Abstract
Background:
Clozapine formulation has been shown to affect plasma concentrations of clozapine and norclozapine. Changes in formulation might result in toxicity or treatment failure.
Methods:
We identified, from electronic records, patients who switched from clozapine tablets to oral liquid or vice versa and compared plasma concentrations before and after the switch.
Results:
We identified 13 patients with 85 blood samples who changed formulation of clozapine. Overall mean standardized clozapine plasma level was 0.67 mg/l daily dose on liquid and 0.87 mg/l daily dose on tablets (p = 0.035).
Conclusion:
Use of clozapine liquid results in lower plasma clozapine concentration than the same dose of tablets.
Introduction
Plasma concentration of clozapine is critical to both clinical efficacy and toxicity. A clear plasma level threshold has been documented with response being much more likely when clozapine exceeds 0.35–0.5 mg/l in plasma. 1 It is assumed that a reduction in plasma levels to below the therapeutic threshold leads to treatment failure.
Switching drug formulation is a common occurrence during treatment. There are various reasons for undertaking this approach. Examples include ensuring medication adherence, in patients with swallowing difficulties and individual patient preferences. In routine clinical practice, bioequivalence is usually assumed. However, one previous study has shown that plasma concentrations are lower in patients receiving oral liquid compared with those receiving tablets. 2 This study was prompted in part by anecdotal observations in practice that switching to oral liquid formulation may lead to psychotic exacerbation.
This study examined clozapine plasma levels in real patients who, for clinical reasons, underwent a change of clozapine formulation from liquid to tablet or tablet to liquid.
Method
We used the United Kingdom (UK) National Health Service ( NHS) online tool (http://www.hra-decisiontools.org.uk/research/) to determine whether our project constituted research requiring ethical approval. Our project was designated as not requiring submission or approval, but was seen as a service evaluation. Service improvement projects involving prescribing changes are considered by the South London and Maudsley NHS Foundation Trust Drugs and Therapeutics Committee, the locally designated committee for all noninterventional prescribing outcome surveys. The committee approved the project and did not consider it necessary to seek formal ethical committee approval because the prescribing change was to take place anyway and this project evaluated the safety of the change. Thus, patients were not required to give informed consent to the study. Their details were seen only by NHS professionals who would normally have access to these data and patient details were anonymized once data were collected and stored on a password-protected NHS computer. This retrospective observational study did not affect treatment choices or outcomes.
In our hospital, patients receiving clozapine oral tablets are registered with the Zaponex® Treatment Access System (ZTAS) and those receiving liquid clozapine with Denzapine® Monitoring Services (DMS). We searched the pharmacy Denzapine® system to identify patients switching from ZTAS to DMS or from DMS to ZTAS over the period January 2013 to April 2017. All patients known to have changed formulation were included.
Using laboratory and pharmacy records, clozapine/norclozapine plasma concentrations (mg/l) and clozapine daily dose at the time that blood samples were taken were recorded.
The switch date was identified. Clozapine and norclozapine levels were standardized as shown below:
Mean and standard deviations for standardized dose sizes, mean plasma concentration levels of clozapine and norclozapine for each patient, separately, were calculated using Microsoft Excel 2016. Means were compared by Student’s t tests and significance tested at the 5% level.
Results
The 13 participants in this study were all male, with an age range from 27 to 58 years old. Patient demographic data are shown in Table 1. All patients had been treated with clozapine for at least 1 year, with minimum of 2 months on each formulation. All patients in the study received a total daily dose of no more than 600 mg. All blood samples were taken between 11 h and 14 h after the previous clozapine dose.
Patient demographic data.
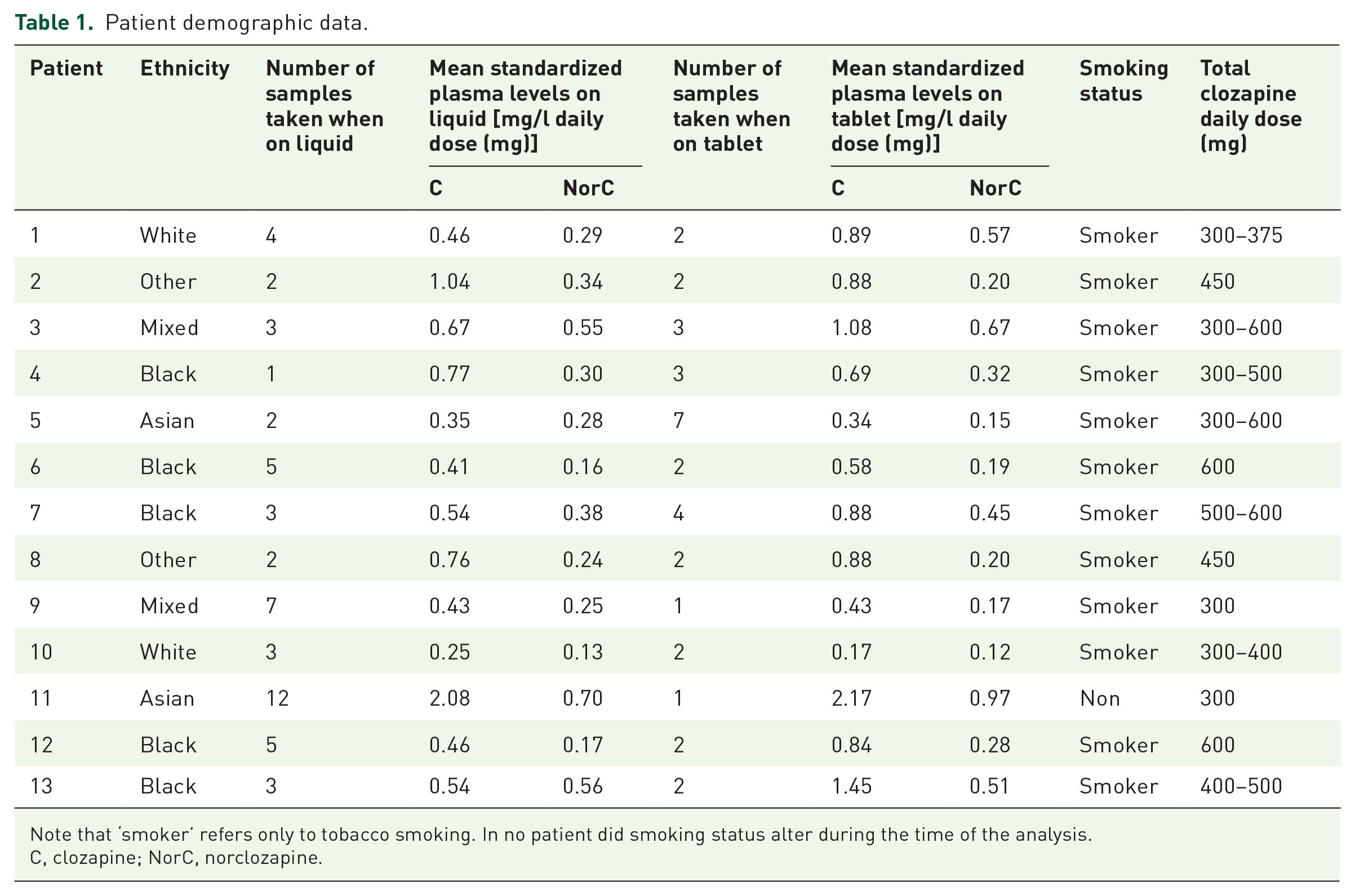
Note that ‘smoker’ refers only to tobacco smoking. In no patient did smoking status alter during the time of the analysis.
C, clozapine; NorC, norclozapine.
Figures 1 and 2 show the mean levels of clozapine and norclozapine when switches were made.
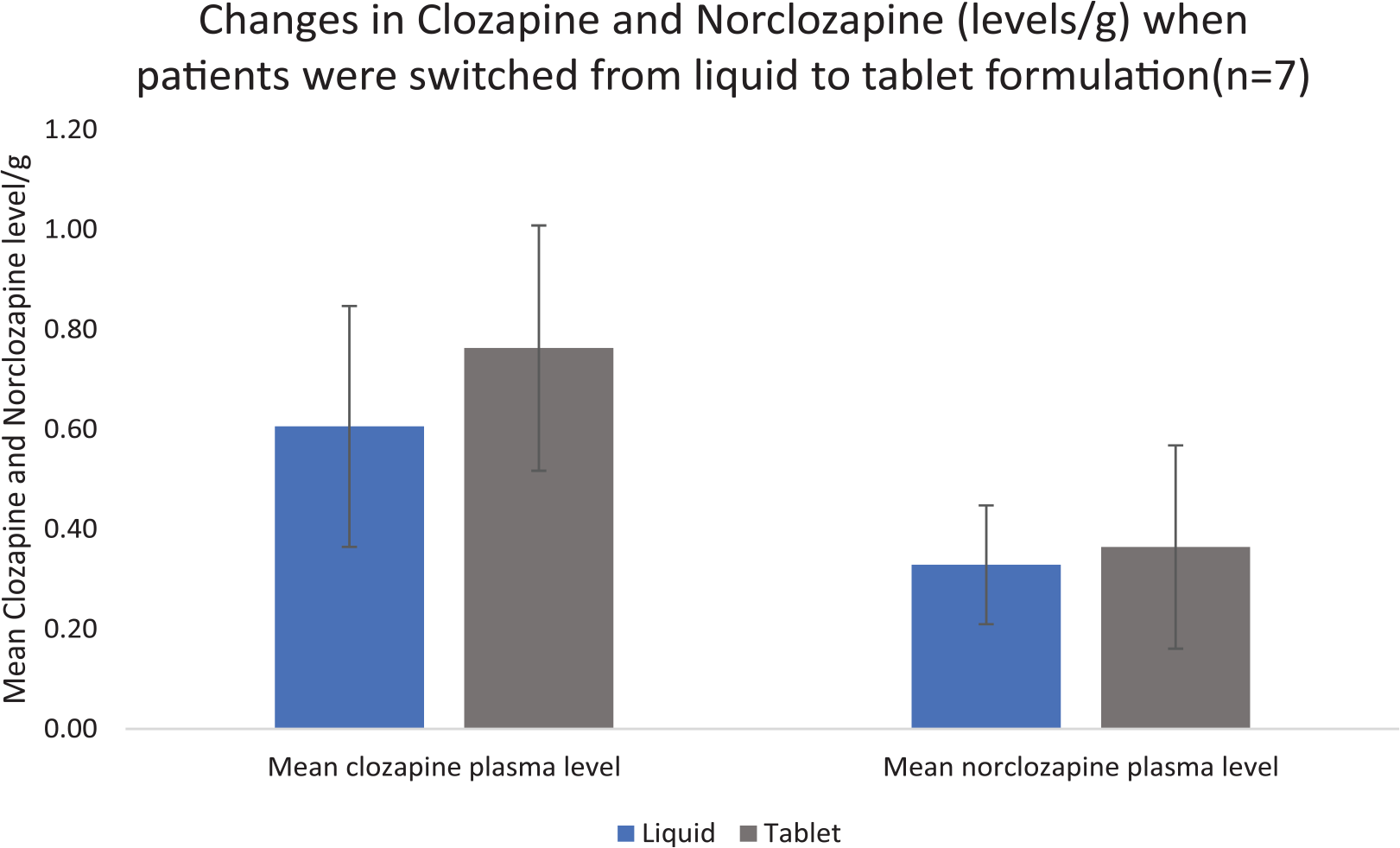
Standardized concentrations of clozapine and norclozapine for patients taking liquid clozapine who were switched to clozapine tablet.
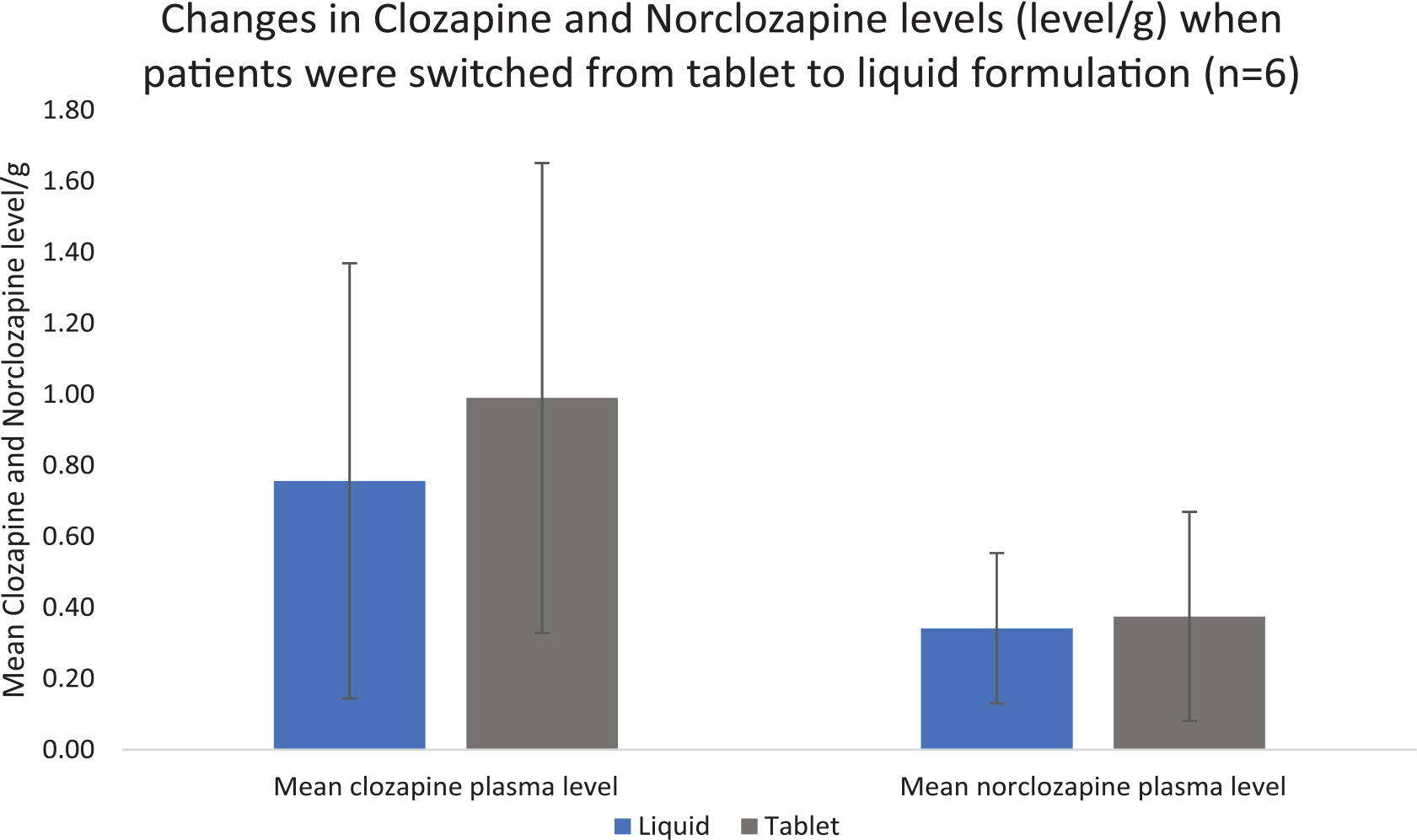
Standardized concentrations of clozapine and norclozapine for patients taking tablet clozapine who were switched to liquid clozapine.
The data set was further analysed using a Spaghetti diagram that showed trends for each patient (see Figures 3 and 4).

Spaghetti diagram showing individuals’ clozapine (left) and norclozapine (right) level changes when they were switched from liquid to tablet formulation, using standardized plasma concentrations.
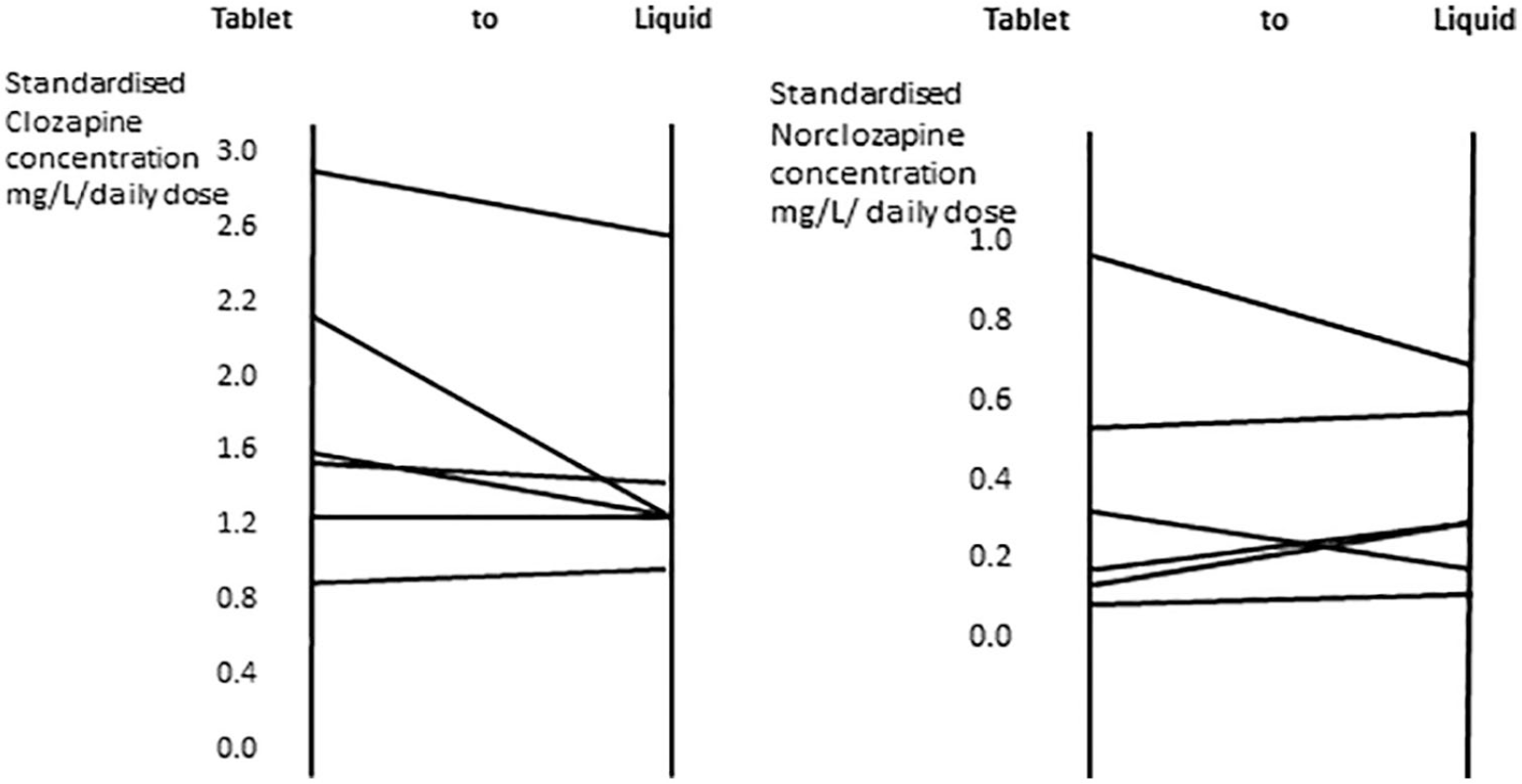
Spaghetti diagram showing individuals’ clozapine (left) and norclozapine (right) level changes when they were switched from tablet to liquid formulation, using standardized plasma concentration.
For the whole cohort (including both directions of switching) the mean dose-standardized clozapine concentration was 0.67 mg/l daily dose (mg) on liquid and 0.87 mg/l daily dose (mg) on tablets (p = 0.035).
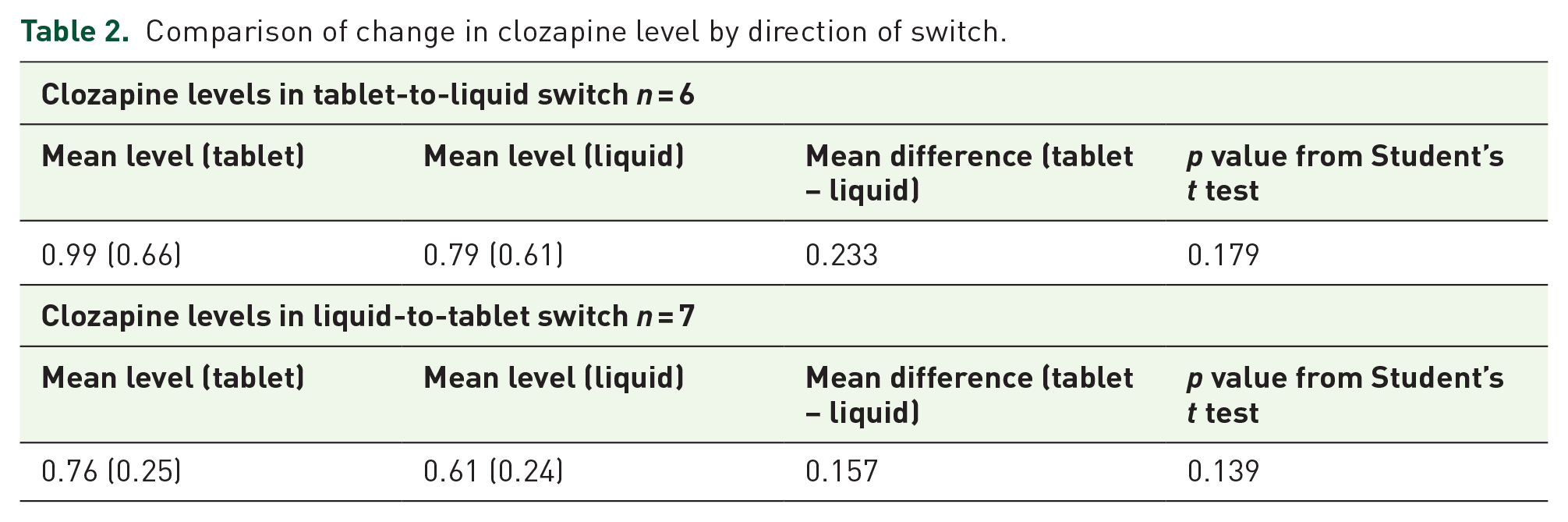
However, analysing the cohort separately according to direction of switch resulted in a nonsignificant change (see Table 2).
Comparison of change in clozapine level by direction of switch.
Standardized norclozapine levels were compared with the simultaneous standardized clozapine level, which is shown in Figure 5.

Dot plot showing all standardized clozapine and norclozapine concentrations for individuals during their treatment.
As seen from Figure 5, norclozapine levels show less variability following formulation changes.
The mean value of the clozapine:norclozapine ratio was calculated using the dose-standardized levels.
Discussion
In this study we found that the use of liquid clozapine led to lower plasma levels of clozapine and norclozapine, regardless of the direction of the switch, whether tablet to liquid or liquid to tablet. The change was less pronounced in norclozapine levels. Clozapine may be less well absorbed in liquid form. However, the liquid formulation is still a valuable method to tackle noncompliance and for those with difficulty in swallowing. 2
Liquid-to-tablet switch
In this group, the general trend was an increase in plasma clozapine levels. However, for some patients (patients 2, 4 and 5) this was not the case. The reason for this is not clear but erratic compliance is possible. A review of the patient notes revealed no obvious pharmacokinetic interactions.
Tablet-to-liquid switch
For those who were taking clozapine tablets and were switched to the liquid form of clozapine, the general pattern was a drop in plasma clozapine levels. Patient 10 was an exception to this general trend; he smoked cannabis and tobacco. This might explain why he had a lower plasma concentration before the switch; however, it is probable that supervised administration of clozapine liquid helped clozapine levels to increase following unsupervised use of tablets, where noncompliance might have been a realistic possibility.
Norclozapine has a longer half-life than its parent drug (clozapine) and is less affected by daily fluctuations than the parent molecule. In addition, sampling time is not as critical due to its longer half-life (see Figure 1).3,4 Different oral formulations of a drug often have different pharmacokinetic profiles. This may be because the excipients affect absorption. Also clozapine is not water soluble and so is presented as an oral suspension form. Thus, drug may not be delivered consistently when the bottle is not shaken well before administration. Thorough shaking is essential to redistribute the suspended particles. Inadequate shaking can lead initially to under dosing and later to overdosing.
There is evidence of plasma level changes after switching between different branded clozapine tablets. For instance a study by Tse and colleagues compared generic and branded clozapine tablets; [the generic clozapine is from Zenith Goldline Pharmaceuticals (Miami, FL, USA), whereas the original ‘brand’ Clozaril® is from Novartis (Basel, Switzerland)]. Switching between these two tablets led to concerns raised by clinicians and researchers because it was found that despite comparable mean plasma concentrations, the pharmacokinetic parameters (such as area under the time–plasma concentration curve) of the two tablets were different in almost 40% of patients. This brought up the issue of average bioequivalence versus individual bioequivalence and implications of interchange ability of the two formulations. This study concluded that if the switch is to be made between generic and brand clozapine tablets, careful patient monitoring is required. 5 Clearly, in this study, individual examples show that the switches do not meet the criteria for generic bioequivalence testing. If different brands of tablets can have different pharmacokinetic profiles then this supports the likelihood that tablet and liquid formulations will also produce different plasma concentrations.
Currently in the UK, the only clozapine liquid formulation available is Denzapine® 50 mg/ml. The Medicines and Healthcare Products Regulatory Agency (MHRA), granted Genus Pharmaceuticals Limited (Newbury, Berks.) Product Licence based on a bioequivalence test between Clozaril® tablets 25 mg and Denzapine® suspension (equivalent to 12.5 mg clozapine) in 24 healthy volunteers in the fasting state. The results were deemed acceptable.6–9 Nonetheless, the MHRA study may not reflect the real-life results because psychotic patients take higher doses of clozapine (in the present study at least 300 mg clozapine every day), most of whom are likely to be taking other drugs. They mostly also smoked and drank alcohol (excessively), quite apart from taking much higher doses of clozapine. These factors and our findings suggest that the existing bioequivalence presumption is not secure.
Pharmacokinetic interactions have an important effect on clozapine plasma levels;10,11 however, we found no suggestions of important drug interactions.
Limitations
The small sample size is perhaps the biggest limitation of this study. It is also likely that blood samples were only drawn at times approximating to trough plasma concentrations. However, recent evidence suggests sampling time (within limits) is not critical to clozapine therapeutic drug monitoring. 12
Another limiting factor was that genetic data were not collected for these patients. For example, there are slow and fast metabolizers of CYP 1A2 and metabolic status critically affects clozapine levels but this was beyond the scopes of this study. However, it should be noted that each participant acted as their own control. Smoking status, medication, caffeine (a CYP 1A2 substrate) consumption and illicit drug use (given their enzyme-inducing properties) at the time of clozapine formulation switch, could also be confounders, although we took steps to reduce their influence.11,13,14 Smoking status did not change, neither were there any reports of changes in caffeine intake. During the period, we kept records of all medication changes. There were no changes with the potential to affect clozapine pharmacokinetics.
Conclusion
From this small sample of patients on clozapine treatment, switching formulation has the potential to result in changes in plasma levels. Switching from tablet to liquid formulation at the same dose may give rise to lower plasma levels. The reasons for this are not clear but oral formulation does seem to affect absorption. We recommend that plasma clozapine levels and patient’s clinical status be monitored closely following a formulation switch.
Footnotes
Acknowledgements
The authors of the study would like to thank all members of the pharmacy department at Maudsley Hospital, UK.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declare that there is no conflict of interest.

