Abstract
Treatment with the antipsychotic clozapine is often complicated by its wide-ranging and sometimes serious adverse effect profile. A link between clozapine therapy and diabetes is well established, although the onset and severity of glucose metabolism abnormalities is variable. Recent literature also suggests there may be an association between clozapine therapy and pneumonia. We review the relevant background literature and present a case of a patient with pre-existing type 2 diabetes mellitus and chronic obstructive pulmonary disease who presented with a diabetic emergency after a relatively short period of treatment with clozapine. He went on to develop pneumonia from which he died. We discuss the implications this case, and others alike, have for early routine physical health monitoring of this patient group.
Background
Clozapine is an important atypical antipsychotic used in treatment-resistant schizophrenia. Despite its known efficacy, it is a medication with many well-known adverse effects. As a result, its use in the UK is limited according to strict guidelines and is closely monitored [National Institute for Health and Clinical Excellence, 2009; Taylor et al. 2009a]. As with certain other second-generation antipsychotics, there is an established relationship between clozapine and impaired glucose metabolism [Henderson et al. 2005; Koller et al. 2001]. Adverse effects seen in clozapine-treated patients range from mild glucose intolerance to diabetes. In rare cases, patients can present with diabetic ketoacidosis (DKA) which may lead to death.
The mechanism of insulin resistance is thought to be via the propensity for clozapine to cause weight gain, which is a known risk factor in the pathogenesis of diabetes [Henderson et al. 2005; Newcomer, 2005]. Recent research however has suggested that in up to a quarter of clozapine-related cases of diabetes, the cause is independent of adiposity [Newcomer, 2005]. Other mechanisms implicated include a potential inhibitory effect of clozapine on glucose transporter proteins [Tovey et al. 2005]. Regarding the timing of presentation of diabetes, a small prospective study examining glucose control found that the majority of patients developed impairments within the first 4 months of clozapine treatment independent of insulin sensitivity [Howes et al. 2004]. Cases of more rapid onset and life-threatening diabetic complications also tend to occur early in the course of treatment, with 61.5% of cases of DKA occurring within 3 months of starting clozapine treatment [Nihalani et al. 2007].
Irrespective of the exact diabetogenic mechanism, the first 6 months of clozapine treatment appear to be a high-risk period and this is reflected in guidelines on the safe use of the drug. In the UK, the commonly used Maudsley Prescribing Guidelines suggest a combination of pretreatment screening of patients at risk and regular monitoring for early detection of diabetes [Taylor et al. 2009a]. The frequency of testing suggested is a fasting plasma glucose (FPG) level at baseline, repeated at 1 month, 6 months and annually thereafter. No specific guidance is given on whether or not patients with pre-existing diabetes require closer monitoring; in these cases clinical judgement is often used.
Recent studies have found an association between antipsychotic therapy and pneumonia. The link is better established in elderly patients with a possible dose-dependent relationship and increased odds ratio in those treated with atypical antipsychotics [Trifirò et al. 2010; Knol et al. 2008]. More specifically, in the adult population an association between fatal pneumonia and clozapine therapy is also suggested. In one study, pneumonia was found to be the primary cause of death in the majority of excess deaths seen in clozapine-treated patients [Taylor et al. 2009b]. The exact pathophysiological association between clozapine and pneumonia is unclear and it is important to note that this later study found that amongst the excess deaths, not one was found to have agranulocytosis at the time of death. Other possible mechanisms include aspiration pneumonia as a result of sialorrhea [Hinkes et al. 1996] or oesophageal dysfunction [Maddalena et al. 2004]; however, the exact process remains uncertain. Currently there is no formal guidance on monitoring or early intervention for this complication in clozapine-treated patients.
We present a case of a 43-year-old patient with schizoaffective disorder and pre-existing diabetes, who in the fourth week of clozapine therapy presented in a hyperglycaemic crisis and subsequently developed fatal pneumonia. We later discuss the clinical implications of safely monitoring these rare but potentially fatal adverse effects.
Case presentation
Mr D was a 43-year-old patient of Afro-Caribbean origin, with an established diagnosis of schizoaffective disorder. He first became unwell aged 24 years and experienced numerous episodes of illness characterized by thought disorder, paranoid delusions and affective symptoms. These episodes were often associated with severe self neglect and aggression and required both voluntary and involuntary admissions to psychiatric wards for management. Despite often successful resolution of his symptoms with psychotropic management, Mr D was difficult to manage in the community because of poor compliance with medication, nonengagement with services and concomitant illicit drug use. In terms of function, he was able to live relatively independently early on in his illness, however he required more support over time. His physical health was also of concern; he developed type 2 diabetes mellitus at the age of 41, had hypertension and a degree of chronic obstructive pulmonary disease (COPD), all of which required medical treatment. Contributory factors were morbid obesity, sedentary lifestyle, poor diet and heavy smoking.
Approximately 18 months before his death, Mr D was admitted to an acute psychiatric ward as an involuntary patient following a relapse in his mental state. He was re-established on oral medications including olanzapine and sodium valproate as well as oral diabetic and antihypertensive medication. After 6 months on this acute ward, Mr D was transferred to our rehabilitation unit in order to thoroughly assess his functional abilities, provide psychoeducation and offer psychological and occupational therapies. Management was however complicated by Mr D’s fragile mental state which showed rapid decompensation into psychosis after brief periods of noncompliance. These unfortunately occurred frequently and were related to chronic positive symptoms as well as poor insight into the management of his illness. During these times he would also significantly neglect his physical health needs by being noncompliant with oral diabetic and antihypertensive medications.
Generally, Mr D’s diabetic control was poor over the first 6 months of this admission, exacerbated by poor compliance with medical and dietary management. Capillary blood glucose (CBG) readings varied from the normal range up to the low 20s and at times he required administration of short-acting insulin. Following referral to a diabetologist and upwards titration of oral hypoglycaemics, his diabetes was brought under control on a regime of gliclazide 160 mg twice daily and metformin 1 g twice daily.
Medical management of Mr D’s schizoaffective disorder included switching olanzapine to quetiapine and also adding a second mood stabilizer, lithium. After months of relative nonresponse, we decided that, because of treatment resistance, we would switch the antipsychotic to clozapine. A standard 2-week clozapine titration, with routine monitoring, was commenced and quetiapine was reduced incrementally to stop over this period.
Mr D tolerated the clozapine titration well. There were no initial side effects apart from hypersalivation, which was treated with hyoscine 300 µg once daily. He was fully compliant with his medication regime. Given his past history of problematic diabetic control, we monitored CBG readings closely, taking random samples twice daily. Figure 1 shows the average daily CBG for the duration of clozapine therapy as well as the 2 weeks preceding the treatment start date. After approximately 3 weeks, Mr D was showing early response to clozapine with less thought disorder and more stable mood. Physically, Mr D appeared well; cardiac observations, weekly full blood count tests and diabetic control showed no signs of complications of therapy.
Graphical illustration of average daily CBG readings prior to and after commencing clozapine.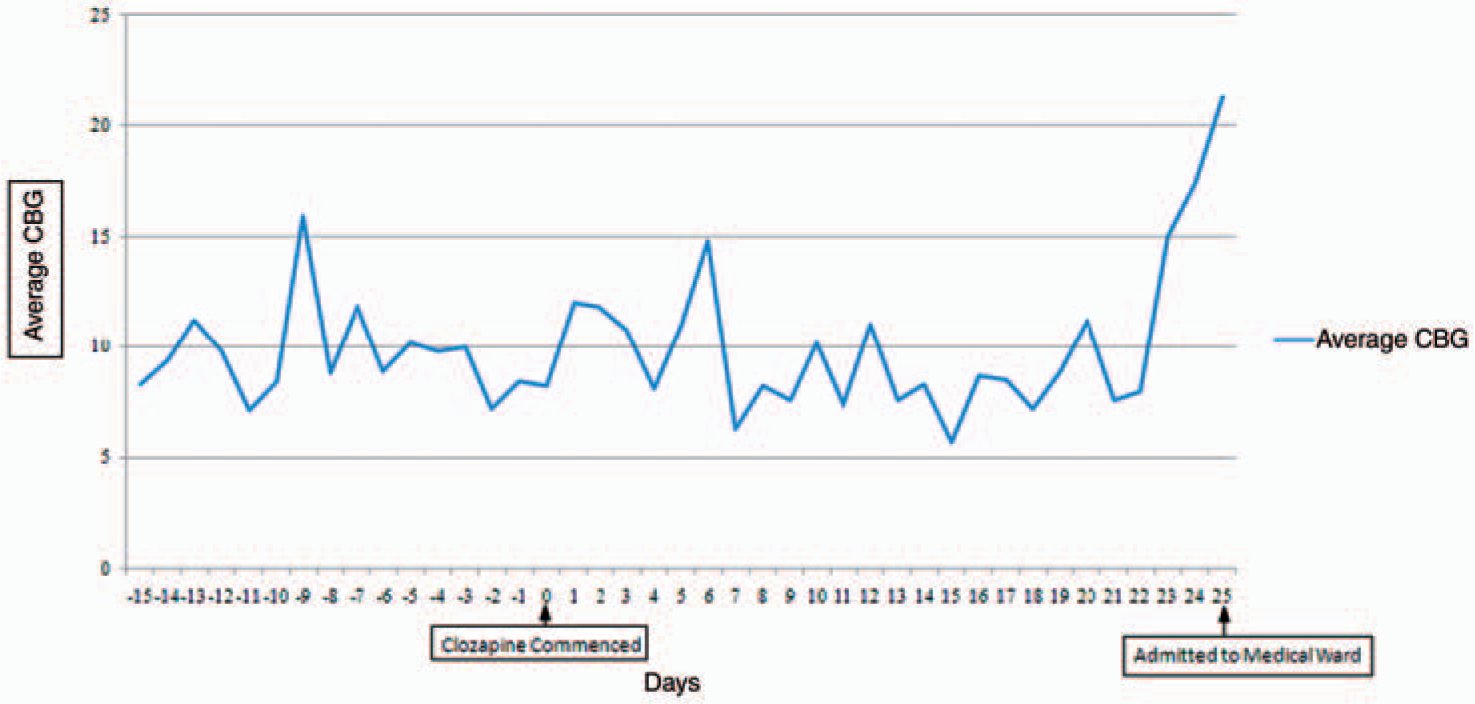
Unfortunately on day 25 of clozapine therapy, Mr D’s physical condition deteriorated. He presented with an unsteady gait, slurred speech and reduced consciousness. His CBG had been rising from normal range up to the mid 20s over a period of 24 h (Figure 1). Mr D was transferred immediately to a local medical hospital.
Initial assessment in the emergency department found a hyperglycaemic state with his CBG ranging between 15 and 22 and arterial blood gases showing a compensated metabolic acidosis. He was treated with a sliding scale of insulin and intravenous fluids. To cover the possibility of an infective exacerbation of his COPD, intravenous benzylpenicillin was commenced. Medical management was complicated by acute confusion and agitation which led to Mr D being unable to tolerate intravenous access for long periods. Eventually, blood glucose levels were brought under control with insulin. Just as Mr D appeared to be showing signs of recovery, he deteriorated once more, developing a sustained pyrexia and respiratory distress. He was treated with further intravenous antibiotics, fluids, steroids and noninvasive ventilation. Sadly, 11 days after his admission, Mr D suffered a respiratory arrest from which he could not be resuscitated. Postmortem examination found the cause of Mr D’s death to be pulmonary oedema secondary to pneumonia.
Discussion
The case presented illustrates rare but serious complications seen in early clozapine therapy. Mr D acutely lost diabetic control after only 24 days of treatment with clozapine, subsequently developing pneumonia from which he died. This occurred despite close monitoring and early intervention in treating his hyperglycaemia. As well as a hyperglycaemic state, the severity of the pneumonia is likely to have been caused by the presence of risk factors, including chronic obstructive airways disease, morbid obesity and heavy tobacco smoking. We cannot say with certainty whether or not the diabetic emergency led to pneumonia or vice versa. However the onset of hyperglycaemia before signs of infection and the presence of a metabolic acidosis on admission suggest that DKA preceded the infection.
In addition to established guidelines, attempts to guide clinicians on glucose monitoring of patients on clozapine therapy have been made in a number of consensus statements and reviews. Most recently, Hasnain and colleagues recommended monitoring for diabetes with FPG testing in patients at high risk of developing diabetes 1 and 2 months after starting treatment with antipsychotics [Hasnain et al. 2010]. The American Diabetes Association consensus statement recognized that clozapine has the highest potential to lead to diabetes [American Diabetes Association, 2004]. A more frequent monitoring regime was suggested, with FPG recommended at baseline then at 4, 8 and 12 weeks after starting treatment. A less stringent monitoring view is taken in Berk and colleagues’ consensus statement, which recommends baseline and 6-monthly FPG testing [Berk et al. 2007]. There is however a proviso that testing should be conducted following dose changes, or if clinically, diabetes is suspected.
In Mr D’s case, monitoring CBG randomly on a twice daily basis allowed us to identify hyperglycaemia at an early stage. Importantly, this occurred before the first recommended FPG test at 4 weeks, suggested by consensus opinion. There is no such established opinion on monitoring or early treatment for pneumonia. As shown in the case presentation, Mr D’s infection was treated promptly and aggressively, albeit unsuccessfully.
Establishing causality between clozapine therapy and pneumonia and diabetic complications in this specific case is of course not possible. It is possible that there was no correlation and that the patient’s death may have been independent of clozapine therapy. Patients with schizophrenia are known to have higher rates of diabetes irrespective of antipsychotic therapy [Holt et al. 2005], reflecting a likely multifactorial association. Mr D’s diabetes had been historically difficult to manage and his deterioration may have been part of his disease course. In addition, Mr D also had risk factors for pneumonia. However, given the current understanding of clozapine’s adverse effects, it is likely that a combination of pre-existing risk factors and clozapine therapy contributed to his death.
Conclusions
Given the evidence in the literature linking clozapine with diabetogenic effects and pneumonia, as illustrated in the case presented, we suggest there is a need to reconsider routine monitoring in early therapy. Guidelines and consensus opinion fail to highlight what may be a high-risk period for clozapine-treated patients, specifically the first month of treatment. Diabetic screening using FPG within the first month of treatment would not represent an extra burden on the patient because it could be incorporated into the weekly full blood count sampling. In patients with pre-existing diabetes, simple CBG could be conducted on a daily basis to monitor diabetic control. We would also suggest that patients with risk factors for developing pneumonia should be closely monitored for infection and steps taken to minimize risk by addressing modifiable risk factors.
It is also important to note that these issues have implications when considering the safety of clozapine titration in the community rather than under closer supervision in the hospital setting.
We appreciate that patients with schizophrenia have high levels of physical comorbidity irrespective of specific iatrogenic mechanisms. Therefore, in clinical practice, the decision to treat patients with clozapine will be based on a risk–benefit analysis. However with further research, high-risk patients requiring closer monitoring, who are more susceptible to presenting with diabetic emergencies and/or pneumonia may be identifiable in the future.
In conclusion, we recommend raised clinician awareness of the potential for serious diabetic and respiratory complications in early clozapine therapy. In addition to closer monitoring and prompt treatment, we suggest a rethink of clinical guidelines reflecting the need for early detection of such cases.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors have no competing interests to declare that are relevant to the content of this submission.
