Abstract
Although exposure therapy is effective in reducing eating-related fears and avoidance in individuals with eating disorders (EDs), there has been little study of this treatment in inpatient settings. To address this gap, we conducted a case series to evaluate the effects of a food-based exposure therapy intervention on eating-related fears and avoidant behaviors among adults being treated for an ED in an inpatient hospital setting. Patients (N = 106) were provided psychoeducation on the treatment approach, aided in the development of an exposure hierarchy containing fear-evoking stimuli related to eating (e.g., feared foods), and guided in completing exposure exercises and gradually progressing through the hierarchy. Results showed that patients experienced significant reductions in eating-related fears and avoidant behaviors from pretreatment to posttreatment. Acceptability of the intervention was reasonably well-established by (a) acceptable rates of treatment participation and completion, (b) favorable ratings of the intervention provided in a posttreatment satisfaction survey, and (c) the absence of any exacerbations in the frequency of compensatory weight control behaviors (e.g., self-induced vomiting) over the course of treatment. Clinical implications of these findings are discussed in the light of clinicians’ concerns about and underutilization of food-based exposure therapy for EDs, particularly in inpatient settings.
Eating disorders (EDs) are very serious mental illnesses that are associated with an extensive array of concerns for afflicted individuals. Of greatest concern is heightened mortality risk, as EDs are the most fatal type of all mental disorders (Arcelus, Mitchell, Wales, & Nielsen, 2011). Importantly, the risk for fatality in individuals with EDs increases as a function of prolonged duration of the disorder (e.g., Franko et al., 2013). Additionally, malnutrition and unsafe behaviors that often accompany EDs are associated with a host of severe medical consequences, such as cardiac abnormalities (e.g., arrhythmias), pancreatitis, and osteoporosis; some of these problems can become chronic and irreversible (Westmoreland, Krantz, & Mehler, 2016). Due to the severe nature of these issues and the commonness of delayed treatment seeking among individuals with EDs (Becker, Arrindell, Perloe, Fay, & Striegel-Moore, 2010; Hepworth & Paxton, 2007), inpatient hospitalization is often necessary.
Compared to other treatment settings, inpatient treatment of EDs is often relatively brief and primarily emphasizes nutritional stabilization and subsequent amelioration of acute medical complications. However, there is considerably less emphasis on addressing core underlying cognitive–behavioral features of EDs, such as eating-related fears and avoidance behaviors (Steinglass et al., 2011). It is thus not surprising that following hospitalization, many patients continue to experience these key ED features, resulting in alarmingly high rates of relapse and need for rehospitalization (Pike, 1998). To illustrate, one study showed that among patients who completed inpatient treatment for EDs, over half had experienced a significant return of ED symptoms at a 1-year follow-up (Walsh et al., 2006). Furthermore, in spite of the high intensity of inpatient treatment programs compared to the standard outpatient treatment, there is some longitudinal evidence that inpatient treatment yields inferior responses (Meads, Gold, & Burls, 2001).
In the light of recent increased utilization of inpatient programs for EDs (Zhao & Encinosa, 2011), as well as limited evidence supporting their long-term efficacy, it is clear that improvements to inpatient treatment are needed. Specifically, it is important to improve treatment in this setting in a manner that addresses key cognitive–behavioral maintenance factors to ensure better long-term outcomes. To illustrate, a core tenet of cognitive behavioral therapy for EDs is that beliefs about the extreme importance of one’s weight and shape contribute to many of the behavioral tendencies commonly seen in EDs. It follows that these behavioral tendencies reinforce the maladaptive beliefs, thereby creating a self-sustaining cycle that maintains the ED (e.g., Fairburn, Cooper, & Shafran, 2003).
Among the most central features involved in the maintenance of EDs are eating-related fears (e.g., exorbitant weight gain) and subsequent avoidant behaviors (Steinglass et al., 2011; Waller, 2008; Waller & Mountford, 2015). Based on the established efficacy of exposure therapy in treating disorders characterized by fear and avoidance, Steinglass et al. (2012) developed an experimental psychotherapy intervention involving gradual exposure to feared/avoided eating-related stimuli coupled with the prevention of avoidant behaviors aimed at neutralizing anticipated consequences (e.g., exercising to thwart weight gain). Although preliminary data suggested this intervention was helpful in reducing eating-related fear and avoidance among inpatients with anorexia nervosa, the intervention contained 12 sessions that were all delivered after patients had completed a period of weight restoration (Steinglass et al., 2014), which represents a substantially longer course of treatment than is typical in the inpatient setting. Thus, there is a need to study briefer exposure-based interventions that can be feasibly implemented in typical ED inpatient settings.
Levinson et al. (2015) developed a similar exposure intervention that was condensed into four therapy sessions delivered over 2 weeks. This intervention was successful in increasing caloric intake as well as decreasing anxiety about eating, food avoidance behaviors, and concerns with feared consequences of eating among a sample of patients receiving treatment in an ED partial hospitalization program (Levinson & Byrne, 2015). It should be noted that these findings were derived from controlled research that allowed for internally valid comparisons between conditions among patients receiving a lower “dosage” of the treatment (i.e., partial hospitalization); however, it is unclear whether a food-related exposure intervention would be effective when implemented naturalistically in an ED inpatient setting.
One potential reason for the lack of examination of food-related exposure therapy interventions in ED inpatient settings is the well-documented concerns among many clinicians that the treatment would backfire, resulting in a worsening of patients’ symptoms (e.g., increased use of compensatory weight control behaviors; Lowe, Bunnell, Neeren, Chernyak, & Greberman, 2011; Turner, Tatham, Lant, Mountford, & Waller, 2014; Waller, 2016; Waller, Stringer, & Meyer, 2012). This concern may be particularly pronounced in inpatient settings, where often the primary objective of treatment is to quickly restore nutritional stability. It follows that food-based exposure therapy is eschewed in ED inpatient settings in favor of a more tranquil approach to facilitating patients’ feeding that actually runs counter to core principles of exposure-based therapy (Becker & Waller, 2017). For example, inpatient treatments often encourage avoidance (rather than approach) of feared stimuli during eating (e.g., by playing games during eating, engaging in distracting conversations, or listening to peaceful music). Given the aforementioned work showing poor long-term outcomes associated with treatment as usual in ED inpatient settings, it is important to empirically investigate whether food-related exposure appears to be well-accepted and tolerated by patients in this setting.
The objective of the present study was to examine the effectiveness and acceptability of a brief food exposure intervention that was adapted for use in an inpatient program for adults with EDs. Following the experimental work of Steinglass et al. (2014) and Levinson et al. (2015), we implemented a food-based exposure therapy approach that primarily involved gradual incorporation of feared/avoided foods into patients’ diets paired with prevention of behavioral attempts to influence weight and/or body shape. Patients were provided psychoeducation on the intervention and were encouraged (but not required) to participate and maintain their engagement throughout treatment. Given their centrality to ED pathology, eating-related fears and avoidant behaviors comprised our primary outcome variables and were assessed at both pretreatment and posttreatment.
Based on previous work demonstrating the benefits of brief food-related exposure for individuals with EDs (e.g., Levinson et al., 2015), we hypothesized that our adapted version of the treatment would demonstrate effectiveness in decreasing patients’ eating-related fears and avoidant behaviors. We also hypothesized that food-based exposure would be well-accepted and tolerated by patients, evidenced by (a) a high rate of participation in treatment among eligible patients, (b) a low rate of discontinuation among patients who initiated the treatment, (c) no significant increases in compensatory weight control behaviors from pretreatment to posttreatment, and (d) favorable treatment satisfaction ratings given at posttreatment.
Method
Participants
Participants (N = 106) included consecutively admitted adult patients who consented to participate in food-based exposure therapy as part of their treatment in an ED inpatient hospital in the Midwestern region of the U.S. between the dates of June 2016 and February 2018. Study inclusion criteria included a principal diagnosis of an ED and a minimum 14-day length of hospital stay. The mean age of the sample was 26.7 years (SD = 10.8, range = 18–64 years), and the mean length of stay (LOS) was 23.8 days (SD = 12.6, range = 14–51 days). Mean body mass index (BMI) for the sample was 18.3 (SD = 4.9, range = 15.3–33.2). The sample was predominantly female (n = 89, 83.0%), and a majority of participants identified their ethnicity as White/Caucasian (n = 95, 89.6%). Principal ED diagnoses according to criteria from the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5; American Psychiatric Association, 2013) were determined by program psychiatrists via semi-structured interview 1 combined with reviewing patient responses on the Eating Disorder Examination Questionnaire (EDE-Q) at admission (see below) and included the following anorexia nervosa, restricting type (n = 43, 40.6%), bulimia nervosa (n = 24, 22.6%), anorexia nervosa, binge-eating/purging type (n = 12, 11.3%), binge Eating Disorder (n = 8, 7.5%), avoidant/restrictive food intake disorder (n = 7, 6.6%), and other specified or unspecified Eating Disorder (n = 12, 11.3%). Table 1 provides a breakdown of demographic variables and baseline symptom severity by diagnosis. Additionally, a majority of the sample was diagnosed with a comorbid mood disorder (n = 58, 54.7%). Patients diagnosed with comorbid anxiety disorders (including obsessive-compulsive disorder and posttraumatic stress disorder) were as follows: at least one anxiety disorder (n = 73, 68.9%), at least two anxiety disorders (n = 40, 37.7%), and three or more anxiety disorders (n = 11, 10.4%). Most patients (n = 88, 83.0%) were prescribed psychotropic medication that was managed by a program psychiatrist.
Demographic variables and pretreatment symptom severity by diagnosis.

Note. AN-R = anorexia nervosa, restricting type; BN = bulimia nervosa; AN-B/P = anorexia nervosa, binge-eating/purging type; BED = binge eating disorder; ARFID = avoidant/restrictive food intake disorder; OSFED = other specified/unspecified feeding or eating disorder; EDE-Q = Eating Disorder Examination Questionnaire; BMI = body mass index; SD = standard deviation.
Measures
Fear of Food Measure
The Fear of Food Measure (FOFM; Levinson & Byrne, 2015) is a 23-item self-report instrument that assesses eating-related fear and avoidance. Each item is a statement describing either an affective (e.g., “I feel anxious when I eat”), cognitive (e.g., “I worry that eating will make me dissatisfied with my body”), or behavioral (e.g., “There are certain foods I avoid because they make me anxious”) feature characteristic of EDs. Participants use a 1 = not at all to 7 = very much so scale to indicate how characteristic each statement is of them. In a previous research with both clinical and nonclinical samples, the FOFM has shown a clear three-factor structure with the following scales demonstrating good internal consistency and reliability: the 8-item Anxiety about Eating (AE) scale, the 6-item Food Avoidance Behaviors (FAB) scale, and the 9-item Feared Concerns (FC) scale (Levinson & Byrne, 2015). These scales have also demonstrated appropriate sensitivity to detect therapeutic change in the context of exposure therapy (Levinson & Byrne, 2015). Consistent with Levinson and Byrne’s (2015) approach, we examined each of the three FOFM scales separately as outcome variables. In the present study, each scale demonstrated good internal consistency at both pretreatment and posttreatment (AE pretreatment α = .86, posttreatment α = .90; FAB pretreatment α = .82, posttreatment α = .83; FC pretreatment α = .83, FC posttreatment α = .80).
EDE-Q, version 6.0
The EDE-Q (Fairburn & Beglin, 2008) is a well-established measure of ED-related pathological features. It is a 28-item questionnaire that assesses 22 attitudinal features of EDs (e.g., weight concerns) and 6 ED-related behaviors (e.g., self-induced vomiting). Participants use a scale ranging from 0 = no days to 6 = every day to indicate the frequency of experiencing each item (e.g., “Have you had a definite fear that you might gain weight?”) during the past 4 weeks. The EDE-Q has previously demonstrated excellent psychometric properties in both clinical and nonclinical samples (Mond, Hay, Rodgers, Owen, & Beumont, 2004). In the present study, the EDE-Q was used to characterize baseline ED severity and assess changes in the frequency of engagement in maladaptive weight control strategies (e.g., self-induced vomiting); it demonstrated good internal consistency (α = .90).
Patient satisfaction survey
As part of the Inpatient Psychiatric Facility Prospective Payment System (IPFPPS) by the Centers for Medicare and Medicaid Services, all patients were asked to complete a satisfaction survey upon their discharge from the program. Whereas the IPFPPS survey is comprehensive and encompasses a variety of aspects related to patient satisfaction (e.g., comfort of the facility), we were particularly interested in patients’ responses to 4 distinct items assessing (a) perceived helpfulness of the individualized treatment plan, (b) degree to which treatment was sensitive to emotional needs, (c) overall rating of the treatment provided, and (d) likelihood of recommending the treatment program to others. Patients used a Likert-type scale ranging from 1 = very poor to 5 = very good to respond to each of these 4 items, which we assessed separately in this study.
Procedure
Within 48 hr of their admission to the inpatient program, patients provided informed consent for research and completed the study measures. Food-based exposure therapy was co-facilitated by a registered dietitian and behavioral therapist, who began by providing psychoeducation on the role of eating-related fears and avoidant behaviors in maintaining ED pathology and describing the rationale for food exposure. These clinicians then worked in collaboration with the patient to develop a fear hierarchy of various eating-related feared stimuli, such as consuming normalized portions of food, eating food at reasonable pace, and gradually including feared food items into the patient’s dietary plan.
Patients met with their clinicians daily to (a) review exposure activities completed the previous day, (b) coordinate repetition with food-related exposures as needed, and (c) facilitate a gradual progression upward in the fear hierarchy to introduce higher fear-evoking exposure activities. Food exposure continued throughout the patient’s entire LOS, which varied from one patient to the next as a function of their nutritional stability and overall readiness for discharge at the discretion of the program psychiatrists. It was explained to patients that they were free to withdraw from food-related exposure therapy at any time during their treatment and instead complete sufficient caloric intake via alternative means (e.g., nasogastric tube feeding, consumption of non-fear-evoking foods, etc.). Ancillary components of the program included nondirective, supportive psychotherapy in a group format, recreational activities (e.g., art making), nutritional education, and family therapy sessions to provide education and periodic updates to patients’ family members. Patients again completed the study measures within 48 hr of their discharge from the program.
Results
Treatment participation
Of the total 142 patients who met study criteria and were provided the opportunity to participate in food-based exposure therapy, 106 patients elected to initiate the treatment (74.6%). Seven of these patients who initiated the treatment chose to discontinue food exposure prior to discharging from the hospital (6.6%). The other 99 patients remained engaged in the treatment throughout the duration of their hospital stay. There were no demographic or LOS differences between patients who remained engaged in food exposure throughout their treatment and patients who either declined participation or chose to discontinue the treatment (all ps > .05).
Eating fears and avoidance
Means and standard deviations for each of the three FOFM scales at both pretreatment and posttreatment are included in Table 2. Consistent with our hypothesis, dependent-samples t-tests showed that patients experienced significant reductions across each FOFM scale from pretreatment to posttreatment. Whereas the effect sizes of these reductions were large for both the AE and FAB scales, the effect size for the reduction in the FC scale score was moderate.
Mean and SDs for FOFM Scales at pretreatment and posttreatment.

Note. FOFM = Fear of Food Measure; AE = Anxiety about Eating; FAB = Food Avoidance Behavior; FC = Feared Concern; SD = standard deviation.
We conducted exploratory follow-up analyses to assess the potential influence of global ED symptom severity, BMI, and LOS on exposure-based treatment outcome. For each of the three FOFM scales, we conducted a repeated-measures analysis of covariance with EDE-Q global score, pretreatment BMI, and LOS entered as covariates. The pattern of results was very similar across these three analyses; there were statistically significant reductions from pretreatment to posttreatment across FOFM scales that were unrelated to each of the covariates. Thus, the extent of reduction in eating-related fear and avoidance throughout treatment did not appear to vary as a function of baseline ED symptom severity, BMI, or LOS.
Treatment acceptability
As a means to assess the acceptability and toleration of food-based exposure in the inpatient setting, we examined changes in the frequency of patients’ engagement in three separate compensatory behaviors measured by the EDE-Q: self-induced vomiting, compulsive exercising, and misuse of laxatives. Means and standard deviations for the frequency of engagement in these behaviors at both pretreatment and posttreatment are presented in Table 3. As hypothesized, there were no significant increases in these behaviors from pretreatment to posttreatment. On the contrary, there were significant reductions in the frequency of both self-induced vomiting and compulsive exercising, and the effect sizes for these differences were moderate to large.
Frequency of engagement in compensatory weight control behaviors at pretreatment and posttreatment.
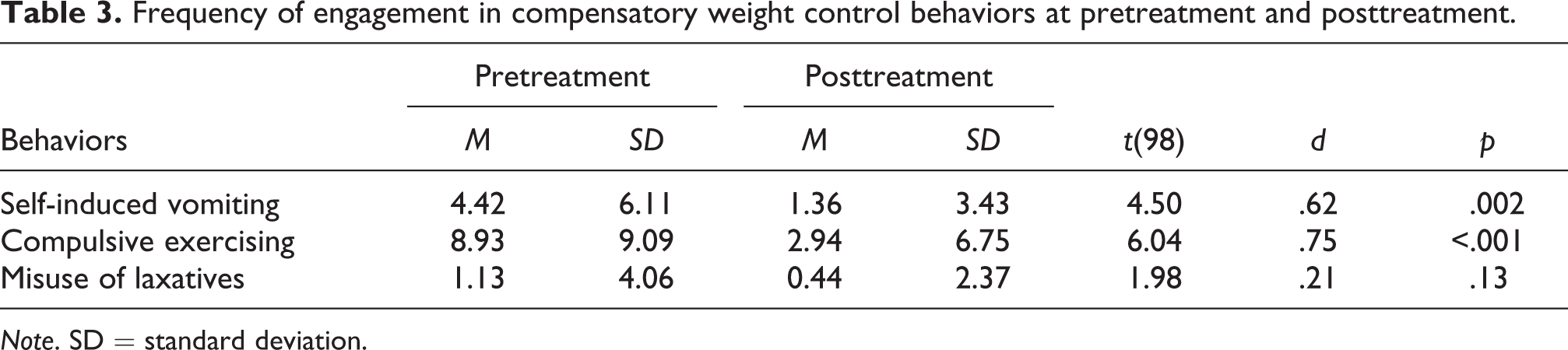
Note. SD = standard deviation.
We also gauged the tolerability of food exposure by examining patients’ responses to 4 separate satisfaction survey items using a 1 = very poor to 5 = very good scale. Average responses indicated favorable ratings and were as follows: (a) perceived helpfulness of the individualized treatment plan (M = 4.21, SD = 0.86), (b) degree to which treatment was sensitive to emotional needs (M = 4.09, SD = 1.12), (c) overall rating of the treatment provided (M = 4.28, SD = 0.90), and (d) likelihood of recommending the treatment program to others (M = 4.11, SD = 0.79).
Discussion
Although inpatient hospitalization is often necessary to address acute medical problems related to EDs, previous work has suggested that long-term maintenance of ED symptom reduction achieved in the inpatient setting may be attenuated by a lack of treatment emphasis on the core cognitive–behavioral features of EDs (e.g., eating-related fears and avoidant behaviors). In the present study, we assessed the initial effectiveness and acceptability of a food-based exposure therapy intervention targeting eating-related fears and avoidance in a case series of adults with EDs who were treated in an inpatient hospital program. As hypothesized, patients on average experienced significant reductions in eating-related fears and avoidant behaviors from pretreatment to posttreatment. Additionally, the treatment appeared to be well-tolerated and accepted by patients, evidenced by a relatively low discontinuation rate, a lack of significant increases in compensatory weight control behaviors, and favorable responses to patient satisfaction survey questions about the treatment experience.
The present findings demonstrating notable reductions in eating-related fear and avoidance in our inpatient sample have several important clinical implications. First, although the primary objective of inpatient hospitalization for EDs is often to establish patients’ nutritional stability and address related medical sequelae, our findings indicate that this objective can be accomplished in concert with addressing several key cognitive–behavioral features known to play a crucial role in the maintenance of EDs. Further, the extent of reduction in eating-related fear and avoidance from pretreatment to posttreatment was not significantly associated with baseline global ED symptom severity, BMI, or LOS. Thus, our findings indicate that food-related exposure may be an efficacious intervention in inpatient settings regardless of symptom severity or body size/weight at the outset of treatment. Finally, it is worth noting that the effect sizes for pretreatment to posttreatment reductions in eating-related fear and avoidance were noticeably larger than the effect sizes reported by Levinson and Byrne (2015). One plausible explanation for this observation is that patients in Levinson and Byrne’s (2015) study received only four sessions of food-based exposure, whereas our inpatient sample was continuously engaged in food exposure activities throughout the duration of their hospital stay. Taken together, the pattern of findings from these studies suggests the presence of a “dose–response” relationship with regard to food exposure and eating-related fear and avoidance; a higher volume of the treatment appears to be associated with more robust symptom relief. Future research is needed to test this possibility. It should also be noted that although patients in the present study experienced greater reduction in eating-related fear and avoidance than those patients in the Levinson and Byrne’s (2015) study, the mean posttreatment FOFM scores in both studies remained considerably higher than mean FOFM scores among normal healthy control individuals (Levinson & Byrne, 2015). Thus, patients with EDs may require a higher volume of food-related exposure in order to facilitate a reduction of eating-related fear and avoidance to a subclinical level.
Our prediction that food-based exposure would be generally well-accepted and tolerated by patients was supported by several outcomes. With regard to treatment participation, nearly three of every four patients who met criteria to undergo the treatment opted to do so. Moreover, among patients who initiated treatment, the vast majority proceeded with the intervention until the time of their discharge from the hospital. Concerning patients’ actual experience with food-based exposure therapy, patient responses to several satisfaction survey questions indicated generally favorable impressions of the treatment, and there was no evidence of any exacerbation of the compensatory weight control behaviors we assessed. In fact, two of these three behaviors (self-induced vomiting and compulsive exercising) showed significant decreases over the course of treatment. These findings are important in the light of recent work showing that many clinicians harbor concerns about employing exposure-based treatments out of fear that they will contribute to exacerbation of maladaptive, high-risk weight control strategies (e.g., self-induced vomiting; Turner et al., 2014). Clinicians who are fearful of utilizing food-related exposure therapy will hopefully take confidence from our findings that the treatment was apparently well-accepted and tolerated among an inpatient sample, which is often a treatment setting characterized by a high degree of ED symptom severity, as well as medical comorbidities. Relatedly, our findings may make an important contribution to ongoing efforts to develop educational interventions aimed at decreasing clinician concerns about utilizing exposure-based therapy for EDs (Waller, D’Souza Walsh, & Wright, 2016).
The present study has several limitations. First, the lack of a control condition precluded our ability to compare the effects of food-based exposure therapy to a credible control or waitlist group. Thus, we cannot rule out the possibility that the therapeutic change observed in this sample was not accounted for merely by patients being hospitalized and encouraged/guided in normalization of eating habits. Future research should aim to replicate the naturalistic treatment elements of this study and extend the findings via inclusion of a credible control group. Relatedly, given that food exposure was delivered in an inpatient program that included other ancillary therapeutic activities (e.g., family therapy sessions, nutritional education groups, etc.), it is unclear to what extent the observed reductions in eating-related fear and avoidance are attributable to food exposure. Future research may address this limitation by assessing the effectiveness of food exposure delivered as a stand-alone intervention in a naturalistic treatment setting. Furthermore, although food exposure was implemented in a hierarchical manner in the present study, recent suggestions to apply inhibitory learning theory principles to exposure therapy for EDs (Reilly, Anderson, Gorrell, Schaumberg, & Anderson, 2017) warrant, among other research, the future study of food exposure in which feared foods are introduced in a variable versus hierarchical manner.
Despite the present study showing promising effects of food exposure therapy in a relatively brief duration of time, the lack of a follow-up assessment precludes conclusions about the durability of the treatment benefit. Similar research studies to ours should consider assessing patients’ maintenance of treatment gains in the period of time that follows discharge from an inpatient ED hospital setting and whether these types of interventions decrease the high rates of relapse often present in this population. The generalizability of the present findings is also unclear due to the possibility of a self-selection bias inherent in our patient sample. It may be that the degree of benefit observed in our study would overestimate the benefit for a patient undergoing food exposure therapy who did not self-select this treatment option. Finally, the limited number of treatment outcome variables in the present study may diminish the confidence that food-based exposure therapy is in fact a key intervention that can facilitate long-term symptom relief when delivered in an inpatient setting. It may be that the “reach” of food exposure does not extend beyond eating-related fear and avoidance to other critical variables known to be influential in the long-term prognosis of EDs (e.g., body image disturbance; Keel, Dorer, Franko, Jackson, & Herzog, 2005).
Strengths of the present study include a large, mixed ED diagnosis sample, the high degree of external validity related to an ecologically valid treatment environment and delivery format, and our multimodal efforts to assess patients’ acceptance and toleration of food-based exposure therapy in addition to its effectiveness. Also, our efforts to translate an experimental psychotherapy intervention that had previously only been studied in more controlled treatment settings to a highly naturalistic treatment setting were a unique strength of the present work. In closing, we hope that our work will encourage others to engage in similar translational research that attempts to apply therapeutic interventions derived from efficacy research to “real-world” clinical practice with the overarching aim of improving long-term treatment outcomes for individuals with EDs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
