Abstract
Background:
Propofol combined with alfentanil is suitable for intravenous anesthesia for day-case hysteroscopy.
Objective:
To investigate the median effective dose (ED50) and 95% effective dose (ED95) of alfentanil compounded with propofol for day-case hysteroscopy.
Design:
In all, 29 patients who volunteered for painless hysteroscopy in 2022 were recruited. 1.5 mg/kg propofol was given as a sedative to all patients. The trial was conducted using the modified Dixon sequential method, with an initial dose of 10 μg/kg of alfentanil, and the subject’s alfentanil dose depended on whether the prior hysteroscopy had failed, which was defined as inadequate cervical dilatation and hysteroscope placement with the patient exhibiting body movement, frowning, or a MOAA/S score >1. If the hysteroscopy failed (i.e. a positive response), the subsequent subject’s alfentanil dosage was raised, and conversely (i.e. a negative response), the dose was decreased, with the adjacent dose ratio always being 1:1.2. The formal test begins with the first crossover wave and lasts until seven crossover waves materialize.
Methods:
The probit method was used to calculate the ED50, ED95, and corresponding 95% confidence intervals (CIs) of alfentanil compounded with propofol for hysteroscopy.
Results:
The ED50 and ED95 of alfentanil combined with propofol for day-case hysteroscopy were 5.701 (95% CI: 3.841–7.069) μg/kg and 8.817 (95% CI: 7.307–20.868) μg/kg, respectively.
Conclusion:
Alfentanil at 8.817 μg/kg in conjunction with propofol is a successful and safe approach for day-case painless hysteroscopy.
Trial registration:
The trial registry name: Modified sequential method to determine the half-effective dose of alfentanil compounded with propofol for ambulatory hysteroscopy. The URL of registration is https://www.chictr.org.cn/showproj.html?proj=171786, where the full trial protocol can be accessed. Registration number: ChiCTR2200061619.
Plain language summary
Why was this study done? Hysteroscopy is a procedure in which a hysteroscope is placed after the cervix is dilated to observe lesions in the uterine cavity. This study explores the effective dose of alfentanil combined with propofol for day-case (i.e., patients admitted and discharged on the same day) hysteroscopy to provide patients with comforting anesthesia (i.e., pain relief) during hysteroscopy.
What did the researchers do? The research team recruited 29 patients who volunteered for painless hysteroscopy in 2022. All patients received alfentanil and propofol for intravenous anesthesia. If anesthesia was not effective, subsequent subjects increased the dose of anesthetic medication, and vice versa, the dose was decreased. According to this method, we can find the optimal dose of anesthetic drugs in hysteroscopy.
What did the researchers find? The effective dose of alfentanil combined with propofol in patients undergoing painless hysteroscopy was 8.817 μg/kg.
What do the findings mean? Alfentanil at 8.817 μg/kg in conjunction with propofol is a successful and safe approach for day-case painless hysteroscopy.
Keywords
Introduction
Hysteroscopy is the gold standard for evaluating intrauterine lesions in patients with abnormal uterine bleeding and as a way to assess the status of the uterine cavity in patients with infertility. 1 Day-case hysteroscopy is widely used in gynecologic patients for its minimal invasiveness and quick recovery, but there remains a disconnect between clinician and patient pain reports. 2 The insertion of instruments and dilatation of the cervix cause varying degrees of stress and pain to the patient, so the choice of anesthesia protocol is critical to the successful performance of day-case hysteroscopy and the management of the patient. In recent years, intravenous anesthesia without tracheal intubation has become increasingly popular as newer anesthetics provide adequate sedation and analgesia during surgery.3–6 During monitored anesthesia care, propofol is commonly used as the primary anesthetic and opioid analgesics as adjunctive analgesics.3–7
Propofol is a short-acting sedative drug with rapid onset and postoperative awakening; however, the analgesic effect is weak. Alfentanil is a short-acting analgesic, but serious side effects such as respiratory depression and postoperative nausea and vomiting have limited its clinical use.8,9 The use of compound anesthesia can provide adequate analgesia while reducing opioid consumption and minimizing opioid-related side effects. However, the minimum effective dose of alfentanil for hysteroscopy when combined with propofol is unknown. The purpose of this study was to investigate the median effective dose (ED50) and 95% effective dose (ED95) of alfentanil combined with propofol for day-case hysteroscopy.
Methods
Participants
We followed CONSORT guidelines (CONSORT Checklist in the Supplemental Material). 10 The protocol was registered with the Chinese Clinical Trials Registry on 29 June 2022 (ChiCTR2200061619), and all experiments were conducted in accordance with the principles of the Helsinki Declaration. The first participant was enrolled in the trial on 30 June 2022, which marked the start of recruitment. Patients who belonged to the American Society of Anesthesiologists (ASA) I–II were between the ages of 18 and 55, had a BMI of 18–25 kg/m2, and had been classified by gynecologists as needing hysteroscopy. The exclusion criteria were as follows: (1) patients with a history of severe cardiac, cerebral, pulmonary, hepatic, renal, or metabolic diseases, (2) heart rate <50 beats/min, (3) history of acute respiratory inflammation not cured within 2 weeks, (4) hypertension [systolic blood pressure (SBP) >180 mmHg] or hypotension (SBP <90 mmHg), and (5) known allergy to emulsions or opioids. The adaptive clinical trial was adapted based on specific criteria and therefore used a seven-step crossover rule to terminate the inclusion of participants. The sample size was not calculated a priori for this study.
Scoring criteria
The Modified Observer’s Assessment of Alertness and Sedation (MOAA/S) scores were as follows11–13: 5 points: the patient responds to regular tone name calls normally; 4 points: the patient is unresponsive to normal-tone name calls; 3 points: the patient responds only to repeated or loud name calls; 2 points: the patient responds only to gentle pricking or shaking; 1 point: the patient responds only to pain from the trapezius squeeze; 0 point: the patient did not respond to pain from the trapezius squeeze.
Study methods
Before surgery, all patients fasted from eating and drinking, and no preoperative medicine was administered. Once the patient entered the operating room, peripheral capillary oxygen saturation (SpO2), noninvasive blood pressure, and electrocardiography were monitored regularly. After the vein was opened normally, anesthesia induction was performed. We did not consult patients or the general public during the study’s design to safeguard patient privacy and safety. The initial dose of alfentanil was determined to be 10 μg/kg based on preliminary pretests and previous research literature.8,9 All patients received more than 30 s of intravenous alfentanil (Yichang Renfu Pharmaceutical Co., Ltd., Yichang, China), followed by 1.5 mg/kg of intravenous propofol (AstraZeneca, London, UK). The modified Dixon sequential method was used to conduct the trial, and the subject’s alfentanil dose depended on whether the prior hysteroscopy had failed, which was defined as inadequate cervical dilatation and hysteroscope placement with the patient exhibiting body movement, frowning, or a MOAA/S score >1.11–14 If the hysteroscopy failed (i.e. a positive response), the subsequent subject’s alfentanil dosage was raised, and conversely (i.e. a negative response), the dose was decreased, with the adjacent dose ratio always being 1:1.2. The formal test begins with the first crossover wave and lasts until seven crossover waves materialize. Failed hysteroscopy (positive reaction) was in the positive group and otherwise in the negative group.
All anesthesia is administered by an anesthesiologist with the rank of attending or higher, and hysteroscopy is administered by an experienced gynecologist with the rank of attending or higher. Additional propofol 1 mg/kg was added intravenously when a failed hysteroscopy occurred. Atropine (0.25–0.5 mg) was administered as needed if heart rate (HR) < 50 beats/min. When SBP < 90 mmHg or SBP decreases >20% of the basal value, give ephedrine 6–12 mg. When SpO2 is lower than 90%, hold up the lower jaw to open the airway and assist breathing when necessary.
Observation indices
The main observations included: alfentanil dose, propofol starting dose, total propofol dose, and operation duration. Secondary observations included visual analog scores (VAS, scores of 0–10 represent different degrees of pain with 0 = painless and 10 = severe pain) at 2 and 6 h postoperatively and various adverse effects, such as nausea, vomiting, choking, dizziness, hypoxemia, bradycardia, and hypotension. NSAIDs (flurbiprofen axetil 50 mg, or parecoxib 40 mg) were given if the VAS score was above 3 until the VAS score was below 3.
Statistical analysis
Statistics were obtained using SPSS (version 25.0, SPSS Inc., Chicago, IL, USA), GraphPad Prism 9.0.0 (GraphPad Software, San Diego, CA, USA), Origin Pro 2021 (Origin Lab Co., Northampton, MA, USA), and Adobe Illustrator (Adobe Systems, Waltham, MA, USA) statistics. Normally distributed variables were presented as means ± standard deviation (SD) and compared between two groups (positive group and negative group) using the student t-test. By the probit method (probability unit regression), the ED50, ED95, and corresponding 95% confidence intervals (CIs) were calculated for alfentanil epidural labor analgesia. Plotting alfentanil sequential diagrams and dose–effect relationship curves. Statistical significance was defined as two-sided p < 0.05 for all analyses.
Results
A total of 29 patients were analyzed in this study, and the flow chart of the experiment is shown in Figure 1. The general demographic and clinical characteristics of the patients are shown in Table 1. Demographic data (age, weight, height, and BMI) were similar between the positive and negative groups, and there were no significant differences in clinical characteristics between the two groups except for alfentanil dose. No intraoperative adverse effects such as bradycardia or hypotension were observed in the participating subjects. Two patients developed respiratory depression (SpO2 higher than 90%) and recovered after the administration of mandibular support without face mask pressure oxygenation. At the postoperative follow-up, there were no adverse effects such as nausea and vomiting, dizziness, or headache. The VAS scores at 2 and 6 h postoperatively were below 3, and no patients requiring additional analgesic medication were seen.
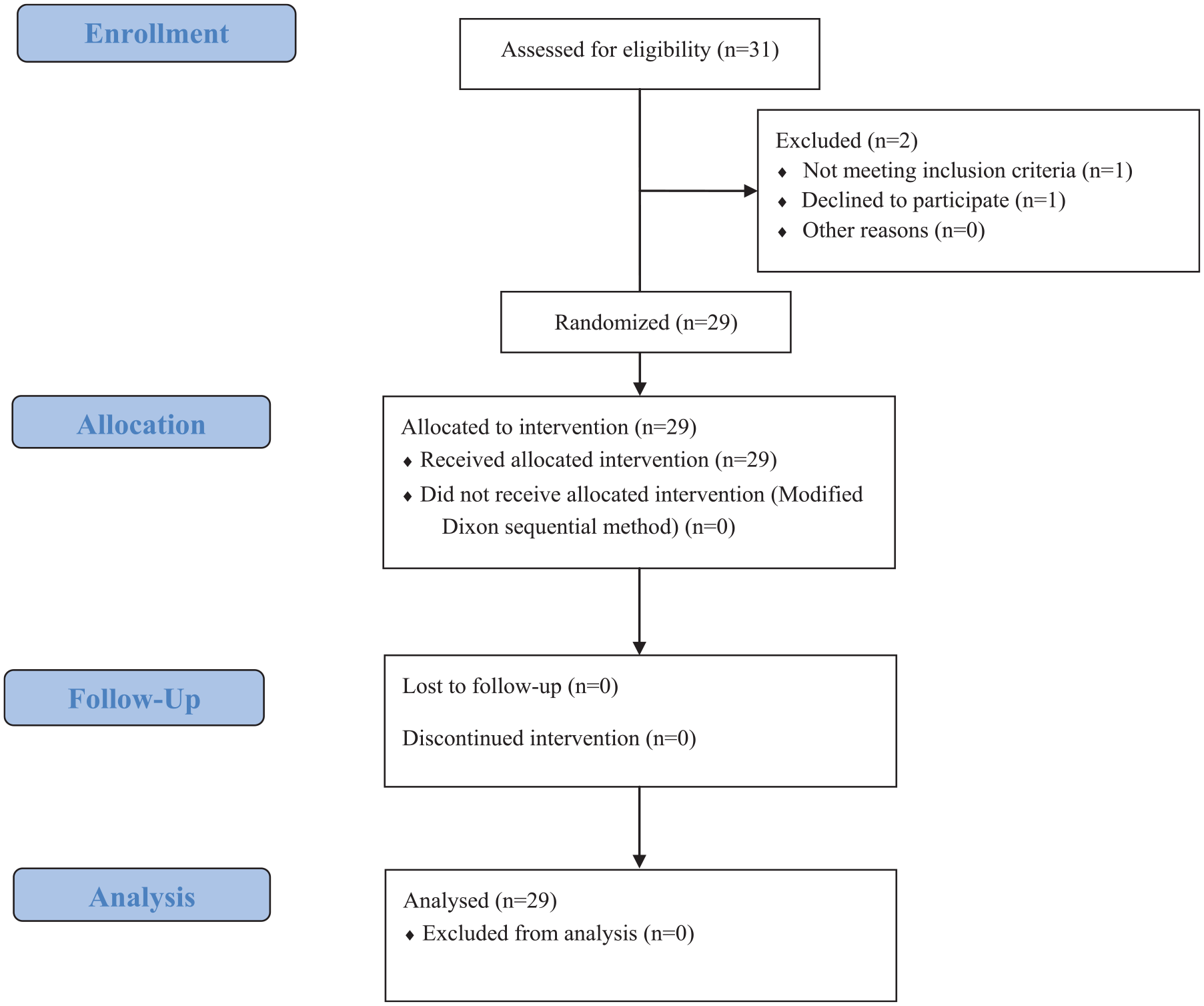
Flow diagram of the study.
Characteristics of patients.

Asterisks denote a statistical difference, two-sided t-test, *p < 0.05.
BMI, body mass index.
The sequence of positive and negative reactions to alfentanil for ambulatory hysteroscopy is shown in Figure 2. The probabilistic unit regression equation fitted according to the probit regression analysis was Probit (P) = −3.010 + 0.528 × alfentanil dose. ‘Goodness of fit test’ χ2 = 1.629 (p = 0.898) indicates the equation was a robust fit of the data. The dose–effect curve of alfentanil for day-case hysteroscopic surgery (Figure 3) showed an ED50 and its 95% CI of 5.701 (3.841–7.069) μg/kg and an ED95 and its 95% CI of 8.817 (7.307–20.868) μg/kg.
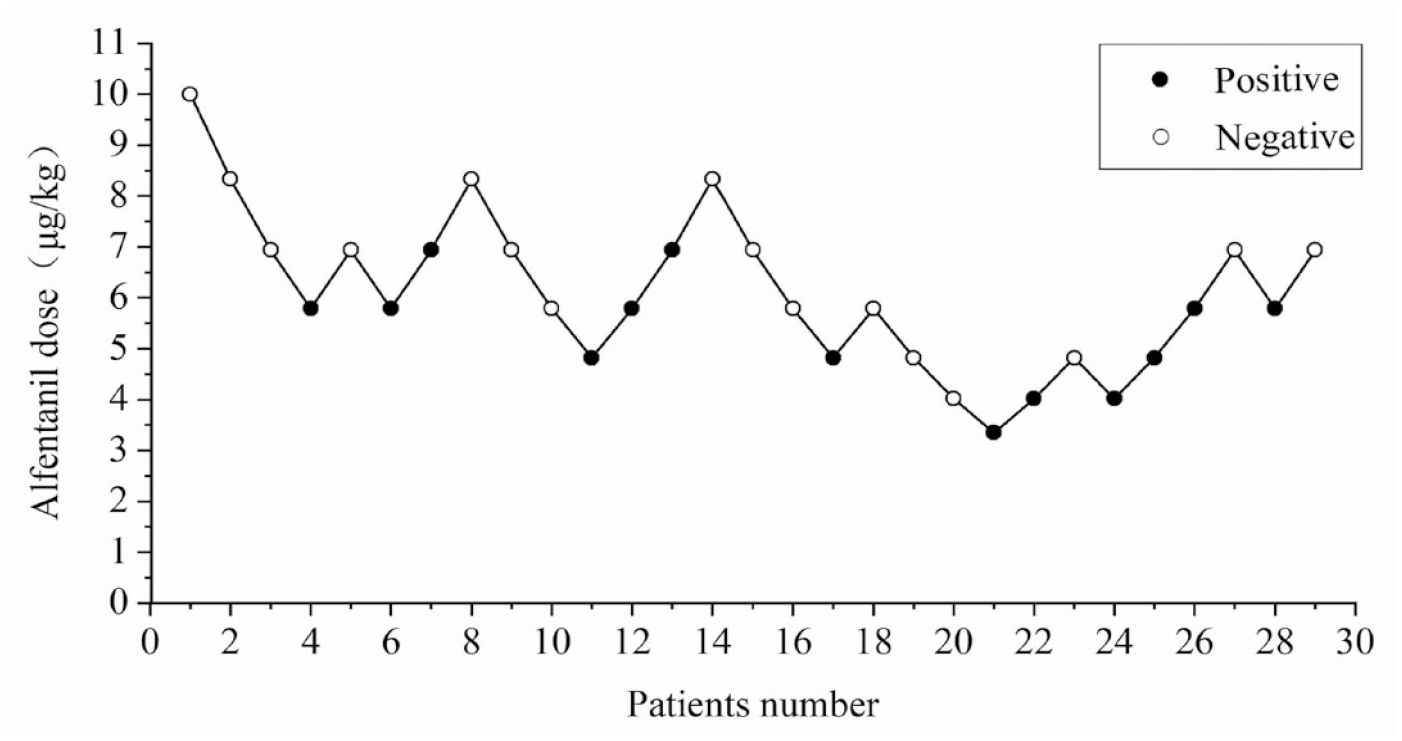
Sequence diagram of alfentanil for day-case hysteroscopy. Solid circles represent positive reactions; hollow circles represent negative reactions.

The dose–effect curve of alfentanil for day-case hysteroscopic surgery.
Discussion
With the benefits of reduced trauma, a quicker recovery, and a shorter hospital stay, hysteroscopic surgery has been used often in gynecological patients as a minimally invasive approach for the diagnosis and treatment of uterine problems. Hysteroscopic operations can be carried out in a day-case hospital setting, which saves time and money and can spare patients from major surgical procedures. Although the procedure is thought to be safe and well-tolerated, preserving patient comfort and satisfaction is nevertheless of utmost importance since cervical dilatation and intrauterine tissue extraction can be quite painful. The choice of the right anesthesia, anesthetic drugs, and dose is particularly crucial to ensuring that patients are induced without choking have stable intraoperative vital signs, recover quickly from surgery, and have no adverse reactions such as nausea and vomiting, headache and dizziness, and abdominal pain, which truly reflect the essence of enhanced recovery after surgery.
Propofol has a wide-ranging central nervous system depressive effect, and its short terminal half-life period speeds up psychomotor recovery, particularly in patients receiving ambulatory surgery. 15 Over the past 30 years, propofol has been the most widely used intravenous anesthetic due to its superior pharmacokinetic (PK) and pharmacodynamic (PD) properties. 15 However, it is undeniable that propofol has time- and dose-dependent depressive effects on the cardiovascular and respiratory systems, which limits its use in basic hospitals with a shortage of anesthesiologists and subpar resuscitation facilities. In conjunction with propofol, opioids (such as fentanyl, 16 sufentanil, 5 and remifentanil 3 ) are frequently used for general intravenous anesthetic during day-case surgery for hysteroscopy. Because PK and/or PD interactions may alter medication dose–effect relationships, they are often not explored separately when thinking about therapeutic applications. 15
When performing a hysteroscopy, two important challenges that anesthesiologists must address are reducing side effects and maintaining appropriate sedation and analgesia. Alfentanil, an opioid analgesic, exhibits the typical side effects of this class of medicines. However, it causes less respiratory depression than an equivalent analgesic dose of fentanyl, probably due to its lower systemic clearance, smaller volume of distribution, and shorter half-life. 17 Alfentanil can be utilized as an additional analgesic for outpatient surgical operations since it has a quicker start of action, a shorter half-life, and greater vagal stimulation qualities than related anesthetics like fentanyl and sufentanil. 17 Propofol combined with nalbuphine has been used for painless hysteroscopy. 7 Nalbuphine, a long-acting opioid, has the major drawback of more considerable postoperative sedation and prolonged recovery time, which is not conducive to the development of day-case surgery. Short-acting opioids such as remifentanil and alfentanil in combination with sedative medications have been widely used for rapid surgery.3,17,18 Previous studies have compared remifentanil and alfentanil for outpatient surgery, and the incidence of nausea and vomiting was significantly higher in the remifentanil group than in the alfentanil group. 19 So this study used alfentanil in combination with propofol for day-case surgery.
Propofol and alfentanil work together to provide analgesia via the adenylyl cyclase pathway. 20 This interaction has a ceiling effect, which means that increasing opioid doses does not strengthen the interaction’s impact. 21 When the combination of these two drugs shows synergistic effects, the two drugs can be used at lower doses, which may result in fewer side effects. 22 On the one hand, the combination significantly reduced the blood propofol concentration at awakening, allowing all patients to breathe adequately at awakening. 23 On the other hand, the plasma concentration of alfentanil increased by 20% in the presence of propofol, which reduced the dosage of alfentanil.20,24 From a PK point of view, propofol may have an inhibitory effect on alfentanil metabolism, which means that propofol may lead to increased toxicity of alfentanil. 24 In addition, alfentanil enhances the depressant effect of propofol on blood pressure, increasing the risk of side effects such as respiratory depression, nausea, vomiting, and prolonged awakening time. 21 As a result, determining the effective dosage of alfentanil coupled with propofol is required to provide a reference for sensible therapeutic usage.
The Dixon sequential method was used in this study to determine the ED50 and ED95 of alfentanil since it is simple and efficient with a small sample size. A single push of 10 μg/kg alfentanil produced analgesia for 7–9 min, which is comparable to the duration of action of propofol.8,9 Referring to the dose of alfentanil required for moderate sedation in previous studies, 10 μg/kg was used for hysteroscopy in this study. 9 In light of the previous hysteroscopy, which used 1.5–2.0 mg/kg propofol alone for anesthesia induction, this study used a combination of 1.5 mg/kg propofol and alfentanil for anesthesia. 25 Patients moved or frowned frequently during hysteroscopy when a score of 2 ⩽ MOAA/S score ⩽ 3. Therefore, a MOAA/S score ⩽1 was used as a valid sedation score in this study.9,11–13 After the hysteroscopy in the 29th instance, there were seven crossover waves of both positive and negative responses. An arithmetic design with seven alfentanil dose groups, including 3.35, 4.02, 4.82, 5.79, 6.94, 8.33, and 10 μg/kg, was used in this experiment according to the requirements of the probit method. We found that the ED50 and ED95 of alfentanil compounded with propofol for daytime hysteroscopy were calculated as 5.701 (95% CI: 3.841–7.069) μg/kg and 8.817 (95% CI: 7.307–20.868) μg/kg, respectively.
In this study, no adverse effects such as bradycardia, hypotension, nausea, vomiting, dizziness, or headache were observed in any of the 29 patients. Both alfentanil and propofol are sedatives with similar rates of airway and respiratory adverse effects, and the transitory respiratory depression in two patients was a combination of the two. 8 All patients had a postoperative VAS score of less than 3, that is, mild pain, and were satisfied with the procedure.
This investigation was limited by the inability to quantify the amount by which the addition of alfentanil lowered the effective dosage of propofol. Therefore, the optimal dose of alfentanil when combined with different doses of propofol needs to be further explored. The same dose of a drug produces different effects and adverse reactions in different populations. The propofol and alfentanil dosages estimated in this study only apply to patients who are ASA I or II, which means that it is necessary to investigate the effective dose of alfentanil for patients with other health conditions (ASA III or IV). Furthermore, despite rigorous adherence to the code of practice, the small sample size of this study may be prone to selection bias, and hence the results of the experiment must be validated by a large-sample and multicenter investigation.
Conclusion
The ED50/ED95 of alfentanil combined with propofol in patients undergoing painless hysteroscopy were 5.701 and 8.817 μg/kg, respectively. As a result, alfentanil at 8.817 μg/kg in conjunction with propofol is a successful and safe approach for day-case painless hysteroscopy.
Supplemental Material
sj-docx-1-taw-10.1177_20420986231214992 – Supplemental material for Modified Dixon sequential method to determine the effective dose of alfentanil compounded with propofol for day-case hysteroscopy
Supplemental material, sj-docx-1-taw-10.1177_20420986231214992 for Modified Dixon sequential method to determine the effective dose of alfentanil compounded with propofol for day-case hysteroscopy by Chenyang Xu, Rui Peng, Xiali Qian, Shanwu Feng and Hongmei Yuan in Therapeutic Advances in Drug Safety
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
