Abstract
Background:
Clozapine (CLZ) stands out for its unique receptor profile, making it more effective than other antipsychotics, especially in treatment-resistant schizophrenia (TRS). There is growing evidence supporting its use in schizoaffective disorder (SZD), although diagnostic challenges and drug characteristics have complicated the implementation of specific clinical trials.
Aim:
Compare efficacy and tolerability of CLZ in real-world patients with TRS and SZD.
Methods:
Prospective, pragmatic, three-month, evaluator-blinded clinical trial with two randomised controlled arms (TRS groups) and a third fixed arm (SZD group). The clinical response was assessed by monthly visits during which the Positive and Negative Syndrome Scale (PANSS), Young Mania Rating Scale (YMRS), Montgomery–Asberg Depression Rating Scale (MADRS), Calgary Depression Scale for Schizophrenia (CDSS), Clinical Global Impression (CGI) and Udvalg für Kliniske Undersogelser were administered. One hundred twenty-seven participants (74.8% men, 25.2% women; 84 TRS, 42 SZD; mean age of 38.53) completed the follow-up.
Results:
Patients treated with CLZ showed a greater reduction in all the PANSS subscales, MADRS and CDSS. In cases of SZD, there was a significant decrease in positive (F: 3.72, p < 0.05) and negative (F: 6.58, p < 0.01) symptoms, the overall score of PANSS (F: 5.64, p < 0.01) and YMRS (F: 12.01, p < 0.01). Patients using CLZ had a better subjective perception of their treatment (χ2: 17.29, p < 0.01). CLZ prescription was the only predictor of better outcomes across all the scales and improved substance use (dual disorders).
Conclusions:
CLZ was effective in reducing psychotic and affective symptoms in patients with dual psychosis, with better outcomes in SZD compared with TRS.
EudraCT protocol trial:
2021-001278-44 (Comparative analysis of the effectiveness of clozapine in resistant schizophrenia and schizoaffective disorder; clinicaltrialsregister.eu/ctr-search/trial/2021-001278-44/ES).
Introduction
Clozapine (CLZ), a second-generation antipsychotic with 5-HT2A antagonism activity and a distinctive pharmacological profile, has a low affinity for dopamine D1 and D2 receptors (<60% occupancy) and a higher affinity for D4, M1, M4, 5-HT2A, 5-HT2C, 5-HT6 and 5-HT7 (Wenthur and Lindsley, 2013). Furthermore, its superiority over other antipsychotics has been demonstrated in patients with schizophrenia (Leucht et al., 2013; Okhuijsen-Pfeifer et al., 2018).
One explanation for its efficacy is the induction of ERK1/2 kinase phosphorylation mediated by the 5-HT2A receptor (Aringhieri et al., 2017) and its ‘biased agonism’ at this receptor: antagonism when G protein activation is involved but agonism in the AKT pathway (Schmid et al., 2014). Furthermore, it inhibits SNAT-2, increasing synaptic glycine to activate NMDA receptors (Javitt et al., 2005) and blocks NOX-2, inactivating microglia and promoting neuroprotection (Jiang et al., 2016).
Treatment-resistant schizophrenia (TRS) is defined by the persistence of core symptoms and functional impairment after two adequate trials of different antipsychotics, ensuring adherence (Howes et al., 2017). Some 20%–33% of people with schizophrenia are resistant to treatment (Agid et al., 2011; Meltzer, 1997), and certain patients may continue to experience symptoms that do not fall into conventional definitions of a neuroleptic response, such as clinical defectiveness or suicidal risk (Meltzer, 1997). Compared to similar molecules, CLZ produces a greater reduction of positive and negative symptoms (Siskind et al., 2016), thereby increasing cognitive performance and global functioning (Jäger et al., 2011), especially in patients with resistant positive symptoms, with the latter being the gold standard of efficacy in patients that have failed to respond to other medications (Vanasse et al., 2016).
Based on the prevalence of TRS and its rate of response to CLZ, it can be estimated that approximately 20% of patients with schizophrenia should receive treatment with CLZ (Bachmann et al., 2017; Siskind et al., 2017). However, even though patterns of use differ between countries, CLZ is still underused (Bachmann et al., 2017), which constitutes a relevant problem in routine clinical practice. Its prescription in Spain is low, in line with surrounding countries (Sanz-Fuentenebro et al., 2019). The contradiction between scientific evidence and clinical practice can be explained by professionals’ perceptions of the complexity of prescription, the potentially serious side effects and the need for pharmacovigilance (Grau-López et al., 2020), making it essential to develop new studies that reinforce CLZ effectiveness.
In addition to its recognised efficacy in TRS, there is growing evidence supporting the use of CLZ in cases with other diagnoses (Young et al., 1997) such as schizoaffective disorder (SZD) (Baethge, 2003; Jäger et al., 2011). However, there is a shortage of specific trials, partly because of the peculiarities of the drug and the diagnostic complexity of SZD. Most trials analyse therapeutic response in SZD as a schizophrenia subgroup, rather than as an independent diagnosis, which undermines the results obtained and limits their interpretability. This implies that there are large differences, in terms of recent available evidence, regarding the effectiveness of CLZ in TRS and SZD; it’s another challenge that should be addressed by research considering the two disorders separately, comparing them with each other.
SZD is an episodic and polyphasic condition characterised by the coexistence of affective disorders of moderate–severe intensity with the core symptoms of schizophrenia, the latter occurring for at least two weeks (Muñoz-Negro et al., 2015). However, there is a lack of consensus on the diagnosis and variability in the criteria used to define SZD (Lermanda et al., 2013), and so the true aetiology of the disease remains uncertain. Nonetheless, the prevalence is estimated at 0.2%–1.1% (Scully et al., 2004) and in other populations around 0.32%, compared to 0.87% for schizophrenia and 0.24% for bipolar disorder (Perälä et al., 2007).
Of note, no specific clinical guidelines are currently available for SZD, which constitutes a knowledge gap in clinical practice. Indeed, most information on SZD available in the academic literature is based on indirect evidence from studies with schizophrenia and other related disorders. Its treatment is based on the use of antipsychotics, although paliperidone is currently the only drug with an authorised indication (Gahr et al., 2011). Previous studies have pointed out the usefulness of CLZ in cases of SZD (Ciapparelli et al., 2003; Rey Souto et al., 2020), for its ability to reduce affective symptoms (Calabrese et al., 1996; Peraire et al., 2024), improve psychotic symptoms (including cognitive dimensions and negative clinical results; Small et al., 2003) and to increase psychosocial functioning (Meltzer et al., 2008).
We intend to reinforce CLZ efficacy to improve its use in routine clinical practice, emphasising the need for early prescription and comparing the drug’s response in two diagnoses that have never been compared independently, to help discriminate between them, offering results to support a new therapeutic tool for SZD.
Thus, the objectives of this current study were (1) to evaluate the efficacy of CLZ in patients with TRS, in association with other antipsychotics and in a representative sample from routine clinical practice, including individuals with comorbidities and substance use; (2) to analyse the effect of CLZ on real-world SZD psychotic and affective symptoms and (3) to compare the response and tolerability to CLZ in both patients with TRS and those with SZD. Therefore, this study aimed to demonstrate the effect of CLZ, both differentiating between these two disorders and by determining the variables that predicted greater efficacy during follow-up.
Methods
Clinical trial
Two cohorts of patients, one with TRS and the other with SZD, with continued refractory symptoms of psychosis. Prospective follow-up period: 3 months. Three intervention arms, with simple randomisation in two of them (TRS–CLZ and TRS–CONTROL). A naturalistic approach was used in the TRS–CONTROL group, as part of routine clinical practice, optimising the dose of the pharmacological regimen according to clinical response and the technical data sheet information for each drug. The randomisation procedure in TRS was completed by block allocation, after defining the two groups, based on a sequential, proportional and rater-hidden alphanumeric series.
The third arm was fixed and included SZD patients treated with CLZ
This arm could not be randomised because SZD is a disorder with low prevalence, especially considering only cases with refractory psychosis, which makes it difficult to obtain a sufficient sample during recruitment. The trial was blinded to the evaluators, who were unaware of the medication prescription, and in addition, the psychiatrists involved in the work were not those treating the participants.
Inclusion criteria
Individuals diagnosed with TRS or refractory SZD, aged over 18 years. Diagnosis by a semi-structured clinical interview according to the DSM-5 criteria. A total score on the Positive and Negative Syndrome Scale (PANSS) greater than or equal to 80 points (moderate–severe impairment), despite the prescribed medication, was considered to be a case with resistant psychotic symptoms (Leucht et al., 2005; Levine et al., 2008).
Exclusion criteria
Contraindications to the prescription of the drug, patients who were pregnant or breastfeeding or intending to become pregnant, participants who had been prescribed more than 150 mg of CLZ daily (this is the minimum therapeutic threshold established for the research, given that it is considered a low dose with limited therapeutic effectiveness and few adverse effects; Subramanian et al., 2017). The use of psychoactive substances (dual disorders) was not an exclusion criterion but was an important factor considered in the evaluation of the patients included in the study. Specifically, subjects were assessed as to whether they consumed the four most frequently used substances (tobacco, alcohol, tetrahydrocannabinol and cocaine) and how much of these substances they consumed. No systematic analysis was conducted to determine whether participants met diagnostic criteria for a substance use disorder, nor was their primary use identified.
The recruitment period was between June 2021 and May 2024, both at the hospital (a Short-Term Hospitalisation Unit and a Medium-Stay Unit) and community levels (Mental Health Units), through intentional, non-probabilistic, consecutive sampling. All these sites are part of the same healthcare network, but their administration depends on the Drugs Research Ethics Committee (CEIM) at the Consorcio Hospitalario Provincial de Castellón (Spain).
After checking the inclusion and exclusion criteria, an initial evaluation (T0) was carried out with an ad hoc questionnaire containing sociodemographic, clinical and therapeutic variables, as well as the Spanish versions of The Positive and Negative Syndrome Scale (PANSS; Kay et al., 1987; Peralta & Cuesta, 1994), Young Mania Rating Scale (YMRS; Colom et al., 2002; Young et al., 1978), Montgomery–Asberg Depression Rating Scale (MADRS; Lobo et al., 2002; Montgomery & Asberg, 1979), Calgary Depression Scale for Schizophrenia (CDSS; Addington et al., 1990; Sarró et al., 2004), Clinical Global Impression–Severity scale (CGI-S; Guy, 1976) and the Udvalg für Kliniske Undersogelser (UKU; Lingjærde et al., 1987).
Family burden calculation
One point for a first-degree relative, 0.5 for a second-degree relative and 0.25 for a third-degree relative. During the follow-up, the patients came for visits every three months (T1, T2 and T3), during which the same battery of tests was administered, except for the sociodemographic questionnaire. Analytical measurements (blood count, blood glucose and lipid profile), physical measurements (blood pressure, weight and body mass index) and complementary measurements (urinary toxins and an electrocardiogram, ECG) were also performed.
The dosage of CLZ was adjusted according to the psychopathology presented by each patient, respecting its therapeutic index (between 200 and 900 mg daily); an effective dose had already been identified from the time of the T1 visit (greater than 150 mg daily). The leucocyte count and absolute neutrophil count were monitored on a weekly basis, according to the technical data sheets. In terms of monitoring the safety of investigational drugs, we complied with the provisions of Spanish Royal Decree 1090/2015, which regulates clinical trials with drugs, the Ethics Committees for Research with Drugs and the Spanish Registry of Clinical Studies.
Regarding the pharmacological intervention, there were no differences between the trial approach applied to participants and the approach that would have been taken in the context of routine clinical practice; the only additional procedure was the administration of the assessment scales, which did not add any additional risk of harm to the participants.
At the ethical level, the principles of the Declaration of Helsinki and the Council of Europe Convention were followed. All participants (or their legal representatives) signed an informed consent to participate. The confidentiality of the participant data was guaranteed according to the General Data Protection Regulation (GDPR) established by Organic Law 3/2018 of 5 December on the Protection of Personal Data and Guarantee of Digital Rights.
The trial was registered in the EudraCT database (2021-001278-44) and the Spanish Agency for Medicines and Health Products (AEMPS) Clinical Trials Registry (REec: protocol CLZ_TRS_TEA). It was also approved by the CEIM of the Provincial Hospital Consortium of Castellón (resolution made on 20/04/2021) and the Clinical Research Ethics Committee (CEIC) of the CEU Cardenal Herrera University of Castellón (CEEI22/346). The trial did not receive funding from the pharmaceutical industry.
To check the CONSORT reporting guidelines (Schulz et al., 2010), see Table 1S (Supplemental Material). The work flowchart is summarised in Figure 1.
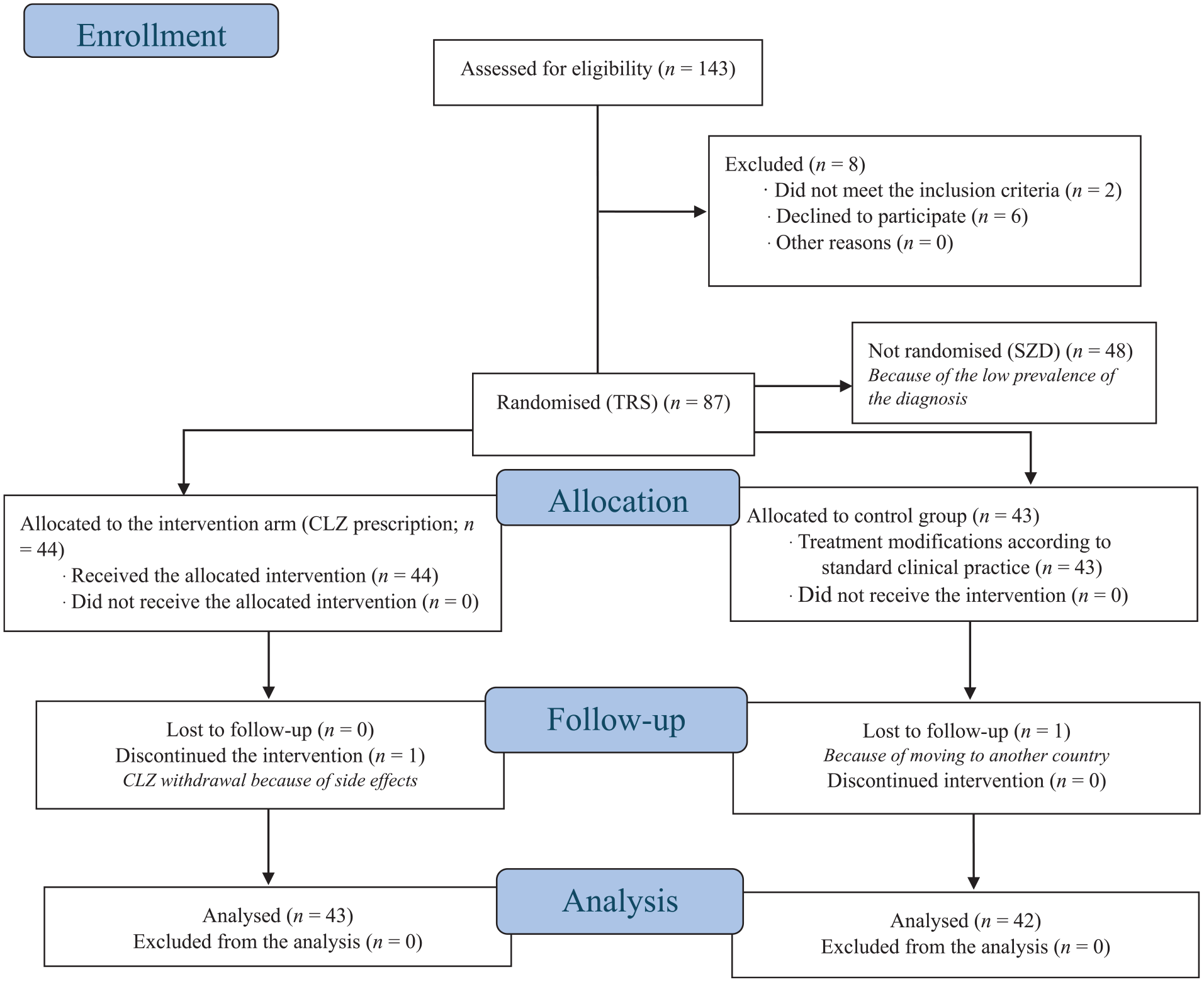
Clinical trial flowchart (according to CONSORT 2010).
Statistical and graphical analyses were performed with SPSS software v.27 (IBM Corporation, Chicago, USA), setting the significance level at 5%. Similar studies have shown decreases of more than 20% in the PANSS score after starting antipsychotic treatment (Brown et al., 2020; Schoemaker et al., 2010) and so for a 95% confidence level and 90% power, we calculated that 38 patients per group would be needed to detect differences of 10 points on the overall PANSS score. Thus, the sample size, adjusted for 5% loss, would be 40 patients per group: 120 patients in total.
After the exploratory and descriptive study, the sociodemographic and clinical variables of the groups were compared using chi squared and corrected standardised residuals for the categorical variables (measurement of the size of Cramer’s V effect) and MANOVA and Turkey or Games–Howell post-hoc tests for the quantitative variables (measurement of the size of the partial eta squared effect). To study the interaction and main effects of group (inter variable) and assessment time (intra variable), generalised linear models (mixed MANOVA) were used, controlling for the variables in which differences were found between groups. To calculate the predictive power of the variables, a generalised linear regression model was used. Both the predictive power of each individual variable (unadjusted model) and the predictive power of each variable controlling for the rest of the variables (adjusted model) were calculated. To minimise overfitting, a main effects model was used.
Trial dropouts, described in the supplementary material, were not included in the final analysis, and missing data were discarded in those analyses involving missing information. No protocol deviations were observed that required consideration.
Results
As shown in Table 1, a sample of 127 participants (74.8% male), with a mean age of 38.53 years (SD: 13.19; range: 18–70; median: 38), was recruited. The majority were Spanish (73.3%), lived in rural environments (63%), and had a primary (42.5%) or secondary (31.5%) education. Table 1 shows the sociodemographic and clinical description of the sample. Most patients were recruited from the hospital environment, either from the Short-Term Psychiatric Hospitalisation Unit (UHB; 83.4%) or the Medium-Stay Psychiatric Unit (UME; 9.4%). They had a mean of 2.71 previous admissions (SD: 2.50; range: 0–11; median: 2).
Sociodemographic and clinical analysis of the sample.
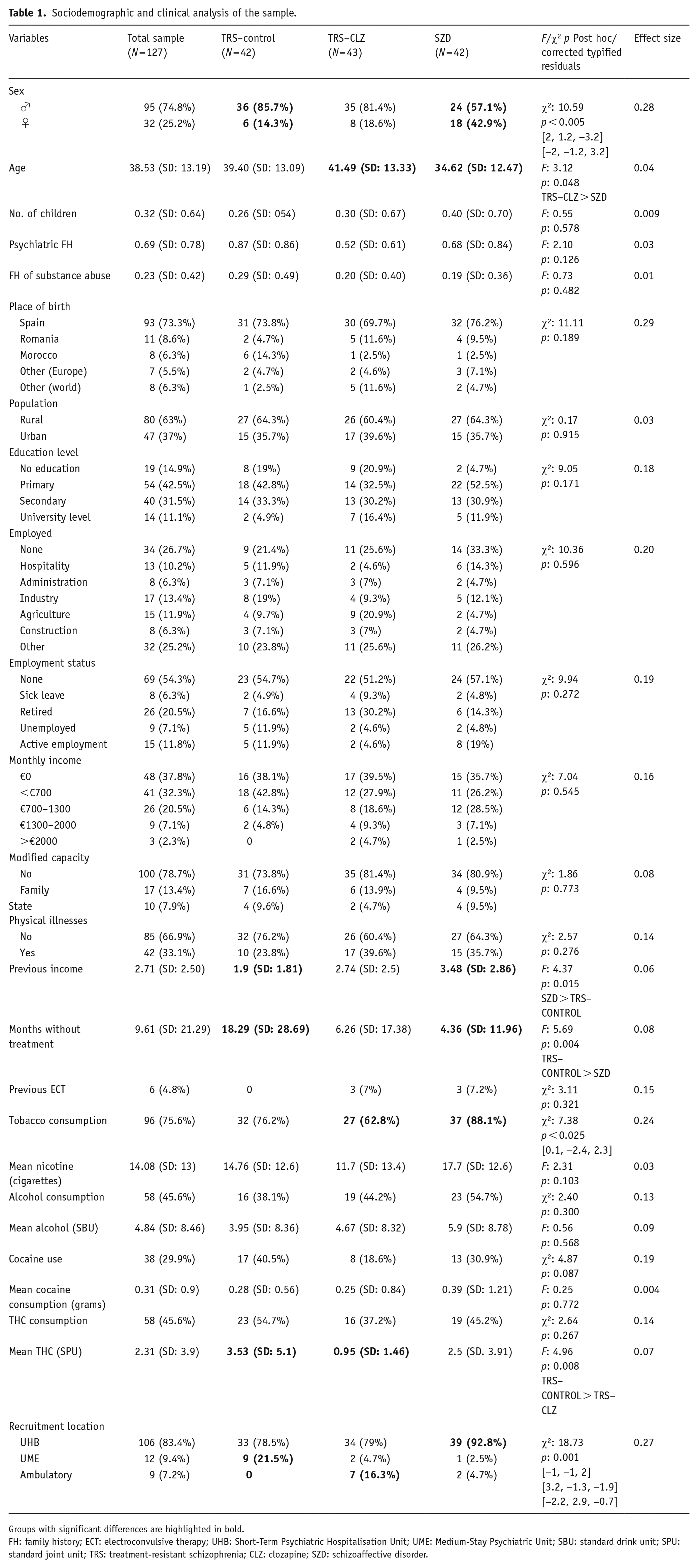
Groups with significant differences are highlighted in bold.
FH: family history; ECT: electroconvulsive therapy; UHB: Short-Term Psychiatric Hospitalisation Unit; UME: Medium-Stay Psychiatric Unit; SBU: standard drink unit; SPU: standard joint unit; TRS: treatment-resistant schizophrenia; CLZ: clozapine; SZD: schizoaffective disorder.
Before being included in the trial, the patients had gone a mean 9.61 months (SD: 21.29) without treatment, with a range between 0 (they were already in treatment) and 72 months. Regarding the consumption of the substances evaluated, 73.2% could suffer from dual disorders, excluding nicotine, rising to 81.9% if tobacco consumption was considered. Some 75.6% were nicotine smokers, 45.6% drank alcohol daily, 45.6% used cannabis and almost 30% abused cocaine.
A description of participants excluded from the trial and those who were not included in the follow-up because of secondary effects is shown in Table 2S (Supplemental Material). Of the 127 participants who completed three months of follow-up, no serious side effects were reported, nor were any criteria detected that would require a downward adjustment of the CLZ dose. Only mild secondary effects appeared (drowsiness, asthenia and hypokinesia), which were controlled by reducing the dose of the other medications. For more information, see Table 2S (Supplemental Material).
The 127 participants were divided into three groups: 42 in the TRS–CONTROL group, 43 in TRS–CLZ and 42 in the SZD group. Table 1 shows the comparative analysis between the three study arms.
We detected significant differences in sex: in the SZD group, more than 40% were women, while in the TRS–CONTROL and TRS–CLZ groups, they did not account for 20%. Participants in the SZD group were also significantly younger. Patients with SZD had significantly more previous hospital admissions, with a mean of 3.48 admissions (SD: 2.86), compared to 1.9 admissions (SD: 1.81) in the TRS–CONTROL and 2.74 (SD: 2.5) in TRS–CLZ. There were also significant differences in the number of months that they had been without treatment: the TRS–CONTROL exceeded 18 months, while the TRS–CLZ group had been without treatment for just over 6 months, and this did not reach 5 months for the SZD group.
Regarding substance abuse, significant differences were only found for tobacco, with SZD patients being more likely to smoke (88.1%). In terms of significant differences in the recruitment site, the patients in the SZD group had mainly been admitted to the UHB (92.8%); the TRS–CONTROL patients mainly came from the hospital environment as a whole, both from the UHB (78.5%) and UME (21.5%), while more than 15% of the TRS–CLZ group were outpatient referrals.
The results regarding the evolution of the patients are summarised in Table 2. In the PANSS (positive, negative symptoms and total psychopathology), YMRS, MADRS, CDSS and CGI scales, it was found that the three groups improved over time (intra effect), but the one that improved the most was the SZD group (interaction effect), so significant differences were found between the groups (inter effect), mainly between the control group and the SZD group. The results from the PANSS showed that there were significant differences in the positive symptoms. Compared with the control group, there was a significant reduction in the SZD group; there were no significant differences between the control and the TRS–CLZ groups. There were also differences on the negative PANSS subscale: in the SZD group, the reduction was significant compared to the TRS–CONTROL. For the total PANSS score, there was a greater reduction in the SZD group compared to the TRS–CONTROL group. There were no differences between the TRS groups.
Analysis of the patient evolution in terms of the psychometric scale results.
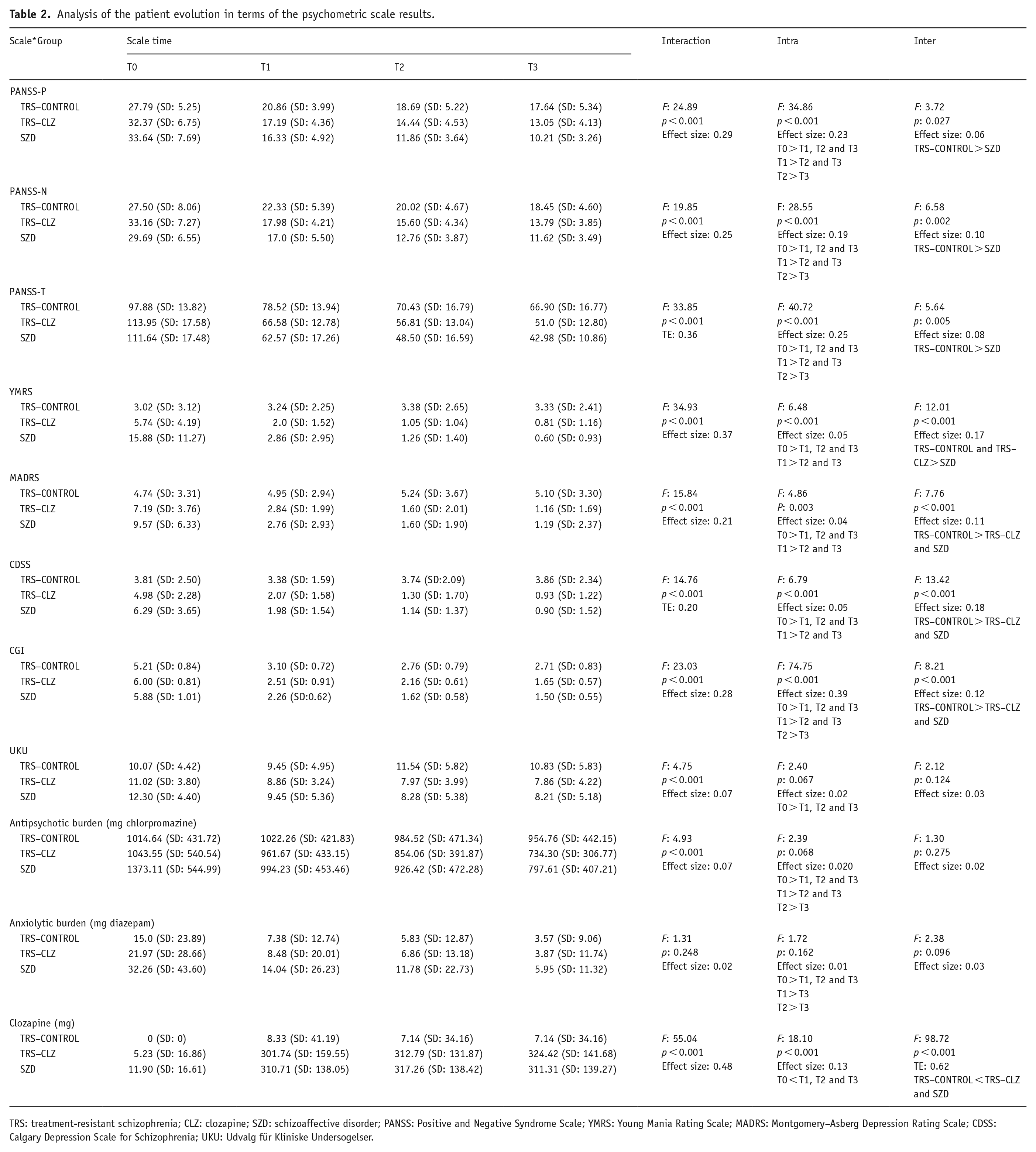
TRS: treatment-resistant schizophrenia; CLZ: clozapine; SZD: schizoaffective disorder; PANSS: Positive and Negative Syndrome Scale; YMRS: Young Mania Rating Scale; MADRS: Montgomery–Asberg Depression Rating Scale; CDSS: Calgary Depression Scale for Schizophrenia; UKU: Udvalg für Kliniske Undersogelser.
On the YMRS, there were significant differences, with a decrease in scores in the SZD group compared to the TRS–CONTROL; between the TRS cohorts, there were no significant differences.
On the MADRS, there was a significant reduction in both groups with CLZ: compared to the TRS–CONTROL, the score in the TRS–CLZ group and SZD had decreased; there were no significant differences between the intervention groups. The comparison using the CDSS obtained similar results: compared to the controls, there was a significant decrease in score in the TRS–CLZ and SZD groups; there were no differences between the intervention groups. The CGI also presented significant results in the intergroup comparison. Regarding the TRS–CONTROL group, there were differences in the SZD and TRS–CLZ groups.
In terms of tolerability (UKU scale), there were no significant differences between the participants, although the subjective perception of those receiving CLZ (both patients with TRS or SZD) was superior to that of the control cohort from the time of the second month, and the significance increased at the last time point in the trial. The patients in the TRS–CONTROL group required treatment for side effects in a significantly higher proportion by the third month of follow-up. For more information on the secondary effects, see Table 3S (Supplemental Material).
Although the mean dose of antipsychotics was reduced to a greater extent in the CLZ arms of the study, the comparison between the groups did not reach statistical significance. The same occurred with the mean load of sedative-hypnotics. In the TRS–CONTROL group, CLZ was prescribed in two participants at doses much lower than the minimum therapeutic dose: mean at T1 of 8.33 (SD: 41.19) and at T2 and T3, 7.14 (SD: 34.16).
The graphical representation of the evolution in the scale responses in which the interaction effects can be seen is shown in Figure 1S (Supplemental Material). In turn, Table 4S (Supplemental Material) reflects the evolution in the mean doses (in milligrams) of the drugs (antipsychotics or sedatives) being used in more than 10% of the participants. Mood stabilisers were not included because of the disproportionate sample size between the study arms (lithium in 2 cases with TRS versus 10 cases with SZD and valproate in 5 cases with TRS versus 15 cases with SZD).
There were significant differences in quetiapine use over time, with the mean dose having been reduced, although these changes could not be attributed to the intervention because there were no differences between the cohorts. In terms of olanzapine, there was a significant reduction in both intervention arms, compared to the TRS–CONTROL group: both the TRS–CLZ and SZD group used reduced mean doses. There were no differences between the CLZ groups.
No significant differences were found in the oral paliperidone doses or the monthly intramuscular. There were differences in the quarterly intramuscular doses, with a reduction in the SZD group compared to the TRS–CONTROL group. There was also a decrease in the mean dose of clotiapine, with this reduction being attributable to time (in T3 compared to previous time points). There were no differences for diazepam or clonazepam use.
There were no differences between the three groups in terms of the oral (N = 6; F: 1.24, p: 0.291) or monthly intramuscular (N = 11; F: 0.69, p: 0.934) doses of aripiprazole, or oral (N = 11; F: 2.55, p: 0.082) or monthly intramuscular (N = 5; F: 0.87, p: 0.421) doses of risperidone. Similarly, there were no differences for cariprazine (N = 9; F: 0.67, p: 0.513), amisulpride (N = 7; F: 0.26, p: 0.772), haloperidol (N = 5; F: 0.19, p: 0.823), lurasidone (N = 5; F: 0.79, p: 0.452) or chlorpromazine (N = 4; F: 0.61, p: 0.94).
The evolution of the analytical parameters is shown in Table 5S (Supplemental Material). There were only significant differences in neutrophil levels between the groups, with a reduction in the TRS–CONTROL compared to the SZD group. Regression models predicting the effect of variables that were significantly different at the time of recruitment, the final CLZ dose, trial arm, and baseline psychometric score, and final PANSS-P, PANSS-N, PANSS-T, YMRS, MADRS and CDSS scores are shown in Table 3.
Regression models on the psychometric scale scores at time point three (T3).
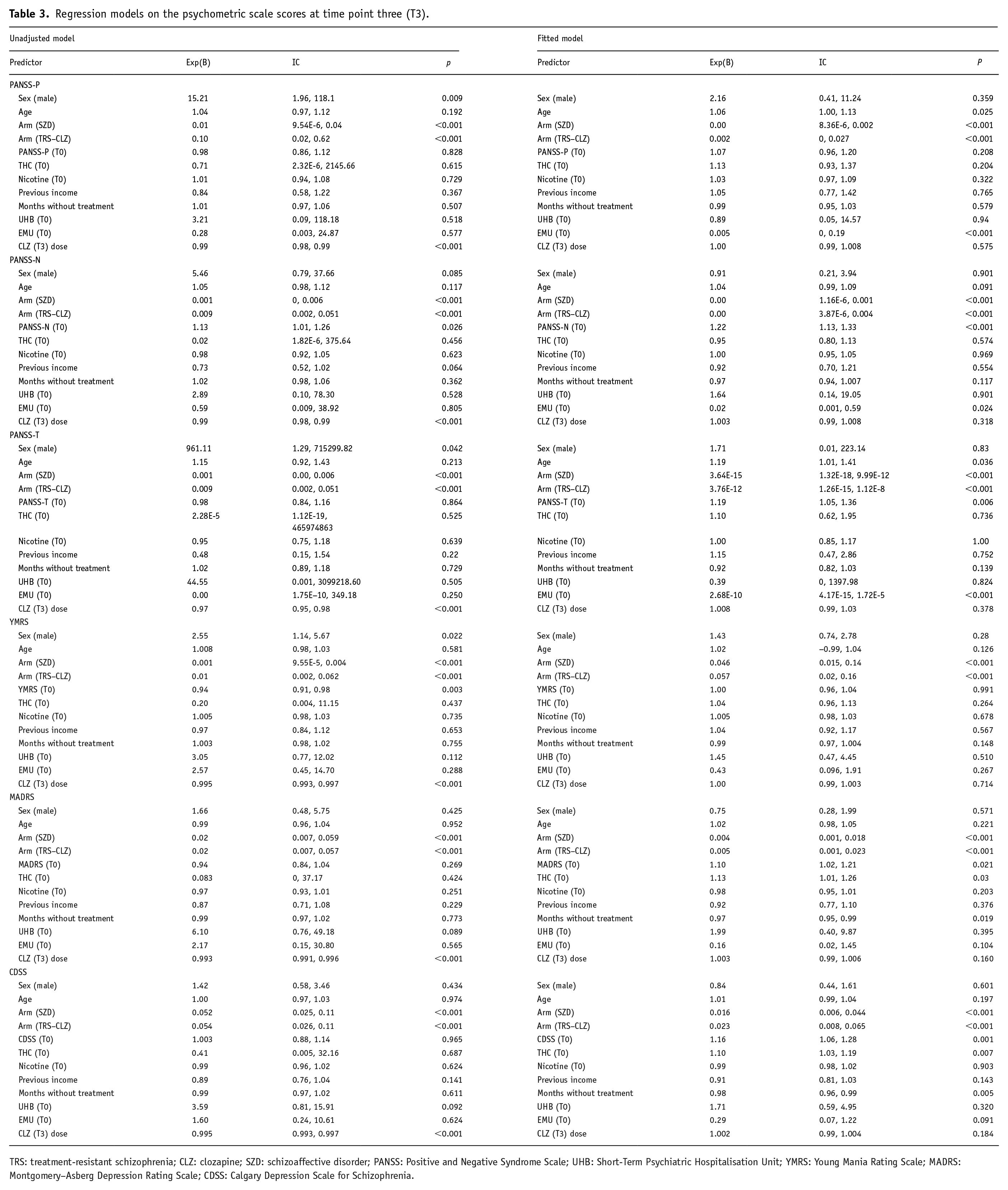
TRS: treatment-resistant schizophrenia; CLZ: clozapine; SZD: schizoaffective disorder; PANSS: Positive and Negative Syndrome Scale; UHB: Short-Term Psychiatric Hospitalisation Unit; YMRS: Young Mania Rating Scale; MADRS: Montgomery–Asberg Depression Rating Scale; CDSS: Calgary Depression Scale for Schizophrenia.
Among the variables that predicted a lower final score on all the scales, belonging to one of the two intervention arms, as well as the CLZ dose, stood out. However, in the adjusted model, CLZ dose lost its predictive power, and the two predictors associated with lower scores on all the scales were belonging to one of the intervention arms (TRS–CLZ or SZD). Recruitment in the UME also predicted a better score on the PANSS (positive, negative and total score). On the PANSS-T, older age and initial score predicted a poorer outcome at T3. For the two depression scales (MADRS and CDSS), in addition to belonging to the intervention cohorts, cannabis use, a high score at T0, and fewer months without treatment predicted a higher final score.
Discussion
This is the first study designed to longitudinally and prospectively compare the response to CLZ in patients with TRS and SZD, considering each entity independently. The results show that CLZ significantly reduced the positive, negative and total scores on the PANSS in both intervention arms. The superiority of CLZ in producing a reduced PANSS score was supported by previous studies (Meltzer et al., 2008; Siskind et al., 2017) and may be explained if the drug has a combined activity including modulation of numerous biogenic amines (including GABA) and involvement of glutamatergic and muscarinic neurotransmission (Sur et al., 2003). Release of pregnenolone in the hippocampus and cortex (Marx et al., 2006), or reversal of hypersensitivity in dopamine receptors (Kim et al., 2018), has also been proposed as potential mechanisms of action.
This trial showed that the response to CLZ in patients with SZD was superior to that of those with TRS in terms of the PANSS-P, PANSS-N and PANSS-T scores, reaching statistical significance with respect to the control cohort, as also shown in previous research (Ciapparelli et al., 2000), and may be because SZD has particular characteristics that make it respond better to medication.
Kasanin coined the term affective psychosis in 1933, based on nine cases of young people with atypical psychosis, good premorbid adjustment, florid sensory-perceptual disorders and marked emotional involvement, whose evolution was better than that of patients with schizophrenia (Kasanin, 1933). The results of this trial, a century later and in more than 100 patients, are in line with those from other work (Harrow et al., 2000; Pinna et al., 2014) and support this description: participants with SZD were younger than those with TRS, there was a higher proportion of women, and more substance use (dual disorder). Furthermore, despite having had more previous admissions and more severe psychopathology (both psychotic and affective), they responded faster and better to CLZ than those with TRS.
The outcomes of patients with SZD were also different from those of those in the two TRS arms in terms of the evolution of the YMRS results, which could explain the relevance of the manic component in the initial diagnosis (Malaspina et al., 2013). In contrast, the response for the two depressive symptom scales (MADRS and CDSS) was comparable in the TRS–CLZ and SZD groups, thereby reinforcing the antidepressant efficacy of the drug, as also suggested by previous research (Tiihonen et al., 2017).
The efficacy of CLZ was not accompanied by an increase in secondary effects but rather by a reduction in the prescription of other antipsychotics and sedative-hypnotics. Decreased polypharmacy was not only not associated with relapses, but also significantly improved symptoms (Shakir et al., 2022). Furthermore, reducing side effects prevented discontinuation: patients with CLZ presented lower discontinuation rates compared to those for other molecules in its class (Guo et al., 2011; Schooler et al., 2016).
In our work, participants with CLZ perceived their side effects as being better, also coinciding with the findings of the evaluators and as reported in previous work based on the UKU (Takeuchi et al., 2016). Indeed, previous studies have shown that patients with schizophrenia taking CLZ had a significantly better subjective experience of their mental health than those receiving other second-generation antipsychotics (Kim et al., 2006; Lewis et al., 2006). Haematological monitoring also confirmed findings from previous studies (Ingimarsson et al., 2016) in that these patients did not show significant reductions in leucocyte or neutrophil levels. The probability of cytopenias was similar between the intervention and control groups, although the TRS–CONTROL presented significantly lower neutrophil levels.
This trial confirms that belonging to the CLZ arms predicted lower scores on psychotic and affective symptom scales. Given that the main intervention was the prescription of the drug, and other covariates were controlled, this efficacy can thus be attributed to CLZ, as also indicated by a recent meta-analysis (Seppälä et al., 2021). The lack of association between the final dose of CLZ and the scores on the adjusted model may have been because of the influence of other variables typical of naturalistic studies or to the fact that the drug dose did not influence its efficacy, meaning that CLZ is effective once the therapeutic dose is reached, regardless of the amount administered.
Recruitment from the UME predicted better PANSS scores, probably because these patients were at a more advanced phase of admission with greater therapeutic adherence and had participated in rehabilitation programmes. In addition, cannabis use predicted increased symptoms of depression at the end of the trial, thereby reinforcing conclusions from previous work (Sorkhou et al., 2024). Less time off treatment predicted higher endpoint scores, because patients with more severe symptoms were detected early and were not off medication for as long; however, this did not prevent them from finishing the trial with poorer outcomes.
Our findings reinforce the evidence base supporting clozapine use, consistent with recent studies (Fabrazzo et al., 2022; Goody et al., 2024). Despite strong clinical evidence, underprescription remains a significant issue, largely due to barriers such as the drug’s complex management requirements, haematological risks and persistent misconceptions among clinicians (Jakobsen et al., 2025). Disseminating accurate information about clozapine’s benefits and teaching appropriate management strategies are crucial to reducing these barriers.
In addition, periodic re-evaluations to establish the minimum effective dose and to minimise side effects are essential for promoting adherence (Xie et al., 2025). Improving pharmacovigilance would also benefit from the creation of a centralised resource compiling clozapine drug–drug interactions, given the discrepancies currently found between sources (Govaerts et al., 2024). Such a resource would optimise the safe combination of therapies, particularly in complex cases such as dual psychosis, where relevant synergies, like those observed with disulfiram, have already been described (Hahl-Häkkinen et al., 2023).
Beyond optimising clozapine use, new therapeutic approaches are emerging. Dopaminergic modulators such as cariprazine show promise for addressing persistent negative and affective symptoms, functional impairments and substance use disorders in patients with dual diagnoses (Szerman et al., 2025). Furthermore, the development of alternative clozapine formulations, including transdermal patches, holds the potential to improve adherence while maintaining therapeutic plasma concentrations and minimising side effects (Qadir et al., 2025).
Finally, addressing these challenges through structured educational initiatives, improved pharmacovigilance and the adoption of novel treatment strategies could enhance clozapine prescription rates and, ultimately, patient outcomes. Future longitudinal studies will be essential to confirm and expand upon the promising results observed during shorter term follow-up periods. This current work had several strengths: it was a prospective, randomised, blinded clinical trial without industry funding, with more than 100 participants whose diagnoses are rarely compared. An important novelty was that a representative sample of patients selected from routine clinical practice was obtained, to whom flexible doses of the drugs under study were administered. This included participants with refractory psychopathologies, multiple previous hospital admissions, polypharmacy and active substance use (dual disorder), factors that are generally exclusion criteria in other trials. Our research offers promising results regarding the use of high-dose antipsychotic polypharmacy for patients with refractory psychosis, an area with few studies and limited evidence (Lawrence et al., 2025).
The limitations of this work include the lack of records of the number of previous trials patients had undergone with antipsychotics (or their duration), which limited the definition of treatment resistance. We also lacked records of the age of disease onset and the duration of psychotic symptoms. Furthermore, we did not study the anti-suicidal effect of the drug, although previous work supported its effectiveness in this regard (Fakra and Azorin, 2012). Moreover, serum CLZ concentration was not analysed because of laboratory limitations: patients with high rates of smoking were included, making interpretation of the results difficult. Similarly, neither thyroid hormones nor prolactin was determined in order to monitor for unwanted side effects.
Previous studies show that dual schizophrenia is associated with worse clinical symptoms and lower quality of life (Benaiges et al., 2012), with differences in daytime functioning (Adan et al., 2024). Still, our research has not compared the therapeutic response between participants with dual disorders or only with mental disorders, which limits the findings of the work. Another limitation is the lack of diagnostic reliability and stability over time, especially in SZD, a controversial label subject to considerable fluctuations across the last decades (Peraire et al., 2023). Its low reliability may alter the included subjects’ characteristics and undermine the findings’ validity. Gender disproportion, with an unequal distribution between arms, is another limitation that can influence the therapeutic response to affective symptoms. Although we hope that our findings contribute to consolidating the SZD clinical characterisation, it would be advisable to promote periodic re-evaluations of participants with SZD in the future and to conduct effective clinical analyses that consider a gender perspective.
Given that this research focused on a single health area and a specific sociocultural environment, caution should be used when generalising the results. Another important limitation is that regression models are overpopulated with predictors relative to the sample size, which risks overfitting and undermines interpretability. Finally, we only carried out a brief patient follow-up, which may not have reflected the full efficacy of CLZ: although the minimum period to evaluate the effect of drugs is usually 3 months (Barnes, 2011), it is still advisable to maintain a follow-up beyond six months (Haddad and Correll, 2018). Our intention was to design a study with a brief follow-up that would minimise participant attrition and ensure robust statistical comparisons, enabling us to establish evidence for CLZ in both diagnoses. This constitutes a significant limitation of our study, as longer follow-up could have increased clinical improvements, so further long-term studies examining our results should be encouraged.
Conclusion
CLZ was effective in reducing psychotic and affective symptoms during a three-month follow-up in patients with SZD or TRS. Results were more favourable in the schizoaffective population, and CLZ prescription was significantly associated with improvements across all psychopathological dimensions. Participants receiving CLZ reported a better subjective perception of side effects and required lower doses of additional antipsychotics and sedative-hypnotics. These findings have direct clinical implications, reinforcing the evidence supporting the use of CLZ in dual psychosis and expanding therapeutic options for SZD, a diagnosis often underrepresented in clinical trials.
Moving forward, efforts to improve clozapine use should focus on enhancing clinician education, standardising knowledge sources and promoting best practices in its management. Additionally, the development of well-designed long-term studies will be essential to consolidate and extend the promising evidence presented in our work.
Supplemental Material
sj-docx-1-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-1-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Supplemental Material
sj-docx-2-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-2-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Supplemental Material
sj-docx-3-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-3-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Supplemental Material
sj-docx-4-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-4-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Supplemental Material
sj-docx-5-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-5-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Supplemental Material
sj-docx-6-jop-10.1177_02698811251355602 – Supplemental material for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder
Supplemental material, sj-docx-6-jop-10.1177_02698811251355602 for Randomised controlled trial of clozapine in resistant schizophrenia and schizoaffective disorder by Marc Peraire, Francisco Arnau-Peiró, Mariano Villar-García, Ana Benito, Iván Echeverria and Gonzalo Haro in Journal of Psychopharmacology
Footnotes
Author contributions
Marc Peraire: conceptualisation, methodology, data curation, funding acquisition, project administration, resources, writing – original draft, writing – review and editing. Francisco Arnau-Peiró: data curation, methodology, investigation, writing – original draft. Mariano Villar-García: writing – review and editing. Ana Benito: conceptualisation, funding acquisition, investigation, writing – original draft. Iván Echeverria: writing – review and editing. Gonzalo Haro: data curation, funding acquisition, project administration, writing – review and editing.
Data availability statement
Research data are available and can be sent to the corresponding author upon request.
Declaration of conflicting interest
The authors declare that doctoral student Marc Peraire is the lead author of the present research and that all authors agree to its inclusion in his PhD thesis by compendium of articles. Non-doctoral authors waive any claim to use this article in future doctoral theses. The authors report no conflicts of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: all the authors acknowledge financial support from the Research Foundation of the Provincial Hospital of Castellon (grant numbers CAFI-24-OCT07, CAFI-25-FEB04). The Research Foundation of the Provincial Hospital of Castellon had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report and in the decision to submit the paper for publication.
Ethical considerations
This trial was registered in the EudraCT database (2021-001278-44) and the Spanish Agency for Medicines and Health Products (AEMPS) Clinical Trials Registry (REec: protocol CLZ_TRS_TEA). It was also approved by the CEIM of the Provincial Hospital Consortium of Castellón (resolution made on 20/04/2021) and the Clinical Research Ethics Committee (CEIC) of the CEU Cardenal Herrera University of Castellón (CEEI22/346).
Consent to participate
It was mandatory to receive an information sheet and sign the consent form in writing to participate in the trial.
Consent for publication
Not applicable, as the manuscript contains no person’s data.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
