Abstract
Background:
Hysteroscopy, a popular outpatient gynecological procedure, often requires sedation due to pain caused by endometrial manipulation and cervical dilation. Propofol, the current standard sedative, has adverse effects like respiratory depression, hypotension, and injection discomfort. Ciprofol, a novel GABA-A receptor agonist and structural analog of propofol, might be a safer choice while maintaining efficacy.
Objectives:
This systematic review and meta-analysis aims to compare the efficacy and safety of ciprofol versus propofol for sedation during hysteroscopy.
Design:
A systematic review and meta-analysis of randomized controlled trials (RCTs).
Data sources and methods:
This systematic review and meta-analysis adhered to Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. RCTs comparing ciprofol and propofol for hysteroscopic sedation were sourced from PubMed, Cochrane, Scopus, Google Scholar, and ClinicalTrials.gov. Data extraction, quality assessment (ROB-2), and analysis were conducted independently. Outcomes included efficacy, safety, and recovery metrics.
Results:
Six RCTs with 1890 patients were met with inclusion. Ciprofol achieved similar sedative success as propofol (risk ratio (RR) = 1.00, 95% CI: 1.00–1.00) and surpassed propofol in important safety outcomes. It significantly reduced intraoperative body movements (RR = 0.53), injection pain (RR = 0.13), hypotension (RR = 0.58), and respiratory depression (RR = 0.67), while increasing eyelash reflex suppression (MD = 0.24). Ciprofol was also associated with a slightly longer recovery time compared to propofol (MD = 0.80 min). Bradycardia incidence and procedure duration did not differ significantly.
Conclusion:
Ciprofol displays a better safety profile, including enhanced hemodynamic and respiratory stability and decreased injection discomfort, and is just as effective as propofol for sedation during hysteroscopy. According to these results, ciprofol is a safe alternative to propofol for hysteroscopy. It is advised that further multicenter studies are required to confirm its effectiveness across broader populations.
Trial registration:
The meta-analysis has been registered with PROSPERO. The registration number is CRD420251091116.
Plain language summary
Hysteroscopy is a common procedure used to examine or treat problems inside the uterus. To make patients comfortable during this procedure, doctors use medications that help them relax or sleep. Two such medicines are Ciprofol and Propofol. In this study, we looked at all available high-quality research trials that compared how well these two drugs work and how safe they are. We focused on how successful the sedation was, how quickly it worked, how long recovery took, and what side effects patients experienced—like low blood pressure, breathing problems, or pain from injection. Our findings suggest that Ciprofol may be just as effective as Propofol, with some benefits like less pain at the injection site and fewer breathing problems. This could make Ciprofol a better option for women undergoing hysteroscopy in certain cases. These results can help doctors choose the most suitable medication to make the procedure safer and more comfortable for patients.
Keywords
Introduction
Hysteroscopy is a mildly invasive gynecological procedure that is mostly used for the diagnosis and treatment of endometrial and other intrauterine diseases. 1 Globally, hysteroscopy is increasingly becoming an everyday practice, with an estimated 600,000 hysterectomies performed yearly in the United States, most of which are preceded by assessment with hysteroscopy (https://www.psmarketresearch.com/market-analysis/us-hysteroscopes-market). European nations perform around 500–800 hysteroscopies/per 100,000 women, reflecting outpatient standardization, while low and middle-income countries show rising urban adoption despite rural gaps, with Pakistan/India performing approximately 50,000–100,000 yearly, primarily for infertility and retained products.
Although the procedure itself is not lengthy, the severe pain from endometrial curettage and cervical dilatation is unbearable.2–4 Consequently, the fear and anxiety patients experience regarding this pain necessitates an anesthetic method in most instances.5,6 While hysteroscopy can technically be performed under various approaches, including general anesthesia, paracervical block, or local anesthesia 7 —achieving the right balance of patient comfort, safety, and procedural efficiency remains a clinical priority.
For decades, propofol, an intravenous anesthetic agent, has been the conventional sedative of choice for hysteroscopic procedures due to its rapid onset, short recovery time, and credible sedative properties. 8 Nevertheless, it also has drawbacks such as injection pain and significant effects on the respiratory and circulatory systems. 9 These limitations directly impact patient experience and safety; injection pain alone affects 28%–90% of adults receiving propofol for procedures like gastroenteroscopy, 10 suggesting similar concerns likely apply in the hysteroscopy setting. Such side effects highlight the need to explore alternative sedatives that maintain propofol’s benefits while minimizing its adverse effects.
The newly developed short-acting gamma-aminobutyric acid (GABA) agonist ciprofol (HSK3486) was officially approved for clinical use by the China National Medical Products Administration on December 15, 2020. The approval was exclusively made for use in sedation for gastrointestinal endoscopy, and it has attracted considerable attention due to persistent clinical challenges and controversies in sedation agents in this use. 11 Ciprofol is gaining rapid popularity as a highly effective substitute for the traditionally used anesthetic compound propofol. This new status is, to a large extent, attributed to the fact that both drugs have the same chemical backbone, with a close structural chemistry relationship, both drugs fall under the general category of 2,6-disubstituted phenols. 12 Due to these similarities, ciprofol has been identified as a safe and effective drug for inducing procedural sedation.
Clinical evidence-wise, findings from a phase II clinical trial evaluating the efficacy and safety of ciprofol specifically in colonoscopy provided a critical set of data. The study established that ciprofol could be used at significantly smaller doses, that is, between one-quarter and one-fifth of the traditional dose of propofol. 13 In addition, ciprofol administration was also associated with fewer residual effects after sedation, an important consideration in patient recovery. Also, patients who received ciprofol did not experience significant pain at the point of injection, a welcome finding compared to other sedation agents. 13 In addition, another large study was conducted to directly compare the efficacy and safety profile of ciprofol with propofol when used for sedation in hysteroscopy. The findings of this study concluded that the success rate for hysteroscopy in both groups, that is, ciprofol group and propofol group, was an astounding 100%. In the subsequent analysis of adverse events, it was noted that the rate of adverse events in the ciprofol group was significantly lower compared to the propofol group. This finding indicates that ciprofol could indeed be a safer sedation agent, with similar efficacy and safety profiles to propofol. 14 Despite the expanding interest of ciprofol in various hysteroscopy procedures, the current evidence remains scattered across individual trials. Although various studies report promising advantages of ciprofol over propofol regarding key clinical outcomes, yet no comprehensive synthesis has yet evaluated it collectively in the context of hysteroscopic sedation. Therefore, a systematic thorough evaluation is warranted to evaluate the efficacy of ciprofol in various clinical practices and address various adverse events.
The major objective of this systematic review and meta-analysis is to compare the efficacy and safety of ciprofol with propofol for sedation in hysteroscopy. The main outcomes that can be expected are onset and recovery times, as well as sedation success rates. By incorporating the available data, this work seeks to offer anesthesiologists and gynecologic practitioners’ evidence-based guidance regarding the optimal sedative choice for hysteroscopic procedures.
Materials and methods
This systematic review and meta-analysis followed Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 15 This study is registered with PROSPERO (ID) CRD420251091116.
Data sources and search strategy:
Different electronic databases such as PubMed (MEDLINE), Cochrane Library via Wiley (all databases), and Scopus were searched from the start of the database indexing until July 2025, using the following search terms: (“Hysteroscopy”[MeSH] OR hysteroscopy) AND (“Ciprofol” OR HSK3486) AND (“Propofol”[MeSH] OR propofol) AND (“Conscious Sedation”[MeSH] OR sedation OR “sedation efficacy” OR “sedation outcome”). The detailed search strategies for each database are provided in Supplemental Table 1. A “ClinicalTrials.gov” search was also conducted to find relevant published or unpublished clinical trials. A manual search of relevant publications and their references was carried out to identify potential articles. No restrictions regarding race, place, sex, ethnicity, language, or dates were applied. Duplicates were removed using Rayyan Systems, Inc. is the U.S.–based private company that develops and operates Rayyan.ai, an AI-powered screening tool for systematic reviews.
Study selection
We included only randomized controlled trials (RCTs) comparing ciprofol (as monotherapy) against propofol (as monotherapy) for procedural sedation in adult women aged 18 years or older undergoing diagnostic OR surgical hysteroscopy. All eligible participants are classified as American Society of Anesthesiologists (ASA) physical status I or II with a body mass index (BMI) ranging from 18 and 30 kg/m2. Other inclusion criteria included:
Both drugs are used as primary sedative agents, with any adjunctive medication used equally across both study arms.
Evaluation of at least one safety outcome (e.g., hypotension, respiratory depression, injection pain, or adverse events) or efficacy outcome (e.g., sedation success rate, onset time, recovery time, or satisfaction)
Studies with a parallel arm design or that provide independently extractable outcome data for each group.
Multi-arm studies are used where data from the ciprofol and propofol arms are available for direct comparison.
We excluded cluster-randomized trials, observational studies (e.g., cohort or case control), uncontrolled trials, and reviews of independently published trials. Clinical trials were also removed if they did not include adult women undergoing hysteroscopy under intravenous anesthesia, had fewer than 10 patients per treatment group, or if anesthesia was combined with other interventions that could invalidate results. Studies that included patients with serious comorbidities, such as severe hepatic, renal or cardiac conditions, known anesthesia drug contraindications, or significant psychiatric illness were also eliminated. In addition, case reports, letters to the editor, commentaries, and cross-sectional studies were also not considered eligible.
Screening process
Two reviewers, S.R. and A.F., independently screened Abstracts and titles using Rayyan. Full paper screening was conducted independently by the same two reviewers. Discrepancies were resolved through discussion. A third reviewer (Z.A.) verified the final decisions. Inter-rater reliability was not explicitly calculated; however, consensus was attained in all cases.
Data extraction and quality assessment
An online Microsoft Excel spreadsheet was created using data from the completed RCTs for the baseline characteristics and outcomes. Two reviewers, HA and FS, carried out the data extraction process, and ZA, a third reviewer, verified it. Discrepancies within the abstract and title, full article screening, and data extraction were resolved through discussion.
The data items extracted for baseline were: total population, total number of patients in ciprofol and propofol groups, mean age, BMI, weight, height, mean arterial pressure (MAP), heart rate (HR), and ASA status. The outcomes were selected to provide a comprehensive evaluation of both efficacy and safety when comparing ciprofol and propofol for procedural sedation.
Primary outcomes were chosen to directly assess the clinical efficacy and immediate safety of sedation:
Sedation success rate
Injection pain
Bradycardia
Hypotension
Respiratory complications
Duration of eyelash response absence
Intraoperative body movement
Secondary outcomes were selected to further evaluate tolerability, recovery profile, physiological stability, and patient-centered measures:
Side effects (hypoxemia)
Recovery time
Hemodynamic and respiratory parameters (heart rate, MAP, systolic blood pressure (SBP), diastolic blood pressure (DBP), oxygen saturation (SpO2))
Hysteroscopy time
A single reviewer (H.H.) undertook a study-level quality assessment. Depending on the study design, the Cochrane tool for risk of bias assessment 2.0 (ROB-2) 16 was used. This tool was considered both reliable and valid, with the review team having prior experience in using it.
Meta-analysis and data synthesis
This meta-analysis was performed using Review Manager (RevMan version 5.1, Cochrane’s Information Management System groups in Copenhagen, Denmark) software. Forest plots were generated, and a random-effects model was applied to calculate risk ratios (RRs) with 95% confidence intervals (CIs) for primary outcomes, which included measures of sedation efficacy and safety, and weighted mean difference or standardized mean difference for continuous outcomes such as onset or recovery time. Heterogeneity was assessed using the I2 statistic, with initial values exceeding 50% indicating substantial heterogeneity. A sensitivity analysis was performed by excluding studies identified as high risk of bias according to the ROB-2 tool as well as those with incomplete outcome data, such as missing sedation success rates, recovery times, or safety outcomes essential for inclusion in the meta-analysis. This exclusion reduced I² below 50%, indicating low to moderate heterogeneity and reinforcing the reliability of the results. Funnel plots were used to assess the presence of publication bias.
Result
Study selection
The database searches yielded a total of 48 articles from PubMed (4), Scopus (33), and the Cochrane Library (11). After removing nine duplicates, six articles remained. These were screened for relevance based on titles and abstracts. Full texts of the remaining studies were assessed for eligibility according to the predefined inclusion criteria. All six studies were deemed suitable and were included in the final meta-analysis. The study selection process is summarized in Figure 1.

PRISMA flowchart illustrating the study selection process according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines.
Characteristics of studies
All six articles included were RCTs conducted on individuals undergoing hysteroscopy. Table 1 (Baseline characteristics) outlines the detailed characteristics of the included studies and the results of each study. This table summarizes six studies (2023–2025) comparing ciprofol and propofol in 1890 patients. Across studies, patients had similar demographics, with average ages ranging from 33 to 43 years and BMIs around 22–23 kg/m2. Weight and height, when reported, were also comparable. MAP ranged from 83 to 95 mmHg, and HR from 71 to 77 bpm. The majority of patients had mild systemic illness, as indicated by their ASA II classification. Importantly, all studies met the inclusion criterion requiring that ciprofol and propofol be used as primary sedative agents with any adjunctive medications administered equally across both groups. This ensured a consistent and unbiased comparison of the primary agents’ safety and efficacy across treatment arms.
Baseline characteristics.
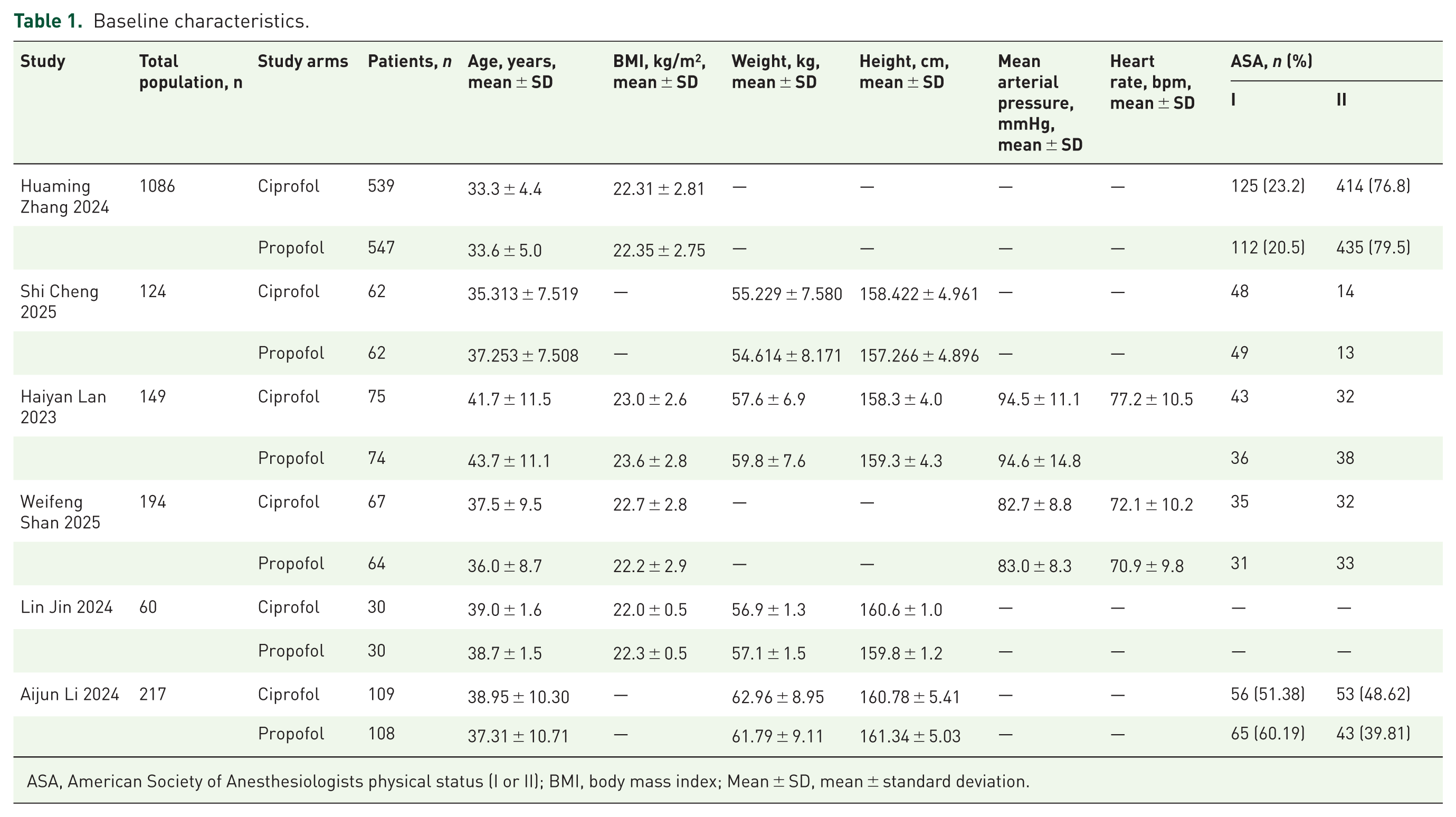
ASA, American Society of Anesthesiologists physical status (I or II); BMI, body mass index; Mean ± SD, mean ± standard deviation.
Risk of bias assessment
The quality of individual studies was analyzed according to the study design using the Cochrane Risk of Bias Tool 2 16 for analyzing the risk of bias in studies. The results of the risk of bias assessment are presented in Supplemental Figure 1. The figure shows that the overall risk of bias across studies is predominantly low. All domains, randomization process, deviations from intended interventions, and missing outcome data, demonstrate 100% low risk, indicating strong methodological integrity in study design and conduct. While the domains of measurement of the outcome and selection of the reported result show some concerns and high risk in a portion of the studies, the majority still fall under low risk. Consequently, the overall bias is primarily low, suggesting that the included studies are generally of good quality, with only minor areas of concern that do not significantly compromise the reliability of the findings.
Certainty of evidence assessment
The table, shown in Supplemental Table 2, summarizes evidence comparing ciprofol and propofol in hysteroscopy, showing that ciprofol generally provides effective sedation with fewer side effects like hypotension, hypoxemia, and injection pain, though some outcomes show uncertainty due to variable data. The certainty of the evidence ranges from high to low based on consistency, precision, and study design.
Meta-analysis
Primary outcomes
Effect of ciprofol versus propofol on sedation success rate
The sedative success rate between the propofol (control) and ciprofol (experimental) groups is assessed using the forest plot in Figure 2 for three different trials. There is no difference in sedation success between the two groups, as indicated by the pooled risk ratio of 1.00 (95% CI: 1.00–1.00) (p = 1.00). All included studies reported very high and nearly identical success rates, reflected in narrow CIs and a perfect overlap in outcomes. The study by Shan et al. 17 contributes the majority of the weight (96.7%), strongly influencing the pooled estimate. There is consistency across all investigations, with no heterogeneity I2 = 0%, p = 1.00). These findings demonstrate that propofol and ciprofol are equally useful for producing a good sedative effect.

Forest plot comparing the sedation success rate between ciprofol and propofol groups across included trials.
Risk of injection pain in ciprofol versus propofol
Figure 3(a) presents the analysis of injection pain. The pooled risk ratio was 0.13 (95% CI: 0.05–0.29), indicating a significantly lower incidence of pain with ciprofol compared to propofol (p < 0.00001). However, heterogeneity was high (I2 = 77%), suggesting variability among studies. Figure 3(b) illustrates the pooled results for injection pain after sensitivity analysis. The ciprofol group had a markedly lower incidence compared to propofol, with a risk ratio of 0.04 (95% CI: 0.01–0.11, p < 0.00001). This indicates a strong and statistically significant reduction in pain at injection, with zero heterogeneity.

(a) Forest plot showing pooled analysis of injection pain between ciprofol and propofol groups; (b) Sensitivity analysis for injection pain showing revised pooled estimates after removing outlier studies.
Risk of bradycardia in ciprofol versus propofol
Figure 4 compares the incidence of bradycardia between ciprofol and propofol. The pooled risk ratio was 1.16 (95% CI: 0.73–1.82), with no statistically significant difference between the groups (p = 0.53). The heterogeneity across the included studies was low (I2 = 26%), indicating consistent findings.
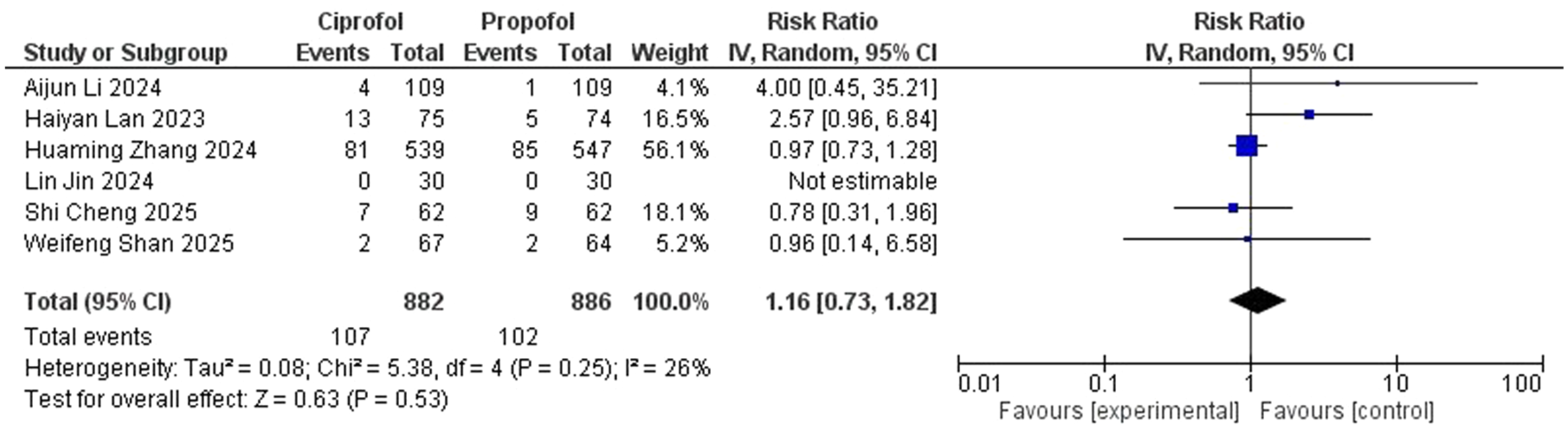
Forest plot comparing the incidence of bradycardia between ciprofol and propofol.
Risk of Hypotension in ciprofol versus propofol
Figure 5 compares the incidence of hypotension between ciprofol and propofol. The pooled mean difference was 0.58 (95% CI: 0.50–0.67), showing a significantly lower risk of hypotension in the ciprofol group (p < 0.000001). Heterogeneity was zero, indicating consistent findings across studies.

Forest plot comparing the incidence of hypotension between ciprofol and propofol groups.
Risk of respiratory complications in ciprofol versus propofol
Figure 6(a) shows the initial pooled analysis of respiratory depression. The risk ratio was 0.51 (95% CI: 0.28–0.92), indicating a statistically significant reduction in respiratory depression with ciprofol (p = 0.03). However, heterogeneity was high (I2 = 79%), raising concerns about study variability. Figure 6(b), after sensitivity analysis, presents a revised risk ratio of 0.67 (95% CI: 0.48–0.92, p = 0.01), with no heterogeneity (I2 = 0%), supporting a more consistent and dependable reduction in respiratory depression with ciprofol.

(a) Forest plot comparing the incidence of respiratory depression between ciprofol and propofol (before sensitivity analysis); (b) Sensitivity analysis for respiratory depression showing revised pooled risk ratio after removing heterogeneous studies.
Effect of ciprofol versus propofol on duration of eyelash response absence
In Figure 7, the forest plot compares the incidence of loss of eyelash reflex between the ciprofol and propofol groups across four studies. The overall pooled mean difference is 0.24 (95% CI: 0.02–0.46), favoring the ciprofol group, indicating a statistically significant difference (p = 0.03). Among the included studies, Lan et al. 14 contribute the greatest weight (89.4%), heavily influencing the overall outcome. Importantly, heterogeneity is low (I2 = 0%, p = 0.44), suggesting consistency across studies. These results suggest that ciprofol may be slightly more effective than propofol in producing the loss of eyelash reflex during induction.
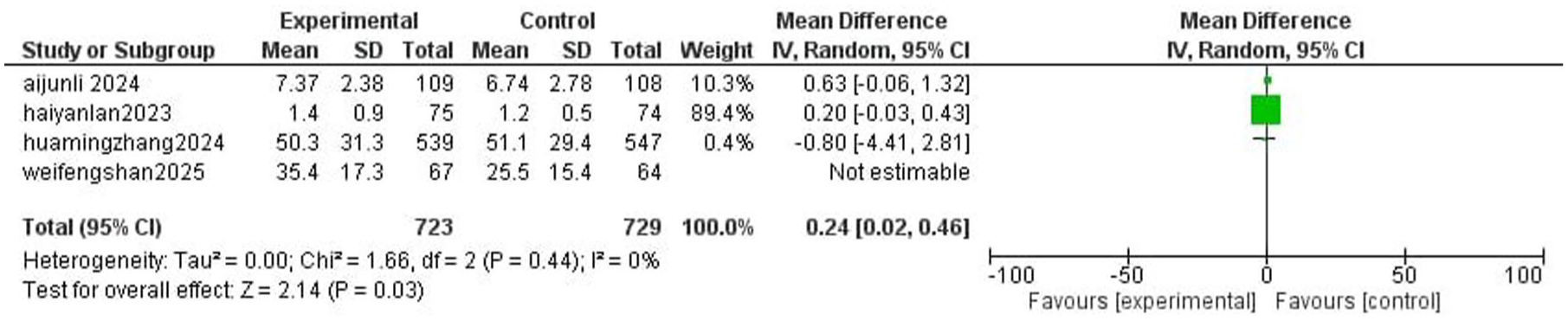
Forest plot comparing duration of eyelash reflex absence between ciprofol and propofol groups.
Effect of ciprofol versus propofol on intraoperative body movement
The forest plot in Figure 8 compares the effect of ciprofol and propofol on intraoperative body movement. The pooled analysis shows a significant reduction in body movement with ciprofol, with a risk ratio of 0.53 (95% CI: 0.39–0.74, p = 0.0002), indicating a 47% lower risk compared to propofol. Two studies—Li et al. 18 (RR: 0.45, 95% CI: 0.23–0.89) and Lan et al. 14 (RR: 0.56, 95% CI: 0.39–0.81)—reported statistically significant reductions in movement with ciprofol. The study by Shan et al. 17 was labeled “Not estimable” in the forest plot because there were zero movement events in both treatment arms; therefore, a risk ratio could not be calculated for that study, and it was excluded from the pooled RR calculation. No heterogeneity (I2 = 0%) confirms ciprofol’s consistent superiority in reducing body movement.
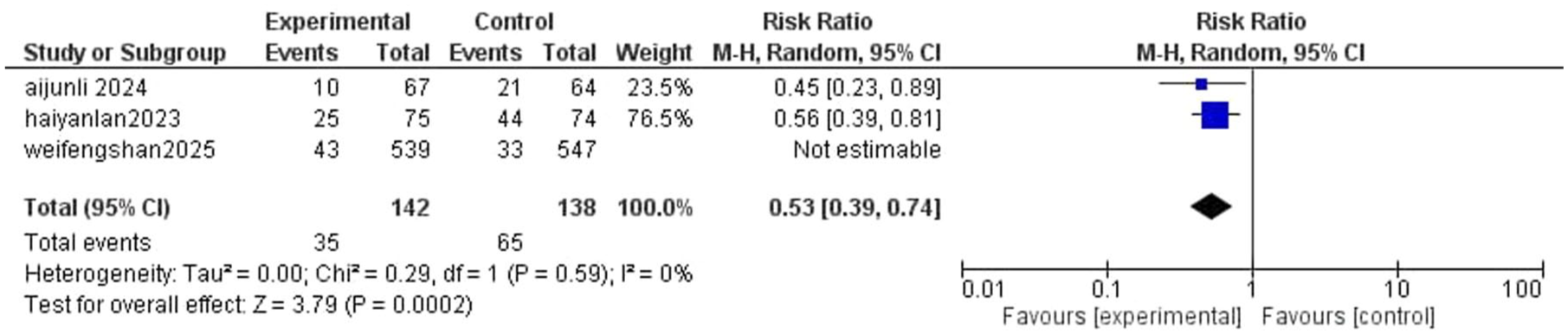
Forest plot comparing the incidence of intraoperative body movement between ciprofol and propofol groups.
Secondary outcomes
Risk of hypoxemia in ciprofol versus propofol
Figure 9 shows the comparison of hypoxemia incidence between the two groups. Ciprofol was associated with a significantly lower risk, with a mean difference of 0.44 (95% CI: 0.25–0.76, p = 0.003). The analysis showed zero heterogeneity, reflecting consistent results across studies.

Forest plot comparing the incidence of hypoxemia between ciprofol and propofol.
Effect of ciprofol versus propofol on recovery time
Figure 10 shows a forest plot analyzing the recovery time between the ciprofol and propofol groups based on three studies. The pooled mean difference is 0.80 (95% CI: 0.05–1.55), showing a statistically significant longer recovery time in the ciprofol group (p = 0.04). Lan et al. 14 is the main contributor to the total outcome, accounting for the full statistical weight (100.0%). The other two studies, Zhang et al. 19 and Shan et al., 17 show wider CIs and contribute minimal weight, possibly due to smaller or more variable sample data. There is no heterogeneity across the studies (I2 = 0%, p = 0.49), suggesting consistency in the findings. These results indicate that ciprofol may be associated with a slightly longer recovery time compared to propofol.

Forest plot comparing recovery time between ciprofol and propofol groups.
Effect of ciprofol versus propofol on heart rate
In Figure 11(a), the mean difference of HR at T1 was −0.69 bpm (95% CI: −1.39 to 0.01), with a borderline non-significant result (p = 0.05). Heterogeneity was zero across studies. Figure 11(b) shows the pooled HR at T2. Although the ciprofol group had a lower heart rate, the difference was not statistically significant (Mean Difference (MD) = −0.80 bpm, 95% CI: −1.82 to 0.23, p = 0.13), with consistent findings across studies (zero heterogeneity). Figure 11(c) displays HR at T3, with a mean difference of −0.23 bpm (95% CI: −0.99 to 0.53, p = 0.55), showing no significant difference between ciprofol and propofol. Heterogeneity remained low. Timepoints for safety assessments were consistent across all studies: preoperatively (T1), intraoperatively (T2), and postoperatively (T3).

(a) Forest plot comparing HR at preoperative time (T1) between ciprofol and propofol groups; (b) Forest plot comparing HR at intraoperative time (T2); (c) Forest plot comparing HR at postoperative time (T3).
Effect of ciprofol versus propofol on MAP
Figure 12(a) shows the comparison of MAP at T1 between the two groups. The mean difference was 1.26 mmHg (95% CI: −0.45 to 2.98), which was not statistically significant (p = 0.15). The studies included showed low heterogeneity, indicating consistent findings. Figure 12(b) illustrates MAP at T2. The ciprofol group showed a significantly higher MAP than the propofol group, with a mean difference of 5.06 mmHg (95% CI: 4.16–5.95, p < 0.00001). Heterogeneity was zero, reflecting consistent outcomes across studies. For MAP at T3, heterogeneity was not applicable.

(a) Forest plot comparing MAP at preoperative time (T1); (b) Forest plot comparing MAP at intraoperative time (T2).
To ensure comparisons were valid, the same three surgical phases—before (T1), during (T2), and after (T3)—were used in every study to record safety issues.
Effect of ciprofol versus propofol on SBP
In Figure 13(a), the pooled mean difference of SBP at T1 was 2.99 mmHg (95% CI: 0.27–5.72), showing a statistically significant increase in SBP in the ciprofol group (p = 0.03). Heterogeneity in the study was zero (I2 = 0%), indicating consistent results. Figure 13(b) illustrates (SBP) at time point T2, comparing ciprofol and propofol. The mean difference was 5.62 mmHg (95% CI: −1.77 to 13.01), which was not statistically significant (p = 0.14). Heterogeneity across studies was low, indicating consistent results. Figure 13(c) displays SBP at time point T3. The ciprofol group showed a significantly higher SBP compared to propofol, with a mean difference of 4.81 mmHg (95% CI: 1.34–8.29, p = 0.007). Heterogeneity was 0%. Timepoints for safety assessments were consistent across all studies: preoperatively (T1), intraoperatively (T2), and postoperatively (T3).

(a) Forest plot comparing SBP at T1 between ciprofol and propofol; (b) Forest plot comparing SBP at T2 between the groups; (c) Forest plot comparing SBP at T3 between the groups.
Effect of ciprofol versus propofol on DBP
Figure 14(a) presents the comparison of DBP at T1 between ciprofol and propofol. The analysis showed a mean difference of 1.41 mmHg (95% CI: −1.51 to 4.34), which was not statistically significant (p = 0.34). Study results were consistent, with low heterogeneity across the included data. Figure 14(b) shows the pooled results for DBP at T3. The ciprofol group had a significantly higher DBP compared to propofol, with a mean difference of 3.82 mmHg (95% CI: 1.54–6.09, p = 0.001). Heterogeneity was zero, supporting the reliability of the result. Timepoints for safety assessments were consistent across all studies: preoperatively (T1), intraoperatively (T2), and postoperatively (T3).

(a) Forest plot comparing DBP at T1 between ciprofol and propofol; (b) Forest plot comparing DBP at T3 between ciprofol and propofol.
Effect of ciprofol versus propofol on SpO2
Figure 15(a) displays studies for SpO2 at T1. The pooled mean difference was 0.05% (95% CI: –0.36 to 0.47), with no statistically significant difference (p = 0.8) and moderate heterogeneity (I2 = 62%; Figure 15(b)). The sensitivity analysis shows the revised estimate after removing the outlier study. The updated mean difference was 0.25% (95% CI: −0.25 to 0.76, p = 0.33), with substantially reduced heterogeneity, improving the consistency of results across studies.

(a) Forest plot comparing SpO2 at T1 between ciprofol and propofol; (b) sensitivity analysis for SpO2 at T1 after excluding an outlier study.
Figure 16(a) presents the studies for SpO2 at T2. The pooled mean difference was 1.30% (95% CI: 0.70–1.89), showing a statistically significant increase in SpO2 with ciprofol compared to propofol (p < 0.0001), but with heterogeneity OF 59%. Figure 16(b) shows that SpO2 at T2 after excluding an outlier in the sensitivity analysis, a revised mean difference of 0.99% (95% CI: 0.41–1.56), maintaining statistical significance (p = 0.0007) and achieving zero heterogeneity, confirming the robustness of the result. Figure 16(c) depicts SpO2 at T3, where the mean difference of −0.08% (95% CI: −0.29 to 0.12) was not statistically significant (p = 0.42). The included studies showed zero heterogeneity. The same three surgical phases—before (T1), during (T2), and after (T3)—were used in every study to record safety issues.

(a) Forest plot comparing SpO2 at T2 between ciprofol and propofol; (b) Sensitivity analysis for SpO2 at T2 after excluding an outlier study; (c) Forest plot comparing SpO2 at T3 between ciprofol and propofol.
Effect of ciprofol versus propofol on hysteroscopy time
Figure 17 displays the forest plot comparing hysteroscopy time between ciprofol and propofol groups across two studies. The pooled mean difference was −0.37 (95% CI: −0.87 to 0.13, p = 0.14), showing no statistically significant difference between the two drugs. The larger study, Zhang et al., 19 contributed most of the weight (97.7%) and reported a small, non-significant reduction (−0.35), while Cheng et al. 20 showed greater variability and a wider CI (−1.28; 95% CI: −4.59 to 2.03). With no heterogeneity observed (I2 = 0%, p = 0.59), the results were consistent, but no clear difference in hysteroscopy time could be established.
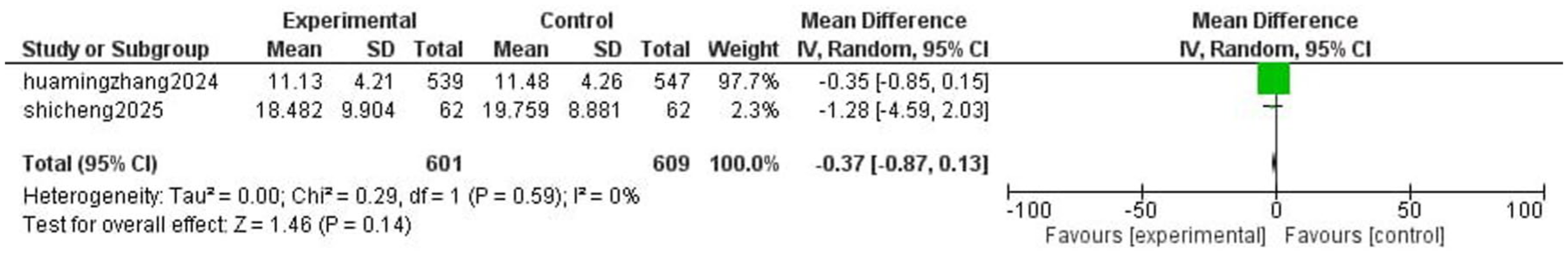
Forest plot comparing hysteroscopy time between ciprofol and propofol groups.
Publication bias
A funnel plot was generated using Comprehensive Meta-Analysis (CMA) version 4 to visually assess publication bias, as shown in Supplemental Figure 2. The funnel plot (Supplemental Figure 2) displays five of the six included studies, as one study had non-estimable values and was excluded from the plot. While some visual asymmetry is noted, suggesting potential publication bias. Egger’s regression intercept yielded a p value of 0.33704, indicating no statistically significant evidence of publication bias in the analysis, as shown in Supplemental Figure 3. Given the limited number of studies, visual interpretation alone may be unreliable; however, Egger’s test provides supportive evidence that bias is unlikely to have substantially influenced the results.
Discussion
Hysteroscopy is a common outpatient procedure for diagnosing and managing intrauterine problems; however, it frequently causes endometrial manipulation and cervical dilatation and results in severe discomfort, which demands strong sedation or anesthesia. 18 The current standard, propofol, is effective but has adverse effects like respiratory depression, hypotension, and injection discomfort. 17 Ciprofol, a novel 2,6-disubstituted phenol derivative with a higher affinity for the GABA receptor, is just as effective but may have fewer adverse effects. 21 Since the anesthesia protocols for hysteroscopy and endoscopic procedures are similar, ciprofol has emerged as a promising substitute due to its better pain management and more stable respiratory and hemodynamic profiles. 18
This meta-analysis and systematic review aims to examine the safety and effectiveness of ciprofol and propofol as sedatives used during hysteroscopy. Six RCTs that assessed a range of clinical characteristics, such as sedation success rates, adverse effect incidence, recovery times, and overall safer experience, are included in the analysis.14,17–21
Among all the noteworthy findings, the most compelling outcome from our pooled analysis was a marked reduction in intraoperative body movements among patients receiving ciprofol (RR = 0.53, 95% CI: 0.39–0.74, p = 0.0002), with a 47% relative decrease in risk, and no variation between studies was noted (I2 = 0%). This interpretation was consistent with some of our included studies by Li et al. 18 and Lan et al., 14 which reported a statistically significant decrease in body movement, establishing ciprofol’s superiority over propofol. Clinically, this reduction leads to better surgical conditions, fewer interruptions, and enhanced patient safety throughout hysteroscopy.
Notably, ciprofol is considered to be four to five times more potent than propofol due to its stronger GABA-A receptor affinity and stereoselectivity, which allows effective sedation at lower dosages.11,22 Conversely, Shan et al. in their study (RR: 1.32, 95% CI: 0.85–2.05) reported no significant difference in the body movements, attributing movements primarily to unsatisfactory anesthetic depths rather than sedative inadequacy. 17
Recovery time is a therapeutically important parameter in ambulatory settings. Our findings showed a statistically significant increase in recovery time with ciprofol compared to propofol, with an average extra time of 0.80 min, and no major differences in outcomes across studies were observed (I2 = 0%, p = 0.49). Ciprofol’s rapid CNS onset and slightly delayed recovery might be attributed to its higher lipophilicity, which allows for broad tissue distribution, slower redistribution from core compartments, and efficient passage across the blood-brain barrier.11,23 Although both drugs are rapidly metabolized, ciprofol’s glucuronidation to the inactive M4 metabolite and renal excretion may contribute to its slightly delayed recovery than propofol, which has a faster hepatic clearance and higher metabolic rate. 24 Supporting our findings, Cheng et al. and Teng et al. in their trials also identified prolonged recovery times in ciprofol groups during hysteroscopy and colonoscopy, respectively, indicating delayed return to full alertness.13,20 In practical terms, such a small delay of <1minute is unlikely to have an affect on patient flow or overall outpatient hysteroscopy effectiveness. From the perspective of the patient, such a small change is unnoticeable and, as a result, has little clinical significance.
In terms of hysteroscopy duration, our analysis aligns with various included studies,14,17,19 all reporting no apparent difference between patients receiving ciprofol and those administered propofol. This was reflected in our pooled analysis showing no significant variation between the two drugs with a mean difference of −0.37 (95% CI: −0.87 to 0.13, p = 0.14), with consistent outcomes across studies and no observed heterogeneity (I2 = 0%). This suggests that the sedative used does not substantially influence procedural duration. Instead, factors such as surgical expertise, uterine anatomy, and complexity of underlying pathology are likely to influence operative time. 25
Our results demonstrated that ciprofol achieved higher rate of eyelash suppression compared to propofol; the average difference was 0.25(95% CI: 0.02–0.45, p = 0.03), favoring ciprofol. The studies were very consistent with each other (I2 = 0%, p = 0.44). This was in line with some of the previous studies,14,17 which also demonstrated the results favoring ciprofol. Greater suppression of the eyelid reflex could mean deeper and more stable sedation, which could indicate that less intraoperative rescue drugs are required. This effect likely stems from ciprofol’s strong stereoselective modulation of GABA-A, particularly the α1β2γ2 subtype present predominantly in the brain, and a raised affinity of ciprofol to this receptor type possibly leads to increased neuronal inhibition in the region governing reflexes.11,26,27
In terms of sedation success rate, all included trials uniformly demonstrated that ciprofol and propofol have similar sedative efficacy. Our results demonstrated a risk ratio of 1.00 (95% CI: 1.00–1.00, p = 1.00), showing no distinction at all, and the findings were perfectly consistent across studies (I2 = 0%, p = 1.00). This supports the use of ciprofol as an effective alternative to propofol.
Injection pain is considered one of the most common adverse effects of propofol administration across different studies and is related to aqueous concentration of propofol, which produces discomfort for patients.14,20,22,28
The reported incidence of injection pain of propofol ranges from 50% to 70%.14,29,30
Our analysis confirms that ciprofol was associated with a significantly lower incidence for injection pain as compared to propofol. Ciprofol shows poor water solubility and increased lipophilicity, and is formulated as an oil-in-water emulsion with a lower concentration of free drug in the aqueous phase than propofol. 31 The intensity of pain is also proportional to the concentration and site of injection. 32 Since ciprofol makes less contact with the venous endothelium, the injection pain is significantly reduced. Due to its deeper level of sedation, chemical resemblance to propofol, lower plasma concentration, and higher hydrophobicity, clinically, it contributes to improved patient comfort and lessens the frequency of body movements.18,19,30
Ciprofol also significantly reduced the risk of hypotension compared to propofol (pooled mean difference = 0.58, 95% CI: 0.50–0.67, p < 0.000001) with no heterogeneity (I2 = 0%). Our findings are in agreement with those of Cheng et al., who discovered that the incidence of hypotension during gastrointestinal endoscopy was significantly decreased by using ciprofol (OR = 0.48, 95% CI: 0.32–0.72, p = 0.0004, I2 = 59%). 33 It suggested that the expansion of peripheral blood vessels and the inhibition of contraction of myocardium were less affected by ciprofol, which provided a more stable hemodynamic environment. 30 This is further supported by data from Chen et al., who show that ciprofol has a little less pronounced effect on the cardiovascular system than propofol, with non-inferior effects on blood pressure and heart rate. 34 Additionally, ciprofol due to its faster metabolism at lower doses and inert metabolites has a shorter duration of cardiovascular suppression than propofol. 23
Clinically, this might reduce the need for continuous monitoring and the use of vasopressor support, especially in high-risk cardiovascular patients undergoing an outpatient hysteroscopy.
In terms of respiratory depression, our results are consistent with those of Lan et al., who found that compared to propofol, ciprofol resulted in fewer airway interventions and a lower incidence of respiratory depression. 14 Similarly, our pooled analysis showed that ciprofol significantly decreased respiratory depression (RR = 0.51, 95% CI: 0.28–0.92, p = 0.03), despite strong heterogeneity (I2 = 79%). However, after sensitivity analysis, the heterogeneity was eliminated (I2 = 0%), the revised RR was 0.67 (95% CI: 0.48–0.92, p = 0.01), suggesting consistent and trustworthy results. This is particularly valuable clinically as ciprofol would be a safer choice for maintaining respiratory stability throughout hysteroscopic procedures and minimizes procedural interruptions. 35 Similarly, in the Shan et al. trial on hysteroscopy, ciprofol produced significantly lower respiratory depression than propofol (7.5% vs 20.3%). 17 This may be due to ciprofol interacting differently with GABA-A receptor subunits, as propofol mainly targets the β3 subunit, which is closely associated with respiratory depression.36–38
Liao et al. also suggest in his study that a central nervous system mechanism could be the cause of the discrepancy. Ciprofol suppresses the respiratory control centers in the medulla oblongata less profoundly at equivalent doses than propofol, which show that it has a weaker inhibitory effect on swallowing function. 39
Changes in heart rate and bradycardia are significant cardiovascular side effects of sedation. According to our analysis, the heart rate mean difference at T1 was −0.69 bpm (p = 0.05), and the differences were statistically non-significant at T2 and T3, with little heterogeneity overall. There was no apparent difference between propofol and ciprofol, as indicated by the bradycardia pooled risk ratio of 1.16 (95% CI: 0.73–1.82, p = 0.53). These findings are consistent with those of Zeng et al., who reported a small, statistically insignificant increase in bradycardia possibly due to reduced coughing and injection pain 30 and with Jin et al., who observed no cases of bradycardia in either group (p > 0.05). 21
These findings suggest that propofol and ciprofol have comparable cardiovascular safety profiles, with neither agent demonstrating a statistically significant effect on heart rate or the incidence of bradycardia; however, further studies are warranted to confirm these observations.
Ciprofol also provided better hemodynamic and respiratory stability during sedation as compared to propofol, showing significantly higher SBP, DBP, MAP, and SpO2 at T2 in our trial. Our results align with those of Li et al. 18 and Zhang et al. 19 who reported that ciprofol raised blood pressure and SpO2 levels. Clinically, this may lead to safer sedation for patients with low cardiopulmonary reserve, and it is essential to highlight that the included studies largely involved relatively healthy patients (ASA I–II) hence, the benefits of ciprofol in the therapy of patients with severe comorbidities require additional investigation.
Several factors may have influenced the outcomes of this meta-analysis beyond the choice of sedative itself. Patient characteristics like age, BMI, baseline hemodynamics may contribute to the risk of hypotension, recovery time, or respiratory depression for some women. Likewise, procedural-related variables, including the complexity of hysteroscopy, the degree of uterine manipulation, or the need for cervical dilation, may affect the intraoperative body movements and sedation depth. Furthermore, slight variations in anesthetic procedures among studies, such as the use of premedication, adjunctive analgesics, or differing target sedation levels may have also affected side effects and recovery durations. Altogether, these factors remind us that sedation outcomes are not determined by the drug alone but also by patient and procedural factors, highlighting the importance of choosing the sedative according to each individual case.
This systematic review and meta-analysis is the first thorough synthesis of current randomized evidence comparing ciprofol with propofol in hysteroscopic procedures. The findings establish that ciprofol has comparable sedative efficacy to propofol while exhibiting greater safety in critical perioperative outcomes such as hypotension, respiratory depression, and injection pain. The analysis also highlights that ciprofol consistently reduced intraoperative body movements and improved hemodynamic stability. Importantly, these benefits were similar across many recent RCTs, reflecting emerging trends in modern sedation practice and identifying ciprofol as a promising next-generation drug for gynecologic procedural sedation.
Clinicians performing hysteroscopy may choose ciprofol as a good alternative to propofol when it comes to patient comfort and minimizing cardiopulmonary or injection-related side effects. Its increased hemodynamic and respiratory stability can be especially useful in outpatient settings where prompt recovery and minimal problems are required. When introducing ciprofol into practice, anesthetic teams should adhere to established dose guidelines, monitor patients using standard sedation protocols, and take into account specific patient characteristics such as baseline cardiovascular state, anxiety levels, and procedural complexity. Although recuperation time may be slightly longer, the difference is clinically insignificant and should not impair workflow in most day-care facilities. Adoption of ciprofol may enhance overall procedural safety and patient experience, while further evidence from multicenter studies will help refine its role across broader and higher risk populations.
Limitations
Our meta-analysis also has several limitations that must be acknowledged.
Firstly, all included RCTs were single-center studies with small sample sizes, which may have introduced bias and limited generalizability of the findings.
The majority of trials only included low-risk female patients (ASA I–II), excluding higher risk populations such as elderly, ASA III–IV patients, or those with obesity and comorbidities, limiting their application to broader clinical settings.
Secondly, subjective scales were primarily used to measure sedation depth, which might have impacted accuracy. Follow-up periods were brief, focusing primarily on the perioperative phase, and no investigation of long-term consequences like delayed recovery, postoperative nausea and vomiting (PONV), or cognitive effects was conducted. Two high-risk trials (Lan et al. 14 and Zhang et al. 19 ) showed methodological concerns, which involve subjective outcome assessment without blinding (Domain 4) and possible selective reporting because there was no pre-specified analysis plan (Domain 5). Due to bias and reporting variability, these issues may affect the internal validity of outcomes like recovery time, sedation depth, and adverse events.
Additionally, the exclusive focus on hysteroscopy limits the applicability of results to other procedures. And because the majority of research was conducted in China, larger, multicenter trials with more diverse populations are required to validate these findings across the globe.
Conclusion
In conclusion, this meta-analysis demonstrates that ciprofol displays a better safety profile, including enhanced hemodynamic and respiratory stability and decreased injection discomfort, and is just as effective as propofol for sedation during hysteroscopy. These findings support the use of ciprofol as a safe and effective alternative for sedation during hysteroscopy. Further large-scale, multicenter trials are warranted to validate these results across broader and more diverse patient populations, including those with higher perioperative risk.
Supplemental Material
sj-docx-1-taw-10.1177_20420986261427077 – Supplemental material for Efficacy and safety of ciprofol versus propofol for sedation in hysteroscopy: a systematic review and meta-analysis
Supplemental material, sj-docx-1-taw-10.1177_20420986261427077 for Efficacy and safety of ciprofol versus propofol for sedation in hysteroscopy: a systematic review and meta-analysis by Zoha Anwar, Hafsa Ahsun, Taha Iqbal, Muhammad Saad khan, Sarah Rehan, Areej Fatima, Fawad Sarwar, Hafsa Haseen, Umaimah Naeem, Kalpana Singh and Aminath Waafira in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
