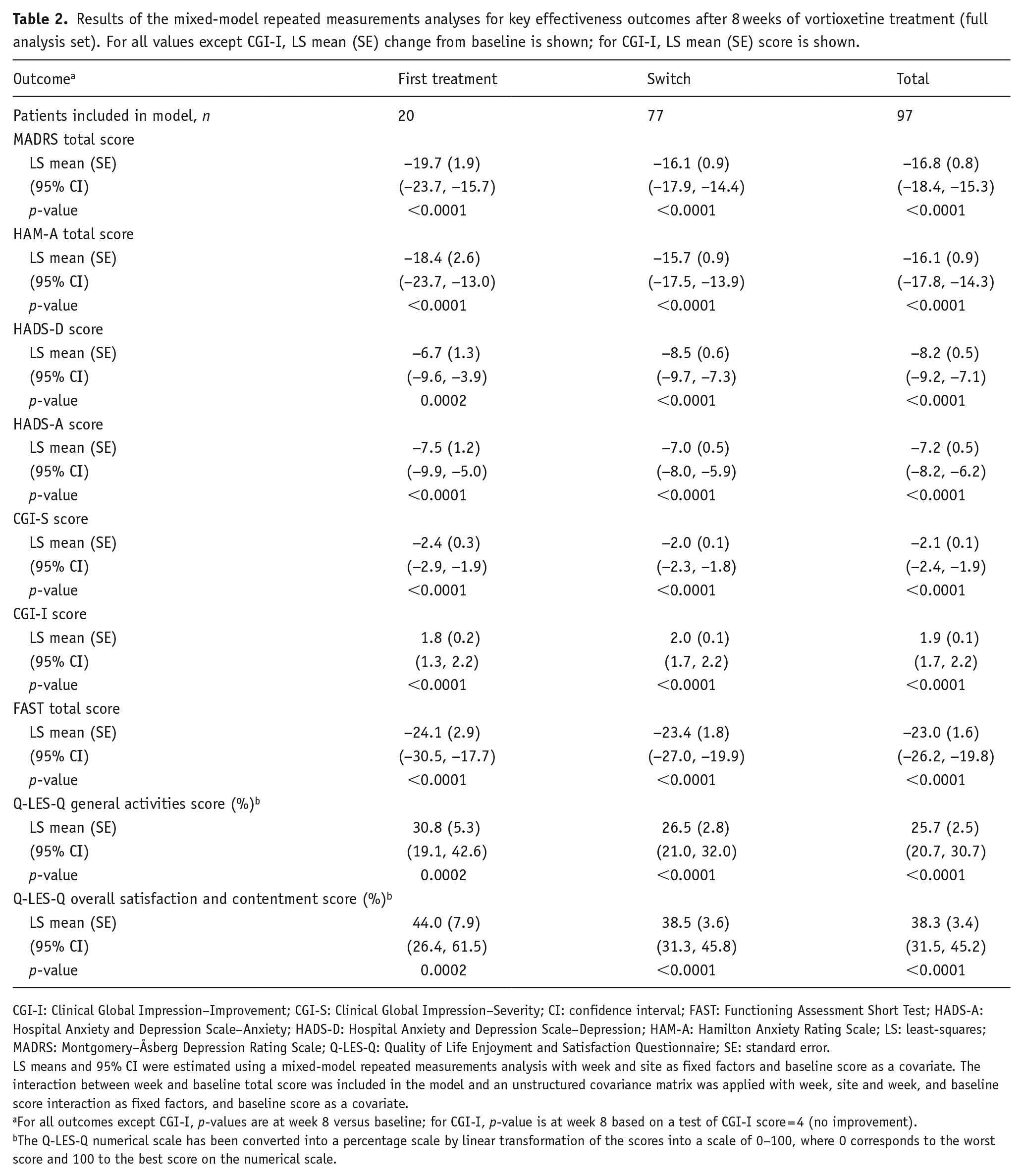
CGI-I: Clinical Global Impression–Improvement; CGI-S: Clinical Global Impression–Severity; CI: confidence interval; FAST: Functioning Assessment Short Test; HADS-A: Hospital Anxiety and Depression Scale–Anxiety; HADS-D: Hospital Anxiety and Depression Scale–Depression; HAM-A: Hamilton Anxiety Rating Scale; LS: least-squares; MADRS: Montgomery–Åsberg Depression Rating Scale; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire; SE: standard error.
LS means and 95% CI were estimated using a mixed-model repeated measurements analysis with week and site as fixed factors and baseline score as a covariate. The interaction between week and baseline total score was included in the model and an unstructured covariance matrix was applied with week, site and week, and baseline score interaction as fixed factors, and baseline score as a covariate.
For all outcomes except CGI-I, p-values are at week 8 versus baseline; for CGI-I, p-value is at week 8 based on a test of CGI-I score = 4 (no improvement).
The Q-LES-Q numerical scale has been converted into a percentage scale by linear transformation of the scores into a scale of 0–100, where 0 corresponds to the worst score and 100 to the best score on the numerical scale.
After 8 weeks of vortioxetine treatment, LS mean (standard error (SE)) change in MADRS total score from baseline was −16.8 (0.8) points (−19.7 (1.9) in the first-treatment group and −16.1 (0.9) points in the switch group) (all p < 0.0001). At week 8, 61% of patients achieved response and 35% achieved remission based on MADRS score. The LS mean (SE) change in HAM-A total score from baseline at week 8 was −16.1 (0.9) points (−18.4 (2.6) in the first-treatment group and −15.7 (0.9) points in the switch group) (all p < 0.0001). At week 8, 55% of patients achieved response and 42% achieved remission based on HAM-A score. In all, 52% of patients achieved response and 31% achieved remission on both the MADRS and HAM-A scales after 8 weeks of vortioxetine treatment.
Significant improvements were also seen in CGI scores and HADS-D and HADS-A subscale scores over the study period (Figure 1). At week 8, LS mean (SE) change in CGI-S score from baseline was −2.1 (0.1) points (p < 0.0001). LS mean CGI-I score at week 8 was 1.9 (0.1) points, with 75.3% of patients considered much or very much improved (CGI-I score ⩽2 points). After 8 weeks of vortioxetine treatment, LS mean (SE) changes in HADS-D and HADS-A scores were −8.2 (0.5) and −7.2 (0.5) points, respectively (both p < 0.0001 vs baseline). CGI-I score and improvements in CGI-S, HADS-D and HADS-A scores after 8 weeks of vortioxetine treatment were generally similar in the first-treatment and switch groups (Table 2).
Statistically significant improvements were seen in FAST total score and across all FAST domains after 8 weeks of vortioxetine treatment (Figure 2; all changes p < 0.0001 vs baseline). At week 8, the LS mean (SE) change in FAST total score from baseline was −23.0 (1.6) points (p < 0.0001), with similar improvements seen in the first-treatment and switch groups (Table 2). Statistically significant improvement was also seen across all Q-LES-Q long-form domains (Figure 3; most changes p < 0.0001). After 8 weeks of vortioxetine treatment, the LS mean (SE) change in Q-LES-Q general activities score from baseline was 25.7% (2.5) (p < 0.0001) (Table 2). Improvement in Q-LES-Q general activities score from baseline over the 8 weeks of vortioxetine treatment was 30.8% (5.3) in the first-treatment group and 26.5% (2.8) in the switch group.
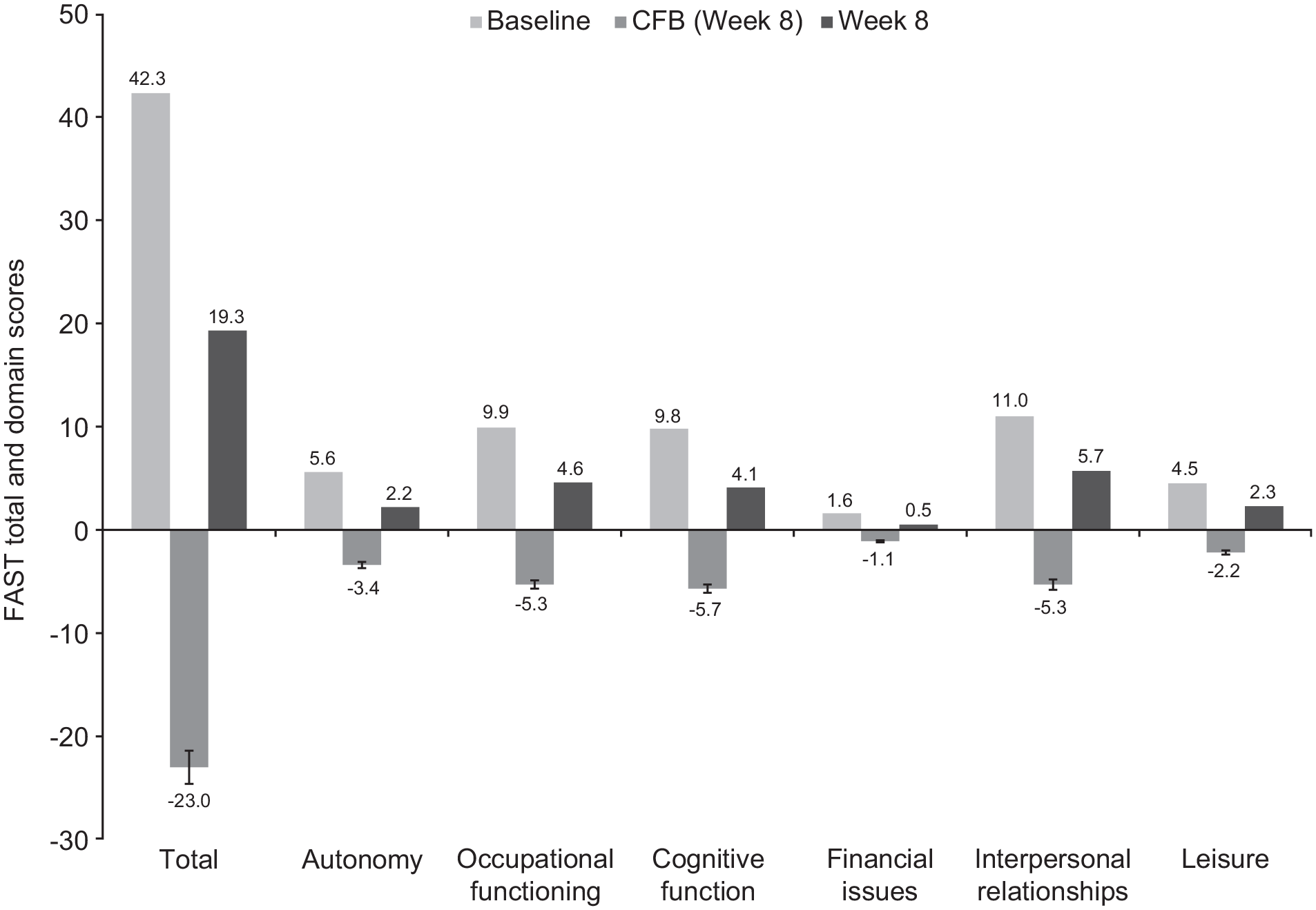
FAST total and domain scores at baseline and change (SE) from baseline after 8 weeks of treatment with vortioxetine (full analysis set; patients with a corresponding baseline score). p < 0.0001 for all changes from baseline.
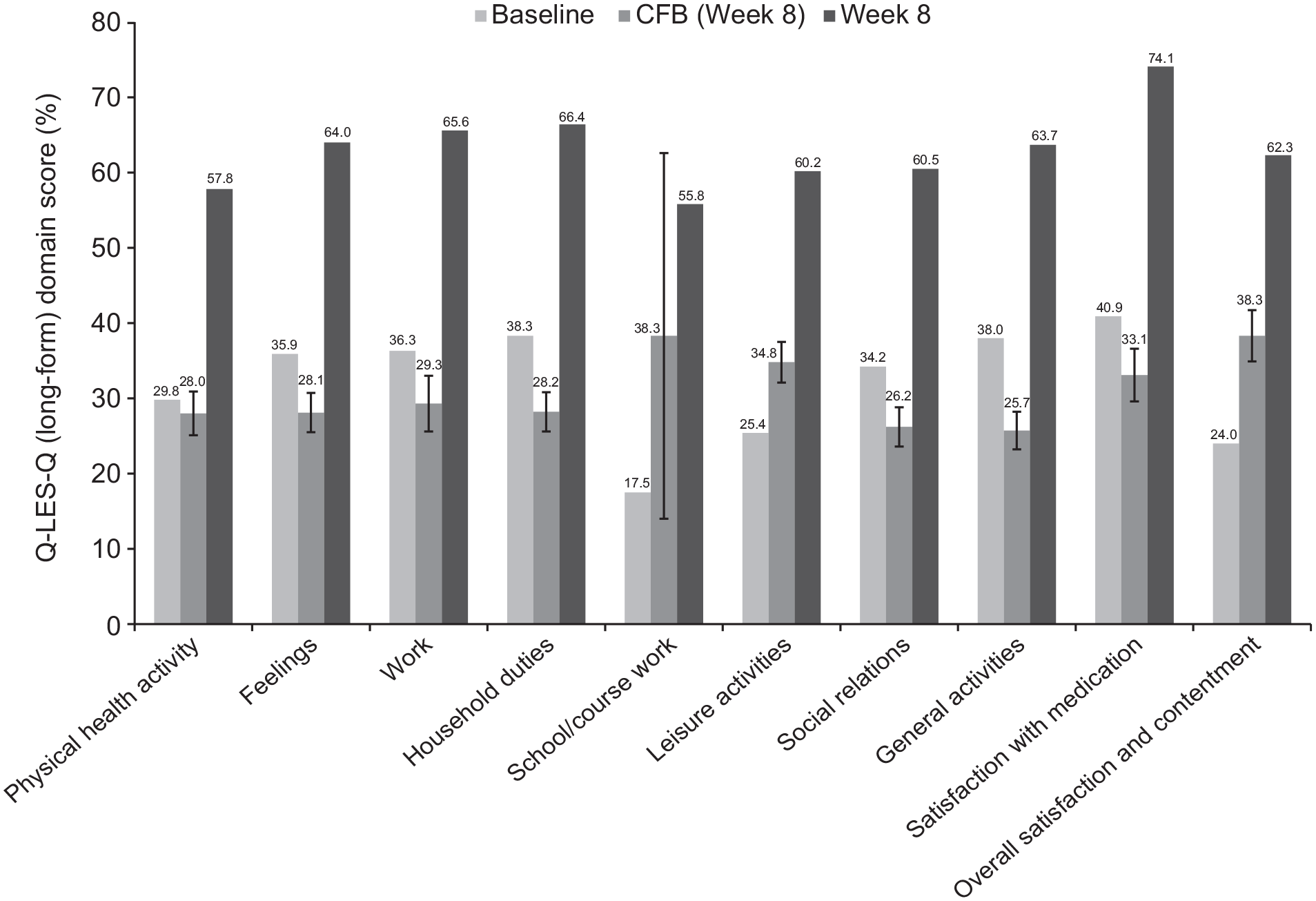
Q-LES-Q long-form domain scores at baseline and change (SE) from baseline after 8 weeks of treatment with vortioxetine (full analysis set, patients with a corresponding baseline score). The Q-LES-Q numerical scale has been converted into a percentage scale by linear transformation of the scores into a scale of 0–100, where 0 corresponds to the worst score and 100 to the best score on the numerical scale. p < 0.0001 for all changes from baseline.
As shown in Table 3, changes from baseline to week 8 in MADRS and HAM-A total scores were significantly and strongly positively correlated with FAST total score (partial r = 0.80 and 0.76, respectively; both p < 0.0001), and significantly and negatively correlated with Q-LES-Q general activities score (partial r = −0.65 and −0.62, respectively; both p < 0.0001).
Correlation analysis of change from baseline to week 8 in key endpoints (full analysis set).
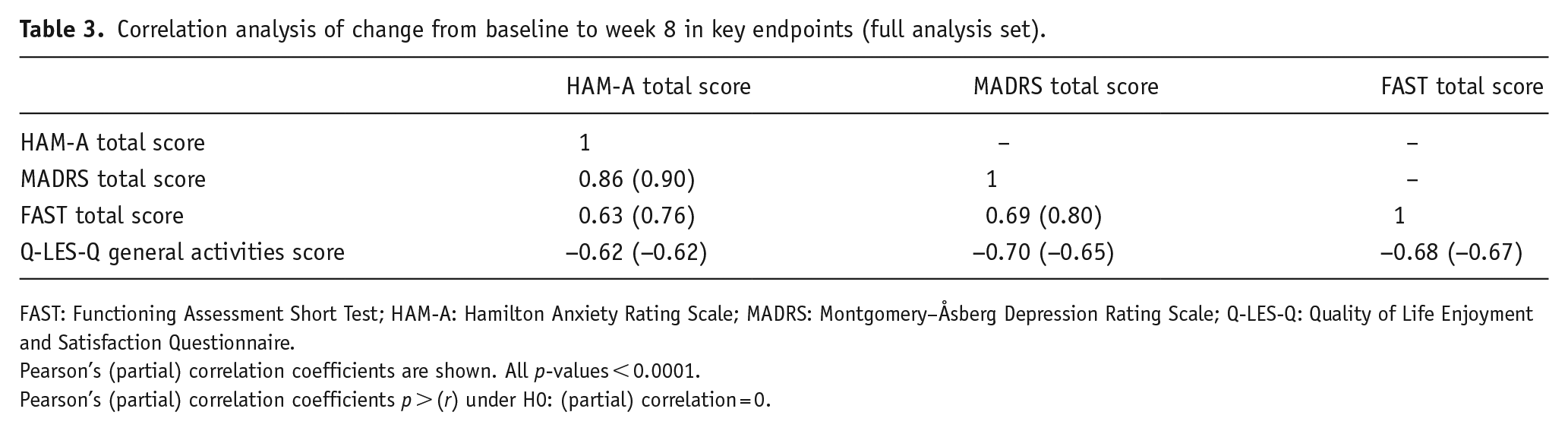
FAST: Functioning Assessment Short Test; HAM-A: Hamilton Anxiety Rating Scale; MADRS: Montgomery–Åsberg Depression Rating Scale; Q-LES-Q: Quality of Life Enjoyment and Satisfaction Questionnaire.
Pearson’s (partial) correlation coefficients are shown. All p-values < 0.0001.
Pearson’s (partial) correlation coefficients p > (r) under H0: (partial) correlation = 0.
Safety
In total, 46 patients (46%) reported a TEAE (Table 4). The most common TEAEs (reported by >5% of patients) were nausea and abdominal pain. All TEAEs were of mild or moderate intensity. No serious adverse events were reported.
Overview of TEAEs occurring in ⩾2% of patients during the 8-week treatment period (all treated patients set).
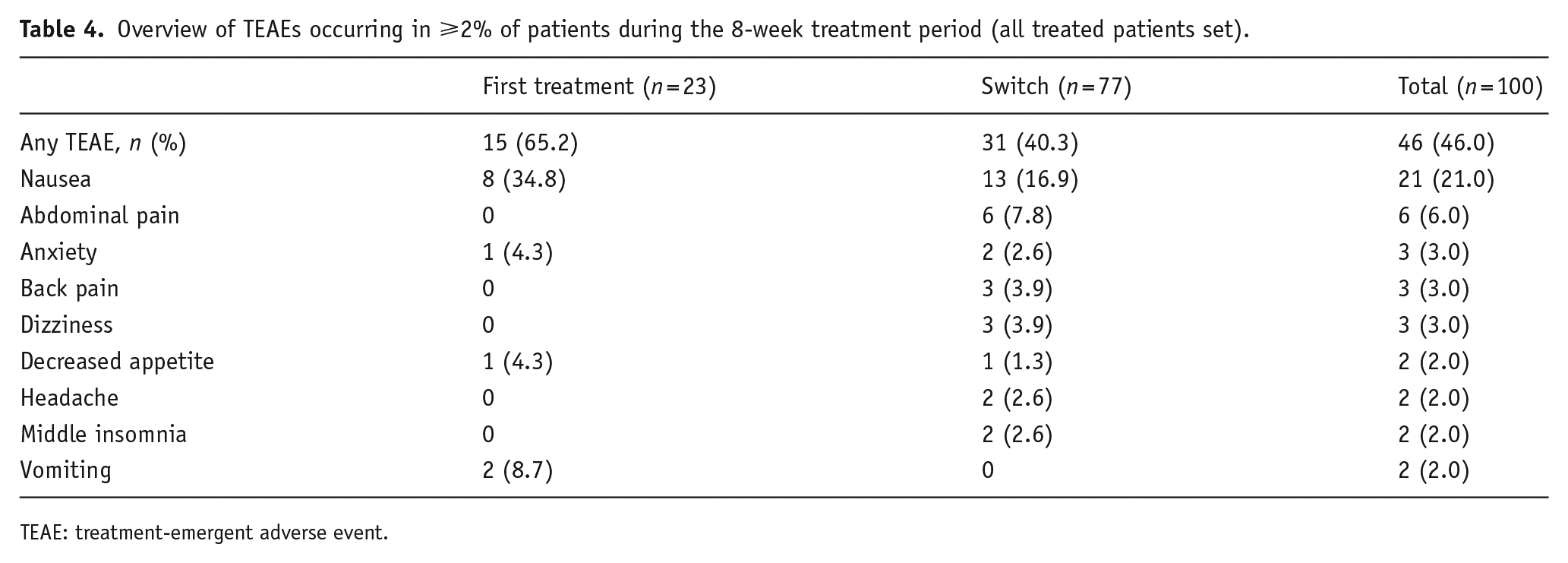
TEAE: treatment-emergent adverse event.
After week 1, TEAEs were reported in 17 of the 86 patients who increased vortioxetine dosage to 20 mg/day and remained on this dosage to study end (most commonly, abdominal pain (five patients) and nausea (four patients)). TEAEs were reported after week 1 in two of the four patients who remained on vortioxetine 10 mg/day throughout the study (headache and hypersensitivity, each reported by one patient).
Discussion
Vortioxetine demonstrated effectiveness in significantly reducing both depressive and anxiety symptoms in this population of patients with severe MDD and severe comorbid GAD, the majority of whom showed inadequate response to prior treatment with another drug for depression. Significant and clinically meaningful effects were already observed after 1 week and continued to increase over the treatment course. At week 8, 35% of patients had achieved remission from depressive symptoms and 42% had achieved remission from anxiety symptoms. The improvement in symptoms of depression and anxiety was accompanied by significant and broad improvement in patients’ daily functioning and their self-perceived ability to perform and enjoy a broad range of activities of daily life. Clinically meaningful improvement in symptoms of depression and anxiety, as well as functioning and HRQoL, was seen in patients receiving vortioxetine as first-line treatment and in those who were switching to vortioxetine due to inadequate response to prior therapy. However, greatest improvements were generally seen when vortioxetine was used as a first-line therapy. Per protocol, vortioxetine dosage was to be up-titrated to 20 mg/day from the starting dose of 10 mg/day after 1 week of treatment. Almost all patients (94%) had their vortioxetine dosage increased at that time, and treatment with vortioxetine 20 mg/day was generally well tolerated.
Vortioxetine is the only drug for depression with a demonstrated dose–response relationship across the full therapeutic dose range (5–20 mg/day) (Baldwin et al., 2016; Christensen et al., 2021; Iovieno et al., 2021; Thase et al., 2016), as is acknowledged in the EU Summary of Product Characteristics (European Medicines Agency, 2020). Available data indicate that an early increase in vortioxetine dosage from 10 to 20 mg/day is generally well tolerated and does not prompt a need for subsequent dosage reduction or treatment withdrawal. In a pooled analysis of data from three randomized, controlled, flexible-dose studies, 48% of vortioxetine-treated patients had their dosage increased to 20 mg/day after the first week of treatment and 64% were receiving vortioxetine 20 mg/day at their last study visit (Christensen et al., 2021). Importantly, vortioxetine dose up-titration did not appear to compromise tolerability; the incidence of adverse events was not increased in patients who received vortioxetine 20 mg/day compared with those who received 10 mg/day (Christensen et al., 2021). In a recent open-label study with flexible vortioxetine dosing in patients with MDD and inadequate response to prior treatment with another drug for depression (Fagiolini et al., 2021), vortioxetine dosage was increased to 20 mg/day after 1 week in 38/143 patients (27%), only three of whom had their dosage subsequently decreased. Half of all patients were receiving vortioxetine 20 mg/day at study end.
The observed improvement in symptoms of both depression and anxiety in patients receiving treatment with vortioxetine in this study is noteworthy as patients with MDD and comorbid GAD are generally considered difficult to treat, typically achieving poorer treatment outcomes than those with either condition alone (Dold et al., 2017; Kelly and Mezuk, 2017; Kessler et al., 2008). Assessment of the clinical effectiveness of drugs for depression in patients with a diagnosis of both MDD and GAD is rare; however, evidence of effectiveness in this patient population is clinically relevant given how frequently these two conditions are comorbid (Montgomery, 2019; Saha et al., 2021).
The beneficial effects of vortioxetine against symptoms of depression and anxiety are most likely due to its multimodal mechanism of action. Vortioxetine has been shown to be a 5-HT3, 5-HT7 and 5-HT1D receptor antagonist, a 5-HT1B receptor partial agonist and a 5-HT1A receptor agonist, as well as an inhibitor of the 5-HT transporter (Bang-Andersen et al., 2011; Mørk et al., 2012; Sanchez et al., 2015). As such, vortioxetine modulates several neurotransmitter systems relevant to the neurobiology of depression and anxiety, including not only the serotoninergic system, but probably also the norepinephrine, dopamine, histamine, acetylcholine, gamma-aminobutyric acid, and glutamate systems (Riga et al., 2016; Stahl, 2015). The effects of vortioxetine on 5-HT1A, 5-HT3 and 5-HT7 receptors and the 5-HT transporter, in particular, are considered responsible for its anxiolytic-like effects.
To fully appreciate the results of this study, it is relevant to consider the results observed in earlier studies with vortioxetine in patients with a diagnosis of either MDD or GAD alone. The observed improvement in depressive symptoms (reduction in MADRS total score, −16.8 points) is comparable to that reported in fixed-dose, randomized controlled trials of vortioxetine 10 or 20 mg/day in patients with MDD alone (Boulenger et al., 2014; Inoue et al., 2020; Jacobsen et al., 2015; Mahableshwarkar et al., 2015; McIntyre et al., 2014; Nishimura et al., 2018), and is of a similar magnitude to that seen in flexible-dose studies of vortioxetine 10–20 mg/day in patients with MDD without comorbid GAD who had inadequate response to prior therapy with another drug for depression (Fagiolini et al., 2021; Montgomery et al., 2014).
With regard to anxiety symptoms, the improvement in HAM-A total score after 8 weeks of vortioxetine treatment in this study (−16.1 points) is greater than that reported in a randomized controlled trial in patients with GAD alone treated with vortioxetine 5 mg/day (−14.3 points) (Bidzan et al., 2012). The reduction in HAM-A total score of −15.7 points seen in patients switching to vortioxetine in this study is also greater than that reported in a randomized, flexible-dose study of vortioxetine 10–20 mg/day in patients with MDD and significant anxiety symptoms who had inadequate response to prior therapy (−11.7 points) (Montgomery et al., 2014). Our findings support earlier evidence of a dose–response effect for vortioxetine for the treatment of anxiety symptoms (Baldwin et al., 2016), as also reported for depressive symptoms (Baldwin et al., 2016; Christensen et al., 2021; Iovieno et al., 2021; Thase et al., 2016).
Substantial and broad improvements were observed in patients’ daily functioning, as comprehensively assessed by the investigator using the FAST scale. The FAST captures not only how well patients are functioning at home, work and in their social life, but also their ability to take care of personal finances and cognitive functioning. Patients had severe functional impairment at baseline, and only mild impairment after 8 weeks of vortioxetine treatment (FAST total score, 42.1 vs 20.1 points). The observed change in FAST total score (−23.0 points) is greater than that reported in a randomized controlled trial in working patients with MDD over 8 weeks of fixed-dose treatment with vortioxetine 10 mg/day or paroxetine 20 mg/day (−13.0 and −11.0 points, respectively, vs −8.3 points for placebo) (Baune et al., 2018). The significant and substantial improvements seen across all domains on the FAST scale in this study are particularly noteworthy, as domains such as work functioning do not necessarily show meaningful improvement after only 8 weeks of treatment.
Although patients had significantly impaired HRQoL at baseline (mean Q-LES-Q general activities score, 38.5%), significant improvements in all HRQoL domains were seen after 8 weeks (mean Q-LES-Q general activities score at week 8, 62.5%). Mean Q-LES-Q general activities scores of approximately 80% have been reported in community samples of healthy individuals with no history of mental illness (Rapaport et al., 2005; Schechter et al., 2007). Improvements in Q-LES-Q general activities scores of 11.9% and 6.8% have been suggested as minimum clinically important changes in patients with bipolar depressive disorder and GAD, respectively, treated with quetiapine for 8 weeks (Endicott et al., 2007; Wyrwich et al., 2009). The LS mean improvement in Q-LES-Q general activities score seen in patients with MDD comorbid with GAD after 8 weeks of vortioxetine treatment in this study (25.7%) was greater than these proposed thresholds and similar to that previously reported in patients with either condition alone following treatment with escitalopram (Demyttenaere et al., 2008).
The Q-LES-Q was described by its developers as a questionnaire that “evaluates patients’ enjoyment and satisfaction with different aspects of their lives” (Endicott et al., 1993). However, it is clear that the scale captures more than these aspects of HRQoL; indeed, it probes in detail about patients’ physical health and their ability to perform activities of daily living and engage in work and social activities. The long-form questionnaire comprises 93 items eliciting information on physical health, subjective feelings, work, household duties, school/course work, leisure time activities, social relationships and general activities. For instance, the 13 questions covering physical health explore whether patients have ‘been free of visual problems’, ‘completely free of aches, pains, or discomfort’ and ‘felt your memory was functioning well’. The 13 questions covering the work domain include ‘solved work problems or dealt with them without undue stress’, ‘been decisive about work, or made decisions’ and ‘communicated and interacted with ease with others while working’. In the subjective feelings domain, the 14 questions assess aspects such as feeling ‘clearheaded’, ‘able to travel about to get things done’ and ‘able to communicate with others’. The marked improvement across all domains of the Q-LES-Q long-form in this study substantiates the clinical relevance of the significant improvements in symptoms of depression and anxiety seen over the 8 weeks of vortioxetine treatment.
This study was conducted in routine practice conditions in a representative population of patients with MDD and comorbid GAD and therefore provides new clinically relevant information about vortioxetine in this patient group. Potential limitations include those generally associated with open-label studies due to lack of blinding, the lack of a placebo or active-comparator arm, and the relatively short duration of treatment (8 weeks). As patients with mood and anxiety disorders generally require long-term treatment, studies involving longer durations of vortioxetine treatment in this patient population would be of clinical interest. Notably, results of a previous study in patients with GAD found vortioxetine 5 or 10 mg/day to be significantly more effective than placebo for the prevention of relapse over a period of up to 56 weeks (Baldwin et al., 2012).
Conclusion
In summary, results of this study demonstrate the effectiveness of vortioxetine 20 mg/day for the treatment of symptoms of both depression and anxiety in patients with severe MDD comorbid with severe GAD. The observed beneficial effects on depressive and anxiety symptoms were already significant after 1 week and continued to increase over the 8 weeks of treatment. The improvements in symptoms of depression and anxiety were significantly correlated with broad improvements in overall functioning and HRQoL. Findings also support increasing vortioxetine dosage to 20 mg/day early in the course of therapy, and show that this is well tolerated.
Supplemental Material
sj-docx-1-jop-10.1177_02698811221090627 – Supplemental material for Effectiveness of vortioxetine in patients with major depressive disorder comorbid with generalized anxiety disorder: Results of the RECONNECT study
Supplemental material, sj-docx-1-jop-10.1177_02698811221090627 for Effectiveness of vortioxetine in patients with major depressive disorder comorbid with generalized anxiety disorder: Results of the RECONNECT study by Michael Cronquist Christensen, Simon Schmidt and Iria Grande in Journal of Psychopharmacology
Footnotes
Acknowledgements
Editorial assistance in the preparation of this manuscript was provided by Jennifer Coward of Piper Medical Communications, funded by H. Lundbeck A/S.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: M.C.C. and S.S. are employees of H. Lundbeck A/S. I.G. is an adviser, consultant and/or speaker for Lundbeck, Otsuka, Angelini, Casen Recordati, Ferrer and Janssen Cilag, and has received research funding from the Instituto de Salud Carlos III, Ministry of Economy and Competitiveness, Spain.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The RECONNECT study was funded by H. Lundbeck A/S.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article. The authors may be contacted for further data sharing.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
