Abstract
Background:
This study examined 5-year outcomes of patients prescribed risperidone long-acting injection (RLAI) or aripiprazole in a clinical setting, using treatment discontinuation as a measure of effectiveness.
Method:
Patients who received RLAI or aripiprazole in the 18 months following their respective UK launches were included. Two-year outcome data were previously reported for these cohorts; this study reported an additional 3 years of follow up for each group. Data were collected from pharmacy records and by retrospective case note review. Patients were classified as continuers or discontinuers at 5 years and reasons for treatment discontinuation noted.
Results:
The number of patients remaining on treatment at 2 years (and included in this study) was 28/84 and 27/92 for RLAI and aripiprazole respectively. Two patients treated with RLAI and three treated with aripiprazole were lost to follow up. Therefore, 5-year outcome data were available for 50 patients. Fifteen patients from each group were continuers at 5 years. Of these, four receiving RLAI and three receiving aripiprazole were coprescribed other antipsychotics at study endpoint. Reasons for discontinuation of RLAI and aripiprazole respectively were lack of effect (
Conclusion:
There was no significant difference between the proportions of patients continuing RLAI or aripiprazole for 5 years. Continuation rates were relatively low (18% and 16% of the original RLAI and aripiprazole cohorts respectively), whilst coprescription of other antipsychotics at endpoint was relatively common. Lack of effectiveness was the most common reason for discontinuation of both compounds. These findings suggested that clinical effectiveness was somewhat disappointing, although the long period of follow up and number of patients previously treated with clozapine in the original cohorts were confounding factors.
Introduction
The antipsychotic treatments risperidone long-acting injection (RLAI) and aripiprazole (tablet formulation) have been available in the UK since October 2002 and July 2004 respectively. When launched, both represented novel approaches to the treatment of psychosis. RLAI was the first available long-acting injectable preparation of an atypical antipsychotic, with the potential advantages of reduced covert nonadherence compared with oral medication [Barnes and Curson, 1994] and reduced propensity for extrapyramidal side effects compared with typical antipsychotics [Leucht et al. 1999]. Aripiprazole had a novel pharmacology (with partial agonist rather than antagonist properties at the dopamine D2 receptor) which suggested that it would be associated with a favourable adverse effect profile [Taylor, 2003] together with the potential associated benefits of improved adherence and outcome [Lambert and Naber, 2004].
Data from randomized controlled trials (RCTs) have provided evidence for the efficacy of RLAI [Kane et al. 2003] and aripiprazole [Potkin et al. 2003] in the treatment of psychosis. However, RCTs typically assess treatment outcome over a short period of time, which may have limited applicability to the clinical management of a chronic illness such as schizophrenia. Furthermore, due to strict inclusion and exclusion criteria, participants may not be representative of patients routinely seen in practice. Consequently, naturalistic patient follow-up studies [e.g. Attard et al. 2014], and studies using pragmatic outcome measures [e.g. Lieberman et al. 2005] have been used to evaluate the effectiveness of a number of different antipsychotics in settings more relevant to clinical practice. Both RLAI and aripiprazole have been the focus of several such studies, with periods of follow up ranging from 6 months [Deslandes et al. 2008; Taylor et al. 2007] to 3 years [Taylor et al. 2009]. Given the chronic and relapsing nature of schizophrenia and the burden of the disease, studies with longer periods of follow up remain important to assess treatment outcome [Turner, 2004].
This study examined 5-year outcomes of patients prescribed RLAI or oral aripiprazole in a clinical setting, using treatment continuation as a measure of effectiveness in order to evaluate their longer term value.
Methods
Design
This was a retrospective, naturalistic follow-up study of patients with a diagnosis of schizophrenia or schizoaffetive disorder, prescribed RLAI or aripiprazole tablets in clinical practice in the 2 years following their respective UK introductions. Two-year outcome data have previously been reported for these cohorts [Deslandes et al. 2009a, 2009b]. This study reported an additional 3 years of follow up for patients continuing either treatment at 2 years. The original study was reviewed by the South East Wales Research Ethics committee panel C (reference number 04/WSE03/25).
Participants and outcome measures
Out of the original cohorts of 176 patients (84 RLAI and 92 aripiprazole), those remaining on treatment at 2 years were identified. Data were collected from pharmacy records and by retrospective case note review during a 6-week period throughout February and March 2013. Patients were retrospectively categorized either as those remaining on treatment (at 5 years), classified as ‘continuers’, or individuals who had discontinued treatment and designated ‘discontinuers’. The reason for discontinuation was recorded for all patients who stopped treatment during the study period. When the reason for discontinuation was recorded as ‘patient refusal’, subjects were either disengaging from mental health services, or exhibited a lack of insight resulting in them refusing treatment. In some cases, adverse effects were the primary reason for a patient refusing (and discontinuing) treatment; such individuals were categorized as discontinuing due to adverse effects. The duration of treatment and the antipsychotic to which patients were switched following discontinuation were also noted. Patients’ drug histories were reviewed in order to determine whether or not they had previously been treated with clozapine. This information was used to provide an indication that their condition had previously been unresponsive to treatment. Due to a possible association between RLAI discontinuation at 2 years in patients who had previously experienced two treatment failures with other antipsychotics [Deslandes et al. 2009a], this information was also recorded. For patients prescribed aripiprazole, inpatient or outpatient status at initiation was recorded. These parameters were used to divide patients into subgroups for further analysis. The maintenance dose upon treatment discontinuation (or at 5 years for continuers) and the details of any coprescribed antipsychotics were recorded for all patients.
Analysis
Outcomes of patient subgroups based upon previous clozapine treatment, two or more previous treatment failures and inpatient
Results
The number of patients remaining on treatment at 2 years (and therefore included in this study) was 28/84 and 27/92 of the original RLAI and aripiprazole cohorts respectively. Out of the 55 patients included, two treated with RLAI and three treated with aripiprazole were lost to follow up. Therefore, 5-year outcome data were available for 50 patients (26 RLAI and 24 aripiprazole). Six patients receiving RLAI and seven receiving aripiprazole had previously been treated with clozapine. Patient demographics, reasons for treatment initiation and mean dose at study endpoint are shown in Table 1.
Patient demographics and reasons for treatment initiation.
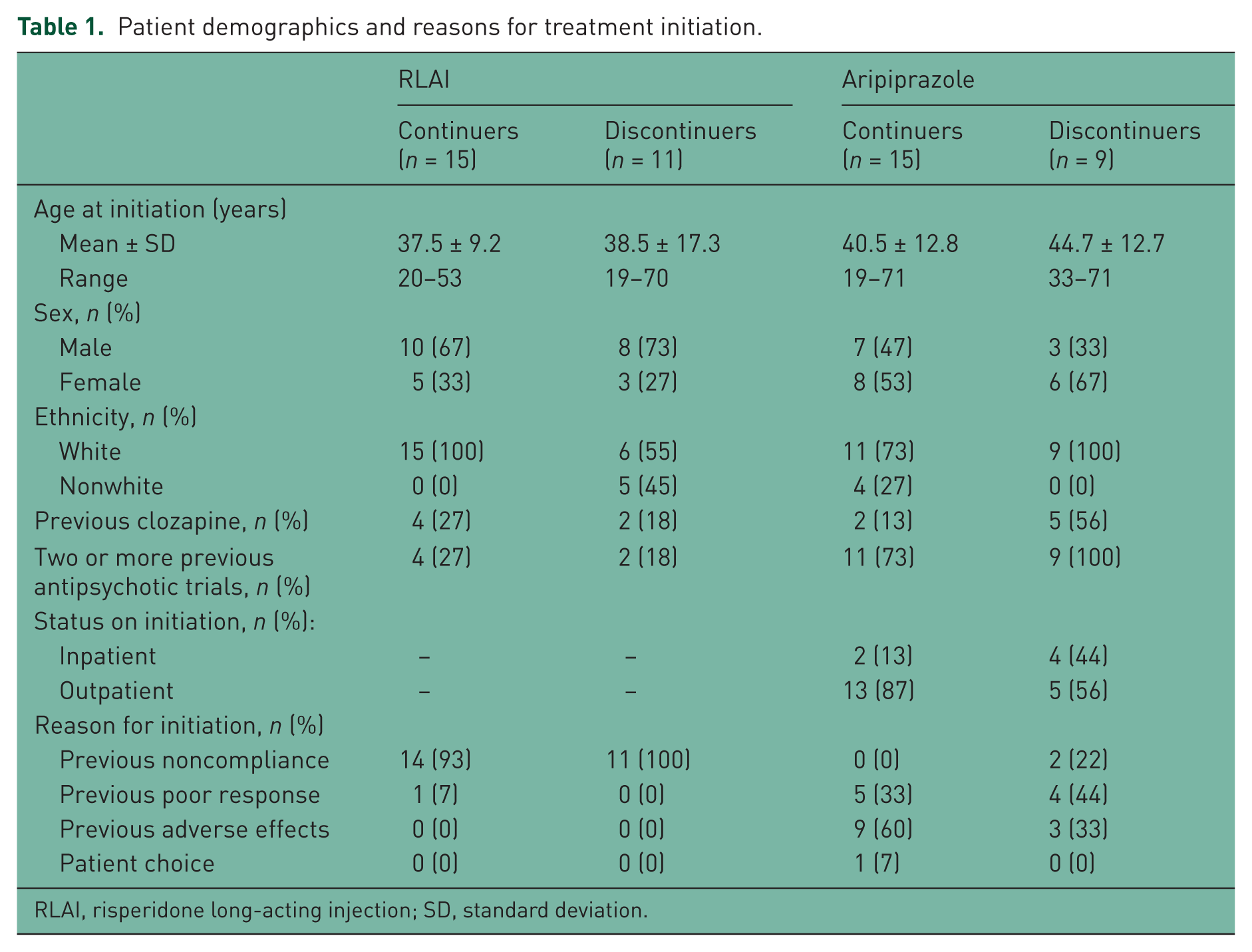
RLAI, risperidone long-acting injection; SD, standard deviation.
Patients continuing treatment
The proportion of patients remaining on treatment over time is shown in Figures 1 and 2. Figure 1 shows the proportion of the original RLAI and aripiprazole cohorts on treatment from time 0 to 5 years, excluding those lost to follow up in the current study (

Proportion of original patient cohorts remaining on treatment over time. RLAI, risperidone long-acting injection.

Proportion of patients remaining on treatment from 2 to 5 years, grouped according to whether they had previously been treated with clozapine (C) or had not previously been treated with clozapine (NC). RLAI, risperidone long-acting injection.
Patients discontinuing treatment
Reasons for treatment discontinuation, dose at discontinuation and the antipsychotic to which patients were switched are shown in Table 2. Patients were categorized by treatment and according to whether or not they had previously received clozapine. Doses for the RLAI group represent mean fortnightly dose. Poor response was the most common reason for discontinuation in both groups.
Reasons for discontinuation.
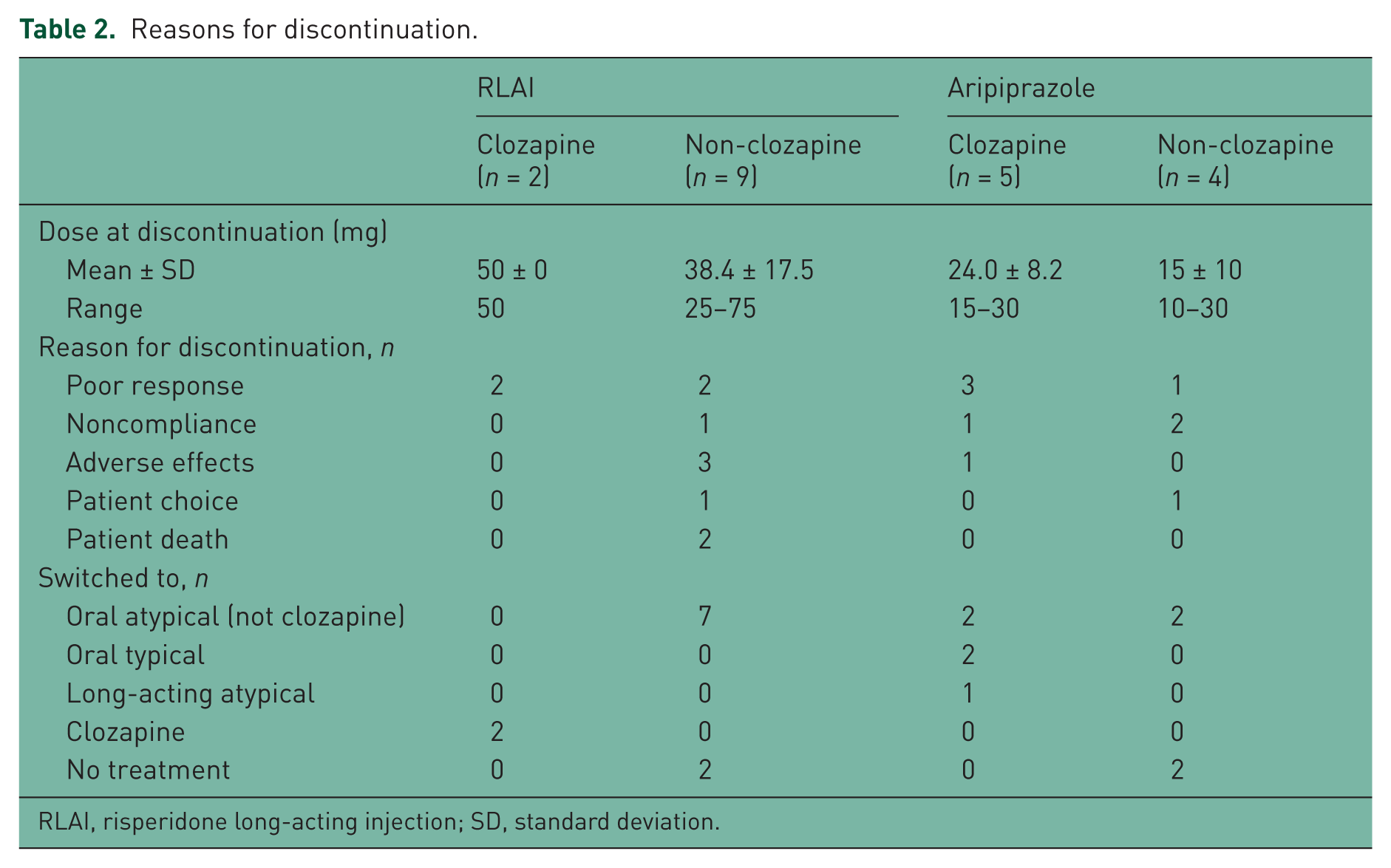
RLAI, risperidone long-acting injection; SD, standard deviation.
Discussion
This study assessed clinical outcomes of patients treated with two novel antipsychotic preparations over an extended period of time. Overall continuation rates from time 0 to 5 years for the original cohorts of 176 patients were relatively low (18% and 16% of RLAI- and aripiprazole-treated patients respectively). However, of the 50 patients remaining on treatment at 2 years (the focus of the present study), 60% completed an additional 3 years of treatment, and were therefore continuers at 5 years. There was no significant difference between the proportions of patients continuing RLAI or aripiprazole at 5 years. Reasons for discontinuation were comparable, with lack of effectiveness being the most common reason for cessation of both compounds. Coprescription of other antipsychotics was relatively common (17% of all continuers at 5 years).
This study was not specifically designed to compare outcomes between patients treated with either RLAI or oral aripiprazole or between subgroups of patients treated with each drug. Indeed, data from the two cohorts were collected over different time periods and the demographics of the two groups (particularly reasons for treatment initiation) were different. However, there was no significant difference in the proportion of patients continuing either drug at 5 years, although it must also be noted that there may not have been sufficient power to detect such a difference. This is consistent with the findings of a 2-year, open-label study examining outcomes with these antipsychotics [Macfadden et al. 2010]. It has been suggested that long-acting injectable formulations of antipsychotics may be more effective than oral formulations of the same drug [Tihonen et al. 2006]. This may be due to a reduction in overt noncompliance associated with the long-acting injections, with subsequent benefits in treatment outcome [Novick et al. 2010]. Whilst overall levels of discontinuation in our study were similar, it is perhaps interesting to note that the proportion of patients discontinuing due to noncompliance (9% and 33% for RLAI and aripiprazole respectively) favoured the long-acting injectable preparation in this regard. Conversely, the proportion of patients discontinuing due to adverse effects (27% and 11% for RLAI and aripiprazole respectively) suggested that the hypothesized improved tolerability profile of aripiprazole [Taylor, 2003] has been realized in clinical practice. It should be noted that in addition to the limitations mentioned above, the number of patients discontinuing due to these reasons was small (
The proportion of patients discontinuing treatment, and the level of antipsychotic coprescribing at 5 years suggested that the clinical effectiveness of both RLAI and aripiprazole was somewhat disappointing. However, the long period of follow up and the large number of patients previously treated with clozapine in the original cohorts were confounding factors. Results with RLAI at 3 years (29% continuation) were consistent with the findings of another naturalistic study, which reported a 33% continuation rate at the same time point [Taylor et al. 2009]. An open-label study comparing RLAI and aripiprazole over 2 years reported continuation rates of 69% and 61% respectively, with 7.9% of RLAI and 11% of aripiprazole patients coprescribed an antipsychotic [Macfadden et al. 2010]. The continuation rate was somewhat higher than that seen in our study at 2 years, which may reflect the differing study designs. At 5 years, 26% of RLAI and 7% of aripiprazole continuers in our study were coprescribed an antipsychotic. The rate of coprescribing with aripiprazole was comparable to that seen by Macfadden and colleagues [Macfadden et al. 2010]. The rate for RLAI was considerably higher, although somewhat lower than the 46% reported by Aggarwal and colleagues in a study of antipsychotic long-acting injection use in clinical practice in the USA [Aggarwal et al. 2012]. The 5-year follow-up period in the present study may explain this difference, as patients requiring supplementation (and therefore presumably showing a suboptimal response to treatment) may have discontinued prior to study endpoint. Oral risperidone was the most commonly coprescribed medicine in the RLAI group. This perhaps suggested that patients showed some response but required a higher plasma level of the drug than that provided by the injectable formulation alone in order to achieve a maximal response.
Whilst discontinuation over the whole 5-year period was high, continuation of treatment at year two appeared to be a relatively good predictor of continuation at year five for both of the treatments studied. Findings from the RLAI cohort showed that previous poor response to antipsychotic treatment was associated with early discontinuation [Deslandes et al. 2009a]. It could be argued that excluding patients who discontinued early (<1 year) in treatment, and assessing response over the subsequent 4 years, would have shown a more positive outcome. This is consistent with the view that in clinical practice newly launched antipsychotics are often prescribed for hard-to-treat patients who failed to respond to other drugs, thereby increasing the likelihood of treatment failure [Young and Taylor, 2006]. However, it should also be noted that longer term studies indicate that patients with schizophrenia tend to stabilize in any event after approximately 5 years through the natural alleviations of the disease and with progressing age and maturity [Harrison et al. 2001]. Despite all of the patients who discontinued RLAI being initiated due to poor compliance, the agent that they were switched to following discontinuation was an oral (non clozapine) atypical antipsychotic. This would appear somewhat illogical if previous poor compliance had been a reason for initiation of long-acting injectable treatment. Subsequent outcomes for this subgroup were not evaluated, but would be of potential benefit in order to explore the effectiveness of this treatment approach.
This study provided evidence for the effectiveness of RLAI and aripiprazole in a real-world clinical setting over an extended time period. Continuation rates with both compounds over the whole 5-year period were somewhat disappointing, which may have been due in part to the long period of follow up and number of patients previously treated with clozapine. However, a relatively large proportion of the two cohorts included in the present study remained on treatment from year two to year five, which highlighted the importance of appropriate prescribing to minimize early discontinuation and achieve good outcomes in a clinical setting.
Footnotes
Acknowledgements
The authors thank Mrs W. Davies for the opportunity to conduct this work.
Conflict of interest statement
PND has received payment for presentations and funding to attend scientific meetings from Janssen-Cilag Ltd, and payment to attend advisory board meetings from Schering-Plough.
Funding
This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
