Abstract
Background:
There have been scarce data on the distribution of clozapine concentrations in comparison with the recommended range (350–600 ng/ml) or their relationship with side effects among patients with treatment-resistant schizophrenia. Furthermore, no studies have assessed the association between side effects and overall exposure to the drug by calculating the 24-h area-under-curve (AUC).
Methods:
In- and outpatients with schizophrenia or schizoaffective disorder (ICD-10) who were receiving a stable dose of clozapine for ⩾2 weeks were included. Side effects were assessed using the Glasgow antipsychotic side-effects scale for clozapine (GASS-C). Using two collected plasma samples, plasma clozapine and norclozapine concentrations at peak and trough and their 24-h AUC were estimated using population pharmacokinetic models.
Results:
A total of 108 patients completed the study (mean ± SD age, 43.0 ± 10.1 years; clozapine dose, 357.5 ± 136.9 mg/day); 33 patients (30.6%) showed estimated trough concentrations of clozapine within the recommended range (350–600 ng/ml) whereas the concentrations were higher and lower than this range among 37 (43.5%) and 28 (25.9%) patients (%), respectively. There were no significant correlations between estimated peak or trough concentrations or 24-h AUC of both clozapine or norclozapine, and GASS-C total or individual scores. No significant differences were found between GASS-C total or individual item scores between the patients with estimated trough concentrations of clozapine of >600 ng/ml and the other subjects.
Conclusion:
The results suggest that clozapine or norclozapine concentrations are not linked directly to the extent of side effects experienced in clozapine-treated patients with treatment-resistant schizophrenia while the cross-sectional study design limits the interpretation of any causal relationships. These findings indicate that side effects associated with clozapine may occur at any dose or concentration.
Introduction
Antipsychotic drugs have played an important role in the treatment of schizophrenia for more than 60 years. Unfortunately, approximately one-third of patients with schizophrenia do not respond to these drugs,1,2 which is referred to as treatment resistance. Clozapine is the only antipsychotic drug that is indicated for treatment-resistant schizophrenia (TRS). While this drug shows consistently superior efficacy over other antipsychotic medications, 3 it causes a variety of side effects in a dose-dependent fashion, including gastrointestinal hypomotility, 4 QTc prolongation,5,6 sedation, 7 hypersalivation, 8 changes on electroencephalogram, 9 and seizure. 10 Besides, use of clozapine is known to be associated with an increased risk of pneumonia. 11 Therefore, routine therapeutic drug monitoring (TDM) is recommended for this drug to minimize such dose-dependent adverse events. 12
One major TDM guideline by the Arbeitsgemeinschaft für Neuropsychopharmakologie und Pharmakopsychiatrie (AGNP) recommend a range of 350–600 ng/ml in trough clozapine blood concentrations for optimal treatment. 13 Since the recommendation of this guideline is based mainly on data from Europe and North America, differences in pharmacokinetic and pharmacodynamic parameters among races should be noted. In fact, Asian patients present with higher blood clozapine concentrations than Caucasian patients. For example, Chong and colleagues reported higher plasma clozapine concentrations in 14 Chinese patients than 59 American patients (mean ± SD, 653 ± 356 ng/ml versus 371 ± 257 ng/ml) although they were receiving comparable doses (mean ± SD, 309 ± 52 mg/day versus 444 ± 188 mg/day). 14 Similarly, Ng and colleagues found comparable plasma clozapine concentrations between Asian (n = 20) and Caucasian patients (n = 20) (mean ± SD, 417.1 ± 290.8 ng/ml versus 415.3 ± 185.8 ng/ml) despite lower doses given to the Asian patients (mean ± SD, 175.6 ± 106.9 mg/day versus 432.5 ± 194.7 mg/day, p < 0.001). 15 In addition, Asian patients were reported to experience adverse events of clozapine such as anticholinergic side effects more frequently in one small study [Korean-American (n = 17) versus Caucasian (n = 7)]. 16 Thus, it is critically important to evaluate the associations between clozapine dose, blood concentrations, and side effects in the ethnic group of focus. Clozapine is metabolized mainly by hepatic enzymes CYP1A2 and excreted in urine and feces.17,18 The principal plasma metabolites are norclozapine and clozapine N-oxide; norclozapine has a lower affinity for D1 receptor than clozapine and a similar affinity for D2 and serotonin 5-HT2 receptors in comparison with clozapine, 19 whereas clozapine N-oxide has little pharmacological activity. 20 Moreover, the proportion of clozapine and norclozapine likely widely differs among individuals due to differences in metabolic parameters. Therefore, both clozapine and norclozapine concentrations should be considered when their association with clinical effects are evaluated. 13 Furthermore, previous studies have assessed concentrations of clozapine at trough, but not overall exposure to the drug by calculating the 24-h area-under-curve (AUC).
We therefore conducted a cross-sectional study to examine the relationship between clozapine dose, plasma concentrations of clozapine and norclozapine, and side effects in Japanese patients with TRS. All patients were receiving treatment with clozapine at Yamanashi Prefectural Kita Hospital or Okayama Psychiatric Medical Center, Japan. These two hospitals were ranked as fourth and third in Japan in regard to the number of patients registered with the Clozaril Patient Monitoring Service (CPMS) in 2017, respectively. 21 To comprehensively assess the exposure to clozapine, a population pharmacokinetic model was utilized to estimate peak and trough concentrations of clozapine and norclozapine and their 24-h AUC. We hypothesized that greater peak and trough concentration would be associated with greater severity and frequency of side effects, and greater 24-h AUC would be related to side effects such as drowsiness and sedation.
Methods
Subjects
In- and outpatients with a diagnosis of schizophrenia or schizoaffective disorder according to the International Classification of Diseases, 10th Edition (ICD-10) (World Health Organization, 1992) who were receiving a stable dose of clozapine for at least 2 weeks, 20 years of age or older, and capable of providing informed consent were included. This study was conducted at Yamanashi Prefectural Kita Hospital and Okayama Psychiatric Medical Center, Japan from September 2018 to June 2019. A series of consecutive patients who fulfilled the inclusion criteria during the study period were approached for participation in the present study. This study was approved by institutional review boards of each hospital, and all subjects provided written informed consent after detailed explanation of the study.
Assessments
The subjects received assessments with the clinical global impression-severity scale (CGI-S), 22 the brief evaluation of psychosis symptom domains (BE-PSD), 23 and the visual analogue scale for distress associated with symptoms - Japanese version (VAS-DAS-J) for symptomatology. 24 Global functioning was assessed using the global assessment of functioning (GAF), 25 and well-being was assessed with the subjective well-being under neuroleptic scale – short form (SWN-K), Japanese version.26,27 Furthermore, clozapine-related side effects were assessed using the Glasgow antipsychotic side-effects scale for clozapine (GASS-C). 28 The BE-PSD is designed to assess five symptom domains (i.e., psychotic symptoms, disorganized thinking, negative symptoms, excitement/mania, and depression/anxiety) on a 7-point (i.e., 0–6) scale according to operational criteria, 23 a higher score indicates greater severity. The GASS-C consists of 16 questions which evaluate 12 domains of common side effects as follows: (1–2) drowsiness and sedation, (3) orthostatic hypotension, (4) tachycardia, (5) myoclonus, (6) hypersalivation, (7–8) anticholinergic side effects, (9–10) gastrointestinal side effects, (11) constipation, (12) nocturnal enuresis, (13–14) diabetes screening, (15) weight gain, and (16) sexual dysfunction.28,29 Each question is scored on a 4-point (i.e., 0–3) scale with higher scores indicating greater frequency of side effects during the past week.
The following information was also collected: sex, age, height, weight, treatment setting (i.e., inpatient or outpatient), smoking status, medications prescribed, duration of clozapine treatment, and ethnicity.
A population pharmacokinetic model was used for estimation of peak and trough plasma concentrations of clozapine and norclozapine and their 24-h AUC. One of the major advantages of the population pharmacokinetic model over the traditional pharmacokinetic model is that this approach allows estimation based on the use of sparsely and randomly sampled data, compared with the traditional method in which only intensive, precisely timed data is collected and used. 30 In the present study, we therefore collected blood samples at two points in time on different days for the measurement of plasma clozapine and norclozapine concentrations. Dates and times of clozapine doses for the past 24 h and blood draw were recorded for population pharmacokinetic analysis. Plasma concentrations of clozapine and norclozapine were assayed in heparinized plasma using liquid chromatography with a limit of quantitation of 100 ng/ml.
Measurement of plasma clozapine and norclozapine concentrations
Sample preparation consisted of a liquid–liquid extraction. To 0.5 ml of plasma in a 10 ml borosilicate glass tube (105 × 15 mm) were added 0.5 ml of 0.2 M NaOH and 0.1 ml of the internal standard loxapine (12.5 µg/ml). The plasma was mixed with a hand vortex mixer (Tube Mixer, As One, Osaka, Japan). Then, 3 ml of chloroform (99.0%)-heptane (99.0%) (30:70, v:v) was added to the preparation and shaken with a vortex mixer (Vortex-Genie2, Scientific Industries, Bohemia, NY, USA) for 3 min. After shaking, the tubes were centrifuged in a table-top centrifuge (Tabletop Refrigerated Centrifuge 2800, KUBOTA, Tokyo, Japan) at 3000 rpm for 5 min. The organic layer was collected in a 10 ml borosilicate glass tube (105 × 15 mm). The organic layer was then evaporated under a gentle stream of nitrogen at 45°C using thermostatic bath (Dry Thermo Unit, TAITEC, Koshigaya City, Saitama, Japan). The residue was dissolved in 0.2 ml of solvent (acetate buffer: acetonitrile = 85:15, v:v), and finally injected into the HPLC system. The injection volume was 150 µl. Clinical personnel collected 5 ml blood samples from patients. Laboratory personnel centrifuged the samples, collected the plasma, and stored the plasma at −20°C. Plasma analyte levels were measured with a high-performance liquid chromatography (HPLC) system featuring ultraviolet detection at 254 nm with loxapine as the internal standard. The HPLC set-up consisted of a 1260 Infinity LC system and an OpenLAB CDS EZChrom Edition chromatography data system (Agilent Technologies, Santa Clara, CA, USA). Fluorinated silica based column (Wakopak® Fluofix-II, FUJIFILM Wako Pure Chemical Co., Osaka, Japan) was used as the solid phase. The mobile phase was composed of acetate buffer and acetonitrile (50:50). Equation of the non-weighted linear regression curve was used for assay calibration (clozapine, y = ax+b, a = 0.572873, b = 0.0121475, R2 = 0.999195; norclozapine, y = ax+b, a = 0.652149, b = 0.0176808, R2 = 0.995656). Intra-assay coefficient of variation (CV) and inter-assay CV were <5% and <9% for clozapine and <4% and <11% for norclozapine, respectively.
Statistical analysis
SPSS version 24.0 (SPSS Inc, Chicago, IL, USA) was used for statistical analysis. NONMEM Version 7.4.3 (ICON Plc, Leopardstown, Ireland) was used for population pharmacokinetics analysis. The population pharmacokinetic model used in the analysis was previously published elsewhere. 30 Briefly, disposition of both clozapine and norclozapine was described with a one-compartment structural model with first-order absorption of clozapine. Elimination of both clozapine and norclozapine was modeled with first-order kinetics, with the assumption of complete conversion of clozapine to norclozapine. Covariate effects were included for clearance of both substances, with smoking status and sex demonstrating proportional and additive effects, respectively. Estimates of peak and trough concentrations were taken from fitted concentration-time profiles, followed by non-compartmental analysis and calculation of 24-h AUCs in R using the open-source package PKNCA.31,32 AUCs were calculated using the linear trapezoidal method for increasing concentrations and the logarithmic trapezoidal method for decreasing concentrations, commonly referred to as the “linear-up log-down” method. Correlations between peak and trough concentrations and 24-h AUC of clozapine or norclozapine and GASS-C total or individual item scores were examined using Spearman’s rank correlation. In addition, peak and trough concentrations and 24-h AUC of clozapine or norclozapine were compared using the analysis of variance (ANOVA) among the following three groups according to severity classification detailed in the GASS-C manual: patients with a GASS-C total score of 0–16 (mild), those with a score of 17–32 (moderate), and those with a score of 33–48 (severe). 28
In addition, GASS-C total and individual item scores were compared between patients whose estimated trough concentrations of clozapine were above the upper limit of the recommended therapeutic concentration (600 ng/ml) 13 and the remaining patients using Student’s t test.
A p value of <0.05 (two-tailed) was considered to indicate statistical significance. Bonferroni correction was applied for multiple comparisons with p values of <0.05/13 (i.e., 12 individual items and total score in the GASS-C) being considered statistically significant.
Results
A total of 112 subjects participated in this study; 4 of them withdrew their consent. Thus, data from 108 subjects were used for the analyses. Clinical and demographic characteristics are shown in Table 1. The mean duration of clozapine treatment was approximately 4 years, three-quarters were outpatients, and the cohort was represented by moderate to markedly ill severity of illness. Measured concentrations as well as estimated peak and trough concentrations and 24-h AUC of clozapine and norclozapine are shown in Table 2. Population pharmacokinetic parameters are summarized in Supplemental Table S1. Clearance of clozapine and norclozapine was greater among smokers and males. The predictive performance of the population pharmacokinetic model is shown in Supplemental Figure S2. A total of 33 patients (30.6%) showed estimated trough clozapine concentrations that were within the recommended therapeutic range of 350–600 ng/ml. Estimated trough concentrations of clozapine were above the upper limit of the recommended range (i.e., 600 ng/ml) among more than 40% of the patients, while it was below the range (i.e., <350 ng/ml) among 26% of the patients.
Clinical and demographic characteristics of the patients (n = 108).
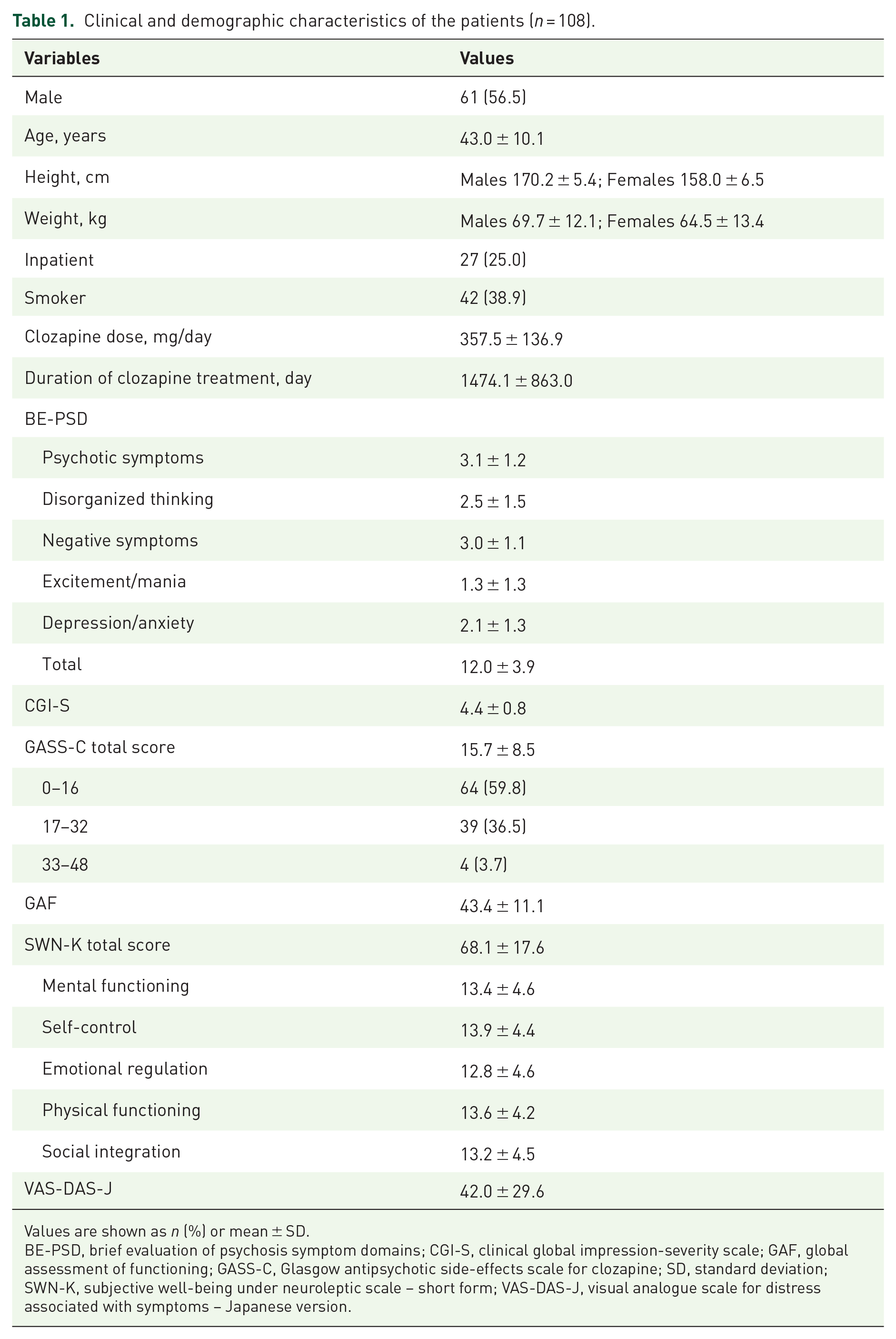
Values are shown as n (%) or mean ± SD.
BE-PSD, brief evaluation of psychosis symptom domains; CGI-S, clinical global impression-severity scale; GAF, global assessment of functioning; GASS-C, Glasgow antipsychotic side-effects scale for clozapine; SD, standard deviation; SWN-K, subjective well-being under neuroleptic scale – short form; VAS-DAS-J, visual analogue scale for distress associated with symptoms – Japanese version.
Plasma concentrations of clozapine and norclozapine.

Values are shown as n (%), mean ± SD (range), or mean ± SD.
AUC, area under the curve; SD, standard deviation.
No significant correlation was found between any of estimated peak and trough concentrations and 24-h AUC of clozapine or norclozapine and GASS-C total or individual item scores (Table 3). Moreover, there were no differences in any of peak and trough concentrations and 24-h AUC of clozapine or norclozapine among the three groups according to the GASS-C total score (Table 4). The distribution of GASS-C total scores and concentrations of clozapine is shown in Supplemental Figure 1.
Correlations between peak and trough concentrations and 24-h AUC of clozapine or norclozapine and GASS-C total or individual item scores.

AUC, area under the curve; GASS-C, Glasgow antipsychotic side-effects scale for clozapine.
Concentrations of clozapine and norclozapine among three groups according to GASS-C total score.
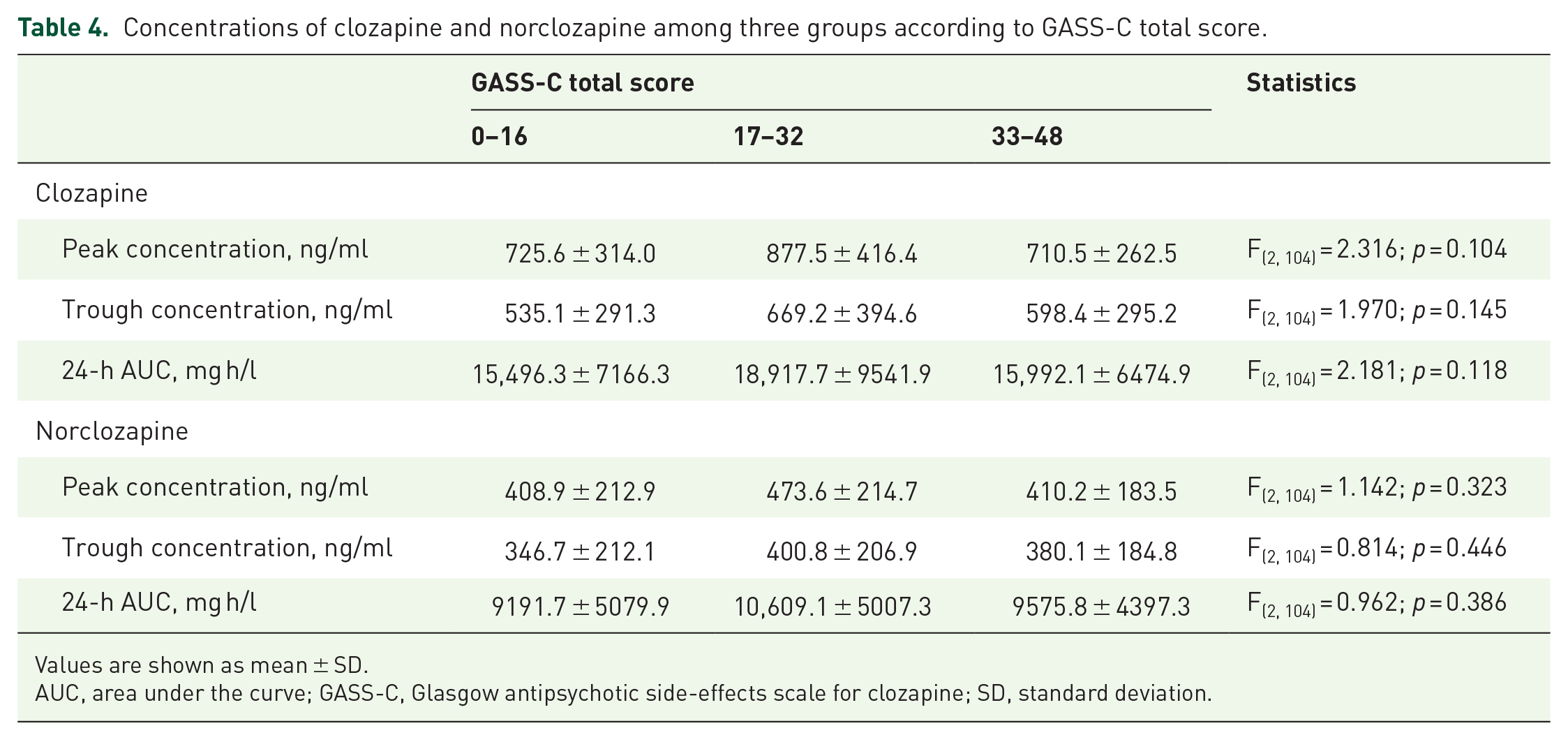
Values are shown as mean ± SD.
AUC, area under the curve; GASS-C, Glasgow antipsychotic side-effects scale for clozapine; SD, standard deviation.
No significant differences were found between GASS-C total or individual item scores between the patients with estimated trough concentrations of clozapine of >600 ng/ml and other subjects (Table 5).
GASS-C scores among patients whose estimated trough concentrations of clozapine of ⩽600 ng/ml and >600 ng/ml.

Values are shown as mean ± SD.
GASS-C, Glasgow antipsychotic side-effects scale for clozapine.
Discussion
In this cross-sectional study, concentrations of clozapine and norclozapine were comprehensively evaluated at two major facilities that provide clozapine treatment in Japan. Approximately 30% of the patients showed estimated trough concentrations of clozapine within the recommended range, whereas the concentrations were higher and lower than this range among 44% and 26% of them, respectively. With regard to the association between drug concentrations and side effects, no significant findings were observed. Collectively, our findings suggest that clozapine or norclozapine concentrations are not directly linked to the extent of side effects experienced in Japanese patients on clozapine treatment.
Our finding that only 30.6% of patients had estimated trough clozapine levels within the recommended therapeutic range (i.e., 350–600 ng/ml) is in line with previous research. The large clozapine TDM data in the UK and Ireland (26,796 patients with 104,127 plasma concentrations) showed that 29.3%, 42.5%, and 28.3% of the concentrations were within, below, and above the recommended range, respectively. 12 According to another TDM study in Belgium, 22.3% of a total of 330 serum trough concentrations collected from 131 patients were within the recommended range, while 67.9% and 9.8% showed values below and above this range, respectively. 33 In a report by Suhas and colleagues on serum trough clozapine concentrations among 142 patients in India, the concentrations were within, below, and above the recommended range among 30.3%, 20.4%, and 49.3% of them. 34 Thus, approximately 20–30% of the samples have been reported to be within the recommended range regardless of geographical region, although samples were taken more than once in some patients in the former two studies.12,33 On the other hand, the proportion of patients presenting with higher concentrations seems greater among Asian patients, as shown in our study and in Indian patients. 34 This may indicate that they require a higher concentration for successful treatment, or that they are excessively dosed in clinical practice.
Contrary to our hypothesis, we did not find a direct relationship between estimated peak, trough concentrations or 24-h AUC of clozapine or norclozapine and side effects. Yusufi and colleagues examined the relationships between side effects and plasma trough concentrations in 103 patients receiving maintenance clozapine treatment in their cross-sectional study (mean ± SD dose, 456 ± 209 mg/day; mean ± SD clozapine concentration, 530 ± 370 ng/ml; median duration of clozapine treatment, 30 months). 35 They found a weak correlation between plasma clozapine concentrations and total scores (r = 0.29, p < 0.004) as well as the number of moderate to severe side effects (r = 0.23, p < 0.03), assessed with the antipsychotic non-neurological side effects rating scale (ANNSERS). Olesen and colleagues found a significant but weak correlation between the UKU side effect scale score and serum trough clozapine concentration in 30 patients with chronic schizophrenia (r = 0.36, p < 0.05) in their cross-sectional study [median (interquartile range; IQR) dose, 350 (228–425) mg/day; median (IQR) clozapine concentration, 351.8 (230–615) ng/ml; mean ± SD duration of clozapine treatment, 5.0 ± 4.4 years]. 36 However, other studies have not shown a clear association between clozapine concentrations and side effects.37 –39 There are several factors that may mask a relationship between clozapine concentrations and side effects. First, the number of subjects included in this study was relatively small. Moreover, the small number of subjects who showed above 1000 ng/ml of clozapine or had a GASS-C score above 33 may have resulted in no correlation between clozapine plasma concentrations and side effects that we observed in this study. Second, it is likely that a subgroup of individuals is sensitive to clozapine, and clinicians tend to prescribe lower doses to these patients in response to the observed clinical sensitivity. Such a possibility was referred to in a previous cross-sectional brain imaging study in which patients with late-life schizophrenia who presented with extrapyramidal symptoms paradoxically showed a lower dopamine D2 receptor occupancy than those who did not. 40 Third, the relatively long duration of clozapine treatment of approximately 4 years, that is, stable patients should be acknowledged since some side effects of clozapine could arise more frequently from faster dose titration of clozapine dose or during the earlier phase of its treatment.9,41 Tolerance may have developed to various side effects on chronic use of clozapine. The relationship between clozapine concentrations and side effects may be clearer in earlier treatment stages. Last, some side effects may be managed effectively with life-style interventions (e.g., high fiber diet for constipation) or concomitant medications (e.g., seizure prophylaxis with anticonvulsants). These confounders should be documented and accounted for in future research.
In terms of clinical implications, our findings indicate that plasma concentrations of clozapine and norclozapine may not be always useful in assessing the side effect burden of clozapine treatment, at least during the maintenance phase. Clinicians should be aware that clozapine-related side effects can occur at any dose or concentration as shown in Supplemental Figure S1. On the other hand, as clozapine is associated with a number of side effects, especially during the initiation phase, future research should examine the utility of measuring clozapine concentrations to manage side effects during early stages of treatment. Furthermore, our study was carried out in clinically stable patients and therefore does not negate the value of assessing clozapine levels in patients presenting with toxicity. Lastly, greater clearance of clozapine and norclozapine among smokers and males indicates that they would need higher clozapine doses to reach appropriate plasma concentrations.
The main strength of this study is the use of the population pharmacokinetic model. While previous studies evaluated mainly trough concentrations, we comprehensively evaluated the association between estimated peak, trough concentrations, and 24-h AUC of clozapine and norclozapine and side effects. However, there are some limitations to be noted in this study. First, due to the nature of cross-sectional study design, any causal relationship could not be determined. Second, adherence to clozapine treatment was not evaluated in this study. In addition, concomitant medications such as laxatives and anticholinergic drugs were not assessed, which could have confounded the assessment of side effects. Third, the GASS-C does not comprehensively assess all potential side effects of clozapine. For example, it does not include evaluation of electroencephalogram abnormalities or seizures although they are common side effects of clozapine.
To conclude, we found no significant associations between estimated clozapine or norclozapine concentrations and adverse events among Japanese patients with treatment-resistant schizophrenia. While our cross-sectional study limits interpretation of any causal relationships, our findings suggest that adverse events associated with the use of clozapine could occur at any dose or concentration. These preliminary observations should be confirmed in prospective studies accounting for duration of clozapine treatment and concomitant medications. Clozapine can be associated with various side effects even at low concentrations, highlighting the importance of timely and comprehensive assessments of adverse events when on this medication.
Supplemental Material
sj-pdf-1-tpp-10.1177_20451253211016189 – Supplemental material for Comprehensive assessment of exposure to clozapine in association with side effects among patients with treatment-resistant schizophrenia: a population pharmacokinetic study
Supplemental material, sj-pdf-1-tpp-10.1177_20451253211016189 for Comprehensive assessment of exposure to clozapine in association with side effects among patients with treatment-resistant schizophrenia: a population pharmacokinetic study by Nobuyuki Nomura, Kohei Kitagawa, Ryuhei So, Fuminari Misawa, Masafumi Kodama, Hiroyoshi Takeuchi, Robert Bies, Thomas Straubinger, Christopher Banker, Yuya Mizuno, Masaru Mimura and Hiroyuki Uchida in Therapeutic Advances in Psychopharmacology
Supplemental Material
sj-pdf-2-tpp-10.1177_20451253211016189 – Supplemental material for Comprehensive assessment of exposure to clozapine in association with side effects among patients with treatment-resistant schizophrenia: a population pharmacokinetic study
Supplemental material, sj-pdf-2-tpp-10.1177_20451253211016189 for Comprehensive assessment of exposure to clozapine in association with side effects among patients with treatment-resistant schizophrenia: a population pharmacokinetic study by Nobuyuki Nomura, Kohei Kitagawa, Ryuhei So, Fuminari Misawa, Masafumi Kodama, Hiroyoshi Takeuchi, Robert Bies, Thomas Straubinger, Christopher Banker, Yuya Mizuno, Masaru Mimura and Hiroyuki Uchida in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
The authors are grateful to Bunta Yoshimura for his advice for this work.
Conflict of interest statement
N. Nomura has received speaker’s honoraria from Dainippon-Sumitomo, Meiji-Seika, and Otsuka Pharma.
K. Kitagawa report no additional financial or other relationship relevant to this article.
R. So has received Health and Labor Sciences Research Grant; the research grant from the Mental Health Okamoto Memorial Foundation; personal fees from Igaku-shoin Co., Ltd., Kagaku-hyoronsha Co., Ltd., Medical Review Co., Ltd., Otsuka Pharmaceutical Co., Ltd., and CureApp Inc. outside the submitted work.
F. Misawa has received speaker’s honoraria from Dainippon-Sumitomo, Eli Lilly, Janssen, Novartis, Otsuka, and Pfizer.
M. Kodama report no additional financial or other relationship relevant to this article.
H. Takeuchi has received research grants from Japan Society for the Promotion of Science (JSPS), Japan Agency for Medical Research and Development (AMED), SENSHIN Medical Research Foundation, and Novartis Pharma; fellowship grants from Astellas Foundation for Research on Metabolic Disorders, the Canadian Institutes of Health Research (CIHR), Centre for Addiction and Mental Health (CAMH) Foundation, and the Japanese Society of Clinical Neuropsychopharmacology (JSCNP); speaker’s honoraria from Kyowa, Janssen, Meiji-Seika, Mochida, Otsuka, Dainippon-Sumitomo Pharma, and Yoshitomiyakuhin; and manuscript fees from Dainippon-Sumitomo Pharma.
R. Bies has served as an expert consultant to Belmore Neidrauer LLP on behalf of Janssen Pharmaceuticals.
Y. Mizuno is supported by fellowship grants from Japan Society for the Promotion of Science, Astellas Foundation for Research on Metabolic Disorders, Japanese Society of Clinical Neuropsychopharmacology, and Mochida Memorial Foundation for Medical and Pharmaceutical Research, and has received manuscript fees from Dainippon-Sumitomo Pharma, and consultant fees from Bracket and MedAvante-ProPhase within the past 3 years.
M. Mimura has received speaker’s honoraria from Byer Pharmaceutical, Daiichi Sankyo, Dainippon-Sumitomo Pharma, Eisai, Eli Lilly, Fuji Film RI Pharma, Hisamitsu Pharmaceutical, Janssen Pharmaceutical, Kyowa Pharmaceutical, Mochida Pharmaceutical, MSD, Mylan EPD, Nihon Medi-physics, Nippon Chemipher, Novartis Pharma, Ono Yakuhin, Otsuka Pharmaceutical, Pfizer, Santen Pharmaceutical, Shire Japan, Takeda Yakuhin, Tsumura, and Yoshitomi Yakuhin within the past 3 years. Also, he received grants from Daiichi Sankyo, Eisai, Pfizer, Shionogi, Takeda, Tanabe Mitsubishi and Tsumura within the past 3 years outside the submitted work.
H. Uchida has received grants from Eisai, Otsuka Pharmaceutical, Dainippon-Sumitomo Pharma, and Meiji-Seika Pharma; speaker’s honoraria from Otsuka Pharmaceutical, Dainippon-Sumitomo Pharma, Eisai, and Meiji-Seika Pharma; and advisory panel payments from Dainippon-Sumitomo Pharma within the past 3 years.
Other authors have nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially supported by the Inokashira Hospital grant for psychiatry research and Kobayashi Magobe Memorial Medical Foundation, Japan.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
