Abstract
Background:
We aimed to assess the quality of physical health monitoring following rapid tranquillisation (RT) for acute behavioural disturbance in UK mental health services.
Methods:
The Prescribing Observatory for Mental Health (POMH-UK) initiated an audit-based quality improvement programme addressing the pharmacological treatment of acute behavioural disturbance in mental health services in the UK.
Results:
Data relating to a total of 2454 episodes of RT were submitted by 66 mental health services. Post-RT physical health monitoring did not reach the minimum recommended level in 1933 (79%) episodes. Patients were more likely to be monitored (OR 1.78, 95% CI 1.39–2.29, p < 0.001) if there was actual or threatened self-harm, and less likely to be monitored if the episode occurred in the evening (OR 0.79, 95% CI 0.62–1.0, p < 0.001) or overnight (OR 0.57, 95% CI 0.44–0.75, p < 0.001). Risk factors such as recent substance use, RT resulting in the patient falling asleep, or receiving high-dose antipsychotic medication on the day of the episode, did not predict whether or not the minimum recommended level of post-RT monitoring was documented.
Conclusions:
The minimum recommended level of physical health monitoring was reported for only one in five RT episodes. The findings also suggest a lack of targeting of at-risk patients for post-RT monitoring. Possible explanations are that clinicians consider such monitoring too demanding to implement in routine clinical practice or not appropriate in every clinical situation. For example, physical health measures requiring direct contact with a patient may be difficult to undertake, or counter-productive, if RT has failed. These findings prompt speculation that post-RT monitoring practice would be improved by the implementation of guidance that integrated and refined the currently separate systems for undertaking and recording physical health observations post-RT, determining nursing observation schedules and detecting acute deterioration in physical health. The effectiveness and clinical utility of such an approach would be worth testing.
Keywords
Introduction
Episodes of acutely disturbed behaviour, with the possibility of self-harm or violence toward others, are common in mental health settings.1–5 Where de-escalation and other situation-specific interventions, which may include oral sedative medication, fail to reduce a patient’s distress, level of behavioural activation or risk of harm to self or others, parenteral medication [rapid tranquillisation (RT)] may be required. For such an indication, the relevant National Institute for Health and Care Excellence (NICE) guideline specifically recommends the use of intramuscular (IM) lorazepam administered alone, or a combination of IM haloperidol and IM promethazine. 5 In addition to these pharmacological strategies, the relevant British Association for Psychopharmacology (BAP) and National Association of Psychiatric Intensive Care Units (NAPICU) joint guidelines also recommend IM promethazine, aripiprazole, droperidol or olanzapine alone, or the combination of IM haloperidol and IM lorazepam. 6
Benzodiazepines, antipsychotic medications and promethazine are all associated with a range of side effects that may complicate the clinical management of acutely disturbed behaviour. For example, the side effects of lorazepam include ataxia, hypotension, respiratory depression and apnoea. 7 Haloperidol may cause dystonia, hypotension, tachycardia, dyspnoea, ventricular arrhythmias and neuroleptic malignant syndrome, 8 while promethazine may cause disorientation, palpitations, hypotension, arrhythmias and muscle spasms. 9 Further, data submitted through the Medicines and Healthcare Regulatory Authority (MHRA) yellow card system suggest that some of these medications can be associated with serious side effects. For example, fatal outcomes secondary to cardiac arrhythmias or respiratory compromise have been reported with lorazepam, haloperidol and promethazine.10–12 In 2015, NHS England issued a Patient Safety Alert mandating NHS organisations to ensure they have systems in place for monitoring vital signs after restrictive interventions, such as seclusion, manual restraint and RT. 13
While it is not always possible to complete a comprehensive physical assessment before RT medication is administered, the risks of such treatment may be increased because the patients for whom it is indicated may be physiologically compromised. This may be related to physical exhaustion associated with overactivity, the recent ingestion of substances or alcohol, dehydration, the side effects of their current regular medication regimen or pre-existing physical illness. The risks associated with comorbid medical conditions are illustrated by the findings of the TREC (Tranquilização Rápida-Ensaio Clínico) studies of RT where, in the haloperidol-promethazine treatment arms, a grand mal seizure occurred in a patient known to have epilepsy, 14 and a seizure and transient hypotension were reported in a patient known to be dehydrated, 15 while, in the lorazepam arm, a patient known to have asthma experienced worsening respiratory difficulty. 16
In recognition of the potential for clinically significant adverse effects associated with the use of parenteral medicines for RT, NICE recommends that blood pressure (BP), pulse, temperature, respiration, level of hydration and level of consciousness should be monitored at least every hour after RT until there are no further concerns, with more frequent monitoring in those patients who may be at an increased risk of complications for any reason. 5 The BAP/NAPICU guideline sets more stringent standards, recommending that, as a minimum, psychiatric observations, respiratory rate, oxygen saturation, systolic BP, pulse, temperature and level of consciousness should be documented every 15 min for 1 h. 6 However, little is known about the extent to which these recommendations are implemented in clinical practice. Loynes and colleagues contacted 58 mental health Trusts in England requesting copies of local audits of RT practice. 17 Only 18 of these Trusts had undertaken any audits of post-RT physical health monitoring, and in all cases practice fell short of that recommended by NICE, usually by a considerable margin. These authors recommended that a national audit of RT practice should be conducted. In 2016, the Prescribing Observatory for Mental Health (POMH-UK) initiated an audit-based quality improvement programme (QIP) focussing on the pharmacological management of acute behavioural disturbance in UK mental health services, thus allowing the quality of post-RT monitoring practice to be explored systematically. We report here on the findings from this work.
Methods
Each year, all National Health Service (NHS) Trusts and other healthcare organisations providing inpatient mental health services in the UK of which POMH-UK are aware are invited to become members and participate in its QIPs. A QIP focussing on the pharmacological management of acutely disturbed behaviour was initiated in 2016. The clinical practice standards for this QIP were derived from the NICE NG10 guideline on the management of violence and aggression, 5 and addressed prescribing and care planning with a focus on the quality of physical health monitoring post-RT, specifically that there should be at least one measure each of BP, pulse, respiratory rate and temperature in the hour after RT.
A baseline clinical audit was conducted in 2016. A customised report on the audit findings was sent to each of the participating mental health services, providing benchmarked data on their prescribing practice in relation to the treatment of acutely disturbed behaviour. A re-audit was conducted in 2018, and an updated customised summary of the data collected was again sent to each participating mental health service.
For both the baseline audit and the re-audit, participating mental health services were asked to identify episodes of acutely disturbed behaviour, occurring over a defined period (July–November 2016 and March–May 2018, respectively), for which additional psychotropic medication had been administered. After each eligible episode, the following data were collected promptly: the patient’s year of birth; gender; ethnicity; psychiatric diagnoses and legal status with respect to mental health legislation; type of clinical service providing care; nonpharmacological interventions used in the episode; symptoms and behaviours displayed at the time of the episode; level of behavioural disturbance (applying the descriptions of the categories in the Behavioural Activation Rating Scale) 18 before and after RT; regularly prescribed medication; medication administered for the episode of acutely disturbed behaviour and route of administration; and the measures of blood pressure, pulse, respiratory rate and temperature recorded after this medication was administered.
For each audit, services were asked to submit data for only one episode for any given patient unless the episodes were separated by at least 7 days; the aim was to avoid the data being skewed by a small number of patients who may have received additional psychotropic medication on multiple occasions over a short period of time. We therefore report information at the level of episodes of disturbed behaviour rather than individual patients.
Data submission and analyses
Anonymised data were submitted online between September–November 2016 and March–May 2018 using Formic software (Formic Limited, Middlesex, UK) and analysed using SPSS (SPSS, Chicago, IL, USA).19,20 To allow the accuracy of data entry to be checked, each participating mental health service was sent a copy of their submitted dataset along with any data-cleaning queries.
The percentage method was used to determine whether regularly prescribed antipsychotic medication was standard or high dose. 21 Where an antipsychotic medication was administered to manage an episode of acute behavioural disturbance, the percentage of its maximum daily dose was added to that of any regularly prescribed antipsychotic medication to determine whether the cumulative dosage reached or exceeded the high-dose threshold on that day.
Binary logistic regression analyses were used to explore any associations between the demographic and clinical variables collected and whether or not the recommended post-RT physical health monitoring (at least one measure in the hour after RT of blood pressure, pulse, respiratory rate and temperature) had been conducted: the dependent variable. The independent variables were gender, ethnicity, age, diagnosis, Mental Health Act status, the clinical service providing care, the day of the week and time of day that the episode occurred, whether substance use was suspected to have contributed to the episode, the level of behavioural activation at the time of the episode [using the behavioural activity rating scale (BARS) descriptors], whether the episode involved aggression towards others or self-harm, the nature of the parenteral medication used to manage the episode, whether the patient received high-dose antipsychotic medication on the day of the episode, whether seclusion or supervised confinement was employed, and the level of behavioural activation observed in the hour after RT had been administered. A set of univariable analyses was performed to examine the associations between these independent variables and the dichotomous dependent variable. Where an association at the level of p < 0.05 was found in the univariable analyses, the relevant variables were then examined in multivariable analyses using a backwards selection procedure to retain only the statistically significant variables. Given that both the baseline audit and the re-audit data related to individual episodes of disturbed behaviour rather than patients, and that clinical practice with respect to post-RT monitoring was similar on the two occasions, the data on all episodes of disturbed behaviour from both audits were analysed together in the regression model.
Results
Demographic and clinical characteristics of the audit samples
At the baseline audit in 2016, 58 mental health services submitted data for 1081 episodes of acutely disturbed behaviour for which parenteral psychotropic medication had been administered (RT). At re-audit in 2018 (18 months later), 54 services submitted data for 1373 such episodes. There were no noteworthy differences in the demographic or clinical characteristics of the patients involved in these episodes or in the clinical management of these episodes at each audit; therefore, the two audit samples were combined to provide a total sample of 2454 episodes of acutely disturbed behaviour that resulted in RT, reported by 66 mental health services in total. The demographic and clinical characteristics of the patients who were involved in these episodes are shown in Table 1. Key clinical features relating to presentation and clinical management of the episodes are shown in Table 2. In the subsample of episodes for which it was possible to derive a post-RT BARS rating (n = 1240), the data indicated that, in a quarter of cases, the patient had remained very behaviourally disturbed (as defined by a derived BARS rating of 6 or 7) in the hour after RT.
Key demographic and clinical characteristics of the patients involved in the 2454 episodes of acutely disturbed behaviour.

All psychiatric diagnoses were collected, therefore some patients had more than one.
ICD10, International Classification of Diseases 10th Revision.
Timing, clinical presentation, management, and outcomes of the 2454 episodes of acutely disturbed behaviour.
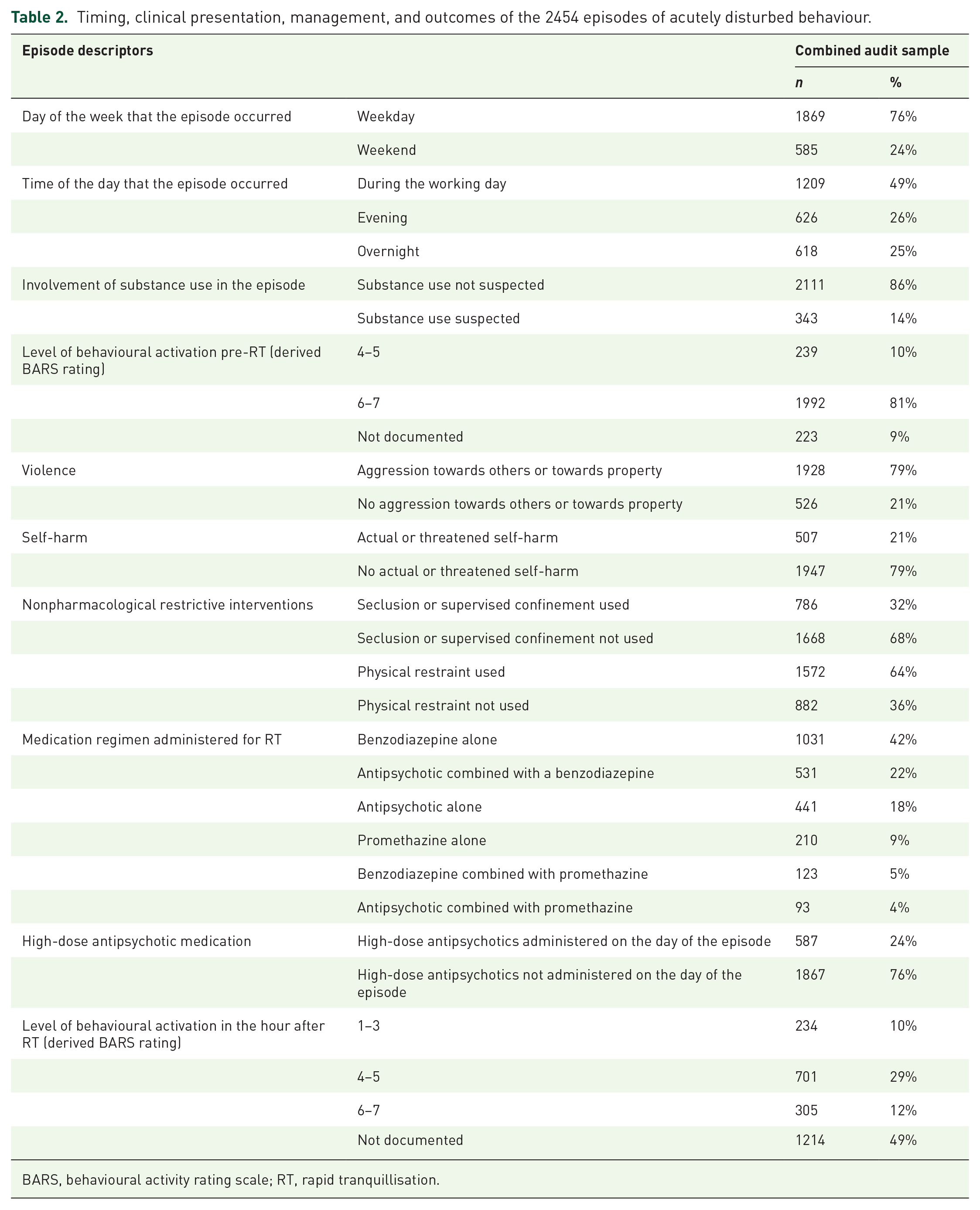
BARS, behavioural activity rating scale; RT, rapid tranquillisation.
Post-RT monitoring
At least one measure each of blood pressure, pulse, respiratory rate and temperature, representing the minimum required physical health monitoring (see Figure 1), was conducted in the hour after RT in 521 [21%; 95% confidence interval (CI) 19–23%] episodes. In 984 (40%; 38–42%) episodes, neither the minimum required physical health monitoring, nor any description of behavioural state was documented. Of the 1240 episodes for which a post-RT BARS rating was derivable, the patient was asleep within an hour of RT being administered in 171 episodes (14%; 12–16%). In this subsample of episodes where the patient fell asleep, the minimum required physical health checks were not documented in 130 episodes (76%; 70–82%), there was no record of any measure of respiratory rate in 95 episodes (56%; 49–63%) and pulse oximetry was used in only 46 episodes (27%; 20–34%). Of the 99 (58%; 51–65%) such episodes occurring in the evening or overnight, there was no documented check of respiratory rate in 61 episodes (62%; 52–72%). In the remaining 72 episodes (42%; 35–49%) where sleep occurred during the day, there was no check of respiratory rate in 34 episodes (47%; 36–59%).

Level of behavioural activation and physical health measures documented in the hour after RT at the baseline audit (n = 1081) and re-audit (n = 1373).
The multivariable analysis revealed that gender, time of day that the incident occurred, whether the incident involved self-harm, the medication regimen administered and the level of behavioural activation after RT was administered were all associated with whether or not the minimum recommended level of physical health monitoring had been carried out and documented. The direction and strength of these associations are shown in Table 3. The presence of risk factors such as recent substance use or RT resulting in high-dose antipsychotic medication being administered on the day of the episode did not predict whether or not such post-RT physical health monitoring was documented.
Multivariable logistic regression model for factors associated with the recommended physical health monitoring being conducted post-RT.

BARS, behavioural activity rating scale; CI, confidence interval; OR, odds ratio; RT, rapid tranquillisation.
Discussion
The minimum level of physical health monitoring recommended by NICE for all episodes of RT is documentation of at least one measure each of pulse, blood pressure, temperature and respiratory rate in the hour after parenteral medication is administered. 5 In our large national sample, this recommended minimum level of monitoring was documented in less than a quarter of RT episodes. Further, there was no documentation of behavioural state post-RT in just under half of episodes, and there were no observations relating to behavioural state or physical health in two-fifths of episodes. In this latter group, there was essentially no clinical documentation relating to the efficacy or tolerability of the RT medication regimen. Given that services chose to participate in these clinical audits to support and inform their local quality improvement endeavours, and that practice at re-audit was not notably different from that assessed at the baseline audit, conducted approximately 18 months earlier, our data suggest that custom and practice in this area of care is static and may be relatively resistant to change interventions.
In our combined sample of RT episodes, there were associations between conducting at least the minimum level of physical health monitoring and variables such as gender, time of the incident, self-harm and the medication regimen administered for RT. While these associations were statistically significant, the numerical differences were relatively small. The NICE guideline recommends an increased level of physical health monitoring in patients who may be at risk of post-RT physical health complications because they have ingested substances, been exposed to high-dose antipsychotic medication or are over-sedated/asleep after parenteral medication has been administered. 5 However, our data suggest that even the minimum recommended monitoring was not more likely to be achieved for episodes involving these risk factors; for example, there was little evidence of any association between over-sedation and level of monitoring.
We also found that episodes of acutely disturbed behaviour that were managed with a combination of IM lorazepam and IM promethazine were more likely to be associated with post-RT physical health monitoring than episodes that were managed with other medication regimens. Given that the number of such episodes was small, a likely explanation for this association is that it is a chance finding. Nevertheless, other possible explanations are that this combination of sedative medicines may be used by a small number of clinical services that have established a more rigorous monitoring practice, or that nurses may have experience of, or concerns about, the potential side effects of this combination.
Why is there a gap between guideline recommendations and clinical practice?
Our data suggest that almost one patient in four may remain behaviourally disturbed after an initial attempt at RT. This proportion was similar for both the baseline audit and re-audit samples. 22 The direct contact required for physical monitoring may be impractical in such cases 17 : patients who are still aroused and disturbed may refuse to cooperate or staff may be wary of approaching them, perceiving that this might be potentially counterproductive and put both the patient and themselves at risk of harm. 6 However, our data provide only very limited evidence that monitoring is less likely to be documented in patients who remain behaviourally disturbed after RT.
The BAP/NAPICU guidelines and Maudsley Prescribing Guidelines, as well as guidance issued by the Care Quality Commission acknowledge that ‘hands-on’ physical health monitoring may not be possible in all cases post-RT.6,23,24 But there is a lack of consensus relating to the appropriate ‘hands-off’ observations that should be undertaken in these circumstances. The BAP guideline suggests respiratory rate, level of consciousness and clinical assessment for pallor, and signs of pyrexia, dehydration, dystonia and akathisia, 6 while the Maudsley Guidelines recommend observing for pyrexia, hypoxia, hypotension, oversedation and general physical wellbeing. 23 But for two episodes out of every five in our sample, the minimum requirement relating to documentation of the level of consciousness and respiratory rate was not met, so it seems unlikely that more general observations relating to physical wellbeing were being undertaken routinely.
The CQC considers that, as a minimum, there should be documentation of the level of consciousness and respiratory rate post-RT, as well as the rationale for not undertaking all the recommended physical health checks. 24 In those cases where a patient remains clearly behaviourally disturbed after RT, clinical staff managing the incident may consider that the reasons for not directly undertaking the required physical health tests are self-evident and do not warrant being documented in the clinical records. Nonetheless, it remains important to keep the patient under direct observation to check for the emergence of any adverse physical signs.
Existing UK guidelines provide recommendations relating to the physical health checks that should be undertaken, and the frequency with which these should be completed.5,6 They also clearly describe the enhanced monitoring that is required in certain circumstances where the patient may be at increased risk of post-RT physical health complications. Our data reveal a common failure to comply with these recommendations, perhaps because clinicians consider them too demanding to implement in routine clinical practice with the available resources or not appropriate in every clinical situation. The findings prompt speculation on other potential barriers to implementation, such as the following: physical health measures that require direct contact with a patient may be difficult to undertake if RT has failed to render the patient calm and approachable; clinicians may have a lack of understanding of the potential risks associated with the medicines used for RT; and there may be a lack of harmonisation between the separate systems for undertaking and recording physical health measures post-RT, determining nursing observation schedules and detecting acute deterioration in physical health.
Our data suggest that checks relating to respiration had been documented in just over half of the episodes, where it was documented that the patient fell asleep in the hour after RT was administered during the day, but this proportion was lower, at just over a third, when RT occurred in the evening or at night. One potential explanation for this apparent lack of targeting of at-risk patients is that while guidelines derive blanket recommendations relating to post-RT monitoring from the evidence base, including the known side-effect profiles of the relevant medicines, monitoring in clinical practice is a task undertaken almost exclusively by ward staff, principally mental health nurses, who may have limited familiarity with the relevant literature and rely on their experience and clinical judgment to manage each episode of RT pragmatically, alongside their other routine clinical duties.
Severe adverse effects after RT are uncommon and the published evidence relating to the incidence of these and any clinical risk factors is scant. 25 Further, the majority of nurses are unlikely to have experience of managing such clinical situations. While the nature of the disturbed behaviour in an individual patient and their response to RT or any underlying physical health problems may be relevant to decisions about post-RT monitoring, training for nurses will have been limited to general recommendations/local protocols for post-RT physical health measures. In an individual patient, the risk of clinically relevant adverse effects post-RT may be perceived as so slight that such monitoring is not a clinical priority. This may particularly be the case where a patient calms down rapidly and appears well; it may not be judged necessary to complete all the physical health measures or document them unless RT was administered in the context of reducing the risk of, or managing actual, self-harm, when monitoring may be prompted by the need to ensure the patient remains unhurt. Most mental health nurses will have experience of managing patients through distressing episodes of self-harm and will draw on this in their efforts to provide best care. Further, when RT is administered in the evening or at night, nurses may consider that it is expected behaviour for the patient to fall asleep. However, it is not necessary to wake a sleeping patient to check respiration, and both the BAP guideline and Maudsley Prescribing Guidelines recommend the use of pulse oximetry in such circumstances.6,23 A measure of oxygen saturation is also a component of the National Early Warning Score, 26 which is part of a system adopted by the NHS to identify patients at risk of clinical deterioration. In our sample, pulse oximetry was used in just over a quarter of the RT episodes where patients were reported to have fallen asleep.
There may also be relevant systems barriers to both conducting and documenting the required physical health checks post RT. For example, where RT is administered in the evening or at night, there may be fewer staff on duty to undertake physical health monitoring. Another practical concern is the drive towards a paperless NHS and the exclusive use of electronic clinical records. It is possible that nurses record physical health observations on pieces of paper and, as other clinical tasks take priority during the course of a shift, the task of entering these observations into the electronic clinical record is no longer a priority. Entering the primary record electronically at the time of the episode may not be possible even if portable IT devices are available as there may be a risk that these could be broken or used as weapons.
Closing the gap between guideline recommendations and clinical practice
Post-RT monitoring may be made more complex in mental health settings as the criteria for determining the level of nursing observations tend to focus exclusively on the risks that the patient poses to themselves and others and physical health is not explicitly considered in organisational guidelines. 27 For example, while a patient who could, at any time, make an attempt to harm themselves or others meets the criteria for level 3 one-to-one observation (within eyesight), a sleeping patient does not; the emphasis is entirely on managing risk related to mental state. A patient who was asleep would, at most, meet criteria for level 2 ‘intermittent’ observations, in which case the clinical care requirement is to check the patient’s location at individually determined intervals. There is, therefore, no direct connection between post-RT monitoring requirements,5,6 general nursing observation schedules (assessing mental and behavioural state) and the use of the NEWS (assessing physical health).26,27
In clinical practice, guidelines relating to post-RT physical health measures, clinical protocols for detecting the deteriorating patient and systems for nursing observations exist as distinct processes and training is provided separately. There may be a case for integrating and refining these protocols into a unified guidance that is more individualised and clinically relevant, with the potential to facilitate care planning. For example, the criteria for level 3 nursing observations could be modified to include patients who fall asleep within an hour of RT being administered; with respect to care planning, the minimum expectation could be that such a patient remains with line of sight and a pulse oximeter is attached (or an equivalent remote monitoring system is put in place) until the patient is awake and mobilising. By documenting the level of consciousness and oxygen saturation using the NEWS system, 26 a developing medical emergency would be immediately identified and subject to the escalation process indicated in the NEWS. The effectiveness and clinical utility of such an approach would be worth testing in practice.
Strengths and limitations of the study
(1) Given the large sample size and involvement of the vast majority of mental health Trusts, the findings are likely to be representative of current RT practice in UK mental health services.
(2) Post-RT monitoring practice was assessed against recommendations in evidence-based, national, clinical guidelines.
(3) The findings regarding post-RT monitoring are based on self-report data from secondary care mental health services.
(4) The findings relate to practice within UK mental health services and may not be confidently extrapolated to other clinical settings.
Key points
(1) The minimum level of post-RT physical health monitoring recommended by NICE was reported for only a small proportion of episodes and not targeted towards at-risk patients.
(2) Post-RT monitoring practice could be improved by the implementation of guidance that integrated and refined the currently separate systems for undertaking and recording physical health observations post-RT, determining nursing observation schedules and detecting acute deterioration in physical health.
Footnotes
Acknowledgements
Acknowledgements are due to the clinicians and other staff in participating services who collected and submitted the audit data. Thanks are also due to members of the POMH-UK team aside from the co-authors of this paper: Gavin Herrington, Charlene Nyanhemwa and Jenny Bari, and to Francis Adzinku for reviewing the contents
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and publication of this article: The work of POMH is funded wholly by subscriptions from mental health services in the UK.
Conflict of interest statement
CP, CA, SD, OD and CO have nothing to declare. TREB has been a member of scientific advisory boards for Sunovion, Otsuka/Lundbeck and Newron Pharmaceuticals, and received speaker fees from Janssen.
Ethical statement
Clinical audit by definition does not involve anything being done to patients beyond their normal clinical management and therefore does not require formal ethical approval.
