Abstract
Cocaine use disorder (CUD) is a debilitating psychopathology, with no recommended medication therapy or specific psychological intervention. Memory-focused cognitive therapy (MFCT) is a novel psychotherapy for CUD, theorized to modify and reconsolidate cocaine craving-related memories for cognitive and behavioral control. A pilot randomized controlled trial indicated that this therapy is associated with reduced craving and cocaine use. With an 80% confidence interval (CI) set for null hypothesis testing, we conducted an exploratory causal mediation analysis with confounder adjustment to determine whether increased cocaine abstinence following MFCT is mediated by reduced craving experience and increased emotion regulation. Participant data on the Difficulties in Emotion Regulation Scale did not meet screening evaluation as a potential mediator. Cocaine craving (assessed by the frequency version of the Craving Experiences Questionnaire) was associated with a total treatment effect of MFCT on cocaine abstinence at follow-up (1.499; 80% CI 1.114 to 1.970; p = .012). A significant natural indirect effect indicated that reductions in cocaine use were strongly mediated by reduced frequency of craving experience (1.753; 80% CI: 1.334 to 2.936; p < .0001). This study provides exploratory evidence in support of the theoretical action for MFCT and underscores the importance of craving as a therapeutic target.
Introduction
Cocaine use disorder (CUD; Diagnostic and Statistical Manual [DSM-5]; American Psychiatric Association, 2013) is a debilitating psychopathology. In DSM-5, CUD has 11 symptoms, spanning increased tolerance, withdrawal symptoms, urges to use cocaine (craving), and several behavioral, health, and social problems associated with chronic consumption.
Among these symptoms, influential models of addiction position the cognitive-affective construct—craving—in a key disorder maintaining role (Robinson & Berridge, 1993; Tiffany, 1990; West, 2006). Craving is a highly subjective construct. It is clear from patient reports that the content and strength of craving varies widely. An episode can be brief or protracted and distressing. Etiologically, craving can be understood as the product of a drug exposure and associative learning process in which previously drug-neutral situations, objects, people, and sensations/moods that are present when cocaine is obtained become conditioned stimuli (CS; O’Brien, Childress, McLellan, & Ehrman, 1992). An encountered CS (or a direct drug-related cue such as the sight of cocaine or cocaine paraphernalia) can trigger a process of cognitive elaboration in which memory of past drug use—often in the form of a vivid sensory mental image (May et al., 2014; May, Andrade, Panabokke, & Kavanagh, 2004) —induces a desire for pleasure or the need to alleviate anxiety, stress, or cocaine-related withdrawal symptoms (Baker, Piper, McCarthy, Majeskie, & Fiore, 2004; Koob, Caine, Parsons, Markou, & Weiss, 1997). Negative reinforcement, in particular, can be very strongly motivating for some people with CUD, leading to problems with the regulation of emotion (Cheetham, Allen, Yucel, & Lubman, 2010; Fox, Axelrod, Paliwal, Sleeper, & Sinha, 2007).
In CUD, cocaine-related imagery can drive the maintenance of pro-drug (approach) thoughts, appraisals, and dysfunctional beliefs (Andrade, May, & Kavanagh, 2012; Conway, Meares, & Standart, 2004). These cue-induced responses can persist long into abstinence and, if not controlled, can cause relapse (Parvaz, Moeller, & Goldstein, 2016). It is also important to recognize that if a person with CUD has immediate access to cocaine, there may be minimal or no craving-related elaboration (Tiffany, 1990). However, if there is a delay in obtaining cocaine, or there is ambivalence with the desire to be abstinent, craving is likely to be strong, distressing, and highly motivating (Kavanagh, Andrade, & May, 2005).
There have been many efforts to develop an effective therapy for CUD. However, to date, there are no licensed medications or guideline-recommended specific psychosocial interventions (National Institute for Health and Care Excellence, 2007). Among the latter, cognitive behavioral therapy (CBT) has been the most widely trialed intervention. A meta-analysis of 53 randomized controlled trials targeting alcohol or another drug disorder calculated that stand-alone CBT is associated with a small overall standardized mean difference for the treatment effect versus control (Hedge’s g = .154; Magill & Ray, 2009). A more encouraging picture emerged when the authors pooled results from trials that combined CBT with another psychosocial intervention (typically general drug counseling). This strategy was associated with a medium effect size (g = .305; p < .005; 19 trials).
The population of cocaine users is heterogeneous. Some use infrequently (typically powder users), but there is a subpopulation who use smokable (crack) cocaine or cocaine powder very intensively and develop CUD; many with considerable unmet treatment need. In England in 2016–2017, it was estimated that there were 760,000 powder cocaine users and 181,000 crack users in the general population (Home Office, 2017; Public Health England, 2017a). In that year, 42,403 people presented to community addiction treatment clinics for help with cocaine-related problems (16,892 powder cocaine and 25,511 crack) of whom 52% had a co-occurring opioid use disorder (Public Health England, 2017b). Community services offer people with CUD general counseling but many do not engage with this intervention or discontinue treatment after a short time. Among those who are retained in treatment, continued crack cocaine use is a significant predictor of poor response to medication for opioid use disorder (Marsden et al. 2012; Marsden et al. 2019).
Against this background—and encouraged by the potential for a CBT intervention to be effective when offered alongside ongoing general counseling and support—we developed a novel cognitive therapy intervention with the goal of helping people to better recognize, modify, and control cocaine craving-related thoughts, emotions, and behavior. Our memory-focused cognitive therapy (MFCT) is a 15-week, outpatient, individual psychotherapy (Marsden et al., 2017). In addition to a formulation-driven assessment and with use of, cognitive restructuring and behavioural experiments, MFCT adapts to CUD the fear memory reliving, imagery rescripting, and memory reconsolidation paradigm successfully used for the treatment of posttraumatic stress disorder (PTSD) (Brewin, Gregory, Lipton, & Burgess, 2010; Grey, Young, & Holmes, 2002; Holmes & Mathews, 2010). Our MFCT therapy protocol also includes a cue-induction procedure to elicit images and affective responses as therapy targets (Hon, Das, & Kamboj, 2016; Xue et al., 2012).
In psychotherapy, an important part of the evidence-gathering process for a new intervention is to address theoretical hypotheses of how treatment causes change (Kazdin, 2007; Murphy, Cooper, Hollon, & Fairburn, 2009). This is typically done by conducting a causal mediation analysis (Hayes, 2014; MacKinnon, 2008). A causal mediation analysis tests whether there is evidence that the psychotherapy exposure is related to changes in a hypothesized mediator and whether the mediator is associated with changes in a subsequent outcome. A randomized controlled trial provides the logical design conditions for causal inference, because participants are randomly allocated to the levels of therapeutic exposure.
Prior to the analysis of the developmental study (Marsden et al., 2018), the statistical analysis plan for the primary and secondary outcome measures was registered (Centre for Open Science; https://www.osf.io/3kfzj/). The results showed that compared to an assessment-only control, the intervention was associated with lower levels of craving (bias corrected Hedge’s g = −1.62; 95% confidence interval (CI) −2.45 to –0.80; the primary outcome measure) and more abstinent days (g = 1.19; 90% CI 0.54 to 1.84; the drug use secondary outcome measure). In that report, we stated our plan to determine if craving and emotion regulation mediate cocaine use, thereby giving evidence of the MFCT’s theoretical change mechanism.
In this article, we present the results of an exploratory causal mediation analysis to estimate the extent to which MFCT is associated with cocaine abstinence through craving experience and emotion regulation. We predict that reduced craving and improved emotion regulation mediate observed treatments effects of MFCT at follow-up.
Method
Design, setting, and participants
Data for the present study were from a completed and published single-site, 15-week, 2-arm, randomized controlled trial contrasting MFCT (the intervention; n = 16) to an assessment and cocaine cue-induction only group (the control; n = 14).
The published protocol provides a detailed description of the study procedures and interventions (Marsden et al., 2017). Briefly, the intervention comprised three 90-min pre-randomization assessments; two 30-min cocaine cue-induction procedures; five 120-min individual MFCT sessions over consecutive days; and three 60-min MFCT-relapse prevention discussions and research follow-ups at 1 week, 1 month, and 3 months conducted as personal interviews at the clinic. The control group received the three-session prerandomization assessment, the two cue-induction procedures, and participated in the three research follow-ups only.
The trial was done at an English National Health Service community addictions clinic operated by South London and Maudsley Trust and at the National Institute for Health and Research and Wellcome Trust Clinical Research Facility at King’s College Hospital, London. Ethical approval for the protocol was granted by the UK National Research Ethics Service.
Patients (aged 18 years and over) receiving ongoing general drug counseling were eligible for the trial if they were diagnosed with CUD (structured clinical interview for DSM-5; First, Williams, Karg, & Spitzer, 2015). Study exclusion criteria were current non-abstinent alcohol use disorder, 1 uncontrolled severe mental health disorder, current PTSD, 2 and suicide planning in the past month or a suicide attempt in the past 6 months. All patients provided their informed written consent.
Measures
The following clinical research measures were used for the analysis (see Figure 1 for timing during the study):
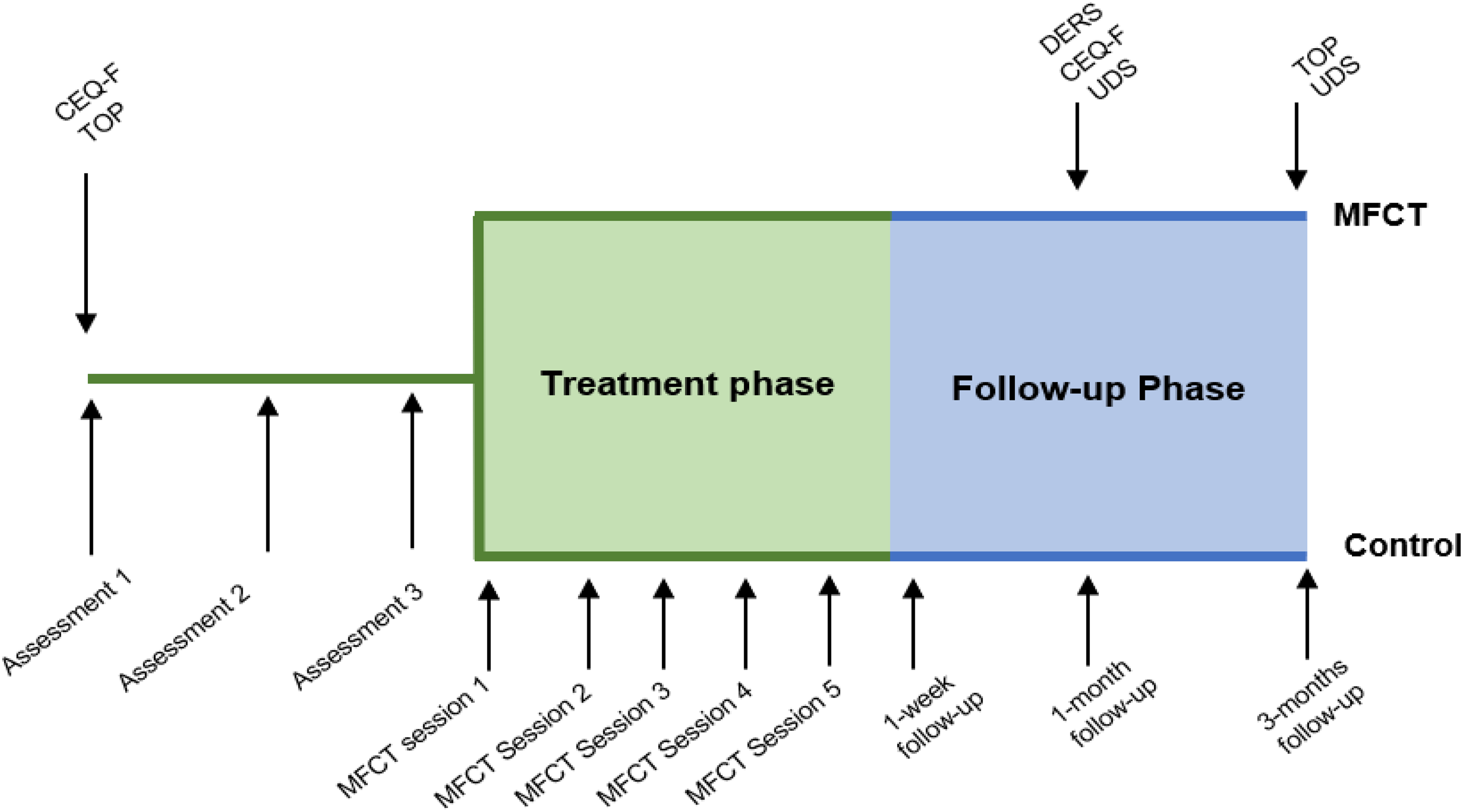
Timing of study measures. CEQ = craving experience questionnaire; DERS = Difficulties in Emotion Regulation Scale; MFCT = memory-focused cognitive therapy; UDS = urine drug screen; TOP = treatment outcome profile (time line follow-back interview for past 28 days).
Difficulties in Emotional Regulation Scale (DERS)
The DERS (Gratz & Roemer, 2004) is a 36-item self-report measure of current emotion regulation. It includes four components: awareness and understanding, acceptance, ability to control impulses in the presence of negative affect, and access to emotion regulation strategies. Items are rated using a 5-point scale (almost never to almost always, 0–5; total score, 36–180). Higher scores indicate more difficulty in emotion regulation. Participants completed the DERS
The Craving Experiences Questionnaire—frequency version (CEQ-F)
The CEQ-F (May et al., 2014) is an 11-item, self-report measure of the frequency of intensity, imagery, and intrusiveness aspects of craving in the past 2 weeks (adapted for the present study). Each item is rated using an 11-point scale (0–10; total score: 0–110). Participants completed the CEQ-F at baseline and at 1-month follow-up and the instrument was screened as a potential mediator for the analysis.
Treatment Outcomes Profile (TOP)
The TOP (Marsden et al., 2008) is a structured, clinician-administered interview for substance use disorder treatment outcome research. It includes a calendar prompt, time line follow-back method to record drug use during the prior 28 days. Participants completed the TOP at baseline and 3-month follow-up. The outcome measure for the analysis was the number (count) of days abstinent (NDA) from cocaine at the 3-month follow-up.
Urine Drug Screen (UDS; Alere Toxicology)
To indicate recent drug use at the 3-month follow-up (and to verify self-report using Cohen’s kappa statistic), we used an instant result immunoassay device to detect the primary cocaine metabolite (benzoylecgonine) in urine.
Statistical analysis
Some description is warranted on our conceptual approach for the causal mediation analysis. Rather than follow the traditional method of assessing mediation (Baron & Kenny, 1986), we used the counterfactual framework (Valeri & Vanderweele, 2013; VanderWeele, 2015).
In Baron and Kenny’s regression equation approach (see Figure 2, Panel 1), Path c′ represents the direct effect of the exposure on the outcome. Path a′ estimates the effect of the exposure on the mediator and Path b′ shows the effect of the mediator on the outcome. Taken together, Paths a′ and b′ express the indirect effect of the exposure on the outcome via the mediator. This approach has served researchers very well—but it cannot estimate exposure-mediator interactions or handle non-continuous outcome measures (Holland, 1986).

NDA = Baron and Kenny mediation model and counterfactual framework. A = exposure (intervention or control group by random allocation); M = mediator (CEQ-F and DERS at 1-month postintervention follow-up); Y = outcome (NDA at 3-month follow-up); RR = rate ratio (continuous mediator and Poisson regression given count-based outcome); CDE = controlled direct effect; NDE = natural direct effect; NIE = natural indirect effect; CEQ-F = craving experience questionnaire (frequency version); DERS = Difficulties in Emotion Regulation Scale; NDA = number of days abstinent.
VanderWeele’s counterfactual framework was developed to address these limitations (see Figure 2, Panel 2). Here, the estimation of effects is achieved by comparing observed and hypothetical outcomes for the intervention and control groups, on the assumption of “exchangeability” (i.e., that people assigned to the control group [A = 0] would respond in the same way as people assigned to the intervention group [A* = 1] had they been assigned to the intervention, and vice versa; Robins & Greenland, 1992).
A causal mediation model decomposes the total effect (TE) into a direct effect (i.e., the effect of treatment [A] on outcome [Y; A = 0 vs. A* = 1]) and the natural indirect effect (NIE). There are two types of direct effect—a controlled direct effect (CDE) and a natural direct effect (NDE). The CDE is computed by holding the mediator to a constant. The NDE is computed by holding the mediator to the unexposed (control) level, allowing for natural variation. The CDE and NDE are equivalent unless there is an exposure-mediator interaction. The NIE captures the effect of the mediation pathway (i.e., the average change in Y if the exposure is fixed to the level of the intervention and the mediator changes accordingly [i.e., A = 0 to A* = 1]; VanderWeele, 2015).
All analysis was done in Stata (version 15.0; StataCorp, 2017) in four steps. First, the data were screened using Little’s test (command: mcartest), with the intention to use multiple imputations (command: mice) to manage missing values provided that missingness was independent of the unobserved and observed data.
Second, we compared baseline and 1-month follow-up values between the groups on variables for the analysis, with a 95% CI for the comparison of measures at 1-month follow-up, because this was the level of precision set for the primary outcome in the original trial.
Third, univariate regression models were fitted to demonstrate significant associations between treatment and mediator; mediator and outcome; and treatment and outcome—each fitted univariably and multivariably to identify potential confounding. At baseline, the following participant measures were used as covariates: sex, age, months of regular cocaine use, months of general drug counseling, and CEQ-F score.
Finally, the causal mediation analysis was done using the command paramed (Dunn et al., 2015). Paramed supports the use of count-based outcome measurement (as used here) and gives standard errors (SE), CI, and parameter estimates for the TE, CDE, NDE, and NIE by the delta method. Given the small sample size and exploratory nature of the analysis, these parameters were estimated by bootstrapping with 80% CIs for hypothesis testing.
Results
Participant enrolment and missing data
Fifty-eight patients were screened for the study, of whom 35 were enrolled. As planned, 30 participants completed the assessment phase and were randomized to the control group (n = 14) and the intervention group (n = 16).
All participants completed the 1-month follow-up, except one member of the control group who declined to complete the DERS. All participants, bar one from each group, completed the 3-month follow-up. A nonsignificant Little’s test statistic supported use of multiple imputation for the missing DERS total score, χ2(16) = 14.50; p = .561, and the two missing values on the NDA outcome, χ2(36) = 11.38; p = 1.000.
Baseline characteristics and group differences on emotion regulation and craving
There was good group balance on baseline characteristics (Table 1). The overall CEQ-F sample mean at baseline was 62.7 (SD 20.2). The CEQ-F had good internal reliability (Cronbach’s α .83). The overall sample mean for the DERS at baseline was 103.3 (SD 26.29) with excellent internal reliability (α .93).
Participant characteristics at baseline.

Note: DSM = diagnostic and statistical manual; CUD = cocaine use disorder; SD = standard deviation; IQR = interquartile range. Data are number of participants, mean (SD), or median (IQR).
Table 2 presents the summary values on the variables for the causal mediation model at baseline and the two follow-ups by the group.
Variables included in the analysis at baseline, 1-month follow-up (potential mediators) and outcome at 3-month follow-up, by study group (n = 30).

Note: M = mean; SD = standard deviation; CI = confidence interval; NDA = number of days abstinent from cocaine past 28 days; CEQ-F = craving experience questionnaire (frequency version), recall period: past 2 weeks (total score, range: 0–110); DERS = Difficulties in Emotion Regulation Scale, recall period past 2 weeks (total score, range: 36–180); Hedge’s g.
aEffect size CI advanced specified at 95% CI.
bEffect size CI advanced specified at 80% CI.
At baseline, there were no group differences on the measures of cocaine use, CEQ-F, and DERS. Compared to the control group, participants in the intervention group had lower CEQ-F scores at 1-month follow-up (g = −1.62; 80% CI: −0.80 to −2.45). The intervention was also associated with greater NDA at the 3-month follow-up (g = .38; 80% CI: 0.30 to 1.28). There was complete concordance between self-report and UDS data (i.e., all participants who reported cocaine use in the past 7 days had a cocaine positive UDS, and all participants who reported no cocaine use in the past 7 days had a negative UDS; κ = 1.00).
For the DERS, there was no statistically significant group difference at 1-month follow-up (g = −0.45; 80% CI: −0.93 to 0.02).
Causal mediation analysis
Assumptions for mediation
Univariate regression models were first fitted to each model pathway. Separate linear regression models tested the direct effect of treatment on the mediator (Path a′) to meet assumptions for mediation. The intervention was associated with lower scores on the CEQ-F at 1-month follow-up, F(1, 28) = 20.99; adjusted R 2 = .408; p < .0001. However, the intervention was not associated with any meaningful change on the DERS, F(1, 28) = 1.63; adjusted R 2 = .021; p = .212.
To test the effect of the CEQ-F on 3-month NDA outcome, a Poisson regression model was fitted to the NDA outcome. Lower CEQ-F scores at 1-month were associated with greater cocaine abstinence, likelihood ratio (LR) X 2(1) = 80.67; p < .0001; pseudo R 2 = .252. The intervention was also associated with increased NDA at 3 months, LR X 2(1) = 21.16; p < .0001; pseudo R 2 = .066. Higher DERS score at 1 month was associated with less abstinence at 3 months, LR X 2(1) = 27.73; p < .0001; pseudo R 2 = .087. However, since the DERS scores at 1 month were not associated with the intervention, it did not meet the minimum requirement for mediation and this variable was dropped from further analysis.
Identification of covariates
Table 3 presents the results of univariable and covariate-adjusted regression models. The univariable models fitted indicated that months of regular cocaine use (p = .035), baseline CEQ-F (p = .030), and trial group (p < .0001) were all predictors of 1-month CEQ-F score. However, in the multivariable model, only trial group was associated with CEQ-F score (adjusted R 2 .479; p = .002).
Unadjusted and covariate-adjusted causal mediation analysis of treatment exposure on cocaine craving and cocaine use outcome at 3-month follow-up.

Note: CEQ-F = craving experience questionnaire (frequency version), recall period: past 2 weeks (total score, range: 0–110); IRR = incidence rate ratio; CI = confidence interval; SE = standard error.
Age (p = .001), months of regular cocaine use (p = .003), baseline CEQ-F (p < .0001), baseline cocaine use (p < .0001), and trial group (p < .0001) were all predictors of change on the outcome. Then, in an adjusted model (pseudo R 2.201), sex (p = .043), baseline CEQ-F (p = .006), baseline cocaine use (p = .001), and trial group (p < .0001) were all associated with outcome.
Based on the adjusted models for 1-month craving and 3-month cocaine abstinence, sex, baseline CEQ, and baseline cocaine use were included in the final model as potential confounders (Table 4). The model was run bootstrapped with 10,000 replications.
Causal mediation model, adjusted for covariates.

Note: CEQ-F = craving experience questionnaire (frequency version); CI = confidence interval; SE = standard error; NDE = natural direct effect, equivalent to CDE without mediator-outcome interaction; NIE = natural indirect effect; TE = total effect; CDE = controlled direct effect.
*Bootstrapped.
Mediation analysis
Overall, there was a significant association of the intervention on NDA outcome (TE 1.499; 80% CI 1.114 to 1.970; p = .012). With no evidence of an exposure–mediator interaction, the CDE and NDE are equivalent, and NDE is reported only herein.
There was no evidence of a significant direct effect (NDE = .855; 80% CI 0.569 to 1.175; p = .242) and a rate ratio coefficient close to 1 indicated little-to-no difference between participants receiving the intervention or control through the model’s direct causal pathway. This was underscored by the Poisson model (coefficient −0.156; 95% CI −0.419 to −0.106; p = .242). The mediating pathway was significant (NIE = 1.753; 80% CI 1.334 to 2.936; p < .0001), indicating that a reduction in craving at 1-month follow-up was associated with increased abstinence in the participants in the intervention group.
Discussion
Using data from a randomized controlled trial, we present an exploratory mediation analysis providing preliminary evidence for a mediating effect of the frequency of intensity, imagery, and intrusiveness aspects of cocaine craving (as measured by the CEQ-F) on cocaine abstinence among patients allocated to MFCT.
Both treatment allocation and craving were independently associated with cocaine use and after covariate adjustment, study data suggest a causal effect of MFCT on cocaine abstinence via reduced craving experience. The significant NIE from the causal mediation model provides evidence in support of our hypothesized therapeutic change mechanism. In spite of the small sample, this was a strong effect: once the effect of craving at 1 month was accounted for, the model showed no statistically significant direct effect of MFCT on outcome.
Based on this analysis, there was no support for the hypothesis that emotion regulation mediates cocaine abstinence after therapy. Reduced DERS scores were associated with the NDA outcome at 3-month follow-up, but this was an independent effect that was not linked to the intervention. However, it would be wrong to miss the opportunity to help patients in this area. Previous studies have suggested that individuals with CUD show stronger responses to emotional stimuli (Aguilar de Arcos, Verdejo-Garcia, Peralta-Ramirez, Sanchez-Barrera, & Perez-Garcia, 2005) and emotion regulation can improve as abstinence is sustained (Fox et al., 2007). It is possible that a lack of association between emotion regulation and treatment in our study may in part be because the DERS (and the CEQ-F) does not capture motivations to use drugs to change emotional state. During assessment and therapy sessions, participants collaboratively explored and addressed craving and cocaine use as a response to positive and negative mood. Substance use as a strategy for coping with negative affect (and associated craving) is likely to play an important role in use and relapse and may be a more relevant predictor than the general aspects of emotion regulation addressed in the DERS.
The present analysis supports assessing and targeting craving experience to inform psychological treatment of CUD (Marsden et al., 2014). While it is not currently feasible to directly target subthreshold, cue-induced responses, MFCT targets cognitive and affective elaborations with the aim of diminishing the strength of future craving experiences. By capitalizing on the malleability of memory reconsolidation processes, our novel psychotherapy appears to reduce drug approach cognitions and responses to cocaine conditional cues, so that subsequent craving experiences are better controlled (Kavanagh et al., 2005). Better cognitive control is likely to increase self-efficacy when exposed to high-risk situations (Sklar, Annis, & Turner, 1999) and may lead to reductions in activity in reward regions in the brain (Volkow et al., 2010).
Nevertheless, as we have noted, craving does not always precede cocaine use. Some people with CUD find that cocaine seeking has become highly automatized and if there is immediate drug access, there may be minimal, if any, craving, and cognitive elaboration. Our efforts to help patients become aware of automatic processes may provide some protection to enable a process of reflection and alternate action.
In the context of the wider literature, there is mixed evidence on the role of craving in predicting cocaine use and a number of studies have concluded that craving does not reliably predict cocaine use (Miller & Gold, 1994; Weiss, Griffin, & Hufford, 1995). However, more recent studies favor craving as a predictor of cocaine-seeking (Da Silveira, Doering-Silveira, Niel, & Jorge, 2006; Preston et al., 2009) and relapse (Crits-Christoph et al., 2007; Paliwal, Hyman, & Sinha, 2008; Rohsenow, Martin, Eaton, & Monti, 2007; Sinha, Garcia, Paliwal, Kreek, & Rounsaville, 2006; Weiss et al., 2003). The present study contributes to and extends this mixed extant literature showing craving as measured by the CEQ-F as a strong predictor of cocaine use.
It is important to note that memory reconsolidation interventions are at a relatively early stage of development (Exton-McGuinness & Milton, 2018; Monfils & Holmes, 2018). However, the literature is growing with encouraging findings from psychological laboratory studies with users of nicotine, heroin, alcohol, and cocaine (Germeroth et al., 2017; Hon et al., 2016; Xue et al., 2012) and pharmacological interventions (e.g., propranolol; Lonergan et al., 2016; Saladin et al., 2013; Xue et al., 2017). Taken together, these interventions suggest that memory-reconsolidation techniques can reduce craving and subsequent drug-seeking.To our knowledge, our study is the first to apply memory reconsolidation approaches in the addiction clinic setting. Replication studies by other groups are now warranted.
Study strengths include the experimental design, a registered analysis plan, statistical control over confounding, and a counterfactual framework analysis of causal mediation. We also acknowledge several limitations. Although in line with recommendations for pilot studies, this was a small sample study and the results must be regarded as exploratory and in need of replication. A longer follow-up period up to 12 months is recommended to examine the robustness of MFCT-based cocaine abstinence against time, given the relapsing nature of CUD. Nevertheless, the present findings are encouraging and give impetus for future superiority randomized controlled trials of MFCT alongside general counseling for people with primary CUD and as an adjunctive intervention for patients with concurrent CUD and opiate use disorder (Marsden et al., 2019).
At present, CUD is a significant and hard-to-treat public health problem with limited treatment options and it is crucial to understand the mechanisms underlying potential new treatments. This study positions craving as an important mediator in reducing cocaine use in participants receiving MFCT.
Footnotes
Acknowledgments
The authors would like to thank the participants and we kindly acknowledge the support from our colleagues at Lorraine Hewitt House (South London and Maudsley National Health Service Foundation Trust) and Elka Giemza and her team at the Clinical Research Facility. We would also like to thank Professor Richard Emsley (Institute of Psychiatry, Psychology and Neuroscience, King’s College London) for advice on causal mediation analysis and using paramed. We dedicate this article to Professor Graham Dunn.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Camille Goetz is supported by a PhD Studentship Award from the National Institute for Health Research (NIHR) Biomedical Research Centre (BRC) at South London and Maudsley National Health Service Foundation Trust (SLaM) and King’s College London (KCL). John Marsden is a Clinical Research and Practitioner Psychologist (licensed with the British Association of Behavioural and Cognitive Psychotherapies). In the past 3 years, he declares grant support from Indivior (via Action on Addiction) for a study of adjunctive psychosocial intervention during opioid agonist treatment (ARC trial: 2015–2019); NIHR (Health Technology Assessments) for a trial of extended-release naltrexone (NEAT trial: 2016–2018); and Indivior for a trial of extended-release buprenorphine maintenance (EXPO trial; 2019–2021). John Marsden acknowledges part-time employment as a Senior Academic Advisor for the Alcohol, Drugs, and Tobacco Division, Health Improvement, Public Health England, and is a Consultant for the Centre for Clinical Trials Network at the U.S. National Institute on Drug Abuse. In the past 3 years, he has received Honoraria Martindale (2017; expert meeting on opioid use disorder); and Indivior (via PCM Scientific) for chairing (2016–2017) for the conference on Improving Outcomes in Treatment of Opioid Dependence. He holds no stocks in any company. Luke Mitcheson declares grant funding for an investigator-led, educational grant from Indivior (administered by Action on Addiction) for the ARC trial. He holds no stocks in any company. John Strang is a researcher and clinician who has worked with a range of types of treatment and rehabilitation service providers. He is supported by the NIHR BRC for Mental Health at SLaM and KCL. He has also worked with a range of governmental and nongovernmental organizations and with pharmaceutical companies to seek to identify new or improved treatments from whom he and his employer (KCL) have received honoraria, travel costs, and/or consultancy payments. This includes work with, during the past 3 years, Martindale, Reckitt-Benckiser/Indivior, MundiPharma, and Braeburn/Camrus (none of these activities relate to the study being reported here). Nick Grey is partly supported by a research grant from the Wellcome Foundation (grant held by Professors Anke Ehlers and David Clark at the University of Oxford).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This article represents independent research funded by the National Institute for Health Research Biomedical Research Centre at South London and Maudsley National Health Service Foundation Trust and King’s College London. The views expressed are those of the authors and not necessarily those of the NIHR or the Department of Health and Social Care.
