Abstract
Background:
Optimal use of lithium involves adjustment of the dose, to keep the plasma level within the narrow, recommended range. Brand-specific prescribing has long been considered critical to achieving this aim, but this is a convention based on very limited data.
Objectives:
To explore the effect of selected demographic and clinical factors on the relationship between lithium dose and plasma level and determine whether there is an independent effect of lithium brand.
Design:
Analysis of clinical audit data collected in 2023 as part of a quality improvement programme addressing the use of lithium, conducted by the Prescribing Observatory for Mental Health.
Methods:
Data were collected from clinical records using a bespoke proforma, submitted online and analysed using SPSS.
Results:
Data were submitted for 4405 patients who had been prescribed solid-dosage formulations of lithium for more than a year. Priadel® was prescribed for 3722 (84%) of these patients, Camcolit® for 112 (2.5%) and the prescription was written generically for 554 (12.5%). Compared with Priadel, where Camcolit was prescribed, the mean daily dose was 10% higher and the mean plasma lithium level was 11% higher. A multivariable analysis was conducted to explore the relationship between selected clinical variables and maintenance lithium dose. This found that in 4213 patients whose most recent plasma lithium level was between 0.3 and 1.19 mmol/L, the variables age, sex, ethnicity, psychiatric diagnosis and the severity of chronic kidney disease were independently associated with dose while the brand of lithium prescribed was not.
Conclusion:
Our findings replicate those of previous studies with respect to the demographic and clinical variables that can be expected to influence lithium dosage in routine clinical practice. This reinforces the need to titrate the dosage for each individual patient, to achieve and maintain the target plasma level. However, the findings suggest that the Priadel and Camcolit brands of lithium are essentially interchangeable.
Introduction
Evidence-based clinical guidelines recommend lithium as a first-line treatment for the prophylaxis of bipolar disorder and as a treatment option for mania1,2 and refractory unipolar depression.3,4 Lithium has a narrow therapeutic range; plasma levels below 0.4 mmol/L are unlikely to be effective, the majority of lithium responders will require a plasma level somewhere between 0.6 and 1.0 mmol/L, and for a very small proportion of patients, a plasma level of 1.0 mmol/L or slightly above may be needed. However, side effects become increasingly common at levels above 0.8 and certainly 1.0 mmol/L.5,6 Levels above 1.5 mmol/L are generally considered to be toxic, requiring urgent intervention. 5 Regular monitoring of plasma lithium level is therefore mandated for all patients who are prescribed this medication so that, with appropriate dosage adjustment the plasma lithium level can be maintained within the narrow recommended range.
Given that tight control of plasma lithium levels is important for maximising efficacy and minimising side effects, any pharmacokinetic differences between brands could potentially compromise the efficacy and/or tolerability of lithium treatment. Given the paucity of pharmacokinetic data comparing different brands of lithium, the Summaries of Product Characteristics (SmPCs) for all formulations recommend that brands should not be switched. 7 Clinical reference sources reinforce this point. 8 Therefore, brand-specific supply chain problems, such as the planned discontinuation of Priadel® in 2020, 9 might be expected to prompt widespread concern about how best to manage the mass-switching of patients from one brand of lithium to another.
We have previously reported on a case series of pre- and post-switch plasma lithium levels in 37 patients switched from solid-dosage formulations of Priadel® to Camcolit® in routine clinical practice. 10 Where the dosage remained the same and the clinical team did not suspect problems with adherence, plasma lithium levels were similar before and after the switch. However, the sample size was small and the findings cannot be considered to be definitive.
In 2008, the Prescribing Observatory for Mental Health (POMH) initiated a quality improvement (QI) programme addressing the monitoring of patients prescribed lithium. This included the collection of clinical audit data to determine the extent to which such monitoring in mental health services was consistent with the recommendations in evidence-based clinical guidelines. The practice standards for the audits related to whether baseline and on-treatment measures of renal and thyroid function, plasma lithium levels and selected physical health checks were documented in the clinical records of patients treated with lithium. Audit findings from this QI programme have been published elsewhere.11–13
In 2023, the seventh audit in this QI programme was conducted, providing the opportunity to explore within routine clinical practice the demographic and clinical factors associated with the dose of lithium carbonate tablets prescribed and whether brand matters.
Methods
POMH is based at the Centre for Quality Improvement at the Royal College of Psychiatrists in the UK. The work of POMH has been described in detail elsewhere.14,15
In 2023, all POMH member Trusts (n = 66) were invited to participate in a clinical audit that focused on the quality of lithium monitoring in patients prescribed this medication. All patients prescribed lithium, irrespective of age, clinical diagnosis or the service providing care, were eligible to be included in Trust audit samples. The data collected, using a standardised, bespoke audit tool, included the following: age; sex; ethnicity; psychiatric diagnosis; current dose of lithium; most recent plasma lithium level on that dose; dosage regimen; formulation of lithium prescribed; whether adherence issues had been noted by the clinical team; and documented co-prescription of medicines known to have a pharmacokinetic interaction with lithium, specifically non-steroidal anti-inflammatory medicines (NSAIDs), cyclo-oxygenase-2 (COX-2) inhibitors, thiazide diuretics, loop diuretics, angiotensin-converting enzyme inhibitors and angiotensin-2 receptor antagonists. The most recent documented estimated glomerular filtration rate (e-GFR) measurement within the last year was also recorded; these measurements were conducted as part of routine clinical care. It is likely that the chronic kidney disease (CKD)-Epidemiology Collaboration (Epi) creatinine method was used in the vast majority of cases as this is the recommended method for estimating e-GFR in adults. 16 Measures of e-GFR are reported in different ways; some laboratories report absolute values in all cases while some report absolute values only when the e-GFR is <60 mls/min or <90 mls/min. We therefore grouped all e-GFR measures >60 mls/min together irrespective of whether they were absolute measures or categorical.
Ethical approval is not required for such audit-based, QI initiatives. 17
Data analysis
Data were submitted online using Formic Clinical audit software, 18 version 5.7.1 and analysed using SPSS, 19 version 26.0.0. The demographic and clinical characteristics of the sample were explored using simple descriptive statistics.
To explore the factors associated with lithium dose, linear regression was used to determine whether potential predictive demographic and clinical variables were associated with lithium dose. These variables were age, sex, ethnicity, psychiatric diagnosis (bipolar disorder, unipolar depression or other), brand of lithium prescribed (Priadel, Camcolit, generic prescription), lithium dosage regimen (once daily at night or other), plasma lithium level, presence of CKD and stage based on the most recent e-GFR, the co-prescription of medication known to interact with lithium and whether the clinical team suspected adherence to lithium to be poor. For these analyses, the effect of each variable was examined separately using a univariable analysis. The joint effect of potentially explanatory variables was then examined in a multivariable analysis, using a backwards selection procedure to retain the statistically significant variables. Statistical significance was defined as p < 0.05. Where patient characteristics were unknown, the values were treated as missing.
An additional, unadjusted analysis was conducted to examine the relationship between plasma lithium level and lithium dose and whether this differed by brand. A significant interaction between brand and dose would imply that this relationship varied depending on the brand of lithium prescribed.
Results
A total of 63 Trusts submitted data for 4405 patients who had been prescribed solid-dosage formulations of lithium for more than a year. For the vast majority of cases (89%–100%), data were available for relevant clinical variables such as ethnicity, diagnosis, renal function, prescription of interacting medication, adherence as assessed by the clinical team, lithium brand, total daily dose, dosage regimen, and plasma lithium level.
The demographic characteristics of the audit sample are shown in Table 1 along with data relating to renal function, prescription of interacting medicines, total daily lithium dose, dosage regimen, plasma lithium level and whether the clinical team suspected poor adherence to lithium treatment.
Demographic characteristics, selected clinical characteristics and details of lithium brand, dosage and plasma levels for patients prescribed solid-dosage formulations of lithium for more than a year (n = 4405).

CKD, chronic kidney disease; e-GFR, estimated glomerular filtration rate.
The Priadel brand of lithium was prescribed in 84% of cases while lithium was prescribed generically in almost 13% of cases. Eleven patients (0.2%) were prescribed more than one brand of lithium. As can be seen in Table 1, those patients prescribed Liskonum® were numerically older than those prescribed other solid-dosage formulations of lithium and also more likely to have some degree of renal impairment; these findings must be interpreted in the context of the number of patients treated with Liskonum being very small. The raw data also show that compared with patients prescribed other brands of lithium, those who were prescribed Camcolit received a numerically higher daily dose of lithium and also had higher plasma lithium levels; for example, the mean prescribed daily lithium dose and mean plasma lithium level with Camcolit were 10% and 11% higher, respectively, than with Priadel. There were no notable differences between brands in any of the other parameters shown in Table 1.
Demographic and clinical variables associated with lithium dose and plasma lithium level
Table 2 shows that mean plasma lithium levels were very consistent and did not vary with age, sex, ethnicity, diagnosis, renal function, co-prescription of interacting medication, adherence as assessed by the clinical team or lithium dosing regimen. However, they were modestly higher with Camcolit than with other solid-dosage formulations of lithium.
Relationship between clinical factors, lithium dose, and plasma lithium level, in the sub-sample of patients with a plasma lithium level of ⩾0.3 mmol/L (n = 3945).

CKD, chronic kidney disease; e-GFR, estimated glomerular filtration rate; ICD10, International Classification of Diseases, 10th Edition.
The doses used to achieve any given plasma lithium level did vary with respect to some demographic and clinical variables: mean lithium doses were lower in women than in men, reduced with advancing age and deteriorating renal function, and were higher in some ethnic groups and when the Camcolit brand of lithium was prescribed. It is possible that some of these factors are inter-related; for example, renal impairment could reasonably be assumed to be more common with increasing age. Multivariable analyses were therefore conducted to explore the relationship between the factors described above and to identify whether any of them were independently associated with dose.
Univariable and multivariable analyses
Cases were identified where the most recent plasma lithium level was between 0.3 and 1.19 mmol/L, as levels outside this range suggest poor adherence or potential toxicity and could potentially confound the relationships between variables. Data from the remaining 4213 cases were entered into a series of univariable analyses to explore the relationship between lithium dose and the following variables: age, sex, ethnicity, psychiatric diagnosis, e-GFR (as CKD severity bands), co-prescription of medication known to have a pharmacokinetic interaction with lithium, lithium brand, dosage regimen (once daily at night, or other) and plasma lithium level.
These analyses found that age, sex, ethnicity, psychiatric diagnosis, e-GFR (as CKD severity bands), co-prescription of medication known to have a pharmacokinetic interaction with lithium and plasma lithium level were significantly associated with lithium dose at p < 0.05. The effect of brand was also significant (p = 0.01), with the dose of Camcolit prescribed being on average 50mg higher than the dose of Priadel.
When these variables were entered into a multivariable analysis, only age, sex, ethnicity, diagnosis and CKD severity remained significant (all at p < 0.001). Lithium level (p = 0.02) and lithium dosage regimen (p = 0.03) also remained significant at the p < 0.05 level. After adjusting for the variables above, there was no longer a significant effect of lithium brand or interacting medication on dosage. The magnitude of the associations retained in the final model can be seen in Table 3. For example, patients who were 80 years of age or older were prescribed, on average, a daily dose of lithium carbonate that was 437 mg lower than that prescribed for patients who were younger than 30 years of age. Women were prescribed, on average, a daily dose that was 96 mg lower than men.
Multivariable analysis: clinical factors associated with lithium dose, excluding cases where the plasma lithium level suggested probable poor adherence (<0.3 mmol/L) or potential toxicity (1.2 mmol/L or above) (n = 4213).

Regression coefficient given for a 0.1 mmol/L increase in plasma lithium level.
e-GFR, estimated glomerular filtration rate.
Relationship between lithium dose, plasma level, and brand of lithium prescribed
In a further, unadjusted analysis, examining the relationship between lithium dose, plasma level, and brand of lithium prescribed, those cases where the daily lithium dose was 1600 mg or higher and those whose e-GFR was below 60 mls/min were also excluded. Those cases prescribed Liskonum were also excluded as this sub-group was very small and the Liskonum® brand of lithium is rarely initiated in the UK. This analysis found no evidence of a significant interaction between lithium dose and lithium brand (p = 0.72), with the shape of the relationship between lithium dose and plasma lithium level being similar for all brands (see Figure 1).
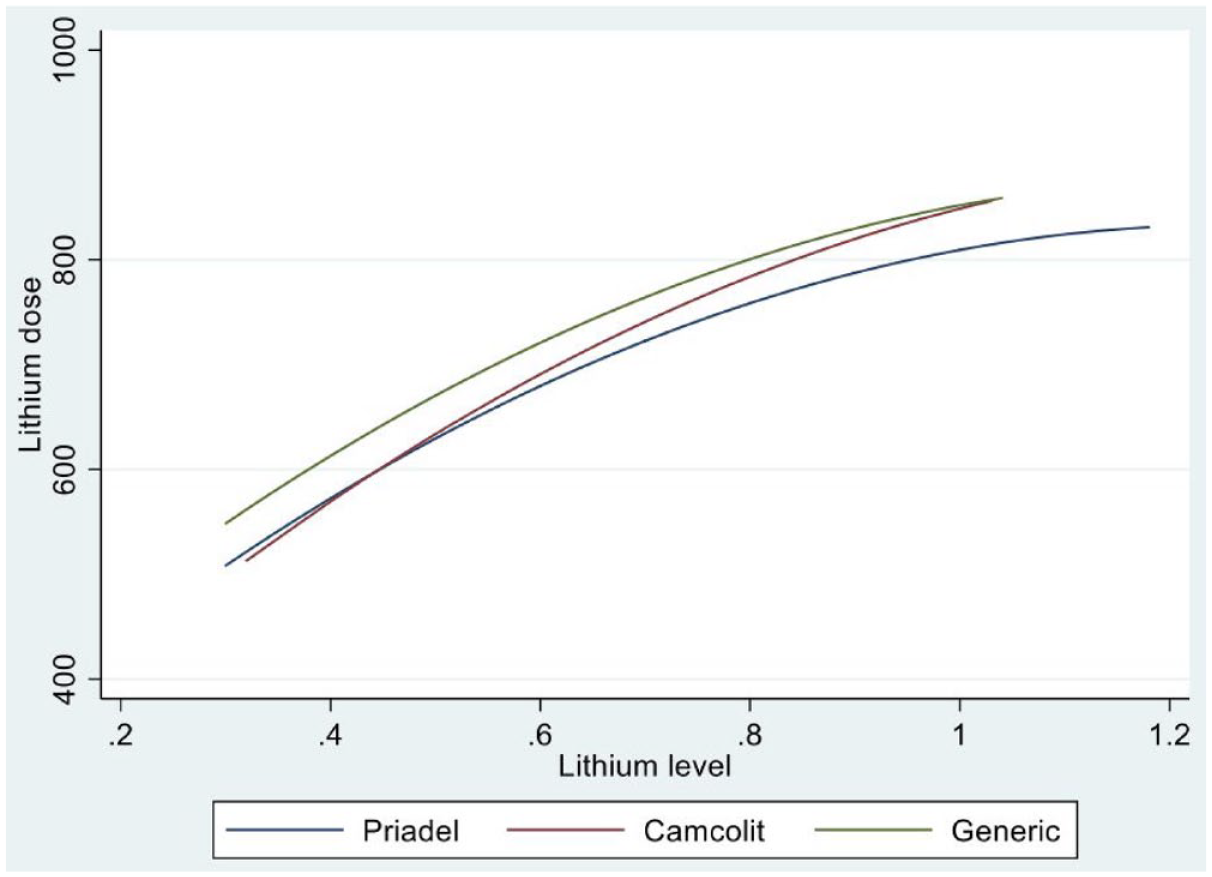
The relationship between lithium level and lithium dose by brand of lithium.
Discussion
Our data suggest that when psychiatrists prescribe lithium, they have a target plasma lithium level in mind and that this target level is not influenced by the patient’s age, sex, ethnicity, diagnosis, renal function, co-prescription of interacting medications or whether lithium is prescribed to be taken as a single daily dose or in multiple daily doses. Rather, the dose of lithium prescribed is tailored to achieve this target level, being inversely associated with age and e-GFR and being lower in women than in men. These findings are consistent with a recent large lithium dosage prediction study that also used routinely collected healthcare data. 20 We also found that the dose of lithium required was lower in those of Asian ethnicity and those who had a psychiatric diagnosis other than bipolar disorder. However, we did not find any differences in the relationship between lithium dose and plasma level between Priadel and Camcolit that were independent of these demographic and clinical variables.
Relationship between renal function and lithium dose
Lithium is almost exclusively renally excreted and it therefore follows that renal function is a major determinant of the dose of lithium required to achieve a target plasma lithium level. 21 Consistent with this, for those patients in our sample whose most recent e-GFR result suggested stage 3a, 3b or 4/5 CKD the mean doses of lithium prescribed were 97, 147 and 260 mg lower respectively than those for patients whose recent e-GFR measure was 60 mls/min or higher. We were unable to determine the effect on the dose of stage 1 or 2 CKD (e-GFR of 60–89) as many UK laboratories only report accurate e-GFR values when these are below 60 mls/min. Values above this cut-off are variably reported as >60 mls/min or >90 mls/min.
Lithium treatment may contribute towards declining renal function, 22 but even if this does occur, the benefits of lithium treatment may still outweigh the risks and continuing treatment still be in the best interests of the patient. 23 In our audit sample, the most recent e-GFR suggested stage 3a CKD in 542 (12%) patients, stage 3b CKD in 200 (5%) and stage 4–5 in a further 43 (1%).
Effect of age, sex and ethnicity on the prescribed daily dose of lithium
Our data suggest that, although the mean plasma lithium level was similar in patients of all ages, the mean dose of lithium that was prescribed to achieve this level decreased with age. Compared with adults younger than 30 years of age, those who were aged 50–59 years and those who were older than 80 years were prescribed a mean total daily dose of lithium that was 138 and 437 mg lower, respectively. This finding is consistent with the recommendations in product prescribing information relating to lower dosage requirements in older adults. 7 Our data also suggest that this effect may be independent of any decline in renal function. This finding is compatible with the known pharmacokinetic profile of lithium: this medicine has a low volume of distribution (Vd) and the total volume of body water is known to decrease with age. Thus, for any fixed dose, the concentration of lithium in plasma would be expected to be higher in older adults. 24
We found that biological sex was independently associated with lithium dose, with women being prescribed a mean total daily dose that was 96 mg less than men. The most likely explanation for this finding is that women have less body water than men, so the mechanism is likely to be similar to that described above for advancing age.
With respect to ethnicity, our multivariable analyses suggest that White patients may be prescribed a daily lithium dose that is on average 77 mg higher than Asian patients and 17 mg higher than Black patients. Asian people have a lower body weight/BMI than those who are White and this may at least partially explain this finding. However, this association should be interpreted with caution as only small proportions of the total national sample (5% and 3% respectively) were from these ethnic communities.
Relationship between diagnosis and lithium dose
The multivariable analyses found that lithium dose was lower in those patients who had a diagnosis other than bipolar disorder. Although this association was statistically significant the mean difference in daily dose was very modest at 19 mg. It is therefore unlikely that this difference in daily lithium dose is clinically meaningful.
Relationship between lithium dose, plasma level, and brand of lithium prescribed
Our data suggest that the relationship between the dose of lithium prescribed and the plasma level does not differ between Camcolit, Priadel, and the generic solid-dosage formulations of lithium carbonate that are available in the UK. This conclusion is based on the fitted relationships illustrated in Figure 1. Rather, the lithium dose is determined by a number of demographic and clinical factors such as those described above. Other clinical factors that we could not control for are also likely to be relevant; these include alcohol consumption and dietary intake, 25 body weight and the presence of co-morbid physical illness. We tried to control for poor adherence by excluding cases from the analyses where the most recent plasma lithium level was sub-therapeutic or suggested toxicity, but this is unlikely to have identified all relevant cases.
The findings we present here are consistent with those we previously reported relating to pre- and post-switch plasma lithium levels in 37 patients who switched from Priadel to Camcolit in routine clinical care, 10 following the planned discontinuation of Priadel in 2020. In this case series, patients acted as their own controls, largely removing the effect of confounding factors such as renal function and co-morbid physical illness. The main finding was that in those cases where the dosage remained the same and the clinical team did not suspect problems with adherence, the switch had little effect on plasma lithium levels.
The data reported here from the POMH audit sample, and in the previously published case series, were collected in routine clinical practice and so can never be considered as robust as those that derive from primary pharmacokinetic studies. However, the two different methodologies we have used lead to the same conclusion: if there are any pharmacokinetic differences between Camcolit, Priadel and the solid-dosage generic formulations of lithium that are available in the UK, these have only a very small impact on plasma lithium levels, compared with factors such as age, sex, and renal function.
Priadel tablets are available as 200mg and 400mg formulations whereas Camcolit is available as 400 mg. The ability to fine-tune doses below 400 mg therefore differs between these brands of lithium and this may at least partially explain our finding of a modestly higher mean dose (and plasma lithium level) when Camcolit was prescribed.
Why do clinical guidelines recommend that lithium is prescribed by brand?
In the UK, three different manufacturers are listed as providing generic solid-dosage preparations each containing 250 mg lithium carbonate; all are immediate release. In addition, two manufacturers provide branded formulations containing 400 mg lithium carbonate: Camcolit, which is described as a controlled release and Priadel, described as prolonged release. Priadel is also available as a 200 mg tablet. A further manufacturer provides a branded formulation containing 450 mg lithium carbonate: Liskonum®, described as modified release. Although it could be argued that these terms are often used loosely and overlap to some degree, the term ‘modified release’ is generally considered to be an umbrella term used to describe formulations that have been modified so as not to release all the active drugs at once. Controlled-release and prolonged-release formulations are sub-types of modified-release preparations: controlled release implies that the active drug is released at an almost constant rate over time whereas prolonged release does not imply that the rate of release is constant.
The limited pharmacokinetic data presented in the various SmPCs 7 further confuses the issue. For example, the time to maximum plasma lithium level (Tmax) is stated to be ‘about 2 hours’ for Priadel (a controlled-release preparation) while it is described as ‘2 to 3 hours’ for lithium carbonate Essential Pharma® (an immediate release preparation). Further, half-life (T1/2) is stated to be ‘about 12-24 hours’ for both Camcolit (a controlled-release preparation) and lithium carbonate Essential Pharma® (an immediate release preparation) suggesting that these values are general and may not have been derived from brand-specific pharmacokinetic studies. There are a handful of very small and methodologically diverse studies conducted in the 1970s and 1980s that compare the pharmacokinetics of different formulations of lithium.26–29 Together, these studies suggest that there is little difference between Priadel and Camcolit or between Priadel and an immediate release preparation 27 but that the prolonged-release preparation Phasal®26,28 and the sustained-release preparation of lithium citrate Litarex®29 may have different release characteristics. The latter two preparations were withdrawn from the UK market many years ago.
Conclusion
In this large sample of patients prescribed continuing treatment with lithium in routine clinical practice, there was no independent effect of brand on the relationship between plasma lithium level and lithium dose.
Implications for clinical practice
When prescribing lithium, lower doses are likely to be required for patients who are female, older and/or have renal impairment. These, and possibly other clinical factors such as body weight and alcohol intake determine the relationship between plasma lithium level and lithium dose. Priadel and Camcolit, along with the other generic solid-dosage formulations of lithium carbonate that are available in the UK do not appear to independently influence the relationship between dose and plasma level and so these formulations can be considered to be interchangeable, negating the need for complex procedures if switching between preparations proves to be necessary.
Strengths and limitations
Given the relatively large sample size and submission of data by the vast majority of NHS mental health Trusts, the data are likely to be representative of prescribing practice relating to lithium in mental health services in the UK.
The dataset is considerably larger than others in the published literature.
The audit data were collected using a standardised, bespoke data collection tool and few data were missing.
Our multivariable analysis confirmed the known relationship between age, gender and renal impairment on the dose of lithium required to achieve any given plasma level, supporting both the internal and external validity of our findings.
The data were obtained from clinical records and the accuracy of these records was not verified.
There was no objective measure of medication adherence.
The sub-sample of patients prescribed Camcolit was relatively small, potentially limiting the certainty of our conclusion.
Supplemental Material
sj-docx-1-tpp-10.1177_20451253241285883 – Supplemental material for Exploring the clinical factors affecting lithium dose and plasma level and the effect of brand
Supplemental material, sj-docx-1-tpp-10.1177_20451253241285883 for Exploring the clinical factors affecting lithium dose and plasma level and the effect of brand by Carol Paton, Paul Bassett, Olivia Rendora and Thomas R. E. Barnes in Therapeutic Advances in Psychopharmacology
Footnotes
Acknowledgements
Thanks are due to the clinicians and clinical audit staff from the UK mental health services who collected and submitted data as part of the POMH quality improvement initiative and to Gavin Herrington, the POMH programme manager.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
