Abstract
Background:
α1-Adrenergic blockers are widely used in older patients, yet nervous system adverse events associated with these agents are often considered nonspecific and remain insufficiently characterized at the population level.
Objectives:
To evaluate nervous system adverse events associated with α1-adrenergic blockers using real-world pharmacovigilance data, assess drug-specific and patient-specific risks, and translate findings into clinically interpretable risk profiles.
Design:
A retrospective, observational pharmacovigilance study
Methods:
We conducted an integrated pharmacovigilance analysis using the U.S. Food and Drug Administration Adverse Event Reporting System (FAERS) as the discovery dataset and the Japanese Adverse Drug Event Report (JADER) database for external validation. Hierarchical signal detection within the Medical Dictionary for Regulatory Activities (MedDRA) nervous system organ class was used to construct a data-driven composite endpoint. Multivariable logistic regression with least absolute shrinkage and selection operator (LASSO)-guided feature selection estimated adjusted associations for five α1-adrenergic blockers while accounting for age, sex, and major comorbidities. Model performance and calibration were evaluated in JADER. Individualized risk portraits were generated across clinically relevant patient profiles.
Results:
Several α1-adrenergic blockers demonstrated significant and heterogeneous associations with nervous system adverse events in FAERS after adjustment. External validation in JADER showed consistent effect directions for drugs with sufficient exposure and acceptable calibration. Risk portrait analyses revealed marked risk stratification by age and comorbidity burden, with the highest predicted reporting probability observed among elderly patients with multiple comorbidities.
Conclusion:
This integrated pharmacovigilance framework provides a structured and clinically interpretable assessment of nervous system safety among α1-adrenergic blockers, highlighting the importance of agent-specific and patient-specific risk evaluation.
Plain language summary
1. A hierarchical pharmacovigilance framework integrating FAERS discovery and JADER external validation identified robust associations between α1-adrenergic blockers and nervous system adverse events.
2. Data-driven signal detection across MedDRA hierarchy levels defined a composite nervous system endpoint based on eight clinically salient preferred terms with consistent hierarchical support.
3. Multivariable analyses demonstrated heterogeneous neurological risk profiles among individual α1-blockers, with drug-specific associations persisting after adjustment for demographics and comorbidities.
4. Individualized risk portraits revealed graded absolute risk stratification across comorbidity subgroups, highlighting the joint contribution of agent-specific effects and cumulative comorbidity burden.
Keywords
Introduction
α1-Adrenergic blockers are widely prescribed for the management of lower urinary tract symptoms associated with benign prostatic hyperplasia.1,2 Historically, earlier generations of these agents, such as terazosin and doxazosin, were developed and remain indicated as antihypertensive therapies. These agents are commonly used in older adults and often administered over long durations.1,3 Their safety profiles have therefore been extensively discussed, particularly with respect to cardiovascular adverse effects such as orthostatic hypotension and syncope. 4 In contrast, adverse events involving the nervous system, including dizziness, headache, and other neurological complaints, are frequently regarded as nonspecific or secondary and have received less systematic evaluation.5,6
Current evidence on nervous system adverse events associated with α1-adrenergic blockers is largely derived from clinical trials, post-marketing case reports, or studies focusing on individual symptoms.7,8 While informative, these approaches are limited in their ability to capture the full spectrum of neurological adverse events and to distinguish drug-related effects from patient-related vulnerability. In addition, α1-adrenergic blockers are often considered as a homogeneous drug class in both clinical practice and safety assessments, despite known differences in receptor selectivity and pharmacokinetic properties among individual agents.9–11
Spontaneous reporting systems (SRS), such as the U.S. Food and Drug Administration Adverse Event Reporting System (FAERS) and the Japanese Adverse Drug Event Report (JADER) database, provide large-scale real-world data suitable for evaluating drug safety beyond the constraints of clinical trials.12,13 However, conventional SRS-based analyses often rely on isolated preferred terms or unadjusted disproportionality measures, which may be particularly problematic for nervous system adverse events that are inherently heterogeneous and hierarchically structured within the Medical Dictionary For Regulatory Activities (MedDRA).14–17 Consequently, clinically meaningful patterns may be obscured, and downstream interpretability remains limited.
To address these challenges, we conducted an integrated pharmacovigilance analysis of α1-adrenergic blocker-associated nervous system adverse events by combining FAERS and JADER data. Our study aimed to construct a data-driven neurological adverse event endpoint, quantify drug-specific associations while accounting for patient characteristics, evaluate the robustness of findings across regulatory systems, and explore structured approaches for translating model outputs into clinically interpretable risk patterns.
Methods
Data sources
This study used two large spontaneous reporting system databases: FAERS as the primary discovery dataset and JADER as an independent external validation dataset. FAERS reports submitted from January 2004 to December 2024 were included, and JADER reports covering the corresponding available period were used for validation. Both databases contain de-identified case-level information, including patient demographics, adverse events coded using MedDRA, drug exposure details, and reporter characteristics. Ethical approval and informed consent were not required because all data were publicly available and anonymized.
Study design overview
We implemented an end-to-end pharmacovigilance framework integrating hierarchical signal detection, multivariable modeling, external validation, and individualized risk visualization. FAERS served as the primary dataset for signal identification, endpoint construction, and model development, whereas JADER was used to evaluate robustness and transportability. The overall workflow is shown in Figure 1. The reporting of this study conforms to the strengthening the reporting of observational studies in epidemiology (STROBE) statement 18 and the READUS-PV. 19

Overall study design and analytical workflow. Schematic overview of the sequential pharmacovigilance workflow used to evaluate nervous system adverse events associated with α1-adrenergic blockers. FAERS was used as the primary discovery dataset to perform hierarchical signal detection within the MedDRA Nervous system disorders SOC, enabling data-driven construction of a composite nervous system endpoint. Multivariable logistic regression was subsequently applied to quantify independent associations while adjusting for demographic characteristics and major comorbidities. The finalized model structure was externally validated in the JADER database to assess effect direction and stability. Finally, FAERS-derived model estimates were translated into individualized risk portraits across clinically relevant combinations of drug exposure and patient characteristics.
Drug exposure definition
Five α1-adrenergic blockers were evaluated: tamsulosin, doxazosin, terazosin, alfuzosin, and silodosin. Drug names were standardized and harmonized across databases using curated dictionaries. In FAERS, exposure was defined when a drug was listed as a primary suspect or secondary suspect. Concomitant and interacting roles were excluded. In JADER, Japanese drug names were mapped to English equivalents using a manually validated dictionary.
Adverse event definition and hierarchical signal detection
The outcome of interest was nervous system adverse events, defined within the MedDRA system organ class “Nervous system disorders.” Hierarchical signal detection was performed across high-level group terms (HLGTs), high-level terms (HLTs), and preferred terms (PTs).
Disproportionality was quantified using the reporting odds ratio (ROR):
where a denotes reports containing both the drug and event, b the drug without the event, c the event without the drug, and d neither. The Information Component (IC) derived from the Bayesian Confidence Propagation Neural Network (BCPNN). Signals were defined as statistically robust if the lower limit of the 95% CI of the ROR > 1 and the lower limit of the 95% CI of the IC (IC025) > 0.
Covariates
Covariates included age, sex, and major comorbidities. Age was categorized as 18–44, 45–64, ⩾65 years, or unknown. Sex was coded as male, female, or unknown. To prevent selection bias commonly associated with non-random missing data in SRS, unknown was retained as a distinct dummy category in the regression models. Hypertension, diabetes mellitus, and chronic kidney disease were identified using MedDRA-coded indication and diagnosis fields. Specifically, to ensure the comprehensive capture of these conditions and avoid under-identification associated with single-term queries, these comorbidities were systematically extracted utilizing relevant Standardized MedDRA Queries (SMQs) alongside a predefined, aggregated list of HLTs and PTs.
Multivariable modeling in FAERS
Multivariable logistic regression was used to estimate independent associations between α1-adrenergic blocker exposure and nervous system adverse events. LASSO regression was applied for variable selection, followed by a generalized linear model with a logit link. 20 Adjusted odds ratios (ORs) and 95% CIs were reported.
External validation in JADER
The FAERS-derived model structure was applied unchanged to JADER. To reconcile database structural differences, JADER’s decadal age categories were harmonized into the FAERS-defined strata by mapping them to the closest corresponding interval. Descriptive analyses characterized exposure patterns and demographics. Model discrimination was assessed using the area under the receiver operating characteristic curve (AUROC) and the area under the precision–recall curve (AUPRC), and calibration was evaluated using the Brier score and decile-based calibration plots.
Individualized risk portrait construction
All clinically relevant combinations of drug, age, sex, and comorbidity status were enumerated, yielding 240 unique profiles. Predicted reporting probability was calculated as:
where η is the linear predictor. 21 Risks were visualized using matrix-based heatmaps with consistent color scales.
Statistical analysis and software
All analyses were conducted using R (version 4.3.0; R Core Team; R Foundation for Statistical Computing, Vienna, Austria) and DuckDB (DuckDB Foundation, Amsterdam, The Netherlands), and reported in accordance with the reporting of external adverse drug events using spontaneous reporting systems – pharmacovigilance (READUS-PV) guidelines. 19 Disproportionality analyses, regression modeling, and visualization were performed using validated R packages. Statistical significance was defined as a two-sided p value <0.05.
Results
Overall study design and analytical workflow
To characterize the association between α1-adrenergic blockers and nervous system adverse events in a manner that is both statistically robust and clinically interpretable, we designed a sequential pharmacovigilance workflow integrating signal detection, adjusted inference, external validation, and individualized risk translation (Figure 1). FAERS served as the primary discovery dataset, in which nervous system-related signals were first delineated within a hierarchical MedDRA framework to inform data-driven endpoint construction. Multivariable modeling was then applied to quantify independent associations while accounting for demographic characteristics and major comorbidities. The resulting model structure was transferred unchanged to the JADER database to evaluate effect stability and directionality in an independent reporting system. Finally, FAERS-derived model estimates were transformed into individualized risk portraits across clinically relevant combinations of drug exposure and patient characteristics, enabling progression from population-level signals to patient-level risk stratification within a single analytical framework.
Descriptive characteristics of α1-blocker-associated reports in FAERS
Across FAERS, a total of 14,458 α1-blocker-associated nervous system reports were identified, with marked heterogeneity in reporting volume across individual agents. Tamsulosin accounted for the largest share of reports (n = 9022), followed by doxazosin (n = 2948), silodosin (n = 1173), alfuzosin (n = 990), and terazosin (n = 325), reflecting substantial differences in real-world exposure and reporting intensity (Figure 2(a) and Table 1). Temporal trend analysis demonstrated a sustained increase in reporting over time for all agents, with tamsulosin consistently dominating annual report counts and a more modest but parallel rise observed for the remaining α1-blockers (Figure 2(b)).

Descriptive characteristics of α1-blocker-associated nervous system reports in FAERS. (a) Distribution of report counts by individual α1-blockers. (b) Temporal trends in annual reporting volume for each α1-blocker. (c) Sex distribution of reports stratified by drug. (d) Age distribution of reports across α1-blockers. (e) Geographic distribution of reports by reporting region.
Baseline characteristics of drug-associated adverse event reports in FAERS and JADER databases.

Age groups reported in FAERS: <18, 18–39, 40–59, 60–74, ⩾75 years.
Age groups reported in JADER: <18, 18–44, 45–64, ⩾65 years.
FAERS, FDA Adverse Event Reporting System; JADER, Japanese Adverse Drug Event Report Database; n, number; —, not applicable (drug absent from database).
Demographic characteristics of α1-blocker-associated nervous system reports demonstrated consistent and structured patterns across agents (Figure 2(c)–(e)). Sex distributions were characterized by a marked male predominance for all α1-blockers, most pronounced for tamsulosin and alfuzosin, while doxazosin exhibited a relatively higher, but still minority, proportion of female reports (Figure 2(c)). Age distributions were heavily weighted toward older individuals, with the majority of reports arising in patients aged ⩾60 years and peak frequencies observed in the 60–74 and ⩾75 years strata across all agents (Figure 2(d)). From a geographic perspective, reports were concentrated in North America and Western Europe, with the United States contributing the largest absolute volume. Agent-specific regional clustering was evident, exemplified by a comparatively higher contribution from France and other European countries for alfuzosin (Figure 2(e)). Collectively, these descriptive patterns delineate systematic variation in demographic composition and regional reporting, thereby providing an empirical basis for subsequent covariate-adjusted analyses rather than serving as independent evidence of differential risk.
Hierarchical signal detection within the nervous system SOC
Hierarchical signal detection within the MedDRA Nervous system disorders SOC identified statistically supported signals across multiple levels. At the HLGT level, 8 of 15 HLGTs met the prespecified signal criteria (a > 20, ROR > 1 with 95% CI lower bound >1; IC025 > 0) (Figure 3(a) and Table S1). The strongest enrichment was observed for Neuromuscular disorders (ROR = 25.83, 95% CI 23.94–27.87; IC = 4.59, IC025–IC975 4.49–4.70), followed by elevated signals in categories such as spinal cord and nerve root disorders and neurological disorders NEC (Table S1). At the HLT level, 19 of 53 HLTs met the same criteria (Figure 3(a) and Table S2). The top HLT signal was autonomic nervous system disorders (ROR = 27.53, 95% CI 25.51–29.71; IC = 4.69, 4.58–4.79). Additional enriched HLTs included Disturbances in consciousness, NEC and other clinically coherent neurological groupings (Table S2).
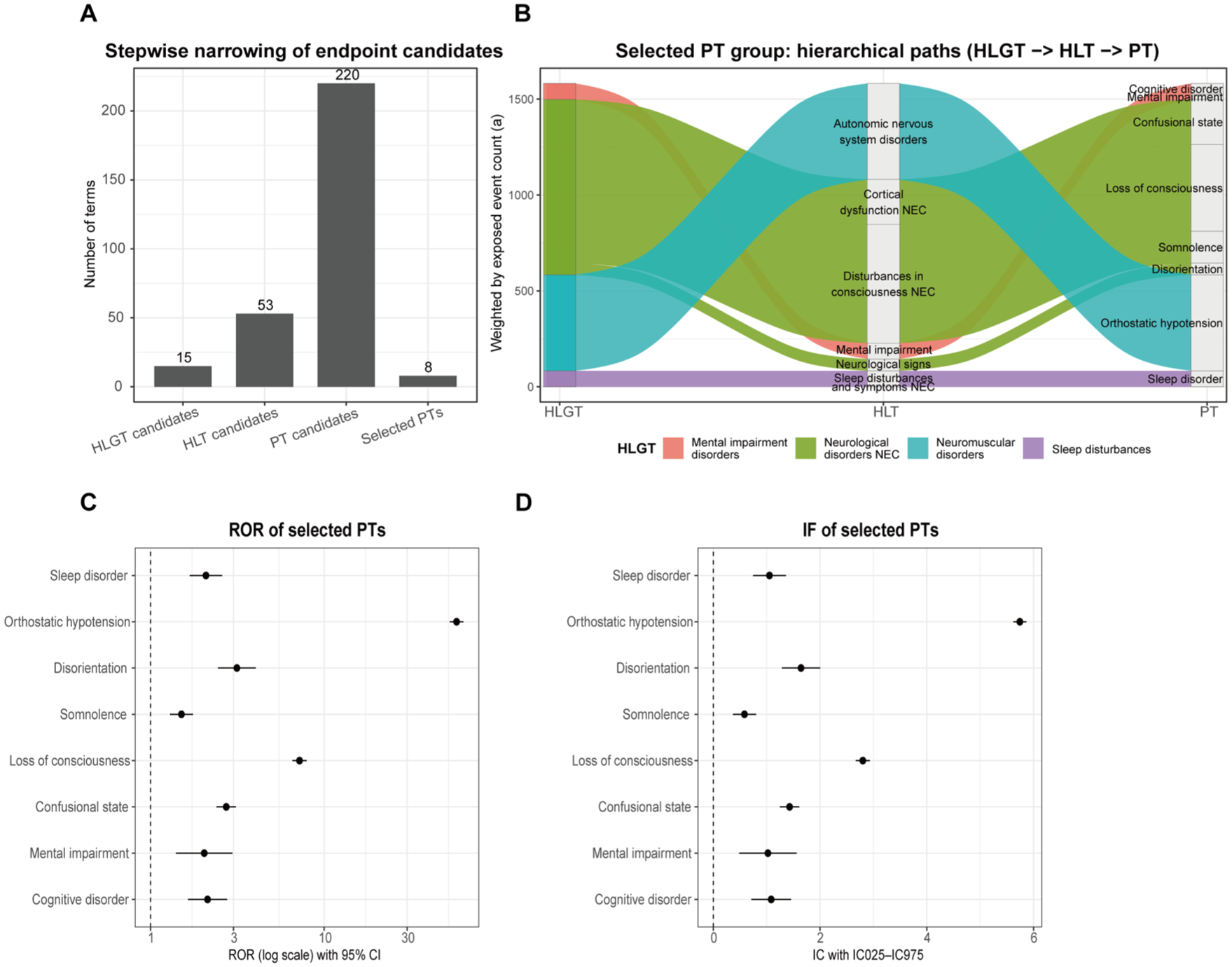
Hierarchical signal detection results for α1-adrenergic blockers within the MedDRA Nervous system disorders SOC. (a) Summary of signal detection at the HLGT, HLT, and PT levels based on predefined statistical criteria (a > 20; ROR > 1 with 95% CI lower bound > 1; IC025 > 0). (b) Hierarchical mapping of the eight clinically salient PTs selected for downstream analyses, demonstrating concordance with their parent HLT and HLGT classifications. (c, d) Signal strength of selected PTs, including reporting odds ratios and information component estimates. The selected PTs were aggregated to define a composite primary nervous system endpoint for subsequent multivariable modeling. (d) The central dots represent the Information Component (IC) value, and the horizontal error bars denote the 95% credibility interval (IC025–IC975).
At the PT level, 29 of 220 PTs met the predefined signal criteria (Figure 3(a) and Tables S3 and S4). Among these, a subset of eight clinically salient PTs was selected a priori for focused downstream analyses based on signal strength, hierarchical consistency, and clinical interpretability, reflecting their concordance with parent HLT and HLGT classifications as illustrated in Figure 3(b). These PTs comprised cognitive disorder, mental impairment, confusional state, disorientation, somnolence, sleep disorder, loss of consciousness, and orthostatic hypotension. Signal magnitude varied across PTs, with orthostatic hypotension exhibiting the strongest association (ROR = 57.80, 95% CI 52.78–63.30; IC = 5.74, IC025–IC975 5.62–5.86), followed by loss of consciousness (ROR = 7.20, 95% CI 6.55–7.90; IC = 2.80, 2.67–2.93), while the remaining PTs demonstrated moderate but statistically robust enrichment within their respective hierarchical categories (Figure 3(c) and (d)). These selected PTs were jointly aggregated to construct the primary nervous system PT group, which served as a composite endpoint for subsequent multivariable modeling and risk stratification analyses.
Multivariable modeling identifies independent risk factors for nervous system adverse events in FAERS
After construction of the primary nervous system adverse event endpoint, multivariable logistic regression was applied to the FAERS cohort to quantify independent associations between α1-adrenergic blocker exposure and nervous system adverse events while adjusting for demographic characteristics and major comorbidities (Figure 4). Among the five agents evaluated, tamsulosin was associated with a significantly increased risk (adjusted OR = 3.43, 95% CI 3.11–3.79), followed by terazosin (OR = 4.49, 95% CI 3.28–6.15), alfuzosin (OR = 3.20, 95% CI 2.50–4.09), and doxazosin (OR = 3.04, 95% CI 2.60–3.54). In contrast, silodosin did not show a statistically significant association after adjustment (OR = 1.52, 95% CI 0.85–2.72), suggesting a weaker or more heterogeneous signal.

Multivariable modeling of α1-blocker-associated nervous system adverse events in FAERS. Adjusted associations between α1-adrenergic blocker exposure and the composite nervous system adverse event endpoint in FAERS. Forest plot displays adjusted odds ratios and 95% confidence intervals derived from multivariable logistic regression, accounting for age, sex, and major comorbidities. Drug-specific associations are shown alongside covariate effects, illustrating independent contributions to neurological adverse event risk.
Age exhibited a non-linear association with nervous system adverse events, with both younger adults (18–44 years) and older individuals (⩾65 years) showing modest but significant increases in risk compared with the reference group, whereas reports with unknown age were associated with lower estimated risk. Sex contributed minimally, with male sex associated with only a marginal increase in risk (OR = 1.01, 95% CI 1.00–1.01). Among comorbidities, hypertension emerged as a strong independent risk factor (OR = 1.40, 95% CI 1.38–1.41), while diabetes mellitus and chronic kidney disease were associated with lower adjusted odds of nervous system adverse events. Overall, these findings indicate that drug-specific associations persist after comprehensive adjustment and are not solely attributable to demographic or comorbidity structure.
External validation in the JADER database
JADER descriptive characteristics of α1-blocker-associated reports
In JADER, α1-adrenergic blocker-associated nervous system reports exhibited distribution patterns that differed quantitatively from FAERS yet remained clinically coherent (Figure 5(a)–(d)). While FAERS included all five α1-adrenergic blockers under study, JADER lacked sufficient representation of the full drug spectrum. Tamsulosin accounted for the largest number of nervous system reports (n = 868), followed by doxazosin (n = 597) and silodosin (n = 475), all of which provided analyzable case volumes for downstream modeling. In contrast, terazosin was represented by only 16 cases, and alfuzosin was not captured in the JADER nervous system dataset, precluding stable or meaningful estimation for these agents (Figure 5(a)). Reporting volumes increased gradually over time, consistent with expanding clinical use rather than abrupt reporting artifacts (Figure 5(b)). Similar to FAERS, reports were predominantly derived from older individuals and showed a marked male predominance, reflecting prescribing patterns in Japan (Figure 5(c) and (d)).

External validation of α1-blocker-associated nervous system adverse events in the JADER database. (a) Distribution of nervous system reports by α1-blocker in JADER. (b) Temporal trends in reporting volume. (c) Sex distribution of reports. (d) Age distribution of reports. (e) Adjusted odds ratios for α1-blockers with sufficient exposure derived from multivariable modeling in JADER. (f) Model calibration plot showing observed versus predicted event rates across deciles of predicted reporting probability.
External validation of the multivariable model in JADER
Applying the FAERS-derived model structure to JADER demonstrated consistent effect directions for α1-adrenergic blockers with sufficient exposure, including tamsulosin, doxazosin, and silodosin (Figure 5(e) and (f)). In JADER, tamsulosin (OR = 7.06, 95% CI 5.44–9.17), doxazosin (OR = 3.92, 95% CI 2.66–5.75), and silodosin (OR = 10.24, 95% CI 7.55–13.90) all showed statistically significant associations with the composite nervous system endpoint after multivariable adjustment. In contrast, terazosin exhibited an unstable and non-significant estimate due to extreme sparsity (OR = 0.001, 95% CI 1.16×10–45–5.80×1038). Although confidence intervals were wider than in FAERS owing to smaller sample sizes and lower reporting density, no reversal of effect direction was observed (Figure 5(e)). Model performance metrics indicated modest discriminative ability, with an AUROC of 0.596 and an AUPRC of 0.021, and overall prediction error was low (Brier score = 0.012), consistent with expectations for spontaneous reporting data. Calibration analysis based on decile-binned predicted reporting probability demonstrated close agreement between mean predicted and observed event rates across all deciles, with absolute differences remaining small and observed risks increasing monotonically from the lowest to highest bins (Figure 5(f)). No systematic overestimation or underestimation was evident at the extremes of predicted reporting probability, supporting the external validity and calibration stability of the model.
Individualized risk portraits for α1-blocker-associated nervous system adverse events
To translate population-level associations into quantitative, patient-level risk estimates, individualized risk portraits were generated from the final FAERS multivariable model and summarized across clinically relevant subgroups (Figure 6). Predicted reporting probabilities were calculated for all combinations of drug exposure, age group, sex, and comorbidity status, yielding a comprehensive risk surface that enabled direct numerical comparison across strata.

Individualized risk portraits for α1-blocker-associated nervous system adverse events. (a) Predicted reporting probabilities in the overall population across combinations of drug exposure, age, and sex. (b) Predicted reporting probabilities restricted to individuals without recorded comorbidities. (c–e) Predicted reporting probabilities stratified by single comorbidity status (hypertension, diabetes mellitus, or chronic kidney disease).
In the overall population, predicted reporting probabilities varied substantially by drug choice, with median predicted reporting probabilities ranging from 0.053 for silodosin to 0.143 for terazosin (Figure 6(a)). When restricted to individuals without any recorded comorbidities, absolute risk levels were uniformly lower across all agents, and the spread of predicted reporting probability was compressed relative to the overall population, indicating attenuation of baseline risk in the absence of comorbidity burden (Figure 6(b)). Across all demographic and comorbidity combinations, terazosin consistently occupied the upper end of the predicted reporting probability distribution (maximum predicted reporting probability 0.205), whereas silodosin demonstrated the lowest and most compressed risk range (maximum predicted reporting probability 0.081). Tamsulosin, doxazosin, and alfuzosin showed intermediate risk profiles, with median predicted reporting probabilities of approximately 0.10–0.11 (Figure 6(a) and (b)).
Stratification by comorbidity burden revealed graded shifts in the predicted reporting probability distribution beyond the no-INDI subgroup. Among individuals without major comorbidities (overall subgroup), the median predicted reporting probability was 0.112, with values spanning from 0.053 to 0.156 depending on drug and demographic characteristics. In patients with a single comorbidity (hypertension, diabetes mellitus, or chronic kidney disease), median predicted reporting probability remained comparable (0.101), but the upper tail of the distribution expanded, reaching a maximum predicted reporting probability of 0.205 (Figure 6(c)–(e)). Dual-comorbidity profiles exhibited a similar median risk (0.107) with sustained elevation of high-risk combinations, while individuals with all three comorbidities demonstrated a concentrated but consistently elevated risk range (median 0.102), indicating attenuation of heterogeneity at uniformly higher baseline risk levels (Figures S1 and S2).
Across all subgroup analyses, age exerted a dominant influence on absolute risk magnitude, with older age strata consistently associated with higher predicted reporting probabilities within each drug–comorbidity combination. Sex-specific differences were numerically modest and did not materially alter the rank ordering of drug-associated risks. Collectively, these data-driven risk portraits demonstrate that neurological adverse event risk associated with α1-adrenergic blockers is jointly determined by agent-specific effects and cumulative comorbidity burden, and that clinically meaningful gradients persist even after multivariable adjustment.
Discussion
In this large-scale pharmacovigilance study integrating FAERS and JADER, we systematically evaluated the association between α1-adrenergic blockers and nervous system adverse events and translated population-level signals into clinically interpretable risk portraits. By combining hierarchical signal detection, multivariable modeling, external validation, and individualized risk visualization, this analysis provides a coherent evidence chain that extends beyond conventional disproportionality approaches and addresses key limitations of SRS-based drug safety research.
From isolated signals to structured neurological risk
Previous pharmacovigilance studies of α1-adrenergic blockers have largely focused on individual adverse events, such as dizziness, syncope, or orthostatic hypotension, often examined in isolation or within narrow clinical contexts.22–25 In contrast, our hierarchical signal detection within the MedDRA Nervous system SOC demonstrated that α1-blocker-associated signals were distributed across multiple HLGTs and HLTs rather than being driven by single PTs (Figure 3). This structured signal architecture suggests that neurological adverse events associated with α1-adrenergic blockers represent a spectrum of related manifestations rather than sporadic reporting artifacts. Leveraging this hierarchy, we constructed a data-driven composite endpoint that better reflects real-world neurological risk and reduces the instability inherent in single-term analyses.
Drug-specific effects beyond class-level assumptions
Although α1-adrenergic blockers are frequently treated as a homogeneous drug class in clinical practice and safety assessments, 26 our multivariable analysis revealed substantial heterogeneity among individual agents. After adjustment for age, sex, and major comorbidities, tamsulosin, doxazosin, terazosin, and alfuzosin showed robust associations with nervous system adverse events, whereas silodosin did not demonstrate a statistically significant association (Figure 4). These findings are consistent with known pharmacological differences among α1-adrenergic blockers, including receptor subtype selectivity, central nervous system penetration, and pharmacokinetic profiles.27–29 Most notably, the distinct lack of a statistically significant association between silodosin and central neurological adverse events in our model can be convincingly explained by its pharmacokinetic properties. Previous pharmacological profiling has demonstrated that silodosin exhibits minimal blood-brain barrier penetrance compared to other agents in its class. 30 This minimal CNS exposure provides a robust mechanistic rationale for its safer neurological profile observed in our real-world cohort. Importantly, the persistence of drug-specific associations after multivariable adjustment argues against simple confounding by indication or demographic structure and supports the need for agent-level safety evaluation rather than reliance on class-wide assumptions.
The modifying role of patient characteristics and comorbidities
Beyond drug effects, patient characteristics substantially shaped neurological risk. Advanced age emerged as a consistent determinant of increased predicted reporting probability, which is biologically plausible given age-related changes in autonomic regulation, cerebral perfusion, and blood-brain barrier integrity.31,32 From a pharmacological perspective, preclinical evidence suggests that the pharmacodynamics of α1-adrenergic blockers are fundamentally altered in older populations, leading to heightened sensitivity to these agents. 33 Furthermore, the amplifying effect of comorbidities such as diabetes and hypertension can be explained by the crucial role of α1-adrenergic receptors in metabolic regulation. Animal models, particularly α1B or α1A knockout models, demonstrate that the loss or blockade of these receptors impairs glucose homeostasis and precipitates metabolic syndrome-like symptoms, potentially exacerbating neurological vulnerabilities.34,35 Additionally, in patients with chronic kidney disease, vascular α1-adrenergic sensitivity is intrinsically enhanced, 36 and long-term α1 blockade has been implicated in salt-sensitive hypertension. 37 These preclinical insights brilliantly align with our clinical risk portraits, explaining why multi-comorbid patients experience compounded neurological risk.
Hypertension further amplified risk across nearly all strata, potentially reflecting both disease-related vascular vulnerability and pharmacodynamic interactions affecting cerebral autoregulation.38,39 In contrast, diabetes mellitus and chronic kidney disease were associated with lower adjusted odds of nervous system adverse events; this pattern may reflect reporting behavior, competing risks, or differential clinical monitoring rather than true protective effects. 40 These observations underscore the importance of cautious interpretation of comorbidity effects and the value of multivariable adjustment in SRS-based research. 41
External validation across regulatory systems
A major strength of this study is the external validation in an independent national SRS. Differences in healthcare systems, prescribing practices, and reporting behavior can limit the generalizability of FAERS-based results.42,43 Despite these differences, we observed consistent effect directions for α1-adrenergic blockers with sufficient exposure in JADER, along with acceptable calibration. Although discrimination metrics were modest, as expected for SRS data, the absence of systematic miscalibration supports model transportability. At the same time, drugs with sparse exposure in JADER could not be reliably evaluated, highlighting a common but often underappreciated limitation of cross-database validation.44,45
Risk portraits as a bridge to clinical interpretability
The most distinctive contribution of this study lies in the individualized risk portraits. Conventional PV outputs-such as ORs or disproportionality scores-are difficult to translate into actionable clinical insights. 42 As illustrated in Figure 6, by enumerating clinically relevant combinations of drug exposure, age, sex, and comorbidity status, we transformed model coefficients into structured visual matrixes that illustrate how risk accumulates across patient profiles. Notably, patients with multiple comorbidities exhibited a concentrated high-risk zone, suggesting a cumulative effect exceeding that of any single risk factor. This visualization-based approach aligns with emerging calls for patient-centered risk communication and decision support in drug safety46,47 and provides a practical pathway for integrating PV findings into clinical reasoning.
Limitations
Taken together, our findings support a shift from isolated signal detection toward integrated, patient-centered risk modeling. Clinically, neurological risk associated with α1-adrenergic blockers should be evaluated in the context of both drug selection and patient profile, particularly among elderly individuals with multiple comorbidities. Methodologically, this study demonstrates how SRS data can be leveraged through hierarchical endpoint construction, multivariable adjustment, external validation, and risk visualization to enhance clinical interpretability.
Several limitations should be acknowledged. First, as with all SRS-based analyses, FAERS and JADER are subject to under-reporting, reporting bias, and incomplete clinical information, which precludes direct estimation of incidence and limits causal inference. Additionally, while JADER was utilized to validate signals in a homogenous Asian population with distinct prescribing practices, we acknowledge that FAERS globally aggregates data, including a proportion from Japan, which introduces a potential limitation of partial data overlap between the discovery and validation cohorts. Second, residual confounding cannot be fully excluded despite multivariable adjustment, as unmeasured factors such as disease severity, concomitant medications, and treatment duration are not reliably captured in SRS data. Third, the composite nervous system endpoint, although constructed using a data-driven hierarchical approach, aggregates heterogeneous neurological manifestations and does not allow assessment of risk for individual clinical syndromes. As correctly noted from a clinical neurology perspective, MedDRA PTs encompass varying degrees of clinical granularity. Terms such as mental impairment or loss of consciousness may reflect distinct pathophysiological processes ranging from transient symptomatic manifestations to well-defined neurological syndromes. Therefore, while our hierarchical approach mitigates the instability of single-term analyses, these grouped terms cannot substitute for rigorous clinical phenotyping, and the interpretability of specific neurological syndromes remains limited. Fourth, external validation in JADER was constrained by limited exposure for certain α1-adrenergic blockers, restricting stable estimation and generalizability for those agents.
Despite these limitations, the consistency of effect directionality across databases and the coherent stratification observed in risk portraits support the robustness of the overall findings. Future studies should pursue prospective validation using electronic health record-based cohorts and longitudinal designs, incorporate richer clinical covariates, and evaluate whether individualized risk portraits can inform personalized prescribing decisions or targeted monitoring strategies in real-world practice.
Conclusion
In conclusion, this integrated pharmacovigilance study provides a comprehensive and clinically interpretable assessment of nervous system adverse events associated with α1-adrenergic blockers. By combining hierarchical signal detection, multivariable modeling, external validation, and individualized risk visualization, we demonstrate that neurological risk varies substantially across individual agents and patient profiles rather than being uniform at the drug-class level. The consistency of effect directionality across FAERS and JADER supports the robustness of the identified associations, while individualized risk portraits translate population-level signals into patient-centered risk patterns. Together, these findings highlight the importance of agent-specific and patient-specific risk evaluation and illustrate a practical framework for enhancing the clinical utility of spontaneous reporting data in drug safety assessment.
Supplemental Material
sj-docx-1-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-docx-1-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-docx-2-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-docx-2-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-3-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-tif-3-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-tif-4-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-tif-4-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-5-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-xlsx-5-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-6-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-xlsx-6-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-7-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-xlsx-7-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Supplemental Material
sj-xlsx-8-taw-10.1177_20420986261444611 – Supplemental material for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER
Supplemental material, sj-xlsx-8-taw-10.1177_20420986261444611 for Drug- and patient-specific neurological risk profiles of α1-adrenergic blockers: evidence from FAERS and external validation in JADER by Xin Hai and Lulu Huang in Therapeutic Advances in Drug Safety
Footnotes
Acknowledgements
The authors would like to thank the FDA for making the FAERS publicly available and the PMDA of Japan for providing access to the JADER database. We thank Wuhan Shengfu Biotechnology Co., Ltd. for providing data analysis support.
Declarations
Supplemental material
Supplemental material for this article is available online.
Declaration of generative AI and AI-assisted technologies in the manuscript preparation process
During the preparation of this work, the authors used ChatGPT 5.2 in order to polish the language. After using this tool, all authors reviewed and edited the content as needed and took full responsibility for the content of the published article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.


