Abstract
Background
Futibatinib, a novel FGFR inhibitor, is a promising therapy for cancers with FGFR alterations. However, its safety profile remains under investigation. This study evaluates futibatinib-associated adverse events using the FDA Adverse Event Reporting System (FAERS).
Methods
A retrospective pharmacovigilance analysis was conducted using FAERS data from January 1, 2013, to March 9, 2025. Disproportionality analysis was performed via OpenVigil 2.1, with signals defined by a reporting odds ratio (ROR) > 2.00 and a 95% confidence interval lower bound >1.00. Events with at least two reports were included after deduplication and standardization.
Results
Strong safety signals were identified. Hyperphosphatasemia showed the highest association (ROR 1612.73; 6 events), followed by elevated blood phosphorus (ROR 603.85; 3 events). Dermatologic toxicities, including onycholysis (ROR 520.15) and onychoclasis (ROR 126.20), were also notable.
Conclusion
These findings highlight the range of adverse events associated with this agent. These events may reflect direct drug effects and individual variability. Further studies are needed to clarify mechanisms and inform clinical management.
Introduction
Targeted therapies have significantly advanced cancer treatment by focusing on specific molecular drivers of tumor growth. 1 Among these targets, fibroblast growth factor receptors (FGFRs) are integral in regulating cell proliferation, differentiation, and survival. 2 Aberrant FGFR signaling has been implicated in the pathogenesis of various malignancies, including cholangiocarcinoma, urothelial carcinoma, and certain lung cancers, establishing FGFRs as attractive therapeutic targets and spurring the development of FGFR inhibitors.3,4 Futibatinib is an irreversible FGFR inhibitor that covalently binds to the receptor's kinase domain, effectively blocking autophosphorylation and downstream signaling pathways contributing to tumor progression. 5 Clinical trials have demonstrated its efficacy in inducing tumor regression in patients with FGFR-altered cancers. A pooled safety analysis encompassing 469 patients across various tumor types revealed that futibatinib has a consistent and manageable safety profile. 6 The most common adverse events (AEs) were mild to moderate in severity, with treatment-related discontinuations being rare and no treatment-related deaths reported. Among the AEs of clinical interest, hyperphosphatemia was the most frequently observed.
The present study leverages data from the FDA Adverse Event Reporting System (FAERS) to quantify the prevalence and strength of associations between futibatinib and a spectrum of adverse events. This analysis aims to inform clinical practice by providing data-driven recommendations for monitoring and managing these toxicities in patients undergoing treatment with futibatinib. By enhancing the understanding of futibatinib's safety profile, healthcare professionals can optimize therapeutic outcomes and improve the quality of life for patients receiving this targeted therapy.
Methods
The methods have been previously described.7–9 In this retrospective pharmacovigilance study, we analyzed data from the FAERS, a publicly accessible database that aggregates spontaneous reports of adverse drug reactions from pharmaceutical companies, healthcare professionals, and consumers, serving as a valuable resource for post-marketing drug safety surveillance. The study period spanned from January 1st, 2013, to March 9th, 2025, encompassing the entire duration of futibatinib's clinical use. OpenVigil 2.1 (OpenVigil, Kiel, Germany), a web-based pharmacovigilance analysis tool specifically designed for FAERS data, was used to extract and preprocess the relevant reports. OpenVigil 2.1 offers advanced capabilities for data cleaning, filtering, extraction, and analysis, ensuring transparency and reproducibility in pharmacovigilance research. To maintain data integrity, several preprocessing steps were implemented: deduplication by identifying and removing duplicate entries through coding individual safety reports (ISRs) and mapping drug names to standardized nomenclature, such as the U.S. Adopted Name (USAN) scheme; and standardization by coding adverse events using the Medical Dictionary for Regulatory Activities (MedDRA) terminology, specifically utilizing the Preferred Terms (PTs) and System Organ Classes (SOCs) to classify and describe AEs consistently. Given futibatinib's recent market introduction and the anticipated limited number of reports, all adverse events with a minimum of two reports were included in the analysis. To identify potential safety signals, we conducted a disproportionality analysis using the Reporting Odds Ratio (ROR), a statistical measure commonly employed in pharmacovigilance to detect associations between drugs and adverse events. The ROR compares the odds of a specific AE occurring with a particular drug to the odds of the same AE occurring with all other drugs in the database. The ROR was calculated using a two-by-two contingency table, and a signal was considered statistically significant if the ROR exceeded 2.00 and the lower limit of the 95% confidence interval (CI) was greater than 1.00. As this is a newer agent, even AEs with few reports were included. All data processing and statistical analyses were performed using OpenVigil 2.1, which facilitates the extraction, cleaning, and analysis of FAERS data. By employing these rigorous methodologies, this study aims to provide a comprehensive assessment of the adverse event profile associated with futibatinib, thereby informing clinical practice and enhancing patient safety. Adverse events reports were filtered for futibatinib as the primary suspect.
Results
The analysis revealed several notable safety signals associated with futibatinib treatment (Table 1). The most pronounced signal was hyperphosphatasemia, with a ROR of 1612.73 (95% CI: 692.56–3755.46), based on six reported cases. Elevated blood phosphorus levels also showed a strong association, with an ROR of 603.85 (95% CI: 188.92–1930.08) from three cases. Dermatologic adverse events were significant; onycholysis had an ROR of 520.15 (95% CI: 126.89–2132.32; two cases), and onychoclasis presented an ROR of 126.20 (95% CI: 30.82–516.69; two cases). Other noteworthy signals included hypercalcemia (ROR 111.64, 95% CI: 34.97–356.39; three cases), decreased blood magnesium (ROR 108.11, 95% CI: 26.41–442.61; two cases), and cytopenia (ROR 66.90, 95% CI: 16.34–273.84; two cases). Additional adverse events such as taste disorders, stomatitis, and gastrointestinal disturbances were observed but had comparatively lower RORs. It is worth noting that reports pertaining to death and disease progression as likely associated with the underlying malignancy.
Statistical data associated with futibatinib.
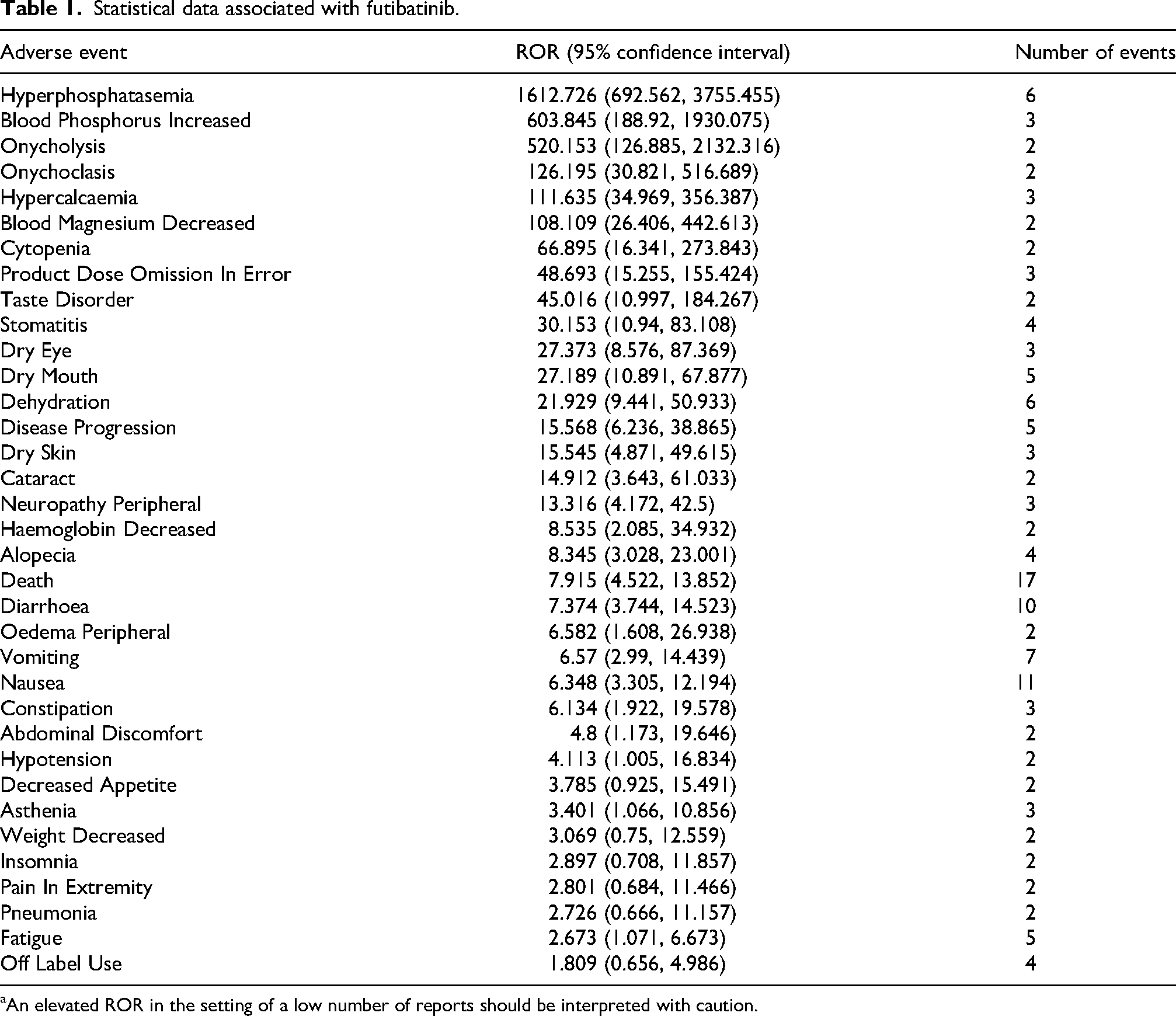
An elevated ROR in the setting of a low number of reports should be interpreted with caution.
Discussion
The findings from this FAERS analysis provide important insights into the adverse event profile of futibatinib. The exceptionally high ROR for hyperphosphatasemia suggests a significant impact on phosphate metabolism, which is consistent with the known effects of FGFR inhibition. Elevated blood phosphorus levels and related metabolic abnormalities further support the possibility of disrupted homeostasis. Dermatologic signals, particularly onycholysis and onychoclasis, raise additional concerns regarding the cutaneous effects of futibatinib, which may compromise patient quality of life.
Several mechanisms may contribute to these adverse events. FGFR inhibitors, by design, block the receptor tyrosine kinase activity of FGFRs, thereby inhibiting downstream signaling that promotes cell proliferation and survival. However, this inhibition is not always perfectly selective. The direct blockade of FGFR signaling can inadvertently interfere with pathways critical for normal metabolic regulation. For example, FGFRs are involved in maintaining phosphate and calcium homeostasis through the FGF23-Klotho axis. 10 Inhibiting these receptors can disrupt this balance, potentially leading to elevated serum phosphate levels and disturbances in calcium and magnesium metabolism. In the FOENIX-CCA2 trial, which investigated the efficacy and safety of futibatinib in patients with intrahepatic cholangiocarcinoma harboring FGFR2 fusions or rearrangements, hyperphosphatemia occurred in 85% of patients, though no reports of hypercalcemia were made. 11 However, 12% of patients in the TPU-TAS-120-101 trial experienced hypercalcemia. 12 There is further evidence of hypercalcemia in this population, including instances of calciphylaxis, a rare but severe complication that has been associated with FGFR inhibitors. 13 These metabolic derangements may manifest as gastrointestinal symptoms if the disruption to electrolytes is severe enough. Genetic differences among patients, such as polymorphisms in drug-metabolizing enzymes, could result in inter-individual variability in the pharmacokinetics of FGFR inhibitors. Patients with slower metabolic clearance may accumulate higher drug concentrations, increasing the risk of toxicity, while others might experience suboptimal dosing and different adverse event profiles. This variability may predispose certain individuals to cytopenias if the drug or its metabolites exert unintended effects on bone marrow function or to gastrointestinal events if the metabolic imbalance affects the gut epithelium or alters local homeostasis.
FAERS is a critical tool for post-marketing surveillance of approved drug and therapeutic biologic products. However, studies utilizing FAERS data face several inherent limitation. 14 Firstly, the system's reliance on voluntary reporting by healthcare professionals and consumers leads to underreporting. Additionally, the data may contain duplicate or incomplete reports, and the information provided is not always medically verified. Importantly, the presence of a report in FAERS does not confirm a causal relationship between the product and the adverse event, as the event could result from underlying diseases or concurrent medications. Moreover, extremely elevated RORs in the setting of few reports should be interpreted cautiously. Furthermore, due to the lack of comprehensive data on the total number of individuals using a particular product, FAERS cannot be used to calculate the incidence of adverse events. As a result, while they are useful for detecting safety signals, they may not fully reflect the frequency or severity of adverse events. To validate these associations, prospective studies are essential. Such studies can systematically collect data and help clarify the causal relationships between futibatinib and its adverse effects. In parallel, detailed mechanistic investigations, including both in vitro studies and preclinical models, are needed to understand the biological pathways involved in futibatinib-induced toxicity.
Moreover, establishing dedicated patient registries would enhance the post-marketing surveillance process by offering a structured way to gather longitudinal data on futibatinib's safety profile. These registries can track long-term outcomes, monitor for rare adverse events, and support more personalized approaches to patient care. Enhanced surveillance systems that integrate data from multiple sources, such as electronic health records, laboratory results, and direct patient reporting, are needed. Together, these measures will enable a more comprehensive evaluation of futibatinib's risks, ultimately ensuring that patient care is optimized and that any potential toxicities are managed proactively.
Footnotes
Acknowledgements
N/a
Ethical approval and consent to participate
As only publicly available patient data were used in this study, no ethics approval was required.
Human ethics
No ethics approval was required.
Consent for publication
C.F. provides consent for publication.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
