Abstract
Background:
This study develops an economic model to evaluate the cost-effectiveness of ethyl-eicosapentaenoic acid (ethyl-EPA) as an adjunct treatment of bipolar I disorder.
Methods:
A 1-year Markov model is used incorporating three health states: euthymic, manic and depressive. The model was populated using outcomes from a clinical trial on clinical efficacy and other published literature.
Results:
The incremental cost-effectiveness ratio (ICER) per quality-adjusted life year (QALY) of ethyl-EPA in comparison with placebo was estimated to be -£2,782 in 2008/09 prices, the negative ICER indicating ethyl-EPA to be a more effective and less costly treatment option than placebo in terms of cost savings of other resource use.
Conclusions:
The sensitivity analysis indicated that the results were robust. Future research covering a longer time period using broader costs of the disease will be required to consolidate these findings.
Introduction
Bipolar disorder (BD) is a chronic illness characterized by recurrent mood episodes resulting in profound negative effects on the interpersonal, social, family and vocational outcomes [Maj et al. 2000].
The economic burden of bipolar disorder is substantial. On average the annual National Health Service (NHS) cost of managing BD has been estimated to be approximately £200 million, of which hospital admissions accounted for 35% [Das Gupta and Guest, 2002]. The direct nonhealthcare cost was estimated to be approximately £90 million annually and the indirect societal cost was estimated to be in the region of £1800 million annually.
Medication is the cornerstone of the treatment for BD but is partially effective for most patients hence the need to evaluate new treatments. One potential treatment option is ethyl-eicosapentaenoic acid (ethyl-EPA). Frangou and colleagues conducted a 12-week double-blind trial to study the efficacy of ethyl-EPA as an adjunct treatment for bipolar disorder [Frangou et al. 2006]. The Markov model developed in this paper used the data from that study.
Methods
The clinical trial
In this double-blinded placebo-controlled clinical trial outpatients with bipolar depression were randomly assigned to adjunctive treatment to mood stabilizers with placebo (n = 26) or with 1 g/day (n = 24) or 2 g/day (n = 25) of ethyl-EPA. The demographic and clinical characteristics of the study participants are given in Table 1. The concomitant medication taken by the participants in each of the arms is given in Table 2. These characteristics did not differ significantly between groups, other than for the use of antipsychotic medication (Fisher’s exact test, p = 0.01). The duration of the trial was 12 weeks and the primary outcome measure was changes in Hamilton Depression Rating Scale (HDRS) score [Hamilton, 1960]. The key finding was the depressive psychopathology as measured by the HDRS was lower by 3.3 (standard error [SE] = 1.40) points for the ethyl-EPA groups as compared with the placebo group. This difference was statistically significant (95% confidence interval [CI] -6.1 to -0.2, p = 0.03). In addition, patients in the placebo arm experienced a mean of 3 days in hospital (due to two patients being admitted) compared with a mean of zero for the ethyl-EPA arm, and this was not statistically significant. The blinding of participants in the study was achieved: there were no group differences regarding participants’ ability to guess their group allocation and only 23% of the placebo group, and 21–24% of the ethyl-EPA groups guessed their allocation correctly. The aim of this study was to specify the cost-effectiveness of adjunctive ethyl-EPA compared with placebo based on the clinical trial data using a Markov model. The parameter values of relative risk (RR) and resource use were taken from the clinical trial. However, the model parameters regarding health state utilities, unit costs and baseline transition probabilities were not available in the clinical trial and were obtained from the published literature.
Demographic and clinical characteristics of 75 study participants (Frangou et al. 2006).

NB: Data relating to preceding 12 months excludes clinical characteristics and states at entry.
SD, standard deviation.
Participants’ concomitant medication at the time of study entry (Frangou et al. 2006).

Model structure
The dynamic nature of the Markov model captures the long-term clinical aspect of BD. The model assumes a hypothetical cohort of 1000 patients entering the model in a stable (euthymic) health state with fixed transition probabilities of moving to manic and depressive states. The length of the cycle is assumed to be 3 months, which is compatible with the NICE Bipolar Guideline CG38 which suggests average length for a manic episode of 9 weeks and 13 weeks for a depressive episode [National Institute for Health and Clinical Excellence, 2006]. At the end of the cycle all of the patients experiencing acute episodes are assumed to transition back to the stable state before developing a subsequent acute episode. A gap of one cycle is assumed between the episodes; this conforms to the clinical aspects of BD. However, this assumption might appear as an oversimplification of reality particularly in the case of rapid cycling patients. The assumption implies that the time period between initiating two episodes is 6 months. The decision tree showing two cycles is given in Figure 1 and the Markov schematic diagram in Figure 2 shows the transition of patients between different states.

Decision tree.
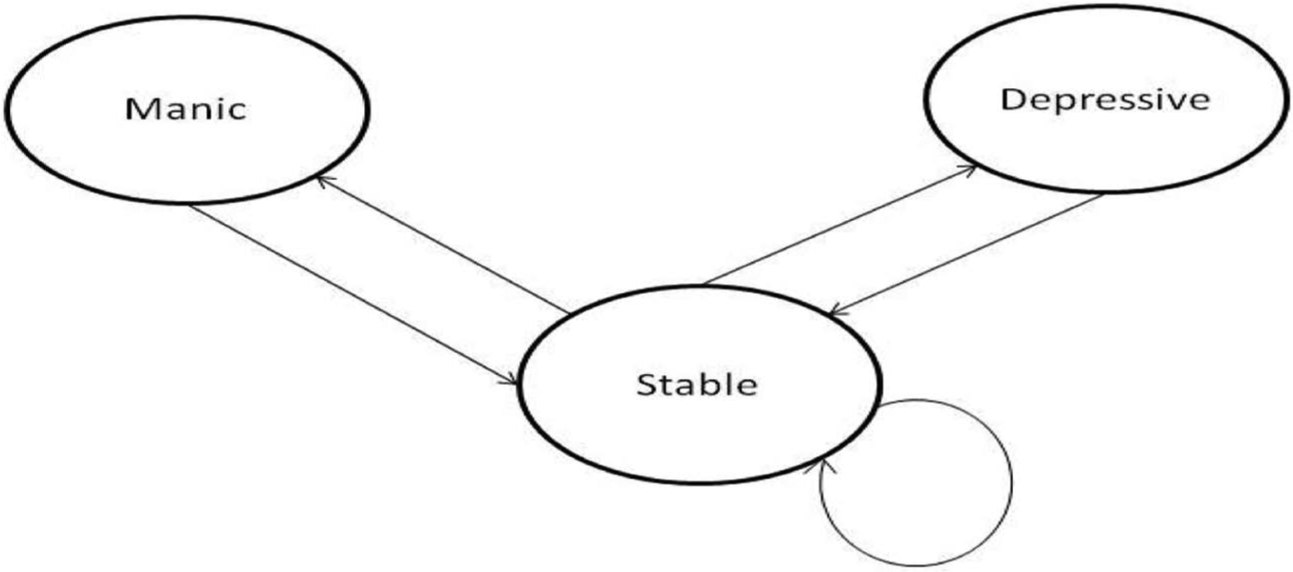
Markov schematic.
Data
Transitional probabilities
Transitional probability estimates are obtained from the published literature and supplemented with the data from the clinical trial. The estimates of transitional probabilities of three states used in the model for the control arm are taken from the work of Fajutrao and colleagues [Fajutrao et al. 2009] after adjustment are given in Table 3. Fajutrao and colleagues estimated transition probabilities using pooled data of clinical trials. The rate of recurrence of mood events was calculated from the data and subsequently transformed to obtain quarterly transition probabilities.
Transitional probabilities.
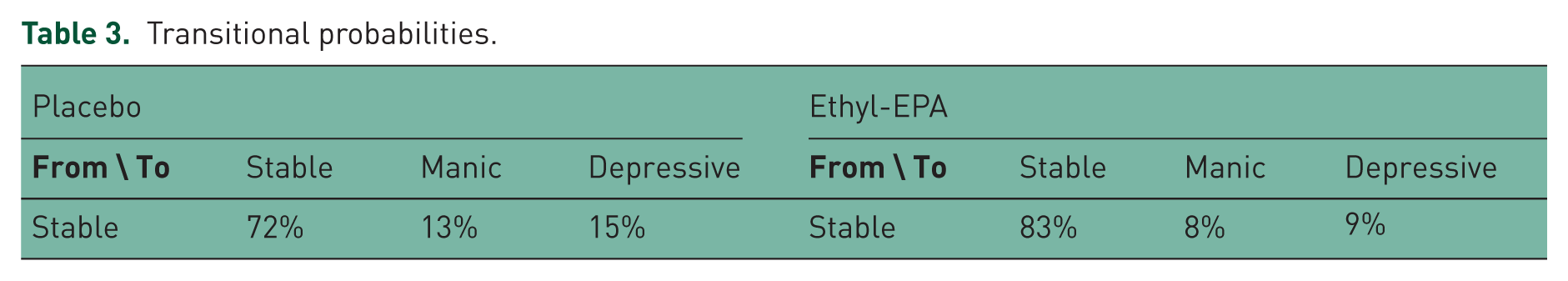
The estimated RR of acute episodes between the two groups (lithium/valproate + ethyl-EPA versus lithium/valproate + placebo) in the Frangou and colleagues [Frangou et al. 2006] clinical trial is 0.6. The transitional probabilities of the group receiving lithium/valproate + ethyl-EPA is estimated by multiplying the estimated RR (0.6) by the quarterly transitional probabilities of the control arm of the trial; these are given below in Table 3. The transition probability of patients from manic/depressive states to the (euthymic) stable state is 100% as the model assumes that the patients transition back to a (euthymic) stable health state after an acute episode.
Health-state utilities
Calvert and colleagues [Calvert et al. 2006] estimated health-state utility values using a standard algorithm and the 36-item form supplemented with published literature [Tengs and Wallace, 2000]. Their analysis assumed 0.8, 0.7 and 0.4 utility values for (euthymic) stable, manic and depressive health states. Fajutrao and colleagues also used same health-state utility values [Fajutrao et al. 2009].
Based on these studies we used 0.8 and 0.4 utility values for the stable and depressive state, respectively. The depressive episodes are frequent and longer in duration and hence have a greater impact on the quality of life. The estimate of manic state utility was adjusted to 0.65 in the model using data from Revicki and colleagues and Soares-Weiser and coworkers [Revicki et al. 2005; Soares-Weiser et al. 2007]. The patients experiencing mania face potential difficulty and tend to elicit higher value to their health state than reality.
Cost and resource use
The clinical resources consumed were estimated using data from the Frangou and colleagues [Frangou et al. 2006] trial. The per unit costs are taken from the work of Netten and colleagues [Netten et al. 2001] and are given in Table 4. The costs are adjusted to 2008/09 prices using the Hospital and Community Health Services (HCHS) index. Patients participating in the Frangou and colleagues [Frangou et al. 2006] trial were only categorized into three health state groups (stable, manic and depressive) irrespective of treatment arm allocation. The average cost of patients in each health state during the 12-week trial gave the estimate of direct cost of one cycle for the patients receiving placebo: £847 for stable, £1439 for manic and £1220 for depressive cycle. The management of an acute manic episode was assumed to be more resource intensive than treatment of depressive episodes, given the higher percentage of hospitalized patients with manic episode (80% manic versus 10% depressive hospitalized) [Soares-Weiser et al. 2007].
Unit costs (2008/09 £s per hour unless stated otherwise).

Service use costs were derived from Netten et al. [2001].
The drug cost £24 for ethyl-EPA was added to reach the estimate of direct cost of one cycle for the patients receiving ethyl-EPA treatment. Costs and quality-adjusted life years (QALYs) were not discounted given the 1-year duration of the model.
Sensitivity analysis
A deterministic sensitivity analysis (one-way and two-way) and probabilistic sensitivity analysis (PSA) were conducted to take into account uncertainty associated with the parameter values used in the model. In the deterministic sensitivity analysis input parameters were varied by 25% in either direction and the results are presented as a tornado diagram. The two-way sensitivity analysis was conducted on the two main input drivers of the ICER estimate, i.e. the utility in the stable health state and the costs of three health states. The PSA attributed appropriate probability distributions to the input parameters.
Results
The ICER estimated from the model was –£2782 at the end of 1 year, which means the use of ethyl-EPA as an adjunct therapy for BD is more effective than placebo and it reduces cost. The main factor contributing toward reduction in cost is the lower transitional probability to manic and depressive episodes for the patients taking ethyl-EPA. This means that fewer ethyl-EPA-treated patients experienced acute episodes as compared with the placebo group. Hence, service use (such as hospitalization) was lower in the ethyl-EPA group and consequently their treatment costs were lower. The additional drug cost of ethyl-EPA was small (£24) per cycle as compared with the reduction of service use elsewhere.
In the Frangou and colleagues [Frangou et al. 2006] trial no inpatient episode was recorded among the patients allocated to ethyl-EPA adjunct treatment as compared with, on average, 3 days of inpatient treatment (daily cost of an inpatient episode of £210) in the placebo arm. The number of inpatient episodes in the case of the placebo group is in line with the RR (0.6) of acute episodes estimated. Two patients in the placebo arm totalled 216 hours of day centre contacts (hourly cost of day centre of £9), while no patients in the ethyl-EPA arm had day centre contacts.
Lower scores of HRSD and Young Mania Rating Scale (YMRS) in the ethyl-EPA compared with placebo group at the week 12 assessment support better quality of life among patients receiving ethyl-EPA, which is estimated in the model as higher number of QALYs.
Greater effectiveness and reduced cost contributed toward negative estimate of the ICER, which implies use of ethyl-EPA as an adjunct treatment for BD is a dominant treatment, falling in the dominant quadrant (II) of the cost-effectiveness plane. Although, the data used from the clinical trial covered a very short time period, the model was extended to 5-year time period, using a discount rate of 3.5% for costs and outcomes, the estimate of 5-year ICER was very close to the 1-year ICER estimate.
Sensitivity analysis
The tornado diagram in Figure 3 shows how the change (25% increase and 25% decrease) in the value of inputs affect the estimate of ICER. The diagram shows that the main input drivers of the ICER estimate are the utility in the stable health state and the costs of three health states. The tornado diagram also shows that the ICER estimate is negative despite a 25% increase or decrease in the values of most of the inputs. The one-way sensitivity analysis shows that the estimate of ICER was robust. A 10% decrease in the stable health-state utility value improved the ICER estimate by 45% and a 10% change in the value of stable health-state cost, in either direction, changed the ICER estimate by 47%. The model used the estimate of RR (0.6), a 10% variation in RR (0.54–0.66) estimated the ICER in the range –£3431 to –£1923 which gives the robust result for the dominant strategy.

Tornado diagram.
A two-way sensitivity analysis showed comparable results. Nearly all of the estimates of ICER were negative as shown in Table 5. The two-way sensitivity analysis of two main input drivers also showed robust ICER estimates of the ethyl-EPA as an adjunct therapy of bipolar disorder.
Two-way sensitivity analysis: (i) cost of stable state (ii) utility of stable state.

There is uncertainty associated with the estimates of inputs and the health related data. PSA takes into account uncertainty by assigning distributions to the input variables. The characteristics of cost data are represented by gamma distribution, transition probabilities and health-state utilities are represented by beta distributions. A total of 10,000 simulations of the model are run on MS Excel, the PSA findings are presented in a cost-effectiveness acceptability curve (CEAC) shown in Figure 4.

Cost-effectiveness acceptability curve (CEAC).
The CEAC shows the percentage of simulations which are cost-effective at different willingness-to-pay (WTP) levels. As we are assuming NHS perspective which is the direct payer (although some of the informal care costs are also added), the lower NHS threshold of £20,000 has 94.67% probability for the ethyl-EPA as an adjunct treatment to be cost-effective. The high probability of cost-effectiveness was expected as the estimated ICER is also negative suggesting a dominant strategy. The mean ICER of 10,000 simulations is –£2421.
Discussion
The results of the model and sensitivity analysis present a strong case for the cost-effectiveness of ethyl-EPA as an adjunct treatment for BD. Ethyl-EPA was dominant in that it resulted in lower costs and better outcomes than the placebo. Other modelling studies have produced evidence of cost-effectiveness for haloperidol in the treatment of mania and to some extent olanzapine [Bridle et al. 2004], olanzapine maintenance treatment for bipolar disorder [McKendrick et al. 2007], and lamotrigine compared with olanzapine, lithium, and no treatment [Calvert et al. 2006]. To aid the development of clinical guidelines for bipolar disorder in the UK a Markov model was constructed to compare drug treatments and this found that valproate dominated olanzapine [National Collaborating Centre for Mental Health, 2006]. A similar model was developed by Soares-Weiser and colleagues and this found valproate to be the least expensive nondominated treatment for patients who had recently experienced a depressive episode [Soares-Weiser et al. 2007]. For those who had had a manic episode, olanzapine dominated all other treatments except for lithium. None of these studies report on the cost-effectiveness of ethyl-EPA.
The results need to be interpreted in light of the limitations of the model. The small sample size and the missing values of resource use did not allow estimation of costs of the six health states individually. Instead the three health-states costs were estimated and the ethyl-EPA drug cost (£24) was added for the three health states of the treatment group. The health-state costs for the placebo and the treatment group may differ due to different resource use and adverse effects of the treatment. Different transitional probabilities for the patients receiving placebo and the patients receiving ethyl-EPA can capture this effect to a certain degree as different numbers of patients have manic and depressive episodes and consequently different level of resource use.
Assigning different utility values for inpatient and outpatient acute episodes and additional health states to reflect the severity of episodes can complicate the model but it can capture the complex nature of the disease. However, it is not possible with the limited data available. The model also assumes that the probability of an event is independent of the previous episode and constant over time. However, in reality a mood episode might be influenced by the previous episode or hospital admission. Soares-Weiser and colleagues showed that patients’ most recent episode is related to the type of their next episode [Soares-Weiser et al. 2007]; the treatments aimed at preventing depression may be more cost-effective in patients with a recent history of depression compared with patients with a recent history of mania and vice versa for treatments aimed at preventing manic episode. In the 1-year model developed in this paper the average number of acute episodes per patient is less than one per patient therefore it is unlikely to have a substantial effect on the results.
Given the chronic nature of BD and the relatively early age of onset [Soares-Weiser et al. 2007], a 1-year time horizon could be considered too short to capture the lifetime costs and benefits of the treatment. A longer time period model is needed to reflect the actual course of BD and lifetime costs and benefits. Economic models frequently make extrapolations from short trials to longer time periods. Here we have made extrapolations to 1-year on the basis of data collected from a 12-week trial. In subsequent analyses we also extended the model to five years and clearly this is a limitation implying that we need to be cautious about the results from the extended model. In addition, adherence to the same treatment is expected to be low after an acute episode therefore the assumption of the same treatment has its own limitations. The cost perspective also needs to be broadened to include wider societal costs of BD.
Conclusion
The economic model estimated cost-effectiveness of ethyl-EPA as an adjunct treatment for BD patients over the time period of 1 year. The model found evidence of high likelihood for adjunct ethyl-EPA treatment to be cost-effective at a very low WTP threshold. The results of the present study are encouraging with regards to the cost-effectiveness of ethyl-EPA in the treatment of BD. However, the ultimate test of the cost-effectiveness of any intervention is how it performs in ordinary care. We could not test this directly as we do not have any observational datasets on the use of ethyl-EPA from routine clinical practice but this should be the next step in future studies.
Footnotes
Acknowledgements
Cost data used in the study were based on earlier calculations by Francis Swaray while an MSc student at City University and on placement at the Institute of Psychiatry. Professor Mireia Jofre-Bonet at City University supervised the research.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
The authors declare that they have no conflicts of interests regarding the content of this research paper.
